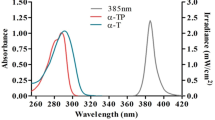
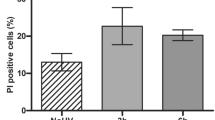
Abstract
Reactive oxygen species (ROS) generated by ultraviolet (UV) irradiation are counterbalanced by endogenous antioxidant systems. To test the hypothesis of a novel photoprotective approach, we irradiated epidermis reconstructed with normal human keratinocytes overexpressing sustainably lentivirus-mediated catalase (CAT), copper/zinc superoxide dismutase (CuZnSOD) or manganese superoxide dismutase (MnSOD) enzymes. We found that following UVB irradiation there was a marked decrease in sunburn cell formation, caspase-3 activation and p53 accumulation in human reconstructed epidermis overexpressing CAT. Moreover, UVA-induced hypertrophy and DNA oxidation (8-oxodeoxyguanosine) were decreased by CAT overexpression. These effects were not achieved by overexpression of CuZnSOD or MnSOD. In conclusion, vector-mediated CAT overexpression could be a promising photoprotective tool against deleterious effects of UV irradiation such skin cancer especially in monogenic/polygenic photosensitive disorders characterized by ROS accumulation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Abbreviations
- CAT:
-
catalase
- CPD:
-
cyclobutane pyrimidine dimer
- CuZnSOD:
-
copper/zinc superoxide dismutase
- E:
-
epidermis
- MnSOD:
-
manganese superoxide dismutase
- NT:
-
non-transduced
- nIr:
-
non-irradiated
- ODC:
-
ornithine decarboxylase
- 8-oxodG:
-
8-oxodeoxyguanosine
- RE:
-
reconstructed epidermis
- SBC:
-
sunburn cell
- SVHK:
-
SV40-transformed human keratinocytes
References
de Gruijl FR, van Kranen HJ, Mullenders LH . UV-induced DNA damage, repair, mutations and oncogenic pathways in skin cancer. J Photochem Photobiol B Biol 2001; 63: 19–27.
Boukamp P . Non-melanoma skin cancer: what drives tumor development and progression? Carcinogenesis 2005; 26: 1657–1667 (Review).
Djavaheri-Mergny M, Mergny JL, Bertrand F, Santus R, Maziere C, Dubertret L et al. Ultraviolet-A induces activation of AP-1 in cultured human keratinocytes. FEBS Lett 1996; 384: 92–96.
Tyrrell R . Redox regulation and oxidant activation of heme oxygenase-1. Free Radic Res 1999; 31: 335–340.
Fuchs J, Packer L . Ultraviolet irradiation and the skin antioxidant system. Photodermatol Photoimmunol Photomed 1990; 7: 90–92.
Danno K, Horio T . Sunburn cell: factors involved in its formation. Photochem Photobiol 1987; 45: 683–690.
Bayerl C, Taak S, Moll I, Jung EG . Characterization of sunburn cells after exposure to ultraviolet light. Photodermatol Photoimmunol Photomed 1995; 11: 149–154.
Schwarz A, Bhardwaj R, Aragane Y, Mahnke K, Riemann H, Metze D et al. Ultraviolet-B-induced apoptosis of keratinocytes: evidence for partial involvement of tumor necrosis factor-alpha in the formation in the formation of sunburn cells. J Invest Dermatol 1995; 104: 922–927.
Aragane Y, Kulms D, Metze D, Wilkes G, Pöppelmann B, Luger TA et al. Ultraviolet light induces apoptosis via direct activation of CD95 (Fas/APO-1) independently of its ligand CD95L. J Cell Biol 1998; 140: 171–182.
Masaki H, Atsumi T, Sakurai H . Detection of hydrogen peroxide and hydroxyl radicals in murine skin fibroblasts under UVB irradiation. Biochem Biophys Res Commun 1995; 17: 474–479.
Peus D, Vasa RA, Beyerle A, Meves A, Krautmacher C, Pittelkow MR . H2O2 is an important mediator of UVB-induced EGF-receptor phosphorylation in cultured keratinocytes. J Invest Dermatol 1999; 112: 751–756.
Murphy G, Young AR, Wulf HC, Kulms D, Schwarz T . The molecular determinations of sunburn cell formation. Exp Dermatol 2001; 10: 155–160.
Matés JM . Effects of antioxidant enzymes in the molecular control of reactive oxygen species toxicology. Toxicol 2000; 153: 83–104.
Vile GF, Tyrrell RM . Oxidative stress resulting from ultraviolet A irradiation of human skin fibroblasts leads to a heme oxygenase-dependent increase in ferritin. J Biol Chem 1993; 268: 14678–14681.
Punnonen K, Puntala A, Ahtupa M . Effects of ultaviolet A and B irradiation on lipid peroxidation and activity of the antioxidant enzymes in keratinocytes in culture. Photodermatol Photoimmunol Photomed 1991; 8: 3–6.
Shindo Y, Witt E, Han D, Tzeng B, Aziz T, Nguyen L et al. Recovery of antioxidants and reduction in lipid hydroperoxides in murine epidermis and dermis after acute ultraviolet radiation exposure. Photodermatol Photoimmunol Photomed 1994; 10: 183–191.
Zhang X, Rosenstein BS, Wang Y, Lebwohl M, Wei H . Identification of possible reactive oxygen species involved in ultraviolet radiation-induced oxidative DNA damage. Free Radic Biol Med 1997; 23: 980–985.
Pelle E, Hung X, Mammone T, Marenus K, Maes D, Frenkek K . Ultraviolet-B-induced oxidative DNA base damage in primary normal human epidermal keratinocytes and inhibition by a hydroxyl radical scavenger. J Invest Dermatol 2003; 121: 177–183.
Maalouf S, El-Sabban M, Drawiche N, Gali-Muhtasib H . Protective effect of vitamin E on ultraviolet B light-induced damage in keratinocytes. Mol Carcinogen 2000; 34: 121–130.
Morley N, Curnow A, Salter L, Campbell S, Gould D . N-acetyl-L-cysteine prevents DNA damage induced by UVA, UVB and visible radiation in human fibroblasts. J Photochem Photobiol B Biol 2003; 72: 55–60.
Cario-Andre M, Briganti S, Picardo M, Nikaido O, Gall Y, Ginestar J et al. Epidermal reconstructs: a new tool to study topical and systemic photoprotective molecules. J Photochem Photobiol B Biol 2002; 68: 79–87.
Rezvani HR, Mazurier F, Cario-André M, Pain C, Ged C, Taïeb A et al. Protective effects of catalase overexpression on UVB-induced apoptosis in normal human keratinocytes. J Biol Chem 281: 17999–18007.
Sander CS, Hamm F, Elsner P, Thiele JJ . Oxidative stress in malignant melanoma and non-melanoma skin cancer. Br J Dermatol 2003; 148: 913–922.
Prunieras M, Régnier M, Schlotterer M . Nouveau procédé de culture des cellules épidermiques humaines sur derme homologue ou hétérologue: préparation de greffons recombines. Ann Chir Plast 1979; 24: 357–362.
Cudre-Mauroux C, Occhiodoro T, Konig S, Salmon P, Bernheim L, Trono D . Lentivector-mediated transfer of Bmi-1 and telomerase in muscle satellite cells yields a Duchenne myoblast cell line with long-term genotypic and phenotypic stability. Hum Gene Ther 2003; 14: 1525–1533.
Géronimi F, Richard E, Redonnet-Vernhet I, Lamrissi-Garcia I, Lalanne M, Ged C et al. Highly efficient lentiviral gene transfer in CD34+ and CD34+/CD38−/lin− cells from mobilized peripheral blood after cytokine prestimulation. Stem Cells 2003; 21: 472–480.
Chen M, Li W, Fan J, Kasahara N, Woodley D . An efficient gene transduction system for studying gene function in primary human dermal fibroblasts and epidermal keratinocytes. Clin Exp Dermatol 2003; 28: 193–199.
Serrano F, Del Rio M, Larcher F, Garcia M, Munoz E, Escamez MJ et al. A comparison of targeting performance of oncoretroviral versus lentiviral vectors on human keratinocytes. Hum Gene Ther 2003; 14: 1579–1585.
Ghazizadeh S, Katz AB, Harrington R, Taichman LB . Lentivirus-mediated gene transfer to human epidermis. J Investig Dermatol Symp Proc 2004; 9: 269–275.
Amstad P, Peskin A, Shah G, Mirault ME, Moret R, Zbinden I et al. The balance between Cu,Zn-superoxide dismutase and catalase affects the sensitivity of mouse epidermal cells to oxidative stress. Biochemistry 1991; 30: 9305–9313.
Takahashi H, Hashimoto Y, Aoki N, Kinouchi M, Ishida-Yamamoto A, Iizuka H . Copper, zinc-superoxide dismutase protects from ultraviolet B-induced apoptosis of SV40-transformed human keratinocytes: the protection is associated with the increased levels of antioxidant enzymes. J Dermatol Sci 2000; 23: 12–21.
Sasaki H, Akamatsu H, Horio T . Effects of a single exposure to UVB radiation on the activities and protein levels of copper–zinc and manganese superoxide dismutase in cultured human keratinocytes. Photochem Photobiol 1997; 65: 707–713.
Hurwitz SA, Spadau DF . Quantitative analysis of UVB-induced apoptosis in human epidermis. Exp Dermatol 2000; 9: 185–191.
Bedner E, Smolewski P, Amstad P, Darzynkiewicz Z . Activation of caspases measured in situ by binding of fluorochrome-labeled inhibitors of caspases (FLICA): correlation with DNA fragmentation. Exp Cell Res 2000; 259: 308–313.
Zuliani T, Denis V, Noblesse E, Schnebert S, André P, Dumas M et al. Hydrogen peroxide-induced cell death in normal human keratinocytes is differentiation dependent. Free Radic Biol Med 2005; 38: 307–316.
Harris CC . p53: at the crossroads of molecular carcinogenesis and risk assessment. Science 1993; 262: 1980–1981.
Shieh SY, Ikeda M, Taya Y, Prives C . DNA damage-induced phosphorylation of p53 alleviates inhibition by MDM2. Cell 1997; 91: 325–334.
Achanta G, Huang P . Role of p53 in sensing oxidative DNA damage in response to reactive oxygen species-generating agents. Cancer Res 2004; 64: 6233–6239.
Giaccia AJ, Kastan MB . The complexity of p53 modulation: emerging patterns from divergent signals. Genes Dev 1998; 12: 2973–2983.
Ko LJ, Prives C . p53: puzzle and paradigm. Genes Dev 1996; 10: 1054–1072.
Levine AJ . p53, the cellular gatekeeper for growth and division. Cell 1997; 88: 323–331.
Steegenga WT, vandereb AJ, Jochemsen AG . How phosphorylation regulates the activity of p53. J Mol Biol 1996; 263: 103–113.
Mitchell DL, Jen J, Cleaver JE . Relative induction of cyclobutane dimers and cytosine photohydrates in DNA irradiated in vitro and in vivo with ultraviolet-C and ultraviolet-B light. Photochem Photobiol 1991; 54: 741.
Doetsch PW, Zasatawny TH, Martin AM, Dizdaroglu M . Monomeric base damage products from adenine, guanine, and thymine induced by exposure of DNA to ultraviolet radiation. Biochemistry 1995; 34: 737.
Cadet J, Anselmino C, Douki T, Voituriez L . Photochemistry of nucleic acids in cells. J Photochem Photobiol B Biol 1992; 15: 277.
Ravanat JL, Douki T, Cadet J . Direct and indirect effects of UV radiation on DNA and its components. J Photochem Photobiol B Biol 2001; 63: 88–102 (Review).
Kulms D, Zeise E, Poppelmann B, Schwarz T . DNA damage, death receptor activation and reactive oxygen species contribute to ultraviolet radiation-induced apoptosis in an essential and independent way. Oncogene 2002; 21: 5844–5851.
Xanthoudakis S, Curran T . Identification and characterization of Ref-1, a nuclear protein that facilitates AP-1 DNA-binding activity. EMBO J 1992; 11: 653–665.
Jayaraman L, Murthy KG, Zhu C, Curran T, Xanthoudakis S, Prives C . Identification of redox/repair protein Ref-1 as a potent activator of p53. Genes Dev 1997; 11: 558–570.
Garmyn M, Degreef H . Suppression of UVB-induced c-fos and c-jun expression in human keratinocytes by N-acetylcysteine. J Photochem Photobiol B Biol 1997; 37: 125–130.
Brenneisen P, Wenk J, Klotz LO, Wlaschek M, Briviba K, Krieg T et al. Central role of ferrous/ferric iron in the ultraviolet B irradiation-mediated signaling pathway leading to increased interstitial collagenase (matrix-degrading metalloprotease (MMP)-1) and stromelysin-1 (MMP-3) mRNA levels in cultured human dermal fibroblasts. J Biol Chem 1998; 273: 5279–5287.
Wang H, Kochevar IE . Involvement of UVB-induced reactive oxygen species in TGF-beta biosynthesis and activation in keratinocytes. Free Radic Biol Med 2005; 38: 890–897.
Lowe N, Koo L, Breeding J . Ultraviolet light induces human epidermal ornithine decarboxylase activity. Br J Dermatol 1980; 103: 18.
Berton T, Mitchell DL, Fischer SM, Locniskar MF . Epidermal proliferation but not the quantity of DNA photodamage is correlated with UV-induced mouse skin carcinogenesis. J Invest Dermatol 1997; 109: 340–347.
Nakamura Y, Murakami A, Ohto Y, Torikai K, Tanaka T, Ohigashi H . Suppression of tumor promoter-induced oxidative stress and inflammatory responses in mouse skin by a superoxide generation inhibitor l′-acetoxychavicol acetate. Cancer Res 1998; 58: 4832–4839.
Ishizaki C, Oguro T, Yoshida T, Wen CQ, Sueki H, Iijima M . Enhancing effect of ultraviolet A on ornithine decarboxylase induction and dermatitis evoked by 12-o-tetradecanoylphorbol-13-acetate and its inhibition by curumin in mouse skin. Dermatology 1996; 193: 311–317.
Schieke SM, Ruwiedel K, Gers-Barlag H, Grether-Beck S, Krutmann J . Molecular crosstalk of the ultraviolet a and ultraviolet B signaling responses at the level of mitogen-activated protein kinases. J Invest Dermatol 2005; 124: 857–859.
Adachi M, Gazel A, Pintucci G, Shuck A, Shifteh S, Ginsburg D et al. Specificity in stress response: epidermal keratinocytes exhibit specialized UV-responsive signal transduction pathway. DNA Cell Biol 2003; 22: 665–677.
Bode AM, Dong Z . Mitogen-activated protein kinase activation in UV-induced signal transduction. Sci STKE 2003; 2003: RE2 (Review).
Casoli V, Cario-Andre M, Costet P, Pain C, Taieb A . Comparison of long-term survival of pigmented epidermal reconstructs cultured in vitro vs xenografted on nude mice. Pigment Cell Res 2004; 17: 87–92.
Sander CS, Chang H, Hamm F, Elsner P, Thiele JJ . Role of oxidative stress and the antioxidant network in cutaneous carcinogenesis. Int J Dermatol 2004; 43: 326–335 (Review).
Kobayashi T, Matsumoto M, Iizuka H, Suzuki K, Taniguchi N . Superoxide dismutase in psoriasis, squamous cell carcinoma and basal cell epithelioma: an immunohistochemical study. Br J Dermatol 1991; 124: 555–559.
Rabilloud T, Asselineau D, Miquel C, Calvayrac R, Darmon M, Vuillaume M . Deficiency in catalase activity correlates with the appearance of tumor phenotype in human keratinocytes. Int J Cancer 1990; 45: 952–956.
F S, Afaq F, Mukhtar H . Photochemoprevention of skin cancer by botanical agents. Photodermatol Photoimmunol Photomed 2003; 19: 56–72 (Review).
Vuillaume M, Calvayrac R, Best-Belpomme M, Tarroux P, Hubert M, Decroix Y et al. Deficiency in the catalase activity of xeroderma pigmentosum cell and simian virus 4-transformed human cell extracts. Cancer Res 1986; 46: 538–544.
Acknowledgements
We thank M Longy (Institut Bergonié, France) for providing anti-p53 antibody.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rezvani, H., Cario-André, M., Pain, C. et al. Protection of normal human reconstructed epidermis from UV by catalase overexpression. Cancer Gene Ther 14, 174–186 (2007). https://doi.org/10.1038/sj.cgt.7701000
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.cgt.7701000
Keywords
This article is cited by
-
Rosiglitazone enhances radiosensitivity by inhibiting repair of DNA damage in cervical cancer cells
Radiation and Environmental Biophysics (2017)
-
Oxidative and Energy Metabolism as Potential Clues for Clinical Heterogeneity in Nucleotide Excision Repair Disorders
Journal of Investigative Dermatology (2015)
-
Antioxidant proteins and reactive oxygen species are decreased in a murine epidermal side population with stem cell-like characteristics
Histochemistry and Cell Biology (2011)
-
Catalase overexpression reduces UVB-induced apoptosis in a human xeroderma pigmentosum reconstructed epidermis
Cancer Gene Therapy (2008)