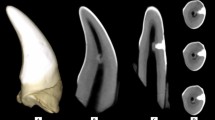
Key Points
-
Provides an overview of important developments in stem cell research and biotherapies.
-
Presents innovative therapeutic approaches for the future of dentistry.
Abstract
The development of new therapies, the biotherapies, can now be envisioned in dentistry thanks to the important development of research in the stem cell and biomaterial fields. Indeed, the recent discovery of stem cells in different tissues of the body, and in particular in the dental pulp, should allow the development of new therapeutic approaches for pulpo-dentinal lesions. In case of moderate pulp injury, the stimulation by biomolecules of pulp stem cells located in the remaining pulp is the approach to be considered. In case of more severe lesions requiring a total removal of pulp tissues, a cell therapy could be considered. The objective of this article is to present these innovative therapeutic approaches for the future of dentistry.
Similar content being viewed by others
Main
Teeth assume an aesthetic and major functional role in the body. They are the keystone of the smile, of the support of the lip and of the dento-alveolar harmony. However, by their anatomical situation they can be the subject of many aggressions such as trauma, cavities, decay or wear. In case of severe trauma or deep cavity the pulp undergoes an irreversible damage. The classic treatment in this case consists of an endodontic eviction of the dental pulp, followed by sealing of the endodontic cavity with a dental material that is supposed to be devoid of biological effect. Dental pulp concomitantly assumes the immune defence, nutrition, sensory and repair functions. A devitalised tooth is devoid of sensitivity and vascularisation. This makes it prone to fractures, often leading to premature tooth loss or infectious complications such as periapical periodontitis. These affections may lead to a bacterial swarming in the body, a door opened to serious diseases, such as infectious endocarditis, nephropathies or rheumatic fever. Medicine today envisioned the development of new therapeutic approaches through biotherapies. This involves the treatment of a damaged organ or tissue by cells (cell therapy), genes (gene therapy) or biomolecules, either combined or not with a biomaterial. Data obtained over the last decade have led to a better understanding of the biological properties of the dental pulp and its cells.1 They now allow considering the development of new biotherapies for some of the dental lesions. Indeed, it has long been known that pulp possess a regenerative potential but the cells and mechanisms involved in the process had remained matter of debate.2 In 2000, the first description of the presence in the pulp of cells endowed with classical mesenchymal stem cell properties3 has allowed understanding of mechanisms involved in pulp repair to progress. Since then, numerous studies have confirmed that a subpopulation of pulp cells, dental pulp stem cells (DPSCs), displayed the classical properties of mesenchymal stem cells that is, self-renewal and the capacity to differentiate towards different lineage, for instance bone, cartilage and adipocytes, similar to bone marrow stromal cells.3,4boxed-text
Highlighting of stem cells in the adult dental pulp has paved the way for research aimed at developing biotherapies that will use the potential of these cells to repair dental damage but also perhaps craniofacial lesions, since DPSCs are also capable of bone repair. To date, depending on the clinical situation, two types of biotherapies can be envisioned in case of injury of the pulpodentinal complex. Upon moderate injury, recruitment and stimulation of the stem cells that persist in the remaining healthy pulp via biomolecules, is the approach to consider. Initially treated by capping with calcium hydroxide and more recently by cements silicate calcium (MTA®, Biodentine®),5,6,7 the addition of biomolecules to these materials could aid the repair process and enable a better treatment of larger pulp injuries. In the case of more severe pulpal lesions, deemed as irreversible and which require a total eviction of the pulp, cell therapy may be considered. It would consist of implantation of an artificial pulp tissue formed from pulp stem cells in the evicted pulp chamber.
Biotherapies by stem cells activating biomolecules
This approach aims at developing biological treatments to restore/regenerate the pulp functions after injury by implanting biomolecules in the pulp, which will enhance its natural repair ability. The extracellular matrix (ECM) proteins of mineralised tissues such as enamel, bone or dentine display protein domains sequence of amino acids with a specific function capable, for some, of inducing or repressing biomineralisation8,9 and for others of inducing pulp cell proliferation10 or of inducing the differentiation of odontoblast precursor cells.9 For now, as the proof of principle of the efficacy of such peptides in the stimulation of dentine repair has been made in animals,9,10,11,12 transfer towards clinical applications requires the establishment of partnerships between academic researchers and industries for financing and organising large scale studies. Such collaborations should allow the development of support materials for biomolecule implantation adapted for the dental practice, but also the initiation of preclinical and clinical studies required to validate and clarify the optimal conditions for their use in human therapy.
Cell biotherapies
Cell therapy aims at restoring the functions of an injured tissue or organ by implanting cells previously amplified in vitro to initiate a repair process. To date, bone marrow stem cells (haematopoietic or mesenchymal) appear as the reference point in this context. While cell therapy approaches have begun to be envisioned in several medical fields, for instance the treatment of major burns, the use of stem cells in odontology is not yet developed. The tooth is an ideal target in the context of cell therapy since it is a relatively isolated organ where the risk of a spreading of implanted cells is restricted as compared to more exposed tissues. Indeed, the uncontrolled swarming of multipotent cells present a risk of ectopic differentiation or, more rarely, of tumour formation. In addition, the dental pulp is a reservoir of mesenchymal stem cells more accessible than the bone marrow. These cells can be obtained without major trauma from, for example, temporary teeth just exfoliated, premolars or molars extracted for orthodontic reasons. Several stem cell populations have been described in the pulp, suggesting the existence, as in the bone marrow, of a hierarchy of progenitors endowed with more or less differentiation potential (Fig. 1).3,13,14,15 Dental pulp stem cells are obtained from the pulp according to isolation procedures that differ between laboratories (enzyme digestion versus explant culture) and selected on the basis of their expression of surface markers common to mesenchymal stem cells (STRO-1+, CD146+...). The stem cells from deciduous teeth and stem cells from human exfoliated deciduous teeth (SHEDs) seem particularly interesting in the context of cell therapy because they present a high proliferation potential and a wider differentiation ability than those isolated from permanent teeth.16,17 Cell therapy of a severely injured pulp would consist of implantation of an artificial tissue formed by a three dimensional matrix seeded with pulp stem cells in the evicted pulp space. The aim would be to maintain the vitality of the tooth due to the formation of a vascularised and innervated neotissue. This approach should evade disadvantages associated with classic endodontic treatment. The development of an artificial pulp tissue however requires:
-
The rapid neovascularisation of the implanted tissue, which is a known prerequisite for tissue maintenance
-
The differentiation of the stem cells in odontoblast-like cells and their deposition of new dentine on the existing dentinal walls of the pulp chamber.
Despite the difficulties and questions still raised by implantation of an artificial tissue in the pulp chamber (enclosed space, severe hypoxia, difficulty in tracking the implanted cell fate by conventional methods of imaging in a highly mineralised tissue), this new therapeutic approach is currently being evaluated by several research teams through the international scientific community and its proof of concept has already been achieved in animals.17,18,19,20 Other uses of DPSCs for cell therapy should also be considered since these multipotent cells are able to engage in osteogenic, chondrogenic, adipocyte, endothelial or neuronal differentiation programmes through specific inductions.1 This ability places the DPSCs as alternative agents to bone marrow mesenchymal stem cells and may extend their therapeutic potential to other tissues than those of the tooth.1 Several pre-clinical and clinical trials involving pulp stem cells are underway.20 The first published trial showed that implantation of collagen sponges seeded with dental pulp cells in a mandibular bone defect allowed a bone to heal faster and more extensively than in the untreated control portion of the mandible.21
Conclusion
Treatment of pulpo-dentinal lesions by biotherapies involving either an activation of pulp stem cells via biomolecules or by direct implantation of such cells opens up innovative therapeutic areas. Depending on the choice of governmental instances (autograft versus allograft) and the position of ethics committees, the therapeutic use of adult pulp stem cells could be developed in humans from the establishment of cell banks potentially managed either by hospital or university centres or other public instances.
References
Huang G T, Gronthos S, Shi S . Mesenchymal stem cells derived from dental tissues vs. those from other sources: their biology and role in regenerative medicine. J Dent Res 2009; 88: 792–806.
Goldberg M, Smith A J . Cells and extracellular matrices of dentin and pulp: a biological basis for repair and tissue engineering. Crit Rev Oral Biol Med 2004; 15: 13–27.
Gronthos S, Mankani M, Brahim J, Robey P G, Shi S . Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci U S A 2000; 97: 13625–13630.
Gronthos S, Brahim J, Li W et al. Stem cell properties of human dental pulp stem cells. J Dent Res 2002; 81: 531–535.
Okiji T, Yoshiba K . Reparative dentinogenesis induced by mineral trioxide aggregate: a review from the biological and physicochemical points of view. Int J Dent 2009; 464: 280.
Dammaschke T, Stratmann U, Wolff P P, Sagheri D, Schäfer E . Direct pulp capping with mineral trioxide aggregate: an immunohistologic comparison with calcium hydroxide in rodents. J Endod 2010; 36: 814–819.
Torabinejad M, Parirokh M . Mineral trioxide aggregate: a comprehensive literature review-part II: leakage and biocompatibility investigations. J Endod 2010 36: 190–202.
Boukpessi T, Gaucher C, Leger T et al. Abnormal presence of the matrix extracellular phosphoglycoprotein-derived acidic serine-and aspartate-rich motif peptide in human hypophosphatemic dentin. Am J Pathol 2010; 177: 803–812.
Chaussain C, Eapen A S, Huet E et al. MMP2-cleavage of DMP1 generates a bioactive peptide promoting differentiation of dental pulp stem/progenitor cell. Eur Cell Mater 2009; 18: 84–95.
Six N, Septier D, Chaussain-Miller C, Blacher R, DenBesten P, Goldberg M . Dentonin, a MEPE fragment, initiates pulp-healing response to injury. J Dent Res 2007; 86: 780–785.
Goldberg M, Lacerda-Pinheiro S, Jegat N et al. The impact of bioactive molecules to stimulate tooth repair and regeneration as part of restorative dentistry. Dent Clin North Am 2006; 50: 277–298.
Goldberg M, Lacerda-Pinheiro S, Priam F et al. Matricellular molecules and odontoblast progenitors as tools for dentin repair and regeneration. Clin Oral Investig 2008; 12: 109–112.
Gronthos S, Zannettino A C, Hay S J et al. Molecular and cellular characterisation of highly purified stromal stem cells derived from human bone marrow. J Cell Sci 2003; 116: 1827–1835.
Kerkis I, Kerkis A, Dozortsev D et al. Isolation and characterization of a population of immature dental pulp stem cells expressing OCT-4 and other embryonic stem cell markers. Cells Tissues Organs 2006; 184: 105–116.
Laino G, d'Aquino R, Graziano A et al. A new population of human adult dental pulp stem cells: a useful source of living autologous fibrous bone tissue (LAB). J Bone Miner Res 2005; 20: 1394–1402.
Miura M, Gronthos S, Zhao M et al. SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci U S A 2003; 100: 5807–5812.
Cordeiro M M, Dong Z, Kaneko T et al. Dental pulp tissue engineering with stem cells from exfoliated deciduous teeth. J Endod 2008; 34: 962–969.
Suzuki T, Lee C H, Chen M et al. Induced migration of dental pulp stem cells for in vivo pulp regeneration. J Dent Res 2011; 90: 1013–1018.
Galler K M, Hartgerink J D, Cavender A C, Schmalz G, D'Souza R N . A Customized Self-Assembling Peptide Hydrogel for Dental Pulp Tissue Engineering. Tissue Eng Part A 2012; 18: 176–184.
Nakashima M, Iohara K . Mobilized dental pulp stem cells for pulp regeneration: initiation of clinical trial. J Endod 2014; 40 (4 Suppl): S26–32.
d'Aquino R, De Rosa A, Lanza V, Tirino V, Laino L, Graziano A, et al. Human mandible bone defect repair by the grafting of dental pulp stem/progenitor cells and collagen sponge biocomplexes. Eur Cell Mater. 2009; 18: 75–83.
Acknowledgements
The authors would like to thank Claudie Damour-Terrasson, publishing director of the Groupe Information Dentaire, Paris, France, for the authorisation of translation and publication of the series in the BDJ and Dr Avijit Banerjee for his support.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaussain, C., Poliard, A. Minimal intervention dentistry: part 8. Biotherapies for the dental pulp. Br Dent J 216, 619–621 (2014). https://doi.org/10.1038/sj.bdj.2014.450
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2014.450