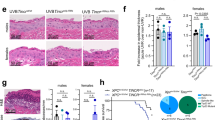
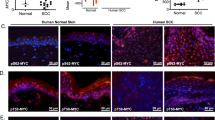
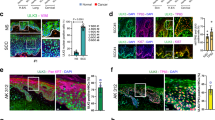
Abstract
We have recently identified a second p53 -related p73L gene, also referred to as p63/p51/p40/KET gene, which encodes the 2 major isoforms p73L and p51 resulting from different exon usage at their amino terminal regions. Although p73L and p51 are suspected to play oncogenic and tumour suppressive roles in mammalian cells, respectively, no evidence of linkage between the expression of these isoforms and human cancers has been reported so far. In this study, we first investigated the expression profile of p51 and p73L in various human tumour cell lines and found that a novel isoform, termed ΔNp73L, was predominantly expressed in squamous cell carcinomas. The expression profile of ΔNp73L/p73L/p51 in primary human skin cancer specimens showed that the expression of p51 was frequently lost (62%) but was detected in all normal skin samples. In p51-expressing skin cancers, ΔNp73L expression was associated at a high frequency (75%) though it was not detected in normal skin tissues. Transient co-transfection data indicate the possibility that ΔNp73L can inhibit p53-, and more preferentially, p51-mediated transactivation. These data suggest that the loss of expression of p51 and/or the expression of ΔNp73L might contribute to the pathogenesis of human squamous cell carcinomas. © 2001 Cancer Research Campaign http://www.bjcancer.com
Similar content being viewed by others
Article PDF
Change history
16 November 2011
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Augustin M, Bamberger C, Paul D and Schmale H (1998) Cloning and chromosomal mapping of the human p53-related KET gene to chromosome 3q27 and its murine homolog Ket to mouse chromosome 16. Mamm Genome 9: 899–902
Di Como CJ, Gaiddon C and Prives C (1999) p73 function is inhibited by tumor-derived p53 mutants in mammalian cells. Mol Cell Biol 19: 1438–1449
Dobbelstein M, Wienzek S, Konig C and Roth J (1999) Inactivation of the p53-homologue p73 by the mdm2-oncoprotein. Oncogene 18: 2101–2106
Greenblatt MS, Bennett WP, Hollstein M and Harris CC (1994) Mutations in the p53 tumor suppressor gene: clues to cancer etiology and molecular pathogenesis. Cancer Res 54: 4855–4878
Hagiwara K, McMenamin MG, Miura K and Harris CC (1999) Mutational analysis of the p63 / p73L / p51 / p40 / CUSP / KET gene in human cancer cell lines using intronic primers. Cancer Res 59: 4165–4169
Hainaut P, Hernandez T, Robinson A, Rodriguez-Tome P, Flores T, Hollstein M, Harris CC and Montesano R (1998) IARC database of p53 gene mutations in human tumors and cell lines: updated compilation, revised formats and new visualisation tools. Nucleic Acids Res 26: 205–213
Hollstein M, Sidransky D, Vogelstein B and Harris CC (1991) p53 mutations in human cancers. Science 253: 49–53
Jost CA, Marin MC and Kaelin WG Jr (1997) p73 is a human p53-related protein that can induce apoptosis. Nature 389: 191–194
Kaelin WG Jr (1999) The emerging p53 gene family. J Natl Cancer Inst 91: 594–598
Kaghad M, Bonnet H, Yang A, Creancier L, Biscan JC, Valent A, Minty A, Chalon P, Lelias JM, Dumont X, Ferrara P, McKeon F and Caput D (1997) Monoallelically expressed gene related to p53 at 1p36, a region frequently deleted in neuroblastoma and other human cancers. Cell 90: 809–819
Kanda T, Segawa K, Ohuchi N, Mori S and Ito Y (1994) Stimulation of polyomavirus DNA replication by wild-type p53 through the DNA-binding site. Mol Cell Biol 14: 2651–2663
Laurenzi VD, Costanzo A, Barcaroli D, Terrinoni A, Falco M, Annicchiarico-Petruzzelli M, Levrero M and Melino G (1998) Two new p73 splice variants, γ and δ, with different transcriptional activity. J Exp Med 188: 1763–1768
Levine AJ, Momand J and Finlay CA (1991) The p53 tumour suppressor gene. Nature 351: 453–456
Marin MC, Jost CA, Irwin MS, DeCaprio JA, Caput D and Kaelin WG Jr (1998) Viral oncoproteins discriminate between p53 and the p53 homolog p73. Mol Cell Biol 18: 6316–6324
Nishi H, Isaka K, Sagawa Y, Usuda S, Fujito A, Ito H, Senoo M, Kato H and Takayama M (1999) Mutation and transcription analyses of the p63 gene in cervical carcinoma. Int J Oncol 15: 1149–1153
Osada M, Ohba M, Kawahara C, Ishioka C, Kanamaru R, Katoh I, Ikawa Y, Nimura Y, Nakagawara A, Obinata M and Ikawa S (1998) Cloning and functional analysis of human p51, which structurally and functionally resembles p53. Nature Med 4: 839–843
Park B-J, Lee S-J, Kim JI, Lee SJ, Lee C-H, Chang S-G, Park J-H and Chi S-G (2000) Frequent alteration of p63 expression in human primary bladder carcinomas. Cancer Res 60: 3370–3374
Parsa R, Yang A, McKeon F and Green H (1999) Association of p63 with proliferative potential in normal and neoplastic human keratinocytes. J Invest Dermatol 113: 1099–1105
Prabhu NS, Somasundaram K, Satyamoorthy K, Herlyn M and El- Deiry WS (1998) p73, unlike p53, suppresses growth and induces apoptosis of human papillomavirus E6-expressing cancer cells. Int J Oncol 13: 5–9
Prives C and Hall PA (1999) The p53 pathway. J Pathol 187: 112–126
Schmale H and Bamberger C (1997) A novel protein with strong homology to the tumor suppressor p53. Oncogene 15: 1363–1367
Senoo M, Seki N, Ohira M, Sugano S, Watanabe M, Inuzuka S, Okamoto T, Tachibana M, Tanaka T, Shinkai Y and Kato H (1998) A second p53-related protein, p73L, with high homology to p73. Biochem Biophys Res Commun 248: 603–607
Steegenga WT, Shvarts A, Riteco N, Bos JL and Jochemsen AG (1999) Distinct regulation of p53 and p73 activity by adenovirus E1A, E1B, and E4orf6 proteins. Mol Cell Biol 19: 3885–3894
Sunahara M, Shishikura T, Takahashi M, Todo S, Yamamoto N, Kimura H, Kato S, Ishioka C, Ikawa S, Ikawa Y and Nakagawara A (1999) Mutational analysis of p51A / TAp63γ, a p53 homolog, in non-small cell lung cancer and breast cancer. Oncogene 18: 3761–3765
Trink B, Okami K, Wu L, Sriuranpong V, Jen J and Sidransky D (1998) A new human p53 homologue. Nature Med 4: 747
Ueda Y, Hijikata M, Takagi S, Chiba T and Shimotohno K (1999) New p73 variants altered C-terminal structures have varied transcriptional activities. Oncogene 18: 4993–4998
Weinberg R (1993) Tumor suppressor genes. Neuron 11: 191–196
Yang A, Kaghad M, Wang Y, Gillett E, Fleming MD, Dotsch V, Andrews NC, Caput D and McKeon F (1998) p63, a p53 homolog at 3q27–29, encodes multiple products with transactivating, death-inducing, and dominant-negative activities. Mol Cell 2: 305–316
Zeng X, Chen L, Jost CA, Maya R, Keller D, Wang X, Kaelin WG Jr, Oren M, Chen J and Lu H (1999) MDM2 suppresses p73 function without promoting p73 degradation. Mol Cell Biol 19: 3257–3266
Author information
Authors and Affiliations
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Senoo, M., Tsuchiya, I., Matsumura, Y. et al. Transcriptional dysregulation of the p73L / p63 / p51 / p40 / KET gene in human squamous cell carcinomas: expression of Δ Np73L, a novel dominant-negative isoform, and loss of expression of the potential tumour suppressor p51. Br J Cancer 84, 1235–1241 (2001). https://doi.org/10.1054/bjoc.2000.1735
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1054/bjoc.2000.1735
Keywords
This article is cited by
-
Expression of p40 (∆Np63) protein in meningiomas, an unexpected finding: immunohistochemical study and evaluation of its possible prognostic role
Journal of Neuro-Oncology (2016)
-
HIPK2 phosphorylates ΔNp63α and promotes its degradation in response to DNA damage
Oncogene (2011)
-
p63 short isoforms are found in invasive carcinomas only and not in benign breast conditions
Virchows Archiv (2010)
-
Clinical relevance of the putative stem cell marker p63 in breast cancer
Breast Cancer Research and Treatment (2010)
-
Characterisation of the proximal airway squamous metaplasia induced by chronic tobacco smoke exposure in spontaneously hypertensive rats
Respiratory Research (2009)