Abstract
Dynamic regulation of the intrinsic apoptosis pathway controls central and peripheral lymphocyte deletion, and may interfere with the pro-apoptotic potency of B-cell lymphoma 2 inhibitors such as ABT-737. By following a T-cell receptor (TCR) transgenic population of alloantigen-specific T cells, we found that sensitivity to ABT-737 radically changed during the course of allo-specific immune responses. Particularly, activated T cells were fully resistant to ABT-737 during the first days after antigen recognition. This phenomenon was caused by a TCR–calcineurin–nuclear factor of activated T cells-dependent upregulation of A1, and was therefore prevented by cyclosporine A (CsA). As a result, exposure to ABT-737 after alloantigen recognition induced selection of alloreactive T cells in vivo, whereas in combination with low-dose CsA, ABT-737 efficiently depleted alloreactive T cells in murine host-versus-graft and graft-versus-host models. Thus, ABT-737 resistance is not a prerogative of neoplastic cells, but it physiologically occurs in T cells after antigen recognition. Reversibility of this process by calcineurin inhibitors opens new pharmacological opportunities to modulate this process in the context of cancer, autoimmunity and transplantation.
Similar content being viewed by others
Main
B-cell lymphoma 2 (Bcl-2) inhibition represents a novel pharmacological principle to control lymphoid malignancies and detrimental immune responses.1, 2 Of particular scientific and potential clinical interest are the small molecule Bcl-2 inhibitors ABT-737 and its bioavailable counterpart Navitoclax (ABT-263).3, 4 ABT-737 binds with high affinity to the anti-apoptotic Bcl-2 family members Bcl-2, Bcl-XL and Bcl-w. By this mechanism, it prevents them from sequestering pro-apoptotic BH3 proteins, and therefore, indirectly initiates the apoptotic cascade. In contrast, ABT-737 has a low affinity to Bcl-2-A1 (A1, Bfl-1), Mcl-1 and Bcl-B. These particular molecular binding characteristics may be important for tissue selectivity and – as a consequence – for the favorable toxicity profile of ABT-737 and ABT-263,5 but limits their therapeutic potency on lymphoma cells expressing A1 and Mcl-1.6, 7
The physiological regulation of apoptosis in lymphocytes has been extensively investigated,8 and may assume a new relevance in the context of therapeutic approaches selectively targeting Bcl-2 proteins. Focusing on the T-cell compartment, it has been shown that the fate of a T cell is linked to the expression of a functional T-cell receptor (TCR)9 and its interaction with antigen-presenting cells (APCs). The combination of signals through the TCR, co-stimulatory molecules (such as CD28 and 4-1BB) and cytokines (such as interleukin (IL)-2 and IL-15) dynamically modulates the intrinsic and the extrinsic apoptosis pathway in T lymphocytes, and eventually controls central and peripheral T-cell selection.10, 11, 12, 13, 14, 15 Of particular interest are previous reports on a TCR-dependent upregulation of A1 in the early phase after antigen recognition, which protects activated thymocytes and splenocytes from apoptosis without interfering with cell proliferation.16, 17 These mechanisms are crucial for the development and maintenance of a functional immune system10 and might be influenced by drugs targeting the apoptosis pathway.
This hypothesis is supported by previous reports about the immuno-modulatory properties of ABT-737 in several experimental models: ABT-737 had a beneficial effect on autoimmunity18 and significantly inhibited solid allograft rejection in mice.2, 19 However, immunosuppression by ABT-737 in a collagen-induced arthritis model was only effective in a preventive setting, but not in mice with established disease.20 Furthermore, the immunosuppressive effect of ABT-737 in a murine skin graft model was rather limited as a single agent, but markedly increased in combination with cyclosporine A (CsA).19 These data suggest that the pro-apoptotic potency of ABT-737 on lymphoid cells is altered in the context of inflammation and T-cell activation.
In this study, we examined the effect of ABT-737 on alloreactive T cells in the setting of host-versus-graft (HvG) and graft-versus-host (GvH) immune reactions. We found a unique selectivity profile of ABT-737 on T lymphocytes over the course of the immune response as a result of a transient, calcineurin-, nuclear factor of activated T cells (NFAT)- and A1-dependent resistance to ABT-737 after antigen recognition. The calcineurin inhibitor CsA blocked A1 upregulation and prevented resistance to ABT-737 in activated T cells, thereby offering new options for effective combination therapies.
Results
Activated T cells are resistant to ABT-737
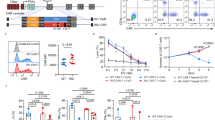
To investigate the impact of allogeneic T-cell activation on the sensitivity to the Bcl-2 inhibitor ABT-737, we used the transgenic mouse strain BM3.3, which expresses on all CD8 T cells a transgenic TCR specific for the major histocompatibility complex (MHC) class I molecule H-2Kb and can be detected by the clonotypic antibody Ti98. In the first experiment, we transplanted BM3.3 bone marrow into non-lethally irradiated CBA mice to create synchimeric mice that express the BM3.3 TCR only on a fraction of the CD8 T-cell pool. This well-defined homogeneous population of alloreactive CD8 T cells could then be followed during the course of an HvG response in the context of an otherwise physiological immune system (Figures 1a and b). Synchimeras received a donor-specific transfusion (DST) (20 × 106 B6 splenocytes, i.v., DST) and treatment with either ABT-737 (50 mg/kg per day) or vehicle, starting at day 2 after DST. At day 5 after priming, mice treated with ABT-737 presented a 75% reduction of T cells in peripheral blood (Figure 1c); CD4 and CD8 T cells were similarly affected by the treatment (Figure 1d). After antigen recognition, the percentage of Ti98+ cells among CD8 T cells increased in both groups, but this effect was markedly enhanced in the ABT-737 group compared with control (Figure 1e). This observation is explained by a selection of activated Ti98+ cells among CD8 T cells under the effect of ABT-737, which may be further enhanced by homeostatic proliferation in a lymphopenic environment.
Alloreactive CD8 T cells are resistant to ABT-737 after DST. (a) Characterization of the BM3.3 model; after 48 h of MLR with BM3.3 responders and CD8-depleted B6 stimulators, all responder (Ti98+) CD8 T cells were activated, as measured by CD25 expression in FACS. (b) Experimental setup: synchimeric mice were generated by bone marrow (BM) transplantation from BM3.3 mice into CBA recipients after non-lethal total body irradiation (3 Gy). After 6 weeks, synchimeras expressed the BM3.3 TCR (Ti98+) on about 6% of the whole CD8-positive population. The mice were primed by i.v. injection of B6 splenocytes, and 2 days later, treatment with ABT-737 was started. (c and d) Exposure to ABT-737 induced a relative and absolute reduction of CD3+ cells in peripheral blood, and similarly affected CD4+ and CD8+ T cells. (e) The increase of the percentage of Ti98+ cells among CD8 T cells was significantly higher in mice exposed to ABT-737 (P<0.01). Statistical comparison of data registered at baseline and at day 5 by paired t-test; *P<0.05, **P<0.01; n=5. Representative results of one of two independent experiments are shown
To limit the confounding effect of homeostatic proliferation, we performed an analogous experiment in a GvH model. The combination of a parent to F1 model with the BM3.3 transgenic system allowed us to specifically analyze a homogeneous population of host-reactive CD8 T cells in the absence of rejection by the recipient and without the effect of any conditioning regime (such as total body irradiation) that may alter the immune response and apoptosis regulation. We minimized the effect of T-cell proliferation by choosing a short protocol: BM3.3 splenocytes were adoptively transferred into (CBAxB6)F1 (allogeneic stimulation) or CBA recipients (syngeneic control). On day 1 after transfer treatment with ABT-737 (50 mg/kg per day) or vehicle was started, and 2 days later, recipient mice were killed for fluorescence-activated cell sorting (FACS) analysis. ABT-737 minimally influenced the activation of Ti98+ cells (Figures 2c and d) and similarly reduced the number of total splenocytes in F1 and CBA recipients by about 30% reduction (Figures 2e and f). However, in the syngeneic combination, Ti98+ cells were equally reduced as total T cells and total splenocytes (Figures 2e and g), whereas in F1 recipients, alloactivated donor-reactive CD8+Ti98+ cells were resistant to ABT-737 (Figure 2h). As a result, the total number of Ti98+ cells in CBA recipients was markedly reduced after ABT-737 treatment, but no difference in the total number of Ti98+ cells between the two groups was registered after allogeneic stimulation (Figures 2g and h). These data strongly suggest that resistance of activated Ti98+ T cells to ABT-737 had developed in both HvG and GvH experiments.
Activated CD8 T cells are resistant to ABT-737 in a GvH model. BM3.3 splenocytes were adoptively transferred to CBA (syngeneic) or (CBA × B6)F1 (allogeneic) recipients to assess the effect of ABT-737 on GvH-reactive cells. After 3 daily injections of ABT-737 (filled circles) or vehicle (open circles), splenocytes were analyzed by FACS. (a and b) Allogeneic CD8 T-cell activation was confirmed by CD25 expression and (c and d) a selective analysis of Ti98+ cells indicated that ABT-737 did not substantially influence this process. (e and f) A similar reduction in the number of total splenocytes was registered in both groups (about 30% reduction, please note the different total number of splenocytes in different recipient strains). (g and h) The total number of Ti98+ cells decreased in the syngeneic, but not in the allogeneic combination, indicating resistance to ABT-737 in allo-activated CD8 T cells. Statistical comparison ABT-737 versus vehicle; *P<0.05, **P<0.01, ***P<0.001; n=5. Representative results of one of two independent experiments are shown
This hypothesis was further tested in vitro in a mixed lymphocyte reaction (MLR) model. BM3.3 splenocytes were cultured with CD8-depleted allogeneic B6 or syngeneic CBA splenocytes during 48 h and then treated with ABT-737 for an additional 12 h. Cell viability analysis by propidium iodide (PI) exclusion in FACS revealed that a 1000- to 10 000-fold higher concentration of ABT-737 was required to induce apoptosis in CD8 T cells after allogeneic stimulation (Figure 3a). To exclude a transgenic artifact, the same experiment was repeated with B6 responders and T-cell-depleted CBA stimulators. Activated (CD25+) CD8 T cells were much more resistant to ABT-737 compared with non-activated (CD25−) cells in the same culture and to syngeneically stimulated (non-activated) T cells. The same phenomenon was observed for CD4 T cells (Figure 3b). Thus, T-cell activation induces resistance to ABT-737 in vitro and in vivo.
Allo-activated T cells are resistant to ABT-737 in vitro. (a) BM3.3 splenocytes were stimulated with CD8-depleted B6 (allogeneic, filled circles) or CBA (syngeneic, open circles) splenocytes during 48 h MLR, and then treated with ABT-737 during additional 12 h of MLR. Much higher concentrations of ABT-737 (1000- to 10 000-fold) were required to induce apoptotis in allo-activated CD8 T cells compared with non-activated T cells. (b) Similarly, B6 splenocytes were stimulated with T-cell-depleted CBA splenocytes during 48 h MLR, and then treated with ABT-737 during additional 12 h of MLR. FACS analysis of responder CD4 and CD8 T cells revealed that activated (CD25+, filled circles) cells were resistant to ABT-737 compared with non-activated (CD25−, filled squares) cells in the same culture and also with syngeneically stimulated cells (open circles). Cell viability was assessed by PI exclusion in FACS in at least three independent experiments. Percentage of cells treated with vehicle is given. Statistical comparison of allo versus syn: *P<0.05, **P<0.01, ***P<0.001
Molecular mechanism of resistance to ABT-737 in activated T cells
The regulation of apoptosis is complex, and several mechanisms may be involved in resistance to ABT-737 after T-cell activation. In MLR experiments using the BM3.3 system, we found that exposure to the ribosome blocker cycloheximide during the stimulation phase prevented the establishment of ABT-737 resistance, indicating that protein synthesis was required to induce this anti-apoptotic state (Figure 4a). Previous studies in tumor models revealed that the expression of anti-apoptotic Bcl-2 proteins with a low binding affinity to ABT-737, such as A1 and Mcl-1, resulted in resistance to this compound.21 Therefore, we first assessed the impact of T-cell activation on the expression of various anti-apoptotic Bcl-2 factors in our system. Analyses by quantitative RT-PCR (qRT-PCR) revealed that T-cell activation rapidly influenced the gene expression pattern of Bcl-2 family members (Figure 4b); among the Bcl-2 members that are not inhibited by ABT-737, expression of A1 was nine-fold higher in alloantigen-stimulated than in non-activated cells. In contrast, expression of Mcl-1 did not change. When looking at time kinetics, we found that resistance to ABT-737 is a transient phenomenon; it rapidly develops after T-cell stimulation, but progressively vanished after 4–5 days of culture (Figure 4c). This evolution strongly correlated with expression of A1 protein over time (Figure 4d), supporting the hypothesis of a crucial role of this particular factor. A selective inhibition of A1 in murine cells is complicated because of the presence of four homologous genes for A1 in the mouse genome. Just one of them – A1-a – was successfully targeted in a knock-out mouse,22 and selective pharmacological A1 inhibitors are currently not available.23 Therefore, we applied a reversed approach using different Bcl-2 inhibitors with a defined binding profile. We found that T-cell activation induced resistance to Bcl-2 inhibition by ABT-737 (no binding of A1 and Mcl-1) and by Antimycin A (no binding of A1 only), but had no impact on the pro-apoptotic potency of the pan-Bcl-2 inhibitor obatoclax (Figure 4e). Thus, A1 upregulation is the crucial factor determining resistance to ABT-737 in activated T cells.
Upregulation of A1 is crucial for resistance to ABT-737. (a) BM3.3 splenocytes were stimulated with CD8 T-cell-depleted splenocytes from B6 (allo) or CBA (syn) donors during 24 h of MLR under different concentration of cycloheximide, and then treated with ABT-737 (1 μM) or vehicle. Alloantigen-stimulated cells without cycloheximide were resistant to ABT-737, but cycloheximide prevented this process in a concentration-dependent manner. (b) After 6 h of MLR, the expression of Bcl-2 family genes was analyzed in BM3.3 CD8 cells by qRT-PCR; we registered a nine-fold upregulation in the expression of A1, and a small increase of Bcl-2 and Bcl-XL in comparison with syngeneically stimulated cells. (c) For a time course analysis, BM3.3 splenocytes were stimulated for up to 6 days with B6 or CBA splenocytes, and then treated with ABT-737 for additional 12 h. Resistance to ABT-737 dynamically changed over time after allogeneic stimulation, reaching a maximum 2–3 days after stimulation and rapidly declining thereafter, whereas syngeneically stimulated cells remained sensitive to ABT-737 over time (one representative time point is shown). Cell viability of BM3.3 CD8 T cell was assessed by PI exclusion in FACS in at least three independent experiments. Percentage of cells treated with DMSO-containing vehicle is given. Statistical comparison of allo versus syn: **P<0.01, ***P<0.001. (d) A parallel analysis of A1 by western blot revealed that ABT-737 resistance and A1 expression strongly correlated over time. (e) The role of different anti-apoptotic Bcl-2 factors in determining resistance to ABT-737 was investigated, comparing the pro-apoptotic effect of ABT-737, antimycin A and obatoclax on activated (filled circles) and non-activated (open circles) BM3.3 CD8 T cells. T-cell activation induced resistance to ABT-737 and to antimycin A, but not to obatoclax, demonstrating a crucial role of A1 in this phenomenon. Percentage of cells treated with DMSO-containing vehicle is given. Half maximal inhibitory concentration (IC50) and 95% confidence interval (95% CI) are reported in the table
T-cell activation and resistance to ABT-737
According to the three-signal concept, physiological T-cell activation is determined by the concurrent stimulation of the TCR (signal 1), together with a costimulatory signal through CD28 and CD154 (signal 2), and by the effect of cytokines such as IL-2 and IL-15 (signal 3).24 The link between resistance to ABT-737 and the different pathways involved in T-cell activation was investigated dissecting the T-cell activation process by blockade of different pathways during the stimulation phase (24 h). We found that resistance to ABT-737 was prevented by blocking signal 1 with the calcineurin inhibitor CsA. In contrast, blocking of CD28 signaling by CTLA4Ig or of CD40 signaling by MR1 or using CD40 knock-out stimulators (data not shown), and blocking of mTOR signaling by rapamycin at a concentration that efficiently inhibited MLR in the same combination, did not influence resistance to ABT-737 (Figure 5a). An important role of the TCR–calcineurin–NFAT (signal 1) cascade was further confirmed by using the alternative calcineurin inhibitor tacrolimus and the cell permeable NFAT-inhibitor VIVIT-R.25 The blockade of this pathway at any level increased the percentage of apoptotic cells in allogeneic, but not in syngeneic cultures (data not shown), and it prevented resistance to ABT-737 (Figure 5b), thereby excluding an off-target effect of CsA and indicating a crucial role for NFAT in preventing T-cell apoptosis in the early phase after antigen recognition. The correlation of these findings with the inhibition of upregulation of A1 by CsA was confirmed at the mRNA and protein level (Figures 5c and d). Thus, antigen recognition induced an NFAT-dependent upregulation of A1 that determined resistance to ABT-737 in alloantigen-activated CD8 T cells, and CsA completely prevented this resistance to ABT-737 in activated cells in vitro.
Signal 1 determines resistance to ABT-737 in activated T cells. (a) Selective inhibitors of signal 1, 2 and 3 were added to an MLR during the stimulation phase with BM3.3 splenocytes reacting against CD8 T-cell-depleted B6 splenocytes to investigate the role of the different T-cell activation pathways for resistance to ABT-737(1 μM, during additional 12 h of culture). The calcineurin inhibitor and signal 1 blocker CsA prevented resistance to ABT-737 in a concentration-dependent manner, whereas inhibition of signal 2 by CTLA4Ig and signal 3 by rapamycin did not influence this process. (b) The results obtained with CsA were confirmed by other inhibitors of this same pathway, namely the alternative calcineurin inhibitor tacrolimus and the NFAT inhibitor VIVIT-R. Cell viability of BM3.3 CD8 T cells was assessed by PI exclusion in FACS in at least three independent experiments. Percentage of cells treated with vehicle is given. Statistical comparison of ABT-737 versus vehicle: *P<0.05, **P<0.01. (c and d) Exposure to CsA during the stimulation phase inhibited the upregulation of A1 as assessed by qRT-PCR (c) and western blot (d)
Reversibility of ABT-737 resistance by CsA in vivo
The critical role of signal 1 for ABT-737 resistance offers the opportunity to prevent resistance to ABT-737, using clinically well-established calcineurin inhibitors. We tested this option using a combination of CsA and ABT-737 in the GvH and HvG models introduced before. For the GvH experiments, BM3.3 splenocytes were transferred to F1 mice under treatment with low doses of CsA (10 mg/kg per day). Similar to the previous experiment (Figure 2), ABT-737 (50 mg/kg per day) was administered at day 1 and 2 after cell transfer, and on day 3, mice were killed for spleen FACS analysis. Concurrent with the in vitro results, the selection of activated donor-reactive CD8+Ti98+ cells observed in mice treated with ABT-737 alone was completely prevented by the addition of CsA (Figure 6a). Because of the general lymphopenia induced by ABT-737, this resulted in a much more pronounced depletion of alloreactive T cells in the combination group (Figure 6b). This effect was even more pronounced in the HvG model, where treatment was continued for 5 days after DST, and the immunosuppressive effect of CsA simultaneously contributed to the inhibition of the allogeneic immune response (Figure 6c). Thus, resistance to ABT-737 after antigen recognition was successfully overcome by combination with low doses of CsA.
CsA prevents resistance to ABT-737 in vivo. (CBA × B6)F1 mice were injected with BM3.3 splenocytes to induce a GvH reaction, and treated with ABT-737 (50 mg/kg per day, i.p.) with or without CsA (10 mg/kg per day, s.c.). After 3 days, the mice were killed and the spleens were analyzed by FACS. (a) Treatment with ABT-737 alone resulted in the selection of alloreactive Ti98+ cells. However, this phenomenon was completely prevented in combination with CsA (statistical comparison with vehicle or CsA alone, respectively). (b) As a result, the total number of alloreactive CD8+Ti98+ cells per spleen was markedly reduced in the combination therapy group, compared with CsA or ABT-737 alone. (c) Similarly, in an HvG combination using synchimeric mice (Figure 1 for experimental setup), the combination of ABT-737 and CsA strongly reduced the number of donor-reactive Ti98+ cells as measured in spleens at day 5 after injection of B6 splenocytes. Statistical comparison to vehicle group: n.s. P>0.05, *P<0.05, **P<0.01, ***P<0.001
Discussion
The regulation of the intrinsic apoptosis pathway in lymphocytes assumes a novel relevance after the advent of small-molecule Bcl-2 inhibitors. Here we present evidence that sensitivity to ABT-737 in T lymphocytes dynamically changes during the course of an immune response; that is, activated T cells are transiently resistant to ABT-737 during the first days after antigen recognition, because of a signal-1-dependent, NFAT-dependent upregulation of A1. As a result, ABT-737 displayed a unique selectivity profile in its pro-apoptotic potency, depleting naive lymphocytes, but sparing T cells after antigen-specific activation.
A1 is upregulated in the first hours after T-cell activation and protects activated thymocytes and splenocytes from premature death.16, 17 A1-dependent resistance to ABT-737 has been previously reported in lymphoma cells.6, 7, 26 Here we show for the first time that physiological upregulation of A1 after antigen recognition and ABT-737 resistance in normal (non-neoplastic) T cells are linked. More precisely, T-cell activation resulted in a 1000- to 10 000-fold resistance to ABT-737, a finding reminiscent of the results obtained by Vogler et al.26 in B-cell lymphoma cells cultured with CD154-expressing fibroblasts and IL-2. Mechanistic analyses revealed that the TCR–calcineurin pathway controlled the A1 upregulation, and that NFAT was the crucial transcription factor in this context,16 thereby mimicking the mechanism regulating A1 in mast cells after IgE receptor stimulation.27 Interestingly, fundamentally different pathways were involved in resistance to ABT-737 in CLL cells (which was signal 2- and 3-dependent and NFκB-mediated), suggesting a different regulation of A1 in B and T lymphocytes. In the complex system of apoptosis regulation, it is possible that additional factors may influence the sensitivity of T cells to Bcl-2 inhibition during the course of the immune response (e.g., Mcl-1 protein stabilization28). However, we demonstrated that blocking A1 was critical to prevent resistance to ABT-737, and inhibiting signal 1 of T-cell activation achieves this goal.
The synergistic effect of CsA and ABT-737 is partially paradoxical because of the anti-apoptotic properties of CsA, which have been previously related to a stabilization of the mitochondrial membrane.29, 30 This effect is presumably of limited relevance in combination with ABT-737. In contrast, the inhibition of the A1-dependent strong anti-apoptotic signal provided by the Ca2+-calcineurin–NFAT pathway was crucial to prevent resistance to ABT-737 in activated T cells, as shown by the similar effect obtained with tacrolimus and VIVIT-R. Interestingly, a dysregulation of the Ca2+–calcineurin–NFAT pathway has been described in several lymphatic and solid tumors, and may therefore influence the anti-neoplastic effect of Bcl-2 inhibitors.31 The most important consequence of the signal 1 dependency of ABT-737 resistance described here is the possibility to easily prevent it by well-established drugs such as calcineurin inhibitors. Characterization of ABT-737 resistance in physiological processes is relevant to find pharmacological strategies to potentiate the effect of Bcl-2 inhibitors, and although the use of immunosuppressive drugs to control cancer may be counterintuitive and potentially dangerous, we speculate that the combination of calcineurin inhibitors with ABT-737 might be beneficial in selected cases.
Because of the different molecular affinities of small-molecule Bcl-2 inhibitors to different members of the Bcl-2 family,1 their immuno-modulatory effect strictly depends on the expression of different members of the Bcl-2 family in distinct lymphocyte subpopulations and during the different phases of an immune response. Myeloid cells express high levels of Mcl-1 and are not affected by ABT-737; in contrast, ABT-737 efficiently induces apoptosis in naive lymphocytes.19, 20 Here we demonstrate that the physiological mechanisms that protect T cells from apoptosis in the initial phase after antigen recognition dramatically influence their sensitivity to ABT-737. Particularly, the low affinity of ABT-737 to A1 determines a selection of antigen-specific T cells in the first days after activation. This explains why ABT-737 as an immunosuppressive agent is not effective during the first days after transplantation and in the acute phase of an autoimmune disease,20 but may find a clinical application for induction therapy before solid organ or stem cell transplantation. In contrast, we speculate that an opposite result would be obtained with a selective A1 inhibitor, but none of the currently available Bcl-2 inhibitors selectively binds to A1. However, the fact that ABT-737, in combination with CsA, efficiently depleted activated T cells in a GvH and an HvG model explains the synergistic effect of ABT-737 and CsA, which we previously observed in a skin graft model,19 and is a reasonable option to potentiate the immunosuppressive effect of Bcl-2 inhibitors.
Finally, the unique selectivity profile of ABT-737 may find a useful application for cell-based immunotherapy. The experimental selection of antigen-specific cells after a short activation time is difficult to achieve and largely limited to the use of transgenic systems. ABT-737 allows selecting polyclonal antigen-specific cells after antigen recognition in vitro and in vivo, with a wide experimental application in the field of infection and cancer immunology, that is, for the generation of virus- or tumor antigen-specific T cells presented by the host MHC. As resistance to ABT-737 depends only on signal 1 activation, antigen-specific T cells can be further influenced by ILs to generate particular subsets of T cells in vitro, such as donor-reactive regulatory T cells or CMV-reactive cytotoxic T cells.32
Thus, in this study, we first described, characterized and found a way to overcome resistance to ABT-737 in activated T lymphocytes. Moreover, we propose a link between the well-established resistance to ABT-737 in tumor cells, and physiological lymphocyte activation after antigen recognition. These findings are relevant for a potential clinical application of Bcl-2 inhibitors as immuno-modulatory and anti-neoplastic agents.
Materials and Methods
Mice
C57BL/6 (B6, H-2b), CBA (H-2k), (CBAxB6)F1 (F1, H-2b/k) and BM3.3 (CBA background, H-2k) mice were housed in specific pathogen-free conditions at the University of Zürich. The BM3.3 mouse,33 which expresses on all CD8 T cells a transgenic TCR selective for a naturally processed octapeptide bound to the allogeneic MHC class I molecule H-2Kb, was kindly provided by A.-M. Schmitt-Verhulst.34 All animal experiments were performed according to protocols approved by the legal authority (Veterinary Office, Canton of Zürich, Switzerland).
Synchimeras and GvH reaction model
Synchimeric animals were generated as previously described.35, 36 Briefly, 5 × 106 bone marrow cells from BM3.3 mice were transplanted into naive CBA mice irradiated with 3 Gy on the same day. After 6–10 weeks, B6 splenocytes were injected, and treatment with ABT-737 (50 mg/kg per day, intraperitoneally (i.p.)) or vehicle was started according to the experimental protocol. Donor-reactive BM3.3 CD8 T cells were monitored in blood using the Ti98 antibody, which selectively binds to the BM3.3 TCR.37
GvH reactions were studied in a parent to F1 model. The F1 mice were generated by breeding CBA females and B6 males, and expressed H-2k and H-2b. After adoptive transfer of 20–25 × 106 BM3.3 splenocytes (H-2k background and therefore not rejected by the host, and selectively reacting against H-2Kb), the GvH-reactive cells were analyzed in the spleen using the Ti98 antibody.
Fluorescence-activated cell sorting
FACS analyses were performed with a BD-FACSCanto II (Becton Dickinson, Basel, Switzerland). Anti-mouse CD3-FITC, CD4-PE, CD8-APC and PI were purchased from eBioscience (Frankfurt, Germany), anti-mouse CD25-PE/Cy7 from Biolegend (Uithoorn, The Netherlands). The Ti98 antibody was kindly provided by A.-M. Schmitt-Verhulst.37 A secondary PE rat anti-mouse IgG was purchased from Becton Dickinson.
Mixed lymphocyte reaction
MLR were performed in 96-well plates with responder splenocytes stimulated by T-cell-depleted (or CD8 T-cell-depleted) splenocytes from allogeneic and syngeneic mice at a final concentration of 4 × 106 cells/ml in RPMI medium containing 10% fetal bovine serum, penicillin 100 U/ml, streptomycin 100 μg/ml, 2-mercaptoethanol 50 μM. Splenocytes were sorted by automatic magnetic cell separation using an autoMACS pro separator according to the protocols of MiltenyiBiotec (Bergisch Gladbach, Germany) to allow a selective analysis of responder CD4 and CD8 T cells in FACS.
Reagents and drugs
ABT-737 was provided by Abbott Bioresearch (Worcester, MA, USA); for in vitro experiments, ABT-737 was dissolved in dimethyl sulfoxide (DMSO) at a concentration of 5 mM, and then diluted in culture medium. For in vivo applications, ABT-737 was dissolved in polyethylene glycol, Tween 80, dextrose solution and DMSO, and injected i.p. at 50 mg/kg per day. Antimycin A, CsA, rapamycin and tacrolimus were purchased from EnzoBiochem (Farmingdale, NY, USA), cycloheximide from Sigma-Aldrich (Buchs, Switzerland), VIVIT-R from Calbiochem (Merck, Darmstadt, Germany), obatoclax (GX15-070) from Selleck (Houston, TX, USA). CTLA4Ig (abatacept) was provided by Bristol-Myers Squibb (Princeton, NJ, USA). The anti-CD154 (CD40L) antibody MR1 was purchased from BioXCell (West Lebanon, NH, USA). For in vivo application, CsA was dissolved in ethanol and cremaphor EL (Sigma-Aldrich), then diluted in PBS and injected subcutaneously (s.c.).
Quantitative RT-PCR
Reverse transcription and qPCR were performed as reported earlier.38 Pre-developed TaqMan reagents were used for mouse Bcl-2 (Mm00477631_m1), Bcl-XL (Mm00437783_m1), Mcl-1 (Mm01257352_g1) and for the housekeeper gene 18S rRNA (Applied Biosystems Europe, Rotkreuz, Switzerland). For mouse A1, the following oligonucleotide primers and probe were designed to simultaneously detect Bcl-2A1a, Bcl-2A1b and Bcl-2A1d: sense primer 5′-ATG GAG GTT GGG AAG ATG G-3′, anti-sense primer 5′-GAG CCA AGG TTC TCT CTG GTC-3′, fluorescence-labeled probe (FAM) 5′-GGC TGG CTG ACT TTT CTG CAG ATG A-3′. The expression of candidate genes in alloantigen-stimulated cells of culture was normalized by 18S rRNA and compared with syngeneically stimulated cells.
Western blot
For western blot allo- and syngeneically stimulated lymphocytes were harvested with Ripa buffer and complete protease inhibitor cocktail (Roche, Mannheim, Germany) at different time points after stimulation in vitro. Extracted proteins were boiled in loading buffer for 5 min, resolved by 15% SDS-PAGE, and transferred to an Immobilon-P membrane (Millipore, Eschborn, Germany). Membranes were blocked overnight with Tris-buffered saline (TBS)/5% fat-free skim milk, then incubated with a polyclonal anti-A1 antibody (Cell Signaling Technology, Danvers, MA, USA) diluted 1 : 1000 overnight at 4 °C and rinsed with TBS that contained 0.1% Tween 20. For detection, an HRP-linked goat anti-rabbit antibody (1 : 4000, 30 min at room temperature; Cell Signaling Technology) and enhanced chemiluminescence substrate (Proteinsimple, Santa Clara, CA, USA) were used. Membranes were also probed with anti-actin antibody (A2066, 1 : 1000, Sigma-Aldrich) as internal loading control.
Statistics
Student’s t-test, Mann–Whitney test and paired t-test were used to compare values between groups as appropriate. IC50-values were calculated using a log (inhibitor) versus response model. P<0.05 was considered significant. Graph Pad Prism Software Version 5.0 was used for calculations (GraphPad Software Inc., La Jolla, CA, USA).
Abbreviations
- APC:
-
antigen-presenting cell
- Bcl-2:
-
B-cell lymphoma 2
- CsA:
-
cyclosporine A
- DMSO:
-
dimethyl sulfoxide
- DST:
-
donor-specific transfusion
- FACS:
-
fluorescence-activated cell sorting
- GvH:
-
graft-versus-host
- HvG:
-
host-versus-graft
- IL:
-
interleukin
- MHC:
-
major histocompatibility complex
- MLR:
-
mixed lymphocyte reaction
- NFAT:
-
nuclear factor of activated T cells
- PI:
-
propidium iodide
- TCR:
-
T-cell receptor
References
Vogler M, Dinsdale D, Dyer MJ, Cohen GM . Bcl-2 inhibitors: small molecules with a big impact on cancer therapy. Cell Death Differ 2009; 16: 360–367.
Carrington EM, Vikstrom IB, Light A, Sutherland RB, Londrigan SL, Mason KD et al. BH3 mimetics antagonizing restricted prosurvival Bcl-2 proteins represent another class of selective immune modulatory drugs. Proc Natl Acad Sci USA. 2010; 107: 10967–10971.
Oltersdorf T, Elmore SW, Shoemaker AR, Armstrong RC, Augeri DJ, Belli BA et al. An inhibitor of Bcl-2 family proteins induces regression of solid tumours. Nature 2005; 435: 677–681.
Tse C, Shoemaker AR, Adickes J, Chen J, Jin S, Johnson EF et al. ABT-263: a potent and orally bioavailable Bcl-2 family inhibitor. Cancer Res 2008; 68: 3421–3428.
Wilson WH, O’Connor OA, Czuczman MS, LaCasce AS, Gerecitano JF, Leonard JP et al. Navitoclax, a targeted high-affinity inhibitor of BCL-2, in lymphoid malignancies: a phase 1 dose-escalation study of safety, pharmacokinetics, pharmacodynamics, and antitumour activity. Lancet Oncol 2010; 11: 1149–1159.
Del Gaizo Moore V, Schlis KD, Sallan SE, Armstrong SA, Letai A . BCL-2 dependence and ABT-737 sensitivity in acute lymphoblastic leukemia. Blood 2008; 111: 2300–2309.
Yecies D, Carlson NE, Deng J, Letai A . Acquired resistance to ABT-737 in lymphoma cells that up-regulate MCL-1 and BFL-1. Blood 115: 3304–3313.
Marsden VS, Strasser A . Control of apoptosis in the immune system: Bcl-2, BH3-only proteins and more. Annu Rev Immunol 2003; 21: 71–105.
Cho JH, Kim HO, Surh CD, Sprent J . T cell receptor-dependent regulation of lipid rafts controls naive CD8+ T cell homeostasis. Immunity 2010; 32: 214–226.
Hughes PD, Belz GT, Fortner KA, Budd RC, Strasser A, Bouillet P . Apoptosis regulators Fas and Bim cooperate in shutdown of chronic immune responses and prevention of autoimmunity. Immunity 2008; 28: 197–205.
Bouillet P, O'Reilly LA . CD95, BIM and T cell homeostasis. Nat Rev Immunol 2009; 9: 514–519.
Fischer SF, Belz GT, Strasser A . BH3-only protein Puma contributes to death of antigen-specific T cells during shutdown of an immune response to acute viral infection. Proc Natl Acad Sci USA. 2008; 105: 3035–3040.
Sabbagh L, Pulle G, Liu Y, Tsitsikov EN, Watts TH . ERK-dependent Bim modulation downstream of the 4-1BB-TRAF1 signaling axis is a critical mediator of CD8 T cell survival in vivo. J Immunol 2008; 180: 8093–8101.
Wojciechowski S, Tripathi P, Bourdeau T, Acero L, Grimes HL, Katz JD et al. Bim/Bcl-2 balance is critical for maintaining naive and memory T cell homeostasis. J Exp Med 2007; 204: 1665–1675.
Fehr T, Lucas CL, Kurtz J, Onoe T, Zhao G, Hogan T et al. A CD8 T cell-intrinsic role for the calcineurin-NFAT pathway for tolerance induction in vivo. Blood 2010; 115: 1280–1287.
Verschelde C, Walzer T, Galia P, Biemont MC, Quemeneur L, Revillard JP et al. A1/Bfl-1 expression is restricted to TCR engagement in T lymphocytes. Cell Death Differ 2003; 10: 1059–1067.
Gonzalez J, Orlofsky A, Prystowsky MB . A1 is a growth-permissive antiapoptotic factor mediating postactivation survival in T cells. Blood 2003; 101: 2679–2685.
Bardwell PD, Gu J, McCarthy D, Wallace C, Bryant S, Goess C et al. The Bcl-2 family antagonist ABT-737 significantly inhibits multiple animal models of autoimmunity. J Immunol 2009; 182: 7482–7489.
Cippa PE, Kraus AK, Edenhofer I, Segerer S, Chen J, Hausmann M et al. The BH3-mimetic ABT-737 inhibits allogeneic immune responses. Transpl Int 2011; 24: 722–732.
Lawlor KE, Smith SD, van Nieuwenhuijze A, Huang DC, Wicks IP . Evaluation of the Bcl-2 family antagonist ABT-737 in collagen-induced arthritis. J Leukoc Biol 2011; 90: 819–829.
Al-Harbi S, Hill BT, Mazumder S, Singh K, Devecchio J, Choudhary G et al. An antiapoptotic BCL-2 family expression index predicts the response of chronic lymphocytic leukemia to ABT-737. Blood 2011; 118: 3579–3590.
Hamasaki A, Sendo F, Nakayama K, Ishida N, Negishi I, Hatakeyama S . Accelerated neutrophil apoptosis in mice lacking A1-a, a subtype of the bcl-2-related A1 gene. J Exp Med 1998; 188: 1985–1992.
Cashman JR, MacDonald M, Ghirmai S, Okolowicz KJ, Sergienko E, Brown B et al. Inhibition of Bfl-1 with N-aryl maleimides. Bioorg Med Chem Lett 2010; 20: 6560–6564.
Matzinger P, Kamala T . Tissue-based class control: the other side of tolerance. Nat Rev Immunol 2011; 11: 221–230.
Aramburu J, Yaffe MB, Lopez-Rodriguez C, Cantley LC, Hogan PG, Rao A . Affinity-driven peptide selection of an NFAT inhibitor more selective than cyclosporin A. Science 1999; 285: 2129–2133.
Vogler M, Butterworth M, Majid A, Walewska RJ, Sun XM, Dyer MJ et al. Concurrent up-regulation of BCL-XL and BCL2A1 induces approximately 1000-fold resistance to ABT-737 in chronic lymphocytic leukemia. Blood 2009; 113: 4403–4413.
Ulleras E, Karlberg M, Moller Westerberg C, Alfredsson J, Gerondakis S, Strasser A et al. NFAT but not NF-kappaB is critical for transcriptional induction of the prosurvival gene A1 after IgE receptor activation in mast cells. Blood 2008; 111: 3081–3089.
Wensveen FM, van Gisbergen KP, Derks IA, Gerlach C, Schumacher TN, van Lier RA et al. Apoptosis threshold set by Noxa and Mcl-1 after T cell activation regulates competitive selection of high-affinity clones. Immunity 32: 754–765.
Hotchkiss RS, Strasser A, McDunn JE, Swanson PE . Cell death. N Engl J Med. 2009; 361: 1570–1583.
Zamzami N, Marchetti P, Castedo M, Hirsch T, Susin SA, Masse B et al. Inhibitors of permeability transition interfere with the disruption of the mitochondrial transmembrane potential during apoptosis. FEBS Lett 1996; 384: 53–57.
Muller MR, A. Rao . NFAT, immunity and cancer: a transcription factor comes of age. Nat Rev Immunol 2010; 10: 645–656.
Sagoo P, Ali N, Garg G, Nestle FO, Lechler RI, Lombardi G . Human regulatory T cells with alloantigen specificity are more potent inhibitors of alloimmune skin graft damage than polyclonal regulatory T cells. Sci Transl Med 2011; 3: 83ra42.
Auphan N, Curnow J, Guimezanes A, Langlet C, Malissen B, Mellor A et al. The degree of CD8 dependence of cytolytic T cell precursors is determined by the nature of the T cell receptor (TCR) and influences negative selection in TCR-transgenic mice. Eur J Immunol 1994; 24: 1572–1577.
Guimezanes A, Barrett-Wilt GA, Gulden-Thompson P, Shabanowitz J, Engelhard VH, Hunt DF et al. Identification of endogenous peptides recognized by in vivo or in vitro generated alloreactive cytotoxic T lymphocytes: distinct characteristics correlated with CD8 dependence. Eur J Immunol 2001; 31: 421–432.
Haspot F, Fehr T, Gibbons C, Zhao G, Hogan T, Honjo T et al. Peripheral deletional tolerance of alloreactive CD8 but not CD4 T cells is dependent on the PD-1/PD-L1 pathway. Blood 2008; 112: 2149–2155.
Fehr T, Takeuchi Y, Kurtz J, Wekerle T, Sykes M . Early regulation of CD8 T cell alloreactivity by CD4+CD25- T cells in recipients of anti-CD154 antibody and allogeneic BMT is followed by rapid peripheral deletion of donor-reactive CD8+ T cells, precluding a role for sustained regulation. Eur J Immunol 2005; 35: 2679–2690.
Buferne M, Luton F, Letourneur F, Hoeveler A, Couez D, Barad M et al. Role of CD3 delta in surface expression of the TCR/CD3 complex and in activation for killing analyzed with a CD3 delta-negative cytotoxic T lymphocyte variant. J Immunol 1992; 148: 657–664.
Lindenmeyer MT, Eichinger F, Sen K, Anders HJ, Edenhofer I, Mattinzoli D et al. Systematic analysis of a novel human renal glomerulus-enriched gene expression dataset. PLoS One 2010; 5: e11545.
Acknowledgements
We thank Anne-Marie Schmitt-Verhulst for providing the BM3.3 mouse and the Ti98 antibody. The project was supported by the Swiss National Science Foundation (Grant 323530-133893 to PEC, 310000-121979 to TF) and the Olga Mayenfisch Stiftung.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PDB is an employee of Abbott, which developed and provided ABT-737. All other authors declare no conflict of interest.
Additional information
Edited by P Salomoni
Rights and permissions
This work is licensed under the Creative Commons Attribution-NonCommercial-No Derivative Works 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-nd/3.0/
About this article
Cite this article
Cippà, P., Kraus, A., Lindenmeyer, M. et al. Resistance to ABT-737 in activated T lymphocytes: molecular mechanisms and reversibility by inhibition of the calcineurin–NFAT pathway. Cell Death Dis 3, e299 (2012). https://doi.org/10.1038/cddis.2012.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cddis.2012.38
Keywords
This article is cited by
-
Venetoclax imparts distinct cell death sensitivity and adaptivity patterns in T cells
Cell Death & Disease (2021)
-
The life and death of immune cell types: the role of BCL‐2 anti‐apoptotic molecules
Immunology & Cell Biology (2017)
-
Synergistic Bcl-2 inhibition by ABT-737 and cyclosporine A
Apoptosis (2013)








