Abstract
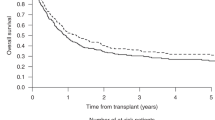
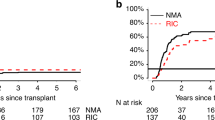
Hematopoietic cell transplantation (HCT) with non-myeloablative (NMA) conditioning for lymphoproliferative diseases (LD) includes fludarabine with and without low-dose TBI. Transplant outcomes were compared among patients aged ⩾40 years with LD who received a HCT with TBI (N=382) or no-TBI (N=515) NMA from 2001 to 2011. The groups were comparable except for donor, graft, prophylaxis for GVHD, disease status and year of HCT. Cumulative incidences of grades II–IV GVHD at 100 days were 29% and 20% (P=0.001) and of chronic GVHD at 1 year were 54% and 44% (P=0.004) for TBI and no-TBI, respectively. Multivariate analysis of progression/relapse, treatment failure and mortality showed no outcome differences by conditioning. Full donor chimerism at day 100 was observed in 82% vs 64% in the TBI and no-TBI groups, respectively (P=0.006). Subsets of the four most common conditioning/GVHD prophylaxis combinations demonstrated higher rates of grades II–IV acute (P<0.001) and chronic GVHD (P<0.001) among recipients of TBI–mycophenolate mofetil (MMF) compared with other combinations. TBI-based NMA conditioning induces faster full donor chimerism, but overall survival outcomes are comparable to no-TBI regimens. Combinations of TBI and MMF are associated with higher rates of GVHD without impact on survival outcomes in patients with LD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
McSweeney P, Niederwieser D, Schizuru J . Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001; 97: 3390–3400.
Maris MB, Niederwieser D, Sandmaier BM, Storer B, Stuart M, Maloney D et al. HLA-matched unrelated donor hematopoieitc cell transplantation after nonmyeloablative conditioning for patients with hematologic malignancie. Blood 2003; 102: 2021–2030.
Niederwieser D, Maris M, Shizuru JA, Petersdorf E, Hegenbart U, Sandmaier BM et al. Low-dose TBI and fludarabine followed by HCT from HLA-matched or mismatched unrelated donors and postgrafting immunosuppression with cyclosporine and MMF can induce durable complete chimerism and sustained remissions in patients with hematologic diseases. Blood 2003; 16: 384–394.
Storb R, Yu C, Wagner JL, Deeg HJ, Nash RA, Kiem HP et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood 1997; 89: 3048–3054.
Pasquini MC, Wang Z. Current use and outcome of hematopoietic stem cell transplantation: CIBMTR summary slides 2013. Available at http://www.cibmtr.org.
Khouri IF, Keating M, Korbling M, Przepiorka D, Anderlini P, O’Brien S et al. Transplant-lite: induction of graft-versus-malignancy using fludarabine-based nonablative chemotherapy and allogeneic blood progenitor-cell transplantation as treatment for lymphoid malignancies. J Clin Oncol 1998; 16: 2817–2824.
Baron F, Baker J, Storb R . Kinetics of engraftment in patients with hematologic malignancies given allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Blood 2004; 104: 2254–2262.
Sorror ML, Storer BE, Maloney DG, Sandmaier BM, Martin PJ, Storb R . Outcomes after allogeneic hematopoietic cell transplantation with nonmyeloablative or myeloablative conditioning regimens for treatment of lymphoma and chronic lymphocytic leukemia. Blood 2008; 111: 446–452.
Kouri IF . Allogeneic stem cell transplantation in follicular lymphoma. Best Pract Res Clin Haematol 2011; 24: 271–277.
Laport G . The role of hematopoietic cell transplantation for follicular non-Hodgkin’s lymphoma. Biolo Blood Marrow Transplant 2005; 12: 59–65.
Besien K . The evolving role of autologous and allogeneic stem cell transplantation in follicular lymphoma. Blood Rev 2006; 20: 235–244.
Banerjii V, Johnston J, Seftel M . The role of hematopoietic stem cell transplantation in chronic lymphocytic leukemia. Transfus Apher Sci 2007; 37: 57–62.
Bacigalupo A, Ballen K, Rizzo D, Giralt S, Lazarus H, Ho V et al. Defining the intensity of conditioning regimens: working definitions. Biol Blood Marrow Transplant 2009; 15: 1628–1633.
Corazziari I, Mariotto A, Capocaccia R . Correcting the completeness bias of observed prevalence. Tumori 1999; 85: 370–381.
Capocaccia R, De Angelis R . Estimating the completeness of prevalence based on cancer registry data. Stat Med 1997; 16: 425–440.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15: 825–828.
Bashey A, Owar K, Johnson JL, Edwards PS, Kelly M, Baxter-Lowe LA et al. Reduced-intensity conditioning allogeneic hematopoietic cell transplantation for patients with hematologic malignancies who relapse following autologous transplantation: a multi-institutional prospective study from the Cancer and Leukemia Group B. Biol Blood Marrow Transplant 2011; 17: 558–565.
Sauter CS, Barker JN, Lechner L, Zheng J, Devline SM, Papdopoulos EB et al. A phase II study of a nonmyeloablative allogeneic stem cell transplant with peritransplant rituximab in patients with B cell lymphoid malignancies: favorably durable event-free survival in chemosensitive patients. Biol Blood Marrow Transplant 2014; 20: 354–360.
Kusumi E, Kami M, Kanda Y, Murashige N, Kishi Y, Suzuki R et al. Reduced-intensity hematopoietic stem-cell transplantation for malignant lymphoma: a retrospective survey of 112 adult patients in Japan. Bone Marrow Transplant 2005; 36: 205–213.
Khouri IF, McLaughlin P, Saliba RM, Hosing C, Korbling M, Lee MS et al. Eight year-experience with allogeneic stem cell transplantation for relapsed follicular lymphoma after nonmyeloablative conditioning wiht fludarabine, cyclophosphamide, and rituximab. Blood 2008; 111: 5530–5336.
Mattsson J, Uzunel M, Remberger M, Ringen O . T cell mixed chimerism is significantly correlated to a decreased risk of acute graft-versus-host disease after allogeneic stem cell transplantation. Transplantation 2001; 71: 433–439.
Huss R, Deeg HJ, Gooley T, Bryant E, Leisenring W, Clift R et al. Effect of mixed chimerism on graft-versus-host disease, disease recurrence and survival after HLA-identical marrow transplantation for aplastic anemia or chronic myelogenous leukemia. Bone Marrow Transplant 1996; 18: 767–776.
Sykes M, Preffer F, McAfee S, Saidman SL, Weymouth D, Andrews DM et al. Mixed lymphohaemopoietic chimerism and graft-versus-lymphoma effects after non-myeloablative therapy and HLA-mismatched bone marrow transplantation. Lancet 1999; 353: 1755–1759.
Ram R, Yeschurun M, Vidal L, Shpilberg O, Gafter-Gvili A . Mycophenolate mofetil vs methotrexate for the prevention of graft-versus-host-disease—systematic review and meta-analysis. Leuk Res 2013; 38: 352–360.
Chen GL, Zhang T, Hahn Y, Abrams S, Ross M, Liu H et al. Acute GVHD prophylaxis with standard-dose, micro-dose. Bone Marrow Transplant 2014; 49: 248–253.
Johnston L, Florek M, Armstrong R, McCune JS, Arai S, Brown J et al. Sirolimus and mycophenolate mofetil as GVHD prophylaxis in myeloablative, matched-related donor hematopoietic cell transplantation. Bone Marrow Transplant 2012; 47: 581–588.
Sabry W, Le Blanc R, Labbe AC, Sauvageau G, Couban S, Kiss T et al. Graft-versus-host disease prophylaxis with tacrolimus and mycophenolate mofetil in HLA-matched nonmyeloablative transplant recipients is associated with very low incidence of GVHD and nonrelapse mortality. Biol Blood Marrow Transplant 2009; 15: 919–929.
Kornblit B, Maloney DG, Storb R . Fludarabine and 2-Gy TBI is superior to 2 Gy TBI as conditioning for HLA-matched related hematopoietic cell transplantation: a phase III randomized trial. Biol Blood Marrow Transplant 2013; 19: 1340–1347.
Acknowledgements
We thank Professor John Klein for the valuable input on the design of this study who had contributed significantly for the development of this study. We would like to dedicate this study to Dr Klein’s memory. The CIBMTR is supported by Public Health Service Grant/Cooperative Agreement U24-CA076518 from the National Cancer Institute (NCI), the NHLBI and the National Institute of Allergy and Infectious Diseases (NIAID); a Grant/Cooperative Agreement 5U10HL069294 from NHLBI and NCI; a contract HHSH250201200016C with Health Resources and Services Administration (HRSA/DHHS); two Grants N00014-12-1-0142 and N00014-13-1-0039 from the Office of Naval Research; and grants from *Actinium Pharmaceuticals; Allos Therapeutics, Inc.; *Amgen, Inc.; Anonymous donation to the Medical College of Wisconsin; Ariad; Be the Match Foundation; *Blue Cross and Blue Shield Association; *Celgene Corporation; Chimerix, Inc.; Fred Hutchinson Cancer Research Center; Fresenius-Biotech North America, Inc.; *Gamida Cell Teva Joint Venture Ltd; Genentech, Inc.;*Gentium SpA; Genzyme Corporation; GlaxoSmithKline; Health Research, Inc. Roswell Park Cancer Institute; HistoGenetics, Inc.; Incyte Corporation; Jeff Gordon Children’s Foundation; Kiadis Pharma; The Leukemia and Lymphoma Society; Medac GmbH; The Medical College of Wisconsin; Merck and Co, Inc.; Millennium: The Takeda Oncology Co.; *Milliman USA, Inc.; *Miltenyi Biotec, Inc.; National Marrow Donor Program; Onyx Pharmaceuticals; Optum Healthcare Solutions, Inc.; Osiris Therapeutics, Inc.; Otsuka America Pharmaceutical, Inc.; Perkin Elmer, Inc.; *Remedy Informatics; *Sanofi US; Seattle Genetics; Sigma-Tau Pharmaceuticals; Soligenix, Inc.; St Baldrick’s Foundation; StemCyte, A Global Cord Blood Therapeutics Co.; Stemsoft Software, Inc.; Swedish Orphan Biovitrum; *Tarix Pharmaceuticals; *TerumoBCT; *Teva Neuroscience, Inc.; *THERAKOS, Inc.; University of Minnesota; University of Utah; and *Wellpoint, Inc. The views expressed in this article do not reflect the official policy or position of the National Institute of Health, the Department of the Navy, the Department of Defense, Health Resources and Services Administration (HRSA) or any other agency of the US Government. *Corporate members.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hong, S., Le-Rademacher, J., Artz, A. et al. Comparison of non-myeloablative conditioning regimens for lymphoproliferative disorders. Bone Marrow Transplant 50, 367–374 (2015). https://doi.org/10.1038/bmt.2014.269
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2014.269