Abstract
Background:
Parental occupational exposures have been associated with childhood brain tumours (CBT), but results are inconsistent. Few studies have studied CBT risk and parental solvent exposure, suggesting a possible association. We examined the association between CBT and parental occupational exposure to solvents in a case–control study.
Methods:
Parents of 306 cases and 950 controls completed detailed occupational histories. Odds ratios (ORs) and 95% confidence intervals (CIs) were estimated for both maternal and paternal exposure to benzene, other aromatics, aliphatics and chlorinated solvents in key time periods relative to the birth of their child. Adjustments were made for matching variables (child’s age, sex and state of residence), best parental education and occupational exposure to diesel exhaust.
Results:
An increased risk of CBT was observed with maternal occupational exposures to chlorinated solvents (OR=8.59, 95% CI 0.94–78.9) any time before birth. Paternal exposure to solvents in the year before conception was associated with an increased CBT risk: OR=1.55 (95% CI 0.99–2.43). This increased risk appeared to be mainly attributable to exposure to aromatic solvents: OR=2.72 (95% CI 0.94–7.86) for benzene and OR=1.76 (95% CI 1.10–2.82) for other aromatics.
Conclusions:
Our results indicate that parental occupational exposures to solvents may be related to an increased risk of CBT.
Similar content being viewed by others
Main
Parental occupational exposures to various chemical agents have been associated with childhood brain tumours (CBTs) in several studies, but results have been inconsistent (Baldwin and Preston-Martin, 2004). Positive associations were reported for a variety of industries and occupations, such as motor vehicle-related occupations, the chemical and petroleum industries, and jobs with frequent exposure to paints (Cordier et al, 1997; Colt and Blair, 1998). The suggested causative agents were polycyclic aromatic hydrocarbons and unspecified solvents (Cordier et al, 1997; Colt and Blair, 1998). Relatedly, a recent meta-analysis showed a positive correlation between parental occupational exposure to pesticides and CBT (Vinson et al, 2011).
Few studies have looked in detail at occupational exposure to solvents. Paternal exposure to chemical solvents (not further specified) was found to be associated with an almost threefold increase of CBT in the 1970s (Peters et al, 1981). Cordier et al (1997) reported an increased risk for maternal occupational exposure to solvents at high levels, with an odds ratio (OR) of 2.4 (95% confidence interval (CI) 1.2–4.9). High exposure was defined by the authors on page 691 of their paper as being classified in either exposure category: (1) ‘job may entail exposure to levels definitely higher than for the general population, but available information does not permit discrimination between exposed and not exposed’ if the probability of exposure was more than 2/3; (2) ‘job entails exposure to the specific agent at levels clearly higher than the general population’; or (3) ‘job entails exposure to the specific agent, and exposure is known to be particular high’ (Cordier et al, 1997).
Solvents comprise a wide range of chemicals: benzene, other aromatics (such as toluene or xylene), aliphatics (such as hexane or ethylene) and chlorinated solvents (such as trichloroethylene or perchloroethylene). Exposures occur, among others, in the chemical, printing and petroleum industries, or in jobs that involve degreasing, painting or dry cleaning. In all previous studies on CBT, solvents were assessed as one group, whereas the different chemical types may have different effects.
We conducted an Australian, nation-wide case–control study of CBT (Aus-CBT) collecting parents’ full occupational histories (Greenop et al, 2013; Peters et al, 2013). In previous analyses we have shown that parental occupational exposure to diesel exhaust may increase the risk of CBT; maternal exposure to diesel exhaust any time before birth showed an OR of 1.77 (95% CI 0.96–3.26), whereas paternal exposure around the time of the child’s conception showed an OR of 1.39 (95% CI 0.97–1.99) (Peters et al, 2013). We also observed increased CBT risks following parental exposure to pest control treatments in the year before the pregnancy (OR 1.54, 95% CI 1.07–2.22) and during the pregnancy (OR 1.52, 95% CI 0.99–2.34) (Greenop et al, 2013).
In this paper we examined the association between parental occupational exposure to specific solvent groups and risk of CBT.
Materials and methods
Study population
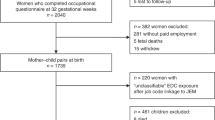
The Aus-CBT study population has been described in detail (Milne et al, 2012). In brief, incident cases (aged 0–14 years, diagnosed between 2005 and 2010) were identified through all 10 paediatric oncology centres in Australia. Approximately three controls, frequency matched by age, sex and state of residence, were recruited for each brain tumour case by random-digit dialling. Controls matched to CBT cases diagnosed in 2005 and 2006 were originally recruited as controls for the Australian childhood acute lymphoblastic leukaemia (Aus-ALL) study (Milne et al, 2009). Written informed consent was obtained from all parents and ethics approval was granted by all participating hospitals.
Data collection
Information about residence, lifestyle factors, medical history and occupation was collected from all parents by a mailed questionnaire. For each job held for more than 6 months, subjects were asked the year started and finished, job title, employer, main tasks and hours worked per week. Job-specific modules with more detailed questions about the tasks undertaken were selected for jobs involving potential exposure to agents of interest. In total, 46 different job modules were available. The selected modules were subsequently asked in computer-assisted telephone interviews. Parents were only included in the current analyses when a job history was available, that is, parents who never had a job before the birth of their child or who did not provide a job history were excluded from the analyses of occupational exposures.
Exposure assessment
We assessed parental occupational exposure to solvents qualitatively (i.e. exposed or non-exposed) based on the job characteristics obtained in the telephone interviews. Based on the questions in the job-specific modules, one of the authors (SP) had written rules to assign exposures (yes/no) to solvents. The preliminary rules were discussed with another author (DG) and modified where necessary to finalise the exposure rules. We assessed the following subtypes of solvents: benzene, other aromatics, aliphatics and chlorinated solvents. For the different exposures, different numbers of questions were selected: benzene was based on 16 questions from 14 job modules; other aromatic solvents based on 31 questions from 16 modules; aliphatic solvents based on 30 questions from 16 modules; and chlorinated solvents based on 20 questions from 11 modules (Peters et al, 2014). The a priori exposure rules were then applied to the information collected in the telephone interviews with the parents. Exposure was assigned blinded to the case–control status.
Exposure variables were created for specific time periods relative to the birth year of the child. These key time periods were exposure at any time before the child’s birth year; exposure in the year 2 years before the birth year (capturing ‘the year before conception’ for fathers only); exposure in the year before the child’s birth year (capturing the time during pregnancy for mothers); and exposure in the year after the child’s birth year (representing the time of breast feeding – mothers only).
Statistical analyses
ORs and 95% CIs were estimated for maternal and paternal occupational exposures to solvents associated with risk of CBT in their offspring, using unconditional logistic regression models. All models were adjusted for the frequency-matching variables. Models were additionally adjusted for best parental education (as a measure of socio-economic status (SES)) and occupational exposure to diesel exhaust. Other potential confounders that were considered, based on their suggested association with CBTs and potential association with occupational exposure to solvents, were any maternal alcohol use during pregnancy, any maternal smoking during pregnancy, maternal age at child’s birth and paternal alcohol use in the year prior to pregnancy. The addition of these variables changed the ORs by <5%, so they were not included in the final models. We have also included occupational exposures to benzene, other aromatics, aliphatics, chlorinated solvents and diesel exhaust in one model (OR2). Analyses were conducted in SAS v.9.3 (SAS Institute Inc., Cary, NC, USA).
Results
Parents of 374 cases consented to participate (65.8% of those invited by their physician). More details of case selection have been published (Peters et al, 2013). Full occupational histories were provided by 293 (78.3%) consenting case mothers and 242 (64.7%) consenting case fathers. In all 3624 families of eligible control children were identified, of whom 2255 (62.2%) agreed to participate. To reach our frequency-matching quotas, 1467 of these families were recruited to the study. Full occupational information was provided by 935 control mothers (63.7% of recruited) and 795 fathers (54.2%). Overall, occupational information of at least one of the parents was available for 306 cases and 950 controls. These families were included in the analyses (Table 1).
Maternal exposure to all solvents combined any time before birth of their child suggested an increased risk of CBT, showing an OR of 1.16, but with a wide CI (Table 2). When analyses were specified to type of solvent, the increased risk was most pronounced with maternal occupational exposure to chlorinated solvents (OR 8.79, 95% CI 0.96–80.7) at any time before birth. Overall, few women were exposed to solvents in the year before birth (Table 2) or in the year after birth (data not shown).
Paternal occupational exposure to all solvents combined any time before the child’s birth appeared to be associated with CBT risk: OR 1.27 (95% CI 0.90–1.79, Table 3). The OR for paternal exposure in the year before conception was 1.55 (95% CI 0.99–2.43). Risk of CBT seemed to be increased with paternal occupational exposure in the year before conception to benzene (OR 2.66, 95% CI 0.92–7.69), other aromatics (OR 1.75, 95% CI 1.10–2.79) and aliphatics (OR 1.42, 95% CI 0.90–2.24) (Table 3). The OR for exposure to chlorinated solvents was also elevated (2.33), but, being based on very small numbers exposed, the CI was wide.
Exposures to other aromatic solvents and aliphatic solvents were strongly correlated among men (RPearson=0.90). When both exposures in the year before conception were included in the same model and adjusted for diesel exhaust exposure, only exposure to other aromatic solvents (OR 4.24, 95% CI 1.10–16.4) was associated with an increased risk of CBT (the OR for aliphatic solvents was 0.39, 95% CI 0.10–1.49) (data not shown). When all exposures were modelled together, the association between CBT risk and paternal exposure to other aromatics appeared to be strongest (OR2 4.47, 95% CI 1.12–17.8; Table 3).
Excluding the Aus-ALL controls from the analyses revealed similar risk estimates (data not shown). When both parents had occupational exposure to solvents any time before the child’s birth, the OR for CBT was 2.04 (95% CI 0.90–4.64, based on 25 exposed controls and 13 exposed cases), compared with the non-exposed case (data not shown).
Discussion
Our results suggest that parental occupational exposure to solvents is associated with an increased risk of CBT. For paternal exposure, this seemed mainly driven by exposure to aromatic solvents in the year before conception. For mothers, a positive association, although based on very small numbers exposed, was observed between CBT risk and exposure to chlorinated solvents any time before the child’s birth.
Our overall findings are consistent with previous reports on the association of CBT with parental exposure to solvents. We observed an OR of 1.16 (95% CI 0.71–1.90) for combined solvent exposure any time before birth for mothers. This risk estimate is lower than the OR of 2.4 (95% CI 1.2–4.9) reported by Cordier et al (1997) for occupational exposure in the 5-year period before the child’s birth. This OR of 2.4 reflected the risk for the highest exposure category. For any exposure, the OR for mothers in their study was 1.3 (95% CI 0.9–2.0) (Cordier et al, 1997). However, we additionally adjusted for occupational exposure to diesel exhaust, whereas without this adjustment the OR for maternal exposure would have been 1.32 (95% CI 0.85–2.06; data not shown). For paternal exposure to combined solvents Cordier et al did not observe an increased risk, while we found an OR of 1.27 (95% CI 0.90–1.79) any time before birth and an OR of 1.55 (95% CI 0.99–2.43) for exposure in the year before conception. Such an increased risk of CBT associated with paternal exposure to solvents was also found in an earlier study (Peters et al, 1981). None of the earlier studies considered the risks by specific type of solvents.
In the model where all solvents and diesel exhaust were mutually adjusted (OR2), the evidence was strongest for an association of CBT with exposure of fathers to aromatic solvents. The association with occupational exposure to diesel exhaust reported previously (Peters et al, 2013) attenuated when modelled together with the specific solvent exposures (OR2). When testing for interactions between exposure to diesel exhaust and solvents, we observed a significant interaction (P=0.049) for paternal exposure in the year before conception. Stratified analyses of the association between paternal occupational exposure to solvents in the year before conception and the risk of CBT revealed an OR of 1.03 (95% CI 0.54–1.99) among fathers not occupationally exposed to diesel exhaust in that year and an OR of 2.48 (95% CI 1.23–4.98) among fathers who were occupationally exposed to diesel exhaust in the same period (data not shown). No interactions were observed for paternal or maternal exposures any time before birth.
The timing of parental exposure relative to the child’s birth can give a clue to the most biologically relevant period of exposure: preconceptional exposure (germ cell effect), perinatal exposure (i.e. intra-uterine or trans-placental) or postnatal exposure (via breast feeding). The detailed job histories obtained from both parents allowed us to assess occupational exposures for the potentially relevant time periods. However, numbers of exposed mothers were generally too low to specify time periods. While we did observe an increased risk for maternal exposure to chlorinated solvents any time before birth, this was based on only three exposed case mothers and four exposed control mothers. None of the mothers was exposed to chlorinated solvents in the year of the pregnancy.
For paternal exposure, the year before conception appeared to have the largest impact as all those ORs were higher than the ORs for any time before birth. This finding suggests a potential effect on sperm cells. Exposure to aromatic solvents such as toluene and xylene showed the strongest relation with CBT in our population. Toluene has been causally linked to DNA damage in the sperm of rats (Nakai et al, 2003), which could potentially lead to carcinogenic effects in the offspring.
The use of organic solvents in Australia and many other developed economies is widespread, including in paints and lacquers, dry cleaning, printing and degreasing. It is recommended that exposures be kept as low as workable and many solvents have occupational exposure standards (e.g. for toluene the 8-h TWA is 50 p.p.m. and the short-term exposure limit is 150 p.p.m. in Australia) (National Occupational Health and Safety Commission, 1990). The median birth year of the children in the current study was 2001 (range 1990–2010), so the relevant paternal exposures in the years before conception will largely have been below this limit. The current use of halogenated solvents should be lower than in previous decades because Australia ratified the Montreal protocol and committed to phasing out the industrial use of particular solvents (e.g. carbon tetrachloride and methyl chloroform) by the end of 1995 and specific uses (e.g. methyl bromide in horticulture) by the end of 2005 (Montreal Protocol, 1987). The Australian government committed to a 90% reduction in the use of hydrochlorofluorocarbons by 2015 and complete phasing out by 2020 (Montreal Protocol, 1987).
Control families were recruited by national random-digit dialling. Approximately 90% of Australian households had a landline telephone connection during the recruitment period (Dal Grande and Taylor, 2010), so families contacted were likely to be representative of the wider population. The participation rates in our study were relatively low, however, among both the cases and the controls. Subjects of highest SES are more likely to participate in a study than subjects of lower SES when recruitment is done via random-digit dialling (Bailey et al, 2010). Previous analyses have shown that the control families used in our study were of higher SES than the general population (Mazloum et al, 2012). We aimed to minimise selection bias by adjusting for SES by means of the highest level of education completed by one of the parents. Adjustment for parental education resulted in slightly lower risk estimates overall, both for paternal and for maternal occupational exposure to solvents, when compared to models not adjusted for education.
Recall bias may have influenced the results, as case parents may reflect on and recall past exposures more than control parents, thus biasing the risk estimates upwards (Schuz et al, 2003). This bias would be particularly likely when questions are asked about specific, known-toxic exposures. We did not ask direct questions about exposure, rather we sought information on jobs and tasks and assigned specific exposures using pre-defined rules (Peters et al, 2014). We cannot rule out, however, greater reporting of jobs or tasks based on case parents’ perceptions of hazards that might be associated with them. ORs may be strongly affected by small changes in the probability of false-positive exposure classifications (i.e. being classified as being exposed when truly non-exposed) when studying low prevalence exposures (Jurek et al, 2008).
When comparing the rule-based exposure assessment method with the more conventional case-by-case expert assessment, it appeared that the assessment of exposure to solvents showed less agreement than pesticides and diesel exhaust, although agreement was still moderate to good for solvents (Peters et al, 2014). Chlorinated solvents showed the lowest agreement (κ=0.26 for men and κ=0.46 for women on a job level), indicating that these solvents are particularly difficult to assess. It is likely that exposure misclassification will have occurred, but without a gold standard we can neither define the specificity of the method nor state whether we under- or overestimated the true exposures.
Exposure misclassification may result in spurious risk estimates; it may therefore be worthwhile applying a more detailed exposure assessment method taking into account exposure levels. In the previous Aus-ALL study, levels of exposure have been studied by applying case-by-case expert assessment (Reid et al, 2011). The same approach could be applied to the Aus-CBT study population, focusing on the specific exposures that appeared to be associated with an increased CBT risk. Additionally, larger study populations may be beneficial when studying a rare disease like CBT in more detail. Pooled analyses of CBT case–control studies where full job histories have been collected may improve our understanding of the effect of occupational exposures on the risk of CBT and its subtypes.
Our results suggest that parental occupational exposures to solvents may be related to CBT. In particular, paternal exposure to aromatic solvents and maternal exposure to chlorinated solvents appeared to be associated with an increased risk of CBT in their offspring.
Change history
26 August 2014
This paper was modified 12 months after initial publication to switch to Creative Commons licence terms, as noted at publication
References
Bailey HD, Milne E, de Klerk N, Fritschi L, Bower C, Attia J, Armstrong BK (2010) Representativeness of child controls recruited by random digit dialling. Paediatr Perinatal Epidemiol 24 (3): 293–302.
Baldwin RT, Preston-Martin S (2004) Epidemiology of brain tumors in childhood—a review. Toxicol Appl Pharmacol 199 (2): 118–131.
Colt JS, Blair A (1998) Parental occupational exposures and risk of childhood cancer. Environ Health Perspect 106 (Suppl 3): 909–925.
National Occupational Health and Safety Commission (1990) Industrial Organic Solvents http://www.safeworkaustraliagovau/sites/SWA/about/Publications/Documents/157/IndustrialOrganicSolvents_1990_PDFpdf (accessed 1 November 13).
Cordier S, Lefeuvre B, Filippini G, Peris-Bonet R, Farinotti M, Lovicu G, Mandereau L (1997) Parental occupation, occupational exposure to solvents and polycyclic aromatic hydrocarbons and risk of childhood brain tumors (Italy, France, Spain). Cancer Causes Control 8 (5): 688–697.
Dal Grande E, Taylor AW (2010) Sampling and coverage issues of telephone surveys used for collecting health information in Australia: results from a face-to-face survey from 1999 to 2008. BMC Med Res Methodol 10: 77.
Greenop KR, Peters S, Bailey HD, Fritschi L, Attia J, Scott RJ, Glass DC, de Klerk NH, Alvaro F, Armstrong BK, Milne E (2013) Exposure to pesticides and the risk of childhood brain tumors. Cancer Causes Control 24 (7): 1269–1278.
Jurek AM, Greenland S, Maldonado G (2008) How far from non-differential does exposure or disease misclassification have to be to bias measures of association away from the null? Int J Epidemiol 37 (2): 382–385.
Mazloum M, Bailey HD, Heiden T, Armstrong BK, de Klerk N, Milne E (2012) Participation in population-based case-control studies: does the observed decline vary by socio-economic status? Paediatr Perinatal Epidemiol 26 (3): 276–279.
Milne E, Greenop KR, Bower C, Miller M, van Bockxmeer FM, Scott RJ, de Klerk NH, Ashton LJ, Gottardo NG, Armstrong BK (2012) Maternal use of folic acid and other supplements and risk of childhood brain tumors. Cancer Epidemiol Biomarkers Prev 21 (11): 1933–1941.
Milne E, Royle JA, de Klerk NH, Blair E, Bailey H, Cole C, Attia J, Scott RJ, Armstrong BK (2009) Fetal growth and risk of childhood acute lymphoblastic leukemia: results from an Australian case-control study. Am J Epidemiol 170 (2): 221–228.
Nakai N, Murata M, Nagahama M, Hirase T, Tanaka M, Fujikawa T, Nakao N, Nakashima K, Kawanishi S (2003) Oxidative DNA damage induced by toluene is involved in its male reproductive toxicity. Free Radic Res 37 (1): 69–76.
Peters FM, Preston-Martin S, Yu MC (1981) Brain tumors in children and occupational exposure of parents. Science 213 (4504): 235–237.
Peters S, Glass DC, Milne E, Fritschi L (2014) Rule-based exposure assessment versus case-by-case expert assessment using the same information in a community-based study. Occup Environ Med 71 (3): 215–219.
Peters S, Glass DC, Reid A, de Klerk N, Armstrong BK, Kellie S, Ashton LJ, Milne E, Fritschi L (2013) Parental occupational exposure to engine exhausts and childhood brain tumors. Int J Cancer 132: 2975–2979.
Montreal Protocol (1987) Montreal Protocol on Substances that Deplete the Ozone Layer. http://www.environmentgovau/topics/environment-protection/ozone-and-synthetic-greenhouse-gases/montreal-protocol (accessed 1 November 2013).
Reid A, Glass DC, Bailey HD, Milne E, Armstrong BK, Alvaro F, Fritschi L (2011) Parental occupational exposure to exhausts, solvents, glues and paints, and risk of childhood leukemia. Cancer Causes Control 22 (11): 1575–1585.
Schuz J, Spector LG, Ross JA (2003) Bias in studies of parental self-reported occupational exposure and childhood cancer. Am J Epidemiol 158 (7): 710–716.
Vinson F, Merhi M, Baldi I, Raynal H, Gamet-Payrastre L (2011) Exposure to pesticides and risk of childhood cancer: a meta-analysis of recent epidemiological studies. Occup Environ Med 68 (9): 694–702.
Acknowledgements
The Aus-CBT consortium conducted the study and the Telethon Institute for Child Health Research (TICHR), University of Western Australia (UWA), was the coordinating centre. Bruce Armstrong (Sydney School of Public Health), Elizabeth Milne (TICHR), Frank van Bockxmeer (UWA), Rodney Scott (University of Newcastle), Lin Fritschi (WA Institute for Medical Research), Lesley Ashton (Children’s Cancer Institute Australia (CCIA)), Nicholas de Klerk (TICHR), Caroline Bower (TICHR), Margaret Miller (Edith Cowan University), John Attia (University of Newcastle), Michelle Haber (CCIA), Murray Norris (CCIA), Judith Thompson (WA Cancer Registry), and Peter Dallas (TICHR) were the research investigators. The authors acknowledge the contribution made by our clinical co-investigators who recruited and cared for the study patients at each participating hospital: Nicholas Gottardo (Princess Margaret Hospital, TICHR); John Heath and Elizabeth Smibert (Royal Children’s Hospital, Melbourne); Tim Hassall and Ross Pinkerton (Royal Children’s Hospital Brisbane); Maria Kirby (Women’s and Children’s Hospital, Adelaide); Stewart Kellie and Luciano Dalla Pozza (Children’s Hospital at Westmead); Frank Alvaro (John Hunter Hospital, Newcastle); Richard Cohn (Sydney Children’s Hospital) and John Daubenton (Royal Hobart Hospital). The authors also acknowledge the Clinical Research Associates at each hospital, and the study coordinators: Jackie Mansour, Somer Dawson, Tamika Heiden and Helen Bailey, for their assistance with data preparation. This work was funded by the National Health and Medical Research Council (NHMRC)-funded Aus-ALL (grant #254539) and Aus-CBT (grant #404089). Lin Fritschi is supported by NHMRC and WA Cancer Council Fellowships.
Author information
Authors and Affiliations
Corresponding author
Additional information
This work is published under the standard license to publish agreement. After 12 months the work will become freely available and the license terms will switch to a Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License.
Rights and permissions
From twelve months after its original publication, this work is licensed under the Creative Commons Attribution-NonCommercial-Share Alike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Peters, S., Glass, D., Greenop, K. et al. Childhood brain tumours: associations with parental occupational exposure to solvents. Br J Cancer 111, 998–1003 (2014). https://doi.org/10.1038/bjc.2014.358
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bjc.2014.358
Keywords
This article is cited by
-
Childhood cancer and residential proximity to petrol stations: a nationwide registry-based case–control study in Switzerland and an updated meta-analysis
International Archives of Occupational and Environmental Health (2022)
-
Socioeconomic differences in the risk of childhood central nervous system tumors in Denmark: a nationwide register-based case–control study
Cancer Causes & Control (2020)
-
Using Decision Rules to Assess Occupational Exposure in Population-Based Studies
Current Environmental Health Reports (2019)
-
Temporal evolution of brain cancer incidence in the municipalities of Navarre and the Basque Country, Spain
BMC Public Health (2015)