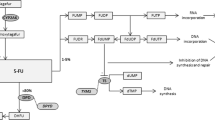
Summary:
Recently, it has been reported that prophylactic administration of ciprofloxacin during cyclophosphamide (CY) conditioning was a high-risk factor for relapse in patients undergoing allogeneic BMT. In the present study, we investigated the possible mechanisms of this interaction in male Sprague–Dawley rats. The kinetics of CY and its active 4-OH-CY metabolite were determined, after 3 days pretreatment with ciprofloxacin (200 mg/kg) and compared to control rats without treatment. CY was administered as a high or low single intravenous dose (150 and 90 mg/kg, respectively). The expression of the CYP2B1, CYP2B2, CYP2C11, CYP3A1 and CYP3A2 genes was evaluated by SYBR Green I Dye real-time PCR for quantification of mRNA. The administration of ciprofloxacin resulted in a significant increase in the AUC (P=0.007) and a significant decrease in clearance (P=0.007) when CY was given as a high dose. In accordance, the metabolic ratio (AUC4-OH-CY/AUCCY) was significantly lower (P=0.007) compared to that found in the control group. Ciprofloxacin significantly suppressed gene expression of CYP2C11 (P=0.01) and CYP3A1 (P=0.04); however, no effect was observed on the gene expression of CYP3A2, CYP2B1 and CYP2B2. Our study revealed that ciprofloxacin interacts with CY and suppressed relevant cytochromes P450 at the transcriptional level. This study may have a great clinical impact when ciprofloxacin is used in therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Boddy AV, Yule SM . Metabolism and pharmacokinetics of oxazaphosphorines. Clin Pharmacokinet 2000; 38: 291–304.
Clarke L, Waxman DJ . Oxidative metabolism of cyclophosphamide: identification of the hepatic monooxygenase catalysts of drug activation. Cancer Res 1989; 49: 2344–2350.
Chang TK, Weber GF, Crespi CL et al. Differential activation of cyclophosphamide and ifosphamide by cytochromes P-450 2B and 3A in human liver microsomes. Cancer Res 1993; 53: 5629–5637.
Weber GF, Waxman DJ . Activation of the anti-cancer drug ifosphamide by rat liver microsomal P450 enzymes. Biochem Pharmacol 1993; 45: 1685–1694.
Huang Z, Roy P, Waxman DJ . Role of human liver microsomal CYP3A4 and CYP2B6 in catalyzing N-dechloro-ethylation of cyclophosphamide and ifosfamide. Biochem Pharmacol 2000; 59: 961–972.
Roy P, Yu LJ, Crespi CL et al. Development of a sub-strate-activity based approach to identify the major human liver P-450 catalysts of cyclophosphamide and ifosfamide activation based on cDNA-expressed activities and liver microsomal P-450 profiles. Drug Metab Dispos 1999; 27: 655–666.
Yu L, Waxman DJ . Role of cytochrome P450 in oxazaphosphorine metabolism. Deactivation via N-dechloroethylation and activation via 4-hydroxylation catalyzed by distinct subsets of rat liver cytochromes P450. Drug Metab Dispos 1996; 24: 1254–1262.
Yu LJ, Drewes P, Gustafsson K et al. In vivo modulation of alternative pathways of P-450-catalyzed cyclophosphamide metabolism: impact on pharmacokinetics and antitumour activity. J Pharmacol Exp Ther 1999; 288: 928–937.
Gervot L, Rochat B, Gautier JC et al. Human CYP2B6: expression, inducibility and catalytic activities. Pharmacogenetics 1999; 9: 295–306.
Chang TK, Yu L, Maurel P et al. Enhanced cyclophosphamide and ifosfamide activation in primary human hepatocyte cultures: response to cytochrome P-450 inducers and autoinduction by oxazaphosphorines. Cancer Res 1997; 57: 1946–1954.
Rae JM, Soukhova NV, Flockhart DA et al. Triethylenethiophosphoramide is a specific inhibitor of cytochrome P450 2B6: implications for cyclophosphamide metabolism. Drug Metab Dispos 2002; 30: 525–530.
Zweers-Zeilmaker WM, Horbach GJ, Witkamp RF . Differential inhibitory effects of phenytoin, diclofenac, phenylbutazone and a series of sulfonamides on hepatic cytochrome P4502C activity in vitro, and correlation with some molecular descriptors in the dwarf goat (Caprus hircus aegagrus). Xenobiotica 1997; 27: 769–780.
Hooper DC, Wolfson JS . Mode of action of the new quinolones: new data. Eur J Clin Microbiol Infect Dis 1991; 10: 223–231.
Hooper DC, Wolfson JS . Fluoroquinolone antimicrobial agents. N Engl J Med 1991; 324: 384–394.
Brouwers JR . Drug interactions with quinolone antibacterials. Drug Saf 1992; 7: 268–281.
McLellan RA, Drobitch RK, Monshouwer M et al. Fluoroquinolone antibiotics inhibit cytochrome P450-mediated microsomal drug metabolism in rat and human. Drug Metab Dispos 1996; 24: 1134–1138.
McLellan RA, Drobitch RK, McLellan H et al. Norfloxacin interferes with cyclosporine disposition in pediatric patients undergoing renal transplantation. Clin Pharmacol Ther 1995; 58: 322–327.
Harder S, Fuhr U, Staib AH et al. Ciprofloxacin–caffeine: a drug interaction established using in vivo and in vitro investigations. Am J Med 1989; 87: 89S–91S.
Wijnands WJ, Vree TB . Interaction between the fluoroquinolones and the bronchodilator theophylline. J Antimicrob Chemother 1988; 22 (Suppl. C): 109–114.
Mulder GJ, Nagelkerke JF, Tijdens RB et al. Inhibition of the oxidative metabolism of theophylline in isolated rat hepatocytes by the quinolone antibiotic enoxacin and its metabolite oxoenoxacin, but not by ofloxacin. Biochem Pharmacol 1988; 37: 2565–2568.
Jolson HM, Tanner LA, Green L et al. Adverse reaction reporting of interaction between warfarin and fluoroquinolones. Arch Intern Med 1991; 151: 1003–1004.
Carlens S, Ringden O, Aschan J et al. Risk factors in bone marrow transplant recipients with leukaemia. Increased relapse risk in patients treated with ciprofloxacin for gut decontamination. Clin Transplant 1998; 12: 84–92.
Griskevicius L, Meurling L, Hassan M . Simple method based on fluorescent detection for the determination of 4- hydroxycyclophosphamide in plasma. Ther Drug Monit 2002; 24: 405–409.
Ibach B, Appel K, Gebicke-Haerter P et al. Effect of phenytoin on cytochrome P450 2B mRNA expression in primary rat astrocyte cultures. J Neurosci Res 1998; 54: 402–411.
Morris DL, Davila JC . Analysis of rat cytochrome P450 isoenzyme expression using semi- quantitative reverse transcriptase-polymerase chain reaction (RT-PCR). Biochem Pharmacol 1996; 52: 781–792.
Moore MJ . Clinical pharmacokinetics of cyclophosphamide. Clin Pharmacokinet 1991; 20: 194–208.
Hassan M, Nilsson C, Olsson H et al. The influence of interferon-alpha on the pharmacokinetics of cyclophosphamide and its 4-hydroxy metabolite in patients with multiple myeloma. Eur J Haematol 1999; 63: 163–170.
Slattery JT, Kalhorn TF, McDonald GB et al. Conditioning regimen-dependent disposition of cyclophosphamide and hydroxycyclophosphamide in human marrow transplantation patients. J Clin Oncol 1996; 14: 1484–1494.
Ayash LJ, Wright JE, Tretyakov O et al. Cyclophosphamide pharmacokinetics: correlation with cardiac toxicity and tumour response. J Clin Oncol 1992; 10: 995–1000.
Sladek NE, Doeden D, Powers JF et al. Plasma concentrations of 4-hydroxycyclophosphamide and phosphoramide mustard in patients repeatedly given high doses of cyclophosphamide in preparation for bone marrow transplantation. Cancer Treat Rep 1984; 68: 1247–1254.
Sladek NE, Powers JF, Grage GM . Half-life of oxazaphosphorines in biological fluids. Drug Metab Dispos 1984; 12: 553–559.
Cooke CE, Sklar GE, Nappi JM . Possible pharmacokinetic interaction with quinidine: ciprofloxacin or metronidazole? Ann Pharmacother 1996; 30: 364–366.
Acknowledgements
This study was supported by grants from the Swedish Cancer Society, Swedish Medical Research Council (04496), Stiftelsen Tornspiran (Sweden) and Anders Otto Swärds Stiftelse, Grant no. PROJ01/059 from Swedish Children Cancer Society and Grant no. 02:119 from the Stockholm's Cancer Foundation (Sweden).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xie, HJ., Griskevicius, L., Broberg, U. et al. Alteration of pharmacokinetics of cyclophosphamide and suppression of the cytochrome P450 genes by ciprofloxacin. Bone Marrow Transplant 31, 197–203 (2003). https://doi.org/10.1038/sj.bmt.1703815
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bmt.1703815
Keywords
This article is cited by
-
A mechanism-based understanding of altered drug pharmacokinetics by gut microbiota
Journal of Pharmaceutical Investigation (2023)
-
Cyclophosphamide bioactivation pharmacogenetics in breast cancer patients
Cancer Chemotherapy and Pharmacology (2021)
-
Microbiome Is a Functional Modifier of P450 Drug Metabolism
Current Pharmacology Reports (2019)
-
Cytochrome P450 2J2, a new key enzyme in cyclophosphamide bioactivation and a potential biomarker for hematological malignancies
The Pharmacogenomics Journal (2015)
-
Molecular genetics and epigenetics of the cytochrome P450 gene family and its relevance for cancer risk and treatment
Human Genetics (2010)