Abstract
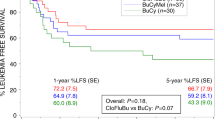
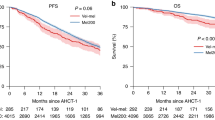
High-risk neuroblastoma is characterised by poor long-term survival, especially for very high-risk (VHR) patients (poor response of metastases after induction therapy). We report the results of an intensified high-dose chemotherapy (HDC) strategy to improve the prognosis of VHR patients. This strategy was based on tandem HDC with thiotepa and busulfan–melphalan (Bu–Mel) followed by autologous stem cell transplantation (ASCT). All data were prospectively recorded in the Gustave Roussy Paediatric ASCT database. From April 2004 to August 2011, 26 patients were eligible for tandem HDC. The median age at diagnosis was 4.4 years (1–15.9). All patients had metastatic disease. MYCN was amplified in 5/26 tumours. Despite the cumulative toxicity of alkylating agents, the toxicity of the intensified HDC strategy was manageable. Thiotepa-related toxicity was mainly digestive, whereas sinusoidal obstruction syndrome was the main toxicity observed after Bu–Mel. The 3-year event-free survival of this cohort was 37.3% (21.3–56.7). This strategy will be compared with combined 131I-mIBG/Bu–Mel in the upcoming SIOPEN VHR Neuroblastoma Protocol.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Matthay KK, Reynolds CP, Seeger RC, Shimada H, Adkins ES, Haas-Kogan D et al. Long-term results for children with high-risk neuroblastoma treated on a randomized trial of myeloablative therapy followed by 13-cis-retinoic acid: a children's oncology group study. J Clin Oncol 2009; 27: 1007–1013 Errata, J Clin Oncol 2014;32:1862–1863.
Cheung NV, Modak S, Ostrovnaya I, Roberts SS, Basu EM, Kramer K et al. When overall survival fails to confirm event-free survival, should the latter be used to set the standard of care? J Clin Oncol 2014; 32: 4173–4174.
Matthay K, London WB, Maris J, Adamson PC, Park JR . Reply to N.-K.V. Cheung et al. J Clin Oncol 2014; 32: 4174–4175.
Berthold F, Boos J, Burdach S, Erttmann R, Henze G, Hermann J et al. Myeloablative megatherapy with autologous stem-cell rescue versus oral maintenance chemotherapy as consolidation treatment in patients with high-risk neuroblastoma: a randomised controlled trial. Lancet Oncol 2005; 6: 649–658.
Pritchard J, Cotterill SJ, Germond SM, Imeson J, de Kraker J, Jones DR . High dose melphalan in the treatment of advanced neuroblastoma: results of a randomized trial (ENSG-1) by the European Neuroblastoma Study Group. Pediatr Blood Cancer 2005; 44: 348–357.
Hartmann O, Valteau-Couanet D, Vassal G, Lapierre V, Brugières L, Delgado R et al. Prognostic factors in metastatic neuroblastoma in patients over 1 year of age treated with high-dose chemotherapy and stem cell transplantation: a multivariate analysis in 218 patients treated in a single institution. Bone Marrow Transplant 1999; 23: 789–795.
Ladenstein R, Poetschger U, Luksch R, Brock P, Castel V, Yaniv I et al. Busulphan-melphalan as a myeloablative therapy (MAT) for high-risk neuroblastoma: results from the HR-NBL1/SIOPEN trial. J Clin Oncol 2011 29(Suppl abstr 2): 2.
Valteau-Couanet D, Le Deley MC, Bergeron C, Ducassou S, Michon J, Rubie H et al. Long-term results of the combination of the N7 induction chemotherapy and the busulfan-melphalan high-dose chemotherapy. Pediatr Blood Cancer 2014; 61: 977–981.
Ladenstein R, Philip T, Lasset C, Hartmann O, Garaventa A, Pinkerton R et al. Multivariate analysis of risk factors in stage 4 neuroblastoma patients over the age of one year treated with megatherapy and stem-cell transplantation: a report from the European Bone Marrow Transplantation Solid Tumor Registry. J Clin Oncol 1998; 16: 953–965.
Decarolis B, Schneider C, Hero B, Simon T, Volland R, Roels F et al. Iodine-123 metaiodobenzylguanidine scintigraphy scoring allows prediction of outcome in patients with stage 4 neuroblastoma: results of the Cologne interscore comparison study. J Clin Oncol 2013; 31: 944–951.
Bostrom B, Enockson K, Johnson A, Bruns A, Blazar B . Plasma pharmacokinetics of high-dose oral busulfan in children and adults undergoing bone marrow transplantation. Pediatr Transplant 2003; 7: 12–28.
Vassal G, Michel G, Espérou H, Gentet JC, Valteau-Couanet D, Doz F et al. Prospective validation of a novel IV busulfan fixed dosing for paediatric patients to improve therapeutic AUC targeting without drug monitoring. Cancer Chemother Pharmacol 2008; 61: 113–123.
Veal GJ, Nguyen L, Paci A, Riggi M, Amiel M, Valteau-Couanet D et al. Busulfan pharmacokinetics following intravenous and oral dosing regimens in children receiving high-dose myeloablative chemotherapy for high-risk neuroblastoma as part of the HR-NBL-1/SIOPEN trial. Eur J Cancer 2012; 48: 3063–3072.
Bouligand J, Boland I, Valteau-Couanet D, Deroussent A, Kalifa C, Hartmann O et al. In children and adolescents, the pharmacodynamics of high-dose busulfan is dependent on the second alkylating agent used in the combined regimen (melphalan or thiotepa). Bone Marrow Transplant 2003; 32: 979–986.
Vassal G, Deroussent A, Hartmann O, Challine D, Benhamou E, Valteau-Couanet D et al. Dose-dependent neurotoxicity of high-dose busulfan in children: a clinical and pharmacological study. Cancer Res 1990; 50: 6203–6207.
Hartmann O . New strategies for the application of high-dose chemotherapy with haematopoietic support in paediatric solid tumours. Ann Oncol 1995; 6: 13–16.
Corbacioglu S, Cesaro S, Faraci M, Valteau-Couanet D, Gruhn B, Rovelli A et al. Defibrotide for prophylaxis of hepatic veno-occlusive disease in paediatric haemopoietic stem-cell transplantation: an open-label, phase 3, randomised controlled trial. Lancet 2012; 379: 1301–1309.
Bearman SI, Appelbaum FR, Buckner CD, Petersen FB, Fisher LD, Clift RA et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
Matthay KK, Shulkin B, Ladenstein R, Michon J, Giammarile F, Lewington V et al. Criteria for evaluation of disease extent by (123)I-metaiodobenzylguanidine scans in neuroblastoma: a report for the International Neuroblastoma Risk Group (INRG) Task Force. Br J Cancer 2010; 102: 1319–1326.
Rothman KJ . Estimation of confidence limits for the cumulative probability of survival in life table analysis. J Chronic Dis 1978; 31: 557–560.
Schemper M, Smith TL . A note on quantifying follow-up in studies of failure time. Control Clin Trials 1996; 17: 343–346.
Brodeur GM, Pritchard J, Berthold F, Carlsen NL, Castel V, Castelberry RP et al. Revisions of the international criteria for neuroblastoma diagnosis, staging, and response to treatment. J Clin Oncol 1993; 11: 1466–1477.
Yanik GA, Parisi MT, Shulkin BL, Naranjo A, Kreissman SG, London WB et al. Semiquantitative mIBG scoring as a prognostic indicator in patients with stage 4 neuroblastoma: a report from the Children's oncology group. J Nucl Med 2013; 54: 541–548.
Gaspar N, Hartmann O, Munzer C, Bergeron C, Millot F, Cousin-Lafay L et al. Neuroblastoma in adolescents. Cancer 2003; 98: 349–355.
Haut PR, Cohn S, Morgan E, Hubbell M, Danner-Koptik K, Olszewski M et al. Efficacy of autologous peripheral blood stem cell (PBSC) harvest and engraftment after ablative chemotherapy in pediatric patients. Biol Blood Marrow Transplant 1998; 4: 38–42.
Hartmann O, Valteau-Couanet D, Benhamou E, Vassal G, Rubie H, Beaujean F et al. Stage IV neuroblastoma in patients over 1 year of age at diagnosis: consolidation of poor responders with combined busulfan, cyclophosphamide and melphalan followed by in vitro mafosfamide-purged autologous bone marrow transplantation. Eur J Cancer 1997; 33: 2126–2129.
Hartmann O, Kalifa C, Beaujean F, Patte C, Flamant F, Jullien C et al. Treatment of advanced neuroblastoma with high-dose melphalan and autologous bone marrow transplantation as consolidation therapy in metastatic neuroblastoma. Cancer Chemother Pharmacol 1986; 16: 165–169.
Kletzel M, Abella EM, Sandler ES, Williams LL, Ogden AK, Pollock BH et al. Thiotepa and cyclophosphamide with stem cell rescue for consolidation therapy for children with high-risk neuroblastoma: a phase I/II study of the Pediatric Blood and Marrow Transplant Consortium. J Pediatr Hematol Oncol 1998; 20: 49–54.
Lucidarme N, Valteau-Couanet D, Oberlin O, Couanet D, Kalifa C, Beaujean F et al. Phase II study of high-dose thiotepa and hematopoietic stem cell transplantation in children with solid tumors. Bone Marrow Transplant 1998; 22: 535–540.
Kletzel M, Katzenstein HM, Haut PR, Yu AL, Morgan E, Reynolds M et al. Treatment of high-risk neuroblastoma with triple-tandem high-dose therapy and stem-cell rescue: results of the Chicago Pilot II Study. J Clin Oncol 2002; 20: 2284–2292.
Grupp SA, Stern JW, Bunin N, Nancarrow C, Ross AA, Mogul M et al. Tandem high-dose therapy in rapid sequence for children with high-risk neuroblastoma. J Clin Oncol 2000; 18: 2567–2575.
George RE, Li S, Medeiros-Nancarrow C, Neuberg D, Marcus K, Shamberger RC et al. High-risk neuroblastoma treated with tandem autologous peripheral-blood stem cell-supported transplantation: long-term survival update. J Clin Oncol 2006; 24: 2891–2896.
Granger M, Grupp SA, Kletzel M, Kretschmar C, Naranjo A, London WB et al. Feasibility of a tandem autologous peripheral blood stem cell transplant regimen for high risk neuroblastoma in a cooperative group setting: A Pediatric Oncology Group study: a report from the Children’s Oncology Group. Pediatr Blood Cancer 2012; 59: 902–907.
Seif AE, Naranjo A, Baker DL, Bunin NJ, Kletzel M, Kretschmar CS et al. A pilot study of tandem high-dose chemotherapy with stem cell rescue as consolidation for high-risk neuroblastoma: Children's Oncology Group study ANBL00P1. Bone Marrow Transplant 2013; 48: 947–952.
Matthay KK, Quach A, Huberty J, Franc BL, Hawkins RA, Jackson H et al. Iodine-131-metaiodobenzylguanidine double infusion with autologous stem-cell rescue for neuroblastoma: a new approaches to neuroblastoma therapy phase I study. J Clin Oncol 2009; 27: 1020–1025.
DuBois SG, Chesler L, Groshen S, Hawkins R, Goodarzian F, Shimada H et al. Phase I study of vincristine, irinotecan, and ¹3¹I-metaiodobenzylguanidine for patients with relapsed or refractory neuroblastoma: a new approaches to neuroblastoma therapy trial. Clin Cancer Res 2012; 18: 2679–2686.
French S, DuBois SG, Horn B, Granger M, Hawkins R, Pass A et al. 131I-MIBG followed by consolidation with busulfan, melphalan and autologous stem cell transplantation for refractory neuroblastoma. Pediatr Blood Cancer 2013; 60: 879–884.
Yanik GA, Villablanca JG, Maris JM, Weiss B, Groshen S, Marachelian A et al. 131I-Metaiodobenzylguanidine with intensive chemotherapy and autologous stem cell transplantation for high-risk neuroblastoma. a new approaches to neuroblastoma therapy (NANT) phase II study. Biol Blood Marrow Transplant 2015; 21: 673–681.
Acknowledgements
We are grateful to Lorna Saint Ange for editing, Sara Calmanti and Imène Hezam for their secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Pasqualini, C., Dufour, C., Goma, G. et al. Tandem high-dose chemotherapy with thiotepa and busulfan–melphalan and autologous stem cell transplantation in very high-risk neuroblastoma patients. Bone Marrow Transplant 51, 227–231 (2016). https://doi.org/10.1038/bmt.2015.264
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.264
This article is cited by
-
A Systematic Review and Meta-Analysis of Studies of Defibrotide Prophylaxis for Veno-Occlusive Disease/Sinusoidal Obstruction Syndrome
Clinical Drug Investigation (2022)
-
Long-term outcomes of the GPOH NB97 trial for children with high-risk neuroblastoma comparing high-dose chemotherapy with autologous stem cell transplantation and oral chemotherapy as consolidation
British Journal of Cancer (2018)
-
Incidence, Survival, and Treatment of Localized and Metastatic Neuroblastoma in Germany 1979–2015
Pediatric Drugs (2017)
-
Busulfan/melphalan/thiotepa
Reactions Weekly (2017)
-
Toxicities of busulfan/melphalan versus carboplatin/etoposide/melphalan for high-dose chemotherapy with stem cell rescue for high-risk neuroblastoma
Bone Marrow Transplantation (2016)