Abstract
Engraftment of clonal hematopoietic precursor cells from patients with myelodysplastic syndrome (MDS) in immunodeficient mice has been difficult to achieve by intravenous (i.v.) injection. We used i.v. coadministration of the human marrow stroma cell line HS27a with CD34+ MDS cells in Nod.cg-Prkdcscid Il2rgtm1wjll (NSG) mice to provide signals that would facilitate engraftment. Hematopoietic cells from 24 MDS patients were transplanted. Cells from all patients were engrafted, and engraftment was documented in 44 of 46 evaluable mice (95%). Immunohistochemistry revealed human HS27a stroma colocalizing with human hematopoietic cells in mouse spleens. Human CD34+ precursors harvested from marrow and spleen of primary murine recipients, when combined with HS27a cells, were also engrafted successfully in secondary NSG recipients, showing persistence of the original clonal characteristics. This observation supports the concept that clonal markers were present in long-term repopulating cells. We suggest that HS27a stroma cells ‘traveled’ in direct contact with hematopoietic precursors and enabled their propagation. An essential signal for engraftment appears to be CD146, which is prominently expressed on HS27a cells. This xenotransplantation model will allow to further dissect signals that control engraftment of MDS cells and should be amenable to in vivo treatment studies.
Similar content being viewed by others
Introduction
Propagation of clonal CD34+ cells derived from the marrow of patients with myelodysplastic syndrome (MDS), in contrast to cells from patients with acute myeloid leukemia (AML),1, 2 has proven difficult in vitro and has met with limited success in xenogeneic transplant models in vivo.3, 4, 5, 6 Extensive research has shown the central importance of the marrow microenvironment in supporting hematopoiesis, and vascular and stromal stem cell niches have been described.7, 8 Importantly, contact with components of the microenvironment not only supports normal hematopoiesis, but also protects malignant cells, for example, in patients with AML, against the effects of cytotoxic therapy.9, 10 Additional data suggest that marrow stroma cells in patients with MDS show abnormal DNA methylation11 and are functionally altered.12, 13 Furthermore, Raaijimarkers et al.8 showed in a murine model that deletion of Dicer 1 in osteoblast progenitors resulted in the development of dysplastic hematopoiesis. Although the clonality of hematopoietic cells in this setting has been challenged, the data support the concept of a crucial role of the microenvironment in the development and propagation of dysplastic hematopoiesis.8 Mesenchymal/stromal cells contribute to the survival and replication of hematopoietic stem/progenitor cells (HPC) by adhesion molecules, by way of extracellular matrix generation and secretion of survival factors.14, 15 That stroma/mesenchymal cells provide support to clonal MDS cells was also shown by Muguruma et al.16 who injected mesenchymal cells together with HPCs, directly into the marrow cavity of NOD/SCID/IL-2Rγnull mice. Particularly, HPCs derived from MDS patients with clonal abnormalities of chromosome 7 could be propagated long term.16
We hypothesized, therefore, that the coadministration of certain marrow stromal cells with MDS marrow-derived HPCs would support the survival of those HPCs. In our previous studies, we achieved low-level engraftment of clonal MDS-derived HPC in NOD/SCID-β2-microglobulin-deficient mice when injected directly into the marrow cavity together with a mix of two stroma cell lines, HS5 and HS27a.3 However, HS5 and HS27a cells, although derived from the same healthy donor, differ substantially in their gene expression profiles and function.17, 18 In fact, we observed prominent apoptosis in HPCs cocultured with HS5 cells, whereas HPCs that remained in contact with HS27a cells remained viable and were propagated. Thus, enhanced engraftment and survival of MDS-derived HPCs might be obtainable if supported by coadministration of HS27a cells.
We conducted the present study to determine whether intravenous (i.v.) injection of stroma and HPCs, similarly, would result in sustained engraftment, thereby addressing not only the survival and propagation of clonal MDS HPCs, but also the question of stroma ‘migration’. Results in Nod.cg-Prkdcscid Il2rgtm1wjll (NSG) mice show that the i.v. coadministration of HS27a cells with HPCs from patients with MDS allowed for engraftment of clonal CD34+ cells of any karyotype. The data further show that HS27a stroma cells were localized with human hematopoietic cells in mouse spleen and marrow. Moreover, clonal MDS cells harvested from the primary recipients were transplanted successfully into secondary recipients. No such success was achieved with unmodified sister cell line HS5.
Taken together, the data indicate that HS27a stroma enabled the engraftment of CD34+ clonal MDS cells in NSG mice, apparently by providing an essential component for the delivery and support of MDS cells in mouse marrow and spleen.
Materials and methods
Patients
MDS cells were obtained from marrow aspirates or (in one case) from peripheral blood (PB) of patients referred to the Fred Hutchinson Cancer Research Center (FHCRC) for consultation or therapy. All patients had given informed consent to participate in research studies as required by the Institutional Review Board of the FHCRC.
Primary cells and cell lines
Bone marrow was aspirated from 23 patients into preservative-free heparin-containing syringes under local lidocaine anesthesia; PB was obtained from one patient by leukapheresis. Bone marrow mononuclear cells and PB cells were separated by Ficoll–Hypaque gradient centrifugation and suspended in RPMI 1640 medium containing 10% heat-inactivated fetal bovine serum until use, or were subjected to magnetic-activated cell sorting to purify CD34+ cells, according to the manufacturer’s protocol (Miltenyi Biotec, Auburn, CA, USA).
All marrow samples were characterized in regard to clonal cytogenetic abnormalities using metaphase G banding, fluorescent in situ hybridization (FISH) or both in the clinical laboratory of the Seattle Cancer Care Alliance/FHCRC.
The human marrow stroma cell lines HS5 and HS27a, derived from the marrow of a healthy volunteer and immortalized by transduction with human papilloma virus E6/E7 constructs,18 were a gift from Dr Torok-Storb (FHCRC, Seattle, WA, USA). These stroma cells were propagated and used for experiments between passages 8 and 24 as recently described.13 KG1a cells (originally derived from a patient with AML) were obtained from American Type Culture Collection (Manasses, VA, USA).
Transplantation and post-transplant studies
Primary transplant recipients
NSG mice, 6–8 weeks of age, were purchased from Jackson Laboratories (Bar Harbor, ME, USA) and maintained according to standard laboratory procedures, including sterile chow and water. Based on dose optimization studies, mice were irradiated with 275 cGy from a 137Cs source, and after 2 h, the mice were injected i.v. with fresh bone marrow mononuclear cells, sorted CD34+ cells or PB mononuclear cells (5 × 106 or 10 × 106 cells per animal), combined with stroma cells, either HS5 or HS27a. The ratio of hematopoietic MDS cells to stroma cells was 10:3 (or 5:1.5). Whenever possible, MDS cells from each patient were injected into at least two recipient mice. In additional experiments, KG1a cells were transplanted.
Fine needle aspirates from the femur were scheduled at 4, 8 and 12 weeks. However, if mice appeared ill they were killed, and studies were carried out at autopsy at the corresponding time points. Spleen and marrow were harvested for in vitro studies and for transplantation into secondary recipients. All experiments were performed in compliance with the guidelines of the Institute for Animal Studies and approved by the Institutional Animal Care and Use Committee of the FHCRC.
Secondary transplant recipients
For transplantation into secondary recipients, bone marrow and spleen cells were collected from the three primary NSG recipients and sorted on the basis of expression of human CD45 (containing variable numbers of CD34+ cells). FACS-sorted human CD45+ cells (purity>98%) were mixed with HS27a cells (10:3) and injected i.v. into three secondary recipients irradiated with 275 cGy. Post-transplant care and analysis were as for primary recipients.
Antibodies
Antibodies for flow cytometry
Antibodies to human CD34 (catalog no. 348057), human CD45 (catalog no. 555482 and catalog no. 555485), CD33 (catalog no. 555626), CD19 (catalog no. 555415), CD14 (catalog no. 340585), CD3 (catalog no. 341091), isotype controls (catalog no. 555743, catalog no. 555748 and catalog no. 550931, respectively), and human CD146 (catalog no. 550315) and isotype control (catalog no. 554680) were purchased from Becton-Dickinson (BD; Sunnyvale, CA, USA). Antibody to bromodeoxyuridine (BrdU) was provided with the BrdU Flow kit (catalog no. 552598; BD). Anti-mouse CD45 (catalog no. 553081) was purchased from BD.
Antibodies for immunohistochemistry
Anti-human CD45 (clone 4H11) was purchased from Biolegend (San Diego, CA, USA) and clone 2B11 (+PD7/26) from DAKO Cytomation (DAKO, Glostrup, Denmark); anti-human ICAM1 (catalog no. MHCD5401) from Invitrogen (Carlsbad, CA, USA) and (catalog no. 4915) from Cell Signaling (Danvers, MA, USA); anti-human CD34 (clone QBEnd-10) and anti-human CD146 (clone N1238) from Leica Microsystems (Buffalo Grove, IL, USA); and anti-GFP (green fluorescent protein) (A11122) from Invitrogen.
Antibodies for western blotting
Rabbit anti-human CD146 antibody (catalog no. 3376-1) was purchased from Epitomics Inc. (Burlingame, CA, USA) and secondary goat anti-rabbit antibody (sc-362272) from Santa Cruz Biotechnology Inc. (Santa Cruz, CA, USA).
Documentation of engraftment
Flow cytometric analysis
To determine engraftment of human MDS cells, mouse spleen and marrow cells were analyzed by flow cytometry as described.3, 4 Briefly, cells were stained with anti-human CD34 and anti-human CD45 (or corresponding isotype controls). At least 30 000 events (cells) were sampled, and the proportions of CD45+ cells (containing CD34+ cells) in comparison to isotype-labeled controls were determined. In several experiments, the bulk of human CD45 cells from primary transplant recipients was used for transplantation into secondary recipients.
Cytogenetic analysis
Marrow and spleen cells harvested from transplanted mice were suspended in phosphate-buffered saline (containing 0.5% bovine serum albumin), and FISH was performed for the previously identified markers.19 Briefly, FACS-sorted human CD45+ cells (containing CD34+) were placed on polylysine-coated glass slides, fixed with 3.7% formaldehyde and permeabilized. Human clonal cells were identified with probes specific for the chromosomal abnormalities present in the respective patients. Commercial probes were used according to the manufacturer’s instructions (Supplementary Table S1). Following FISH, the tissue samples were mounted in Tris-buffered 90% glycerol (pH 8.8, DABCO antifade, Sigma, St Louis, MO, USA) and imaged on an Olympus IX71 microscope ( × 100/1.40, PL Apo oil objective (Scientific Imaging Facility, FHCRC)) equipped with a cooled Photometrics Coolsnap HQ scientific-grade CCD camera using Deltavision SoftWorx software V3.5.1 (Applied Precision Inc., Issaquah, WA, USA). FISH spots were counted on two-dimensional projections of deconvolved image stacks (SoftWorx V3.5.1). Images were transferred to Adobe Photoshop CS4 (Adobe Systems, San Jose, CA, USA) and analyzed.
Localization of KG1a cells, primary MDS HPC and stroma cells in transplanted mice
KG1a cells
To test for the localization of injected hematopoietic cells and stroma, in a first series of short-term experiments, KG1a cells and HS27a (or HS5) stroma cells were coinjected i.v. into mice. After 72 h, spleen and bone marrow samples were harvested and snap-frozen in O.T.C buffer. Sections were then examined for localization of KG1a cells (labeled with Alexa Fluor 647-conjugated anti-human CD45 antibody) and possible colocalization with HS27a (or HS5) stroma cells, identified by expression of human ICAM1 as determined by FITC-conjugated specific antibody, using a Zeiss LSM 51 microscope (Zeiss, Jena, Germany). KG1a cells do not express ICAM1, even after extended culture. Slides were rinsed with phosphate-buffered saline and treated with anti-fading Vectashield+DAPI mounting medium (Vector Laboratories, Burlingame, CA, USA). In additional experiments using GFP-transfected HS27a cells, sections were exposed to anti-GFP antibody for analysis. Images were acquired by confocal microscopy with a Plane Apochromat × 63 NA14 objective.
Primary MDS cells
Bone marrow and spleen cells from NSG mice that had been coinjected with MDS marrow and HS27a (or HS5) stroma cells were harvested at the time of killing of the mice at 12–13 weeks. Tissue was fixed in 10% formalin buffer for 72 h, and stained with anti-human ICAM1-FITC antibody and anti-human CD45 Alexa Fluor 647 (FHCRC Experimental Pathology facility). Results were analyzed using confocal fluorescent microscopy (Zeiss LSM 510, Scientific Imaging Facility, FHCRC).
In addition, serial sections of spleens and marrows were incubated with anti-human CD34, CD45 and ICAM1 antibody, separately, and analyzed by microscopy (Nikon E800, Scientific Imaging Facility, FHCRC)
To further investigate the engraftment of human stroma cells, we also determined expression of CD146, a niche marker that is not expressed on HPC.20, 21 In ancillary experiments, the CD146 antibody was tested on human and mouse control samples to ascertain specificity for human cells. Additional experiments verified that the anti-CD146 antibody did not detect endogenous immunoglobulins in the immunodeficient mice.
Tissues were formalin fixed for 72 h and paraffin-embedded (8 h protocol), and 4-μm sections were cut, deparaffinized and rehydrated in Tris-buffered saline containing 0.1% Tween-20 (TBS-T). Antigen retrieval used a Black and Decker steamer (Towson, MD, USA) with a 20-min exposure in preheated Trilogy buffer (Cell Marque, Rocklin, CA, USA) followed by 20-min cooling. Slides were rinsed three times in wash buffer and subsequent staining was performed at room temperature using a DAKO Autostainer (Carpinteria, CA, USA). Endogenous peroxide was blocked with 3% H2O2 for 8 min, followed by avidin/biotin blocking (Biocare Medical, Concord, CA, USA). Slides were then placed for 10 min in 15% horse serum (Vector Laboratories) in TBS containing 1% bovine serum albumin. CD146 antibody was diluted in a ratio of 1:50 (0.42 μg/ml), incubated on the tissue for 60 min and washed with wash buffer. Antibody staining was detected using biotinylated horse anti-mouse antiserum (BA-2000, Vector Laboratories) at 1:200 for 30 min, followed by horseradish peroxidase-labeled streptavidin (016-030-084, Jackson ImmunoResearch, West Grove, PA, USA) at a dilution of 1:2000 for 30 min. Staining was visualized with 3,3′-diaminobenzidine (DAKO) for 8 min, and slides were counter-stained with a 1:4 dilution of hematoxylin (DAKO) for 2 min. An irrelevant, concentration-matched, isotype-stained slide was evaluated for background staining for each tissue sample.
Stroma cells
To determine organ localization of stroma cells, 5 × 106 HS27a (or HS5) cells were injected i.v. (without HPC) into NSG mice irradiated with 275 cGy. In addition to spleen and liver, lungs and long bones were collected from mice killed at 48 h, 96 h or 7 days. Tissue sections were stained with anti-human ICAM1 antibody, or in the case of GFP-transfected cells, with anti-GFP antibody, and results were analyzed by microscopy.
Expression of CD146 on HS27a stroma cells
To further characterize the potential role of CD146 in HS27a function, the human small interfering RNA (siRNA) set for CD146 knockdown (Qiagen, Valencia, CA, USA), was used to transfect HS27a cells as previously described.13 A lentiviral vector to overexpress CD146 was purchased from Open Biosystem (Lafayette, CO, USA) and used as described.13 To determine the success of CD146 inhibition by knockdown or, conversely, overexpression, total cell lysates were prepared by sonication for 4 min and fractions were cleared by centrifugation at 20 000 g for 10 min. Protein concentrations were quantified by bicinchoninic acid assay (Pierce Biotechnology Inc., Rockford, IL, USA), and equal amounts of protein (20 μg) from each lysate were diluted in Laemmli sodium dodecyl sulfate (SDS) sample buffer and resolved by electropheresis on 4–12% Bis-Tris pre-cast NuPage gels (Invitrogen) in running buffer (50 mM 2-(N-morpholino) ethane sulfonic acid, 50 mM Tris base, 0.1% SDS and 1 mM EDTA) as per the manufacturer's instructions. Protein was then transferred to polyvinylidene difluoride membranes for immunoblotting. The membranes were blocked in 5% nonfat dry milk diluted in TBS-T buffer for 1 h at room temperature and incubated overnight at 4 °C in 5% nonfat dry milk/TBS-T containing rabbit anti-human CD146 antibody. Secondary goat anti-rabbit antibody (1:2000) conjugated to horseradish peroxidase was used for enhanced chemoluminescence (Pierce Biotechnology), and membranes were exposed to film.
BrdU uptake of HPC in coculture with stroma cells
CD34+cells from healthy donors or from patients with MDS, as well as CD34+KG1a cells were cocultured with unmodified HS27a (or HS5) cells, with HS27a stroma cells with CD146 knockdown or with HS5 cells overexpressing CD146. After 48 h, CD34+ cells were sorted and exposed to BrdU for 45 min. The extent of BrdU uptake was determined by flow cytometry, following labeling of cells with anti-BrdU antibody as per the BrdU Flow kit (catalog no. 552598; BD, Franklin Lakes, NJ, USA).
Statistical analysis
Student’s t-test was used to compare continuous variables between two groups.
Results
Intravenous coadministration of HS27a, but not HS5 stroma cells, supports engraftment of clonal human CD34+ MDS cells in NSG mice
Previous studies had shown that the combined coinjection of two stroma cell lines, HS5 and HS27a, together with MDS marrow cells into the marrow cavity of immunodeficient mice, was associated with an inconsistent graft-enhancing effect.3 As HS5 and HS27a stroma cells differ markedly in regard to gene expression, surface characteristics and function,17, 18, 21, 22 we aimed at determining the impact of HS5 and HS27a individually.
Human HPC from all 24 patients (100%) when injected together with HS27a cells, engrafted in at least one mouse, and only two mice failed to show evidence of engraftment of human cells (engraftment success 95%) (Table 1; Figures 1a–d). Very few hematopoietic cells were detected in mice coinjected with HS5 stroma and marrow cells from the same donors as mice that were coinjected with HS27a cells (Figures 1a and b and d). No hematopoietic cells were detectable in mice not coinjected with stroma (Supplementary Figure S1). Human hematopoietic cells isolated from transplanted mice were CD45+ and contained a population of CD34+ cells (Figure 1c).
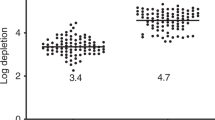
Human MDS marrow cells in spleen and bone marrow of mice coinjected with MDS marrow and HS27a or HS5 stroma cells, respectively. (a and b) Spleen and marrow cells were harvested at weeks 12 or 13 after transplantation and analyzed by flow cytometry. The percentages of cells in the respective quadrants are indicated. The flow analysis shows results obtained with cells from patients 14 and 18, respectively (Table 1). (c) Immunohistochemical staining of spleen and femur from NSG mouse engrafted with MDS marrow. Labeling with anti-human CD34 and CD45 antibodies appears as brown signals. (d) Summary of engraftment of human HPC in four experiments comparing the effects of HS5 and HS27a (box plot: lowest, 1st quartile, median, 3rd quartile, highest value). BM, bone marrow (Comparison by Student’s t-test).
As shown in Figure 2a, engraftment of CD34+ cells carrying the patient’s clonal chromosomal marker was readily documented. The highest proportions of human cells observed in spleen and marrow were 73.2% and 30.3%, respectively, comparable to results seen with intramedullary injection.3, 6, 23 We also determined the engraftment ratio of clonal versus non-clonal cells for four MDS marrows. The proportion of clonal cells tended to be higher in marrows than in spleens (Table 2). The spectrum of detectable human cells, determined in mice transplanted from three different patients, included CD34+, CD33+, CD19+, CD14+ and CD3+ subpopulations (Table 1a).
Extent of engraftment and clonality of human MDS marrow cells in secondary NSG recipients: analysis by flow cytometry and FISH. (a) FISH detection of del 5q (two green and one red signal identify cells with 5q-), trisomy 8 (three red signals indentify cells with +8), del 20q (two red and one green signal indentify cells with 20q-) and a CBFB/MYH11 dual fusion translocation (one green and one red merging indentifies a cell with the CBFB/MYH11 translocation). (b) Flow cytometric profiles of marrow cells recovered from secondary NSG recipients 13 weeks after transplantation of CD34+ human cells that had been harvested from the primary recipients 12 weeks after the original transplant. Human CD45+ and CD34+ cells are present. The percentages of cells in the respective quadrants are indicated (human cells were derived originally from patients 14 and 18, respectively; Table 1). (c) FISH shows del 5q (two green and one red signal identifying cells with 5q-) in the spleen of the secondary recipient. BM, bone marrow.
Engraftment of clonal CD34+ MDS cells in secondary NSG recipients
To determine whether clonal MDS cells that engrafted in primary NSG recipients had long-term self-renewal potential, we carried out transplants into secondary recipients. Cells were harvested from the bone marrows and spleens of two primary recipient NSG mice (transplanted with cells from the same patient and killed at 12–13 weeks), human CD45+ cells (Supplementary Figure S2) were pooled and 7–10 × 106 human CD45+ cells, containing 4–5% CD34+ cells, were injected together with HS27a cells (as used in primary recipients) into two secondary recipients. Overall, three such pairs of secondary recipient mice were injected with cells harvested from six primary recipients. Spleens and marrows of all secondary recipients, harvested at 12–13 weeks after injection, contained human CD45+/CD34+ cells (Figure 2b), which expressed the FISH marker originally identified in the patient (Figure 2c). Thus, those patient cells had been propagated over 25–26 weeks in mice. These data support the concept that the clonal karyotype was expressed in cells with long-term repopulating capacity.
Organ localization of human stroma and hematopoietic cells
Coinjection of stroma with hematopoietic cells
As illustrated in Figure 3a, in NSG mice injected with KG1a cells (which express both CD45 and CD34), labeled with anti-human CD45-Alexa Fluor 647 and HS27a cells, identified by labeling with antibody to human ICAM1, the two cell populations colocalized. In contrast, no colocalization of KG1a with coinjected HS5 cells was detectable (data not shown).
Localization and colocalization of human HS27a stroma and hematopoietic cells in murine marrow (BM) and spleen. (a) Confocal microscopy showing colocalization of KG1a cells and HS27a stroma in fresh frozen sections of spleen (original magnification: × 40): HS27a cells are labeled with FITC-conjugated anti-human ICAM1 antibody. Red indicates human CD45+ KG1a cells, whereas blue shows nuclear staining with DAPI (both murine and human nuclei stain with DAPI). The right lower panel represents the merged picture. Superimposition of the FITC signals of the anti-ICAM1 antibody and the Alexa 647 signals of the CD45 antibody results in a yellow hue of signals. White arrows indicate examples of staining for colocalizing HS27a stroma and CD45+ cells. (b) Spleen sections (formalin fixed) stained with anti-human ICAM1 antibody (green) and anti-CD45 antibody (red), merged in the right panel (original magnification= × 40). The lower panels show isotype controls. (c) Immunohistochemical determination of the distribution of primary MDS cells (labeled with anti-human (h) CD34 and CD45 antibodies) and HS27a stroma (labeled with anti-human ICAM1 antibody) in the bone marrow and spleen of NSG mice. White arrows indicate identical coordinates on sequential sections (section distance=4 μm; orignal magnification= × 40). (d) Immunohistochemical staining of formalin-fixed spleen sections labeled with anti-human CD146 antibody (original magnification= × 40). Dark brown identifies 3,3′-diaminobenzidine chromagen linked to the CD146 antibody, identifying HS27a cells; blue represents counter staining with hematoxylin. Samples from two mice injected with primary CD45+ MDS cells from two different patients. Each figure represents one example of 2–4 similar experiments. (e) Flow cytometric analysis of bone marrow (BM) and spleen cells harvested from mice transplanted with human MDS marrow without coinjection of stroma (without stroma) or coinjected with unmodified HS5 or HS27a stroma. Cells positive for human ICAM1and CD146 (typical for HS27a stroma; see also Figure 4a and Supplementary Figure S4b) were identified in marrow and spleen from mice injected with HS27a.
We next performed dual immunofluorescent staining of sections of spleen obtained at the time of autopsy of mice transplanted with primary MDS cells. As illustrated in Figures 3a–c, human HPC (labeled with anti-human CD45 antibodies) colocalized with human HS27a stroma (labeled with anti-human ICAM1). No colocalization of human CD45+ cells with HS5 stroma was detectable (data not shown).
Considering the possibility that ICAM1 had been induced on transplanted HPC in response to undefined signals rather than identifying colocalizing HS27a cells, we carried out additional experiments using antibodies to CD146, which is not expressed on HPC, but has been recognized as a marker of marrow niche cells.20 As illustrated in Figure 3d, staining for human CD146 was present in the spleens of NSG mice injected concurrently with human HS27a stroma and HPC. CD146 staining was prominent in some mice (as shown), whereas in others only infrequent CD146-positive cells were identified.
Efforts to label human CD146 and CD45 (or CD34) simultaneously were unsuccessful: First, the CD146 antibody failed to conjugate with the detection reagent using either the FHCRC Mouse-on-Mouse kit or the commercially available DAKO ARK kit. Second, amplification of the signal with horse-anti-mouse secondary antibody resulted in concurrent amplification of endogenous immunoglobulin, which completely obliterated the image. Results were similarly unsuccessful with a second anti-CD146 antibody purchased from Qiagen.
Being unable to effectively colabel cells, we carried out the following experiment: four mice were injected with MDS marrow cells (from one of two different donors) and either HS5 or HS27a stroma. After killing the mice, marrow and spleen cells were labeled with antibodies to human CD146 and human ICAM1, both of which are expressed on HS27a, and analyzed by flow cytometry. Figure 3e shows one example (mice injected with cells from patient 24 and killed at 4 weeks). In the mouse coinjected with HS27a stroma both marrow and spleen contained cells staining for human CD146 and ICAM1. In contrast, only a rare cell staining for human CD146 and ICAM1 was identified in the marrows and spleens of mice coinjected with HS5 stroma.
Injection of stroma without hematopoietic cells
The localization of HS27a cells to the spleen raised questions as to the migration of those cells. To determine the distribution of stroma cells in vivo when administered without HPC, we injected 5 × 106 HS27a cells (unmodified or transfected with a GFP construct) i.v. and harvested spleen, liver, lung and long bones from mice at 48 h, 96 h or 7 days. As shown in Supplementary Figure S3, cells expressing human ICAM1 or GFP were present, as expected, in the lung (at 48 and 96 h), but not in spleen, liver or long bones. At 7 days, no human ICAM1- or GFP-positive cells were detectable in any organ. The data suggest that in the coinjection experiments, HS27a stroma cells reached the spleen traveling as partners of hematopoietic cells, similar to the recently described model of fibroblasts traveling with metastatic cells.24 This observation also suggests a strong interaction of hematopoietic cells with HS27a (but not to HS5) stroma, possibly via CD146.
Inhibition of CD146 expression in HS27a stroma and response of hematopoietic cells in stroma coculture
The discrepant results achieved with HS27a and with HS5 stroma likely reflect the substantial differences in gene expression and function between these two cell lines.17, 18, 25 Because of the documented functional role of CD146 in marrow niches described in other models,20, 26 we examined the expression of CD146 and showed significantly higher levels of CD146 on HS27a than on HS5 cells (Figure 4a), confirming the report by Pillai et al.21 Knockdown with four CD146-specific siRNA constructs reduced CD146 expression in HS27a cells to levels comparable to those in HS5 cells (Figure 4a). Conversely, stable overexpression of CD146 in HS5 cells using lentiviral vectors enhanced CD146 expression to levels comparable to those in HS27a cells (Figure 4a).
Effect of CD146 expression in stroma on BrdU uptake by cocultured CD34+ hematopoietic cells in vitro, and on engraftment of CD34+ MDS marrow cells in NSG mice in vivo. (a) Flow cytometric analysis showed prominent expression of CD146 on HS27a, but not on HS5 stroma. Knockdown (KD) of CD146 in HS27a cells using four different siRNAs (no. 1–4 in comparison with a scrambled siRNA) reduced CD146 expression to levels comparable to those in HS5 cells. Conversely, overexpression of CD146 (over-CD146) in HS5 cells using a lentiviral construct (pLOC vector, over-CD146, in comparison to a scrambled vector (SCR)) increased CD146 expression to levels comparable to those in unmodified HS27a cells (see also Supplementary Figure S4b; upper row). (b) BrdU uptake by hematopoietic cells after coculture with either unmodified HS27a stroma wild type (WT), HS27a cells with KD of CD146 (146 KD), or HS5 cells overexpressing CD146 (over-CD146), in comparison with unmodified (WT) HS5 cells. BrdU uptake was highest in KG1a cells, followed by MDS-derived CD34+ cells and CD34+ cells from healthy donors. Results of coculture with HS27a cells with KD of CD146 approached those with unmodified HS5 cells, whereas, conversely, BrdU uptake in coculture with CD146 overexpressing HS5 cells did not differ significantly from that in coculture with unmodified HS27a cells (Student’s t-test; mean±s.e.m. of three experiments ). (c) Engraftment of CD45+ marrow cells from two patients with RAEB-2 and RAEB-1, respectively, coinjected with unmodified HS5 stroma (HS5), unmodified HS27a stroma (HS27a) or HS5 cells overexpressing CD146 (HS5-CD146), in marrow and spleen of NSG mice, determined at 5–7 weeks after transplantation. The table shows, in addition, the proportions of human clonal and non-clonal CD34+cells (from patient 23) in mouse marrows and spleens after coinjection with HS27a and HS5-CD146 cells (day 35), respectively. Additional data on FISH and flow cytometric analysis, as well as immunohistochemical analysis of ICAM1 and CD146 expression are shown in Supplementary Figure S4. RAEB-1 or 2, refractory anemia with excess blasts 1 or 2, respectively; NBM, normal bone marrow.
Unmodified and modified HS27a and HS5 cells were then tested in coculture with primary CD34+ cells from healthy donors, patients with MDS or KG1a cells for their effect on proliferation as measured by BrdU uptake. At 36–48 h, primary CD34+ cells or KG1a cells were sorted from the cocultures, exposed to BrdU for 45 min, and BrdU uptake was determined by labeling with anti-BrdU antibody and flow cytometric analysis. As shown in Figure 4b, BrdU uptake by hematopoietic cells that had been cocultured with HS27a cells with CD146 knockdown was decreased significantly (in comparison to unmodified HS27a cells) and was comparable to values obtained in cells cocultured with unmodified HS5 cells. Conversely, HS5 cells overexpressing CD146 facilitated BrdU uptake in cocultured hematopoietic cells to an extent similar to that seen in unmodified HS27a cells.
On the basis of those findings, we coinjected HS5 cells overexpressing CD146 with marrow cells from two patients with RAEB-2 and RAEB-1, respectively, into two NSG mice each. At 6 weeks, engraftment was determined (by flow cytometry) and compared with results in mice injected with marrow from the same patient, but in combination with unmodified HS27a or unmodified HS5 cells. As shown in Figure 4c (and Supplementary Figure S4), engraftment with CD146 overexpressing HS5 cells was increased significantly in comparison with results with unmodified HS5 cells in all mice studied, a pattern identical to that achieved with HS27a cells. Notably, HS5-CD146 stroma cells were detectable by flow analysis (Figure 3e and Supplementary Figure S4b) in support of stroma cells traveling with hematopietic cells. These results are consistent with a role of CD146 expression in facilitating the engraftment and propagation of cotransplanted clonal hematopoietic cells.
Discussion
In vivo models of human diseases offer many advantages over in vitro studies by allowing longitudinal observations and possible treatment interventions in a ‘physiological’ environment. Numerous human solid tumors have been engrafted in immunodeficient mice. Similarly, cells from patients with AML are generally propagated successfully.27 A xenogeneic human in vivo model of MDS has been more difficult to establish,4, 5, 6 whereas several autologous murine models have been described.3, 16 Recently, Muguruma et al.1, 2, 16 showed, similar to our previous observations,3 propagation of human MDS-derived cells, in particular from patients with clonal chromosome 7 abnormalities, when injected along with mesenchymal cells directly into the marrow cavity.
In the present study, we show sustained engraftment of human MDS clones after i.v. injection. Engraftment was independent of the patient’s karyotype. Strikingly, engraftment success was achieved by coinjection of one human stroma cell line, HS27a, but not by another, HS5, derived from the same healthy donor,18 a reflection of the heterogeneity of cells forming the marrow microenvironment. Strikingly, however, overexpression of CD146, a previously identified niche marker,20 that facilitates homing and renewal of HPCs26 modified the function of HS5 cells, such that their in vitro effects and in vivo engraftment-facilitating capacity were similar to those observed with HS27a cells.
HS27a stroma cells, identified by labeling with anti-human ICAM1 and CD146 antibodies, were present in the spleens of mice coinjected with a mix of HS27a and hematopoietic cells, whereas they were found only in the lungs when injected alone, consistent with the concept that stroma cells do not migrate. However, recent data suggest that under certain conditions fibroblast-like stroma cells are able to ‘travel.’ In a model of cancer metastasis, Duda et al.24 showed that fibroblast-like stroma cells accompanied tumor metastases and facilitated the establishment of metastatic lesions in distant target organs. Although MDS is a liquid rather than a solid tumor, our studies suggest that close interactions of clonal MDS cells with HS27a stroma may allow for a similar scenario and thereby provide support for the propagation of the MDS clone by HS27a cells. The fact that coinjection of HS5 stroma cells modified to overexpress CD146 also facilitated engraftment of HPC suggests that CD146 is involved in the transport of HPC to the murine hematopoietic organs. Nevertheless, the amount of HS27a cells in the spleen, identified by ICAM1 or CD146 staining, varied considerably between mice, ranging from very prominent to infrequent. Also, there was no strong correlation of the number of HS27a cells documented in marrow and spleen, and the extent of engraftment of human HPC. Thus, it is likely that additional, possibly humoral signals provided by the coinjected HS27a cells, directly or indirectly, contributed to engraftment.11, 28 Support for such a mechanism comes also from a study by Thanopoulou et al.,23 who showed that engraftment of clonal MDS cells was enhanced if recipient NOD/SCID β2 microglobulin-deficient mice were rendered transgenic for human interleukin-3, granulocyte–macrophage colony-stimulating factor and stem cell factor. Similarly, a recent report by Takagi et al.29 shows that NSG mice transgenic for human stem cell factor achieved higher rates of engraftment of human CD45+HPCs and CD45+CD33+ myeloid development than non-transgenic controls. The observation of a trend of clonal HPC to localize preferentially to marrow rather than spleen will merit future studies to determine microenvironmental signals that may favor such a distribution.
Earlier studies in sublethally irradiated NOD/SCID (NOD/LtSz-scid/scid) mice were unsuccessful in reproducibly engrafting clonal MDS cells.4 In NOD/SCID β2 microglobulin-deficient mice, coinjection of a mix of the two human stroma cell lines, HS5 and HS27a, along with MDS marrow directly into the marrow cavity, did allow for low-level engraftment of MDS marrow, but only rarely were clonal HPC identified.3 We now interpret those data as a reflection of the proapoptotic effect of HS5 cells12 that out-competed the hematopoiesis supporting function of HS27a cells. Although HS27a cells, as reported previously, are also capable of conveying proapoptotic signals to MDS cells,12 HPC that maintained a sustained contact with HS27a did not undergo apoptosis, an important observation in the context of the present results, which suggest that close interaction between the stroma and HPC was essential for engraftment.12, 13, 24, 30, 31
The success of transplanting clonal MDS HPC (along with HS27a) into secondary recipient mice provides support for the concept that the clonal cytogenetic abnormalities that characterized a patient’s MDS were present in long-term repopulating cells. Although earlier studies showed higher numbers of colony forming cells produced from CD38low HPCs in in vitro cocultures with HS5 than in those with HS27a, HS27a did support cobblestone area forming cells,18 consistent with our interpretation of the present data as being indicative of support for self-renewing, long-term repopulating HPC.
In summary, we describe a model of predictable engraftment of human MDS clones in NSG mice. Engraftment was achieved after i.v. injection and did not require intramedullary inoculation as described by us3 and others16 before. Engraftment was not restricted to a particular clonal cytogenetic abnormality, but was achieved with clonal MDS karyotypes encompassing the characteristic broad spectrum of patients with MDS.32 Although determination of the precise mechanism leading to colocalization of stroma cells with HPCs requires further study, the data suggest that stroma cells traveled with HPC, a phenomenon presumably related to the characteristics of HS27a stroma (CD146 expression) and, conceivably, features of clonal MDS cells that allowed them to serve as ‘carriers.’ Our observations also suggest that HS27a cells provide an essential component of the functional microenvironment for HPC support. The differences between the effects of HS5 and HS27a cells underscore the fact that marrow-derived stroma comprises a spectrum of functionally highly distinct cell populations.17, 33
Further characterization of differences between clonal MDS cells (including recently identified mutations34) that achieve higher levels versus those that achieve lower levels of engraftment may identify novel parameters that determine the survival of MDS clones and may be of relevance when choosing between therapeutic options aimed at eliminating the clone(s).
References
Sanchez PV, Perry RL, Sarry JE, Perl AE, Murphy K, Swider CR et al. A robust xenotransplantation model for acute myeloid leukemia. Leukemia 2009; 23: 2109–2117.
Agliano A, Martin-Padura I, Mancuso P, Marighetti P, Rabascio C, Pruneri G et al. Human acute leukemia cells injected in NOD/LtSz-scid/IL-2Rgamma null mice generate a faster and more efficient disease compared to other NOD/scid-related strains. Int J Cancer 2008; 123: 2222–2227.
Kerbauy DMB, Lesnikov V, Torok-Storb B, Bryant E, Deeg HJ . Engraftment of distinct clonal MDS-derived hematopoietic precursors in NOD/SCID-β2microglobulin-deficient mice after intramedullary transplantation of hematopoietic and stromal cells. Blood 2004; 104: 2202–2203.
Benito AI, Bryant E, Loken MR, Sale GE, Nash RA, Gass MJ et al. NOD/SCID mice transplanted with marrow from patients with myelodysplastic syndrome (MDS) show long-term propagation of normal but not clonal human precursors. Leuk Res 2003; 27: 425–436.
Nilsson L, Astrand-Grundstrom I, Arvidsson I, Jacobsson B, Hellstrom-Lindberg E, Hast R et al. Isolation and characterization of hematopoietic progenitor/stem cells in 5q-deleted myelodysplastic syndromes: evidence for involvement at the hematopoietic stem cell level. Blood 2000; 96: 2012–2021.
Nilsson L, Astrand-Grundstrom I, Anderson K, Arvidsson I, Hokland P, Bryder D et al. Involvement and functional impairment of the CD34+CD38-Thy-1+ hematopoietic stem cell pool in myelodysplastic syndromes with trisomy 8. Blood 2002; 100: 259–267.
Raaijmakers MH, Scadden DT . Evolving concepts on the microenvironmental niche for hematopoietic stem cells. Curr Opin Hematol 2008; 15: 301–306.
Raaijmakers MHGP, Mukherjee S, Guo S, Zhang S, Kobayashi T, Schoonmaker JA et al. Bone progenitor dysfunction induces myelodysplasia and secondary leukaemia. Nature 2012; 464: 852–857.
Konopleva M, Konoplev S, Hu W, Zaritskey AY, Afanasiev BV, Andreeff M . Stromal cells prevent apoptosis of AML cells by up-regulation of anti-apoptotic proteins. Leukemia 2002; 16: 1713–1724.
Jenkins MK, Khoruts A, Ingulli E, Mueller DL, McSorley SJ, Reinhardt RL et al. In vivo activation of antigen-specific CD4 T cells. Annu Rev Immunol 2001; 19: 23–45.
Bhagat TD, Spaulding E, Sohal D, Mo Y, Marcondes M, Zhou L et al2008 MDS marrow stroma is characterized by epigenetic alterations. Blood 112: 3635 (Abstract).
Mhyre A, Marcondes AM, Spaulding EY, Deeg HJ . Stroma-dependent apoptosis in clonal hematopoietic precursors correlates with expression of PYCARD. Blood 2009; 113: 649–658.
Li X, Marcondes AM, Gooley TA, Deeg HJ . The helix-loop-helix transcription factor TWIST is dysregulated in myelodysplastic syndromes. Blood 2010; 116: 2304–2314.
Mendez-Ferrer S, Michurina TV, Ferraro F, Mazloom AR, Macarthur BD, Lira SA et al. Mesenchymal and haematopoietic stem cells form a unique bone marrow niche. Nature 2010; 466: 829–834.
Bhatia R, McGlave PB, Dewald GW, Blazar BR, Verfaillie CM . Abnormal function of the bone marrow microenvironment in chronic myelogenous leukemia: role of malignant stromal macrophages. Blood 1995; 85: 3636–3645.
Muguruma Y, Matsushita H, Yahata T, Yumino S, Tanaka Y, Miyachi H et al. Establishment of a xenograft model of human myelodysplastic syndromes. Haematologica 2011; 96: 543–551.
Graf L, Iwata M, Torok-Storb B . Gene expression profiling of the functionally distinct human bone marrow stromal cell lines HS-5 and HS-27a. Blood 2002; 100: 1509–1511.
Roecklein BA, Torok-Storb B . Functionally distinct human marrow stromal cell lines immortalized by transduction with the human papilloma virus E6/E7 genes. Blood 1995; 85: 997–1005.
Croft JA, Bridger JM, Boyle S, Perry P, Teague P, Bickmore WA . Differences in the localization and morphology of chromosomes in the human nucleus. J Cell Biol 1999; 145: 1119–1131.
Tormin A, Li O, Brune JC, Walsh S, Schutz B, Ehinger M et al. CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood 2011; 117: 5067–5077.
Pillai MM, Yang X, Balakrishnan I, Bemis L, Torok-Storb B . MiR-886-3p down regulates CXCL12 (SDF1) expression in human marrow stromal cells. PLoS One 2010; 5: e14304.
Dolan ME, Pegg AE, Moschel RC, Vishnuvajjala BR, Flora KP, Grever MR et al. Biodistribution of O6-benzylguanine and its effectiveness against human brain tumor xenografts when given in polyethylene glycol or cremophor-EL. Cancer Chemother Pharmacol 1994; 35: 121–126.
Thanopoulou E, Cashman J, Kakagianne T, Eaves A, Zoumbos N, Eaves C . Engraftment of NOD/SCID-β2 microglobulin null mice with multilineage neoplastic cells from patients with myelodysplastic syndrome. Blood 2004; 103: 4285–4293.
Duda DG, Duyverman AM, Kohno M, Snuderl M, Steller EJ, Fukumura D et al. Malignant cells facilitate lung metastasis by bringing their own soil. Proc Natl Acad Sci USA 2010; 107: 21677–21682.
Li L, Milner LA, Deng Y, Iwata M, Banta A, Graf L et al. The human homolog of rat Jagged1 expressed by marrow stroma inhibits differentiation of 32D cells through interaction with Notch1. Immunity 1998; 8: 43–55.
Sacchetti B, Funari A, Michienzi S, Di Cesare S, Piersanti S, Saggio I et al. Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 2007; 131: 324–336.
Bonnet D, Dick JE . Human acute myeloid leukemia is organized as a hierarchy that originates from a primitive hematopoietic cell. Nat Med 1997; 3: 730–737.
Ramakrishnan A, Torok-Storb BJ . The role of the marrow microenvironment in hematopoietic stem cell transplantation. Cell Ther Transplant 2010; 2: 7–12.
Takagi S, Saito Y, Hijikata A, Tanaka S, Watanabe T, Hasegawa T et al. Membrane-bound human SCF/KL promotes in vivo human hematopoietic engraftment and myeloid differentiation. Blood 2012; 119: 2768–2777.
Kerbauy DMB, Mhyre A, Bryant E, Deeg HJ . Do MDS-derived clonal hematopoietic precursors require human stroma support for survival. Curr Res Hematol 2007; 1: 1–13.
Marcondes AM, Li X, Gooley TA, Milless B, Deeg HJ . Identification of DJ-1/Park-7 as a determinant of stroma-dependent and TNFα-induced apoptosis in MDS by using mass spectrometry and phosphopeptide analysis. Blood 2010; 115: 1993–2002.
Schanz J, Steidl C, Fonatsch C, Pfeilstocker M, Nosslinger T, Tuechler H et al. Coalesced multicentric analysis of 2351 patients with myelodysplastic syndromes indicates an underestimation of poor-risk cytogenetics of myelodysplastic syndromes in the international prognostic scoring system. J Clin Oncol 2011; 29: 1963–1970.
Akavia UD, Veinblat O, Benayahu D . Comparing the transcriptional profile of mesenchymal cells to cardiac and skeletal muscle cells. J Cell Physiol 2008; 216: 663–672.
Bejar R, Stevenson K, Abdel-Wahab O, Galili N, Nilsson B, Garcia-Manero G et al. Clinical effect of point mutations in myelodysplastic syndromes. N Engl J Med 2011; 364: 2496–2506.
Acknowledgements
We thank the staff of Animal Health and Flow Cytometry shared resources of the FHCRC for their assistance, Mark Groudine for allowing us to use his laboratory to carry out the FISH analysis, Julie Randolph-Habecker for helping us with immunohistochemical studies, and Helen Crawford and Bonnie Larson for helping us with manuscript preparation. This work was supported by grants DK085156, DK056465, HL036444 and HL095999 from the National Institutes of Health, Bethesda, and by a donation from S. and S. Briscombe, MD. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health nor its subsidiary Institutes and Centers.
Author contributions
Xiang Li designed the study, carried out most experiments, interpreted the data and wrote the manuscript. A Mario Marcondes carried out experiments and provided critique of the manuscript. Tobias Ragoczy carried out the FISH experiments, assisted in data analysis and provided critique of the manuscript. Agnes Telling carried out FISH experiments. H Joachim Deeg designed the study, assisted in data interpretation and revised the manuscript. All authors read and provided input to the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on Blood Cancer Journal website
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 3.0 Unported License. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/3.0/
About this article
Cite this article
Li, X., Marcondes, A., Ragoczy, T. et al. Effect of intravenous coadministration of human stroma cell lines on engraftment of long-term repopulating clonal myelodysplastic syndrome cells in immunodeficient mice. Blood Cancer Journal 3, e113 (2013). https://doi.org/10.1038/bcj.2013.11
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bcj.2013.11
Keywords
This article is cited by
-
Loss of bisecting GlcNAcylation on MCAM of bone marrow stoma determined pro-tumoral niche in MDS/AML
Leukemia (2023)
-
Screening differentially expressed proteins from co-cultured hematopoietic cells and bone marrow-derived stromal cells by quantitative proteomics (SILAC) method
Clinical Proteomics (2019)
-
Stem cell origin of myelodysplastic syndromes
Oncogene (2014)