Abstract
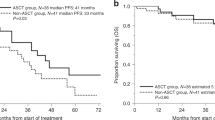
Autologous stem cell transplant (Auto-SCT) is increasingly used in older patients with multiple myeloma (MM), despite lack of phase 3 trials in this age-defined population. For 207 consecutive MM patients who underwent Auto-SCT and were 70 years or older at transplant (study cohort), data were analyzed and compared with a younger cohort (1764 Auto-SCT patients <70 years old). The proportion of Auto-SCT in the older patients increased from 7.8% of all transplants in 1998–2006 to 12.9% in 2007–2015. Sixty percent of patients required stem cell mobilization with chemotherapy or plerixafor. Full-dose melphalan conditioning was given to 55% of the older patients compared with 93% of the younger patients (P<0.001). Older patients were more likely to be hospitalized (64% vs 55%; P=0.01), but hospitalization duration was comparable. For newly diagnosed patients, median PFS was 33.5 months for the older cohort and 33.8 months for the younger cohort (P=0.91), and median overall survival was 6.1 and 7.8 years, respectively (P=0.11). Presumably, a smaller fraction of patients, age 70–76, is selected for Auto-SCT, but the benefits are comparable to those seen for younger patients. Reduced-dose melphalan was given to approximately half the patients to avoid excessive toxicity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Palumbo A, Bringhen S, Bertola A, Cavallo F, Falco P, Massaia M et al. Multiple myeloma: comparison of two dose-intensive melphalan regimens (100 vs 200 mg/m2. Leukemia 2004; 18: 133–138.
Child JA, Morgan GJ, Davies FE, Owen RG, Bell SE, Hawkins K et al. Medical Research Council Adult Leukaemia Working Party. High-dose chemotherapy with hematopoietic stem-cell rescue for multiple myeloma. N Engl J Med 2003; 348: 1875–1883.
Attal M, Harousseau JL, Stoppa AM, Sotto JJ, Fuzibet JG, Rossi JF et al. Intergroupe Francais du Myelome. A prospective, randomized trial of autologous bone marrow transplantation and chemotherapy in multiple myeloma. N Engl J Med 1996; 335: 91–97.
Passweg JR, Baldomero H, Peters C, Gaspar HB, Cesaro S, Dreger P et al. European Society for Blood and Marrow Transplantation EBMT. Hematopoietic SCT in Europe: data and trends in 2012 with special consideration of pediatric transplantation. Bone Marrow Transplant 2014; 49: 744–750.
Palumbo A, Cavallo F, Gay F, Di Raimondo F, Ben Yehuda D, Petrucci MT et al. Autologous transplantation and maintenance therapy in multiple myeloma. N Engl J Med 2014; 371: 895–905.
Kyle RA, Gertz MA, Witzig TE, Lust JA, Lacy MQ, Dispenzieri A et al. Review of 1027 patients with newly diagnosed multiple myeloma. Mayo Clin Proc 2003; 78: 21–33.
Winn AN, Shah GL, Cohen JT, Lin PJ, Parsons SK . The real world effectiveness of hematopoietic transplant among elderly individuals with multiple myeloma. J Natl Cancer Inst 2015; 107: 1–6.
Wildes TM, Finney JD, Fiala M, Gao F, Vij R, Stockerl-Goldstein K et al. High-dose therapy and autologous stem cell transplant in older adults with multiple myeloma. Bone Marrow Transplant 2015; 50: 1075–1082.
El Cheikh J, Kfoury E, Calmels B, Lemarie C, Stoppa AM, Bouabdallah R et al. Age at transplantation and outcome after autologous stem cell transplantation in elderly patients with multiple myeloma. Hematol Oncol Stem Cell Ther 2011; 4: 30–36.
Muta T, Miyamoto T, Fujisaki T, Ohno Y, Kamimura T, Kato K et al. Fukuoka Blood and Marrow Transplant Group (FBMTG). Evaluation of the feasibility and efficacy of autologous stem cell transplantation in elderly patients with multiple myeloma. Intern Med 2013; 52: 63–70.
Kumar SK, Dingli D, Lacy MQ, Dispenzieri A, Hayman SR, Buadi FK et al. Autologous stem cell transplantation in patients of 70 years and older with multiple myeloma: results from a matched pair analysis. Am J Hematol 2008; 83: 614–617.
McCarthy PL Jr, Hahn T, Hassebroek A, Bredeson C, Gajewski J, Hale G et al. Trends in use of and survival after autologous hematopoietic cell transplantation in North America, 1995-2005: significant improvement in survival for lymphoma and myeloma during a period of increasing recipient age. Biol Blood Marrow Transplant 2013; 19: 1116–1123.
Auner HW, Szydlo R, Hoek J, Goldschmidt H, Stoppa AM, Morgan GJ et al. Trends in autologous hematopoietic cell transplantation for multiple myeloma in Europe: increased use and improved outcomes in elderly patients in recent years. Bone Marrow Transplant 2015; 50: 209–215.
Gertz MA, Dingli D . How we manage autologous stem cell transplantation for patients with multiple myeloma. Blood 2014; 124: 882–890.
Gertz MA, Ansell SM, Dingli D, Dispenzieri A, Buadi FK, Elliott MA et al. Autologous stem cell transplant in 716 patients with multiple myeloma: low treatment-related mortality, feasibility of outpatient transplant, and effect of a multidisciplinary quality initiative. Mayo Clin Proc 2008; 83: 1131–1138.
Durie BG, Harousseau JL, Miguel JS, Blade J, Barlogie B, Anderson K et al. International Myeloma Working Group. International uniform response criteria for multiple myeloma. Leukemia 2006; 20: 1467–1473.
Sharma M, Zhang MJ, Zhong X, Abidi MH, Akpek G, Bacher U et al. Older patients with myeloma derive similar benefit from autologous transplantation. Biol Blood Marrow Transplant 2014; 20: 1796–1803.
Kumar SK, Dispenzieri A, Lacy MQ, Gertz MA, Buadi FK, Pandey S et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia 2014; 28: 1122–1128.
Pozotrigo M, Adel N, Landau H, Lesokhin A, Lendvai N, Chung DJ et al. Factors impacting stem cell mobilization failure rate and efficiency in multiple myeloma in the era of novel therapies: experience at Memorial Sloan Kettering Cancer Center. Bone Marrow Transplant 2013; 48: 1033–1039.
Morris CL, Siegel E, Barlogie B, Cottler-Fox M, Lin P, Fassas A et al. Mobilization of CD34+ cells in elderly patients (⩾70 years) with multiple myeloma: influence of age, prior therapy, platelet count and mobilization regimen. Br J Haematol 2003; 120: 413–423.
Silvennoinen R, Anttila P, Saily M, Lundan T, Heiskanen J, Siitonen TM et al. A randomized phase II study of stem cell mobilization with cyclophosphamide+G-CSF or G-CSF alone after lenalidomide-based induction in multiple myeloma. Bone Marrow Transplant 2015; 51: 372–376.
Lin TL, Wang PN, Kuo MC, Hung YH, Chang H, Tang TC . Cyclophosphamide plus granulocyte-colony stimulating factor for hematopoietic stem cell mobilization in patients with multiple myeloma. J Clin Apher, (e-pub ahead of print 5 September 2015; doi:10.1002/jca.21421).
Hamaker ME, Jonker JM, de Rooij SE, Vos AG, Smorenburg CH, van Munster BC . Frailty screening methods for predicting outcome of a comprehensive geriatric assessment in elderly patients with cancer: a systematic review. Lancet Oncol 2012; 13: e437–e444.
Palumbo A, Bringhen S, Mateos MV, Larocca A, Facon T, Kumar SK et al. Geriatric assessment predicts survival and toxicities in elderly myeloma patients: an International Myeloma Working Group report. Blood 2015; 125: 2068–2074.
Johnson LS, Juhlin T, Engstrom G, Nilsson PM . Reduced forced expiratory volume is associated with increased incidence of atrial fibrillation: the Malmo Preventive Project. Europace 2014; 16: 182–188.
Buch P, Friberg J, Scharling H, Lange P, Prescott E . Reduced lung function and risk of atrial fibrillation in the Copenhagen City Heart Study. Eur Respir J 2003; 21: 1012–1016.
Sureddi RK, Amani F, Hebbar P, Williams DK, Leonardi M, Paydak H et al. Atrial fibrillation following autologous stem cell transplantation in patients with multiple myeloma: incidence and risk factors. Ther Adv Cardiovasc Dis 2012; 6: 229–236.
St Bernard R, Chodirker L, Masih-Khan E, Jiang H, Franke N, Kukreti V et al. Efficacy, toxicity and mortality of autologous SCT in multiple myeloma patients with dialysis-dependent renal failure. Bone Marrow Transplant 2015; 50: 95–99.
Acknowledgements
Author contributions
EM and MAG designed the study, collected the data, analyzed the data, wrote the first draft, and approved the final version of the manuscript; DD, SK, FKB, AD, SRH, WJH, NL, PK, MQL and SVR provided patient management, critically reviewed the first draft and approved the final version of the manuscript; RCW and RC collected data, provided critical review and approved the final version of the manuscript; and DAG provided patient management, collected data, provided critical review and approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
SK: Celgene Corp. (consultancy and research funding), Millennium Pharmaceuticals Inc. (consultancy and research funding), Novartis International AG (research funding), Onyx Pharmaceuticals Inc. (consultancy and research funding), AbbVie Inc. (research funding), Janssen Pharmaceuticals, Inc. (consultancy and research funding) and Bristol-Myers Squibb Co. (consultancy and research funding); AD: Celgene Corp (research funding), Millennium Pharmaceuticals Inc. (research funding), Pfizer Inc. (research funding and travel grant) and Janssen Pharmaceuticals Inc. (research funding); PK: Millennium Pharmaceuticals Inc. (Takeda Pharmaceutical Co. Limited) (research funding), Celgene Corp. (research funding) and Onyx Pharmaceuticals Inc. (Amgen Inc.) (research funding); MQL: Celgene Corp. (research funding); MAG: Celgene Corp (honoraria), Millennium Pharmaceuticals, Inc. (consultancy and honoraria), Onyx Pharmaceuticals Inc. (honoraria), Novartis International AG (honoraria) and GlaxoSmithKline PLC (honoraria). The remaining authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Bone Marrow Transplantation website
Supplementary information
Rights and permissions
About this article
Cite this article
Muchtar, E., Dingli, D., Kumar, S. et al. Autologous stem cell transplant for multiple myeloma patients 70 years or older. Bone Marrow Transplant 51, 1449–1455 (2016). https://doi.org/10.1038/bmt.2016.174
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2016.174
This article is cited by
-
Autologous hematopoietic stem cell transplantation activity for lymphoma and multiple myeloma in China
Bone Marrow Transplantation (2023)
-
Melphalan 200 mg/m2 does not increase toxicity and improves survival in comparison to reduced doses of melphalan in multiple myeloma patients
Bone Marrow Transplantation (2021)
-
Autologous stem cell transplant for patients with multiple myeloma between ages 75 and 78
Bone Marrow Transplantation (2021)
-
Outcomes with autologous stem cell transplant vs. non-transplant therapy in patients 70 years and older with multiple myeloma
Bone Marrow Transplantation (2021)
-
Real-world utilisation of ASCT in multiple myeloma (MM): a report from the Australian and New Zealand myeloma and related diseases registry (MRDR)
Bone Marrow Transplantation (2021)