Abstract
Lead (Pb) is a metal of special importance because of its long history of commercial and industrial use, global atmospheric contamination accelerated by the use of gasoline additives, and health effects, with children being especially vulnerable. Global atmospheric Pb pollution reached its zenith in the 1970’s, but subsequent impacts on freshwater aquatic systems are poorly understood. Employing metal-free sampling and handling protocols, we show that snowmelt from the Athabasca bituminous sands region is an insignificant source of dissolved Pb to the Athabasca River (AR). Total Pb in the AR is low, and almost entirely in particulate form. Lead in the suspended solids in the AR exactly follows thorium (Th), a conservative lithophile element, and a linear regression of Pb against Th (Pb = 1.6 × Th + 0.0; R2 = 0.99) yields a slope identical to the Pb/Th ratio in the Upper Continental Crust. In the “dissolved” fraction, the Pb/Th ratio is equivalent to that of deep, open ocean seawater; and dominated by colloidal forms. Taken together, these results show that the efforts of recent decades to reduce anthropogenic Pb to the environment have been successful: Pb loading to the river can now be explained predominantly by natural processes, namely erosion plus chemical weathering.
Similar content being viewed by others
Introduction
Among the long list of contemporary environmental contaminants, lead (Pb) is one of the most problematic potentially toxic trace elements because atmospheric contamination by this metal became a global phenomenon1,2. The geochemical cycle of Pb has been impacted by anthropogenic activities more than any other metal3. The use of leaded gasoline around the world from ~1920 to ~1970 was the last but by far the largest episode of the most significant Pb sources to the environment after smelting and refining of Pb and other non-ferrous metals, coal combustion and cement production4. Environmental impacts have been studied extensively, and evidence of Pb in both polar regions suggested no area on the earth’s surface has been unaffected by anthropogenic Pb1. However, the phase-out and eventual elimination of leaded gasoline in North America and Europe since 1970 and 1980, respectively has precipitated a dramatic decrease in the concentrations of atmospheric Pb4,5. While the fall in air Pb concentrations has been accompanied by a corresponding drop in human blood Pb6, there is no known safe level and blood Pb values are now “seen by society as evidence of its commitment to its own health”7.
While declining air Pb concentrations have been documented in environmental archives affected exclusively by atmospheric inputs such as polar snow and ice as well as ombrotrophic (i.e. rain-fed) peat bogs8,9,10 the consequences for surface waters have received less attention. Anthropogenic Pb inputs to global oceans were documented by Clair Patterson11 and declines in Pb concentrations in surface seawater subsequent to the phaseout of leaded gasoline have been clearly illustrated for the Atlantic Ocean by Ed Boyle12,13. In respect to freshwaters, pioneering work on Pb in surface and groundwaters in the 1970’s documented the significant contributions of anthropogenic Pb to aquatic ecosystems at that time14. It became clear from these studies that the key to understanding the biological significance of this additional Pb was to distinguish between dissolved and particulate forms: it is the dissolved (<0.45 μm fraction) that is potentially mobile, bioaccessible, bioavailable and toxic14. Coincidentally, almost all of the anthropogenic Pb released to the atmosphere from high temperature combustion processes is in the sub-micron fraction15. Thus, as far as potential impacts of Pb on human and ecosystem health is concerned, in both air and water, it is the sub-micron fraction which has the greatest relevance. However, studies show that within the conventional dissolved fraction (<0.45 μm) not all the Pb is bioavailable and it can be partitioned into colloidal and mainly ionic (truly dissolved) Pb16,17,18. Ionic Pb is the only fraction that can directly impact the aquatic life19. If it were not for the various industrial inputs, almost all of the Pb would be in the particulate fraction, both in the atmosphere14 as well as in water20, with wind and water erosion, respectively, being the dominant sources of Pb to the surficial environment.
The accurate and precise determination of Pb in the dissolved fraction of natural freshwaters, however, poses several considerable challenges. First, the natural abundance of dissolved Pb in freshwaters is extremely low. Pioneering work from the Patterson lab showed long ago that dissolved Pb in remote streams of the Sierra Nevada watershed is commonly below 15 ng/l21. The low concentrations of Pb pose two additional difficulties: 1), the need for tremendous analytical sensitivity, and 2), the grave risks of contamination which are encountered at virtually every stage of the sampling, handling, and analysis process22,23,24,25,26. While some labs have understood and met these challenges, most have not27. In the intervening period there have been many studies of Pb in rivers and lakes28,29,30,31,32,33,34,35,36,37,38,39,40,41, but there are remarkably few studies providing reliable data for the dissolved fraction. Studies employing the clean lab procedures and protocols developed for polar snow and ice as well as sector-field inductively-coupled plasma mass spectrometer (SF-ICP-MS) have reported Pb values in the dissolved fraction of surface waters and groundwaters in the range of a few ng/l or less8,42,43. Any studies of dissolved Pb in surface waters remote from industrial activity, therefore, need to be able to reliably measure Pb well below the part per trillion (ng/L) concentration range. Studies reveal that the dissolved Pb fraction (<0.45 μm) consists of both colloidal and free (ionic) forms, but it is the ionic Pb fraction that is biologically significant for aquatic organisms19. Depending on the relative importance of the colloidal fraction, ionic forms of Pb may be far less abundant than the fraction traditionally defined as “dissolved” i.e., <0.45 μm. The reliable determination of ionic forms of Pb in natural waters, therefore, presents considerable analytical challenges.
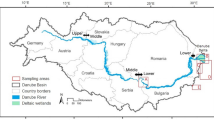
To better develop an understanding of the contemporary geochemical cycle of Pb in a major river system and to determine the impact of mining and upgrading activities of the Athabasca bituminous sands on dissolved Pb in the AR, we employ the state-of-the-art metal-free ultra-clean analytical sample collection, handling and analytical procedures developed at the University of Heidelberg for measuring Pb and Pb isotopes in Arctic snow and ice4,44. These ultra-clean sample collection, handling and analytical procedures have been successfully employed at the SWAMP lab, University of Alberta45. We compare total, particulate (>0.45 μm), dissolved (<0.45 μm), colloidal (1 kDa to 0.45 μm) and mainly ionic (<1 kDa) Pb concentrations in the lower reaches of Athabasca River (AR, ~1500 km) in northern Alberta, one of the largest rivers in Canada (http://www.awc-wpac.ca/). The section of the AR selected for study is in the heart of the industrialized region where open pit mines and upgraders of the Athabasca Bituminous Sands (ABS) are found. It was claimed that these commercial activities are a significant source of Pb to air and waters of this region46, so samples were collected upstream, downstream, and within the industrialized zone. The main objective of this study is to quantify the impacts of human activities on Pb dissolved in the river. From October 7 to 17, 2014 water samples and suspended solids were collected from thirteen sites along the main stem of the AR, upstream of Fort McMurray and along a transect ~125 km downstream, as well as from five tributary streams and five groundwater sampling locations (Fig. 1). In addition to the water and suspended solids, in March, 2015 snow samples were also collected from peat bogs in the area to characterize Pb in contemporary atmospheric deposition (Fig. S1 and Table S1).
Terrain conductivity data is taken from Gibson et al.71, and ranges from 12 mS m−1 (dark blue) to 76 mS m−1 (light pink). The map is created using ArcGIS Desktop (ESRI 2011: Release 10.3. Redlands, CA: Environmental Systems Research Institute, http://www.esri.com/software/arcgis/arcgis-for-desktop) by taking the basemaps and reference layers information through basemap imagery (Source: Esri, DigitalGlobe, GeoEye, i-cubed, USDA, USGS, AEX, Getmapping, Aerogrid, IGN, IGP, swisstopo, and the GIS User Community).
Dissolved Pb concentrations for the main stem and tributaries from this sampling campaign are taken from Shotyk et al.45, and the rest of the data produced in this study. Sampling, handling and Pb determination in surface and groundwaters are described in detail in the supporting information (SI).
Lead in the Athabasca River (AR) in perspective
Total Pb in the main stem of the AR (127 ± 58 ng L−1; n = 13, Fig. 2A) and its tributaries (119 ± 112 ng L−1; n = 5, Fig. 2B) are low: approximately 25 to 55 times below the guideline values established for the protection of aquatic life by the Canadian Council of Ministers of the Environment (CCME)47 and ~75% lower than the values for this section of the river reported previously48. Total Pb in the AR on average is ~80 times lower than the World Health Organization (WHO) guideline value (10 μg L−1) for drinking water. To help put these values into perspective, total Pb in the AR is considerably lower than dissolved Pb in the Nippissing River which was sampled in the most remote section of Algonquin Provincial Park in southern Ontario (dissolved Pb: 306 ± 34 ng L−1; n = 3); waters were sampled from this nature reserve (7,653 km2) in the autumn of 2006, using the identical sampling and handling protocols described here, but measured in the metal-free, ultraclean lab at the University of Heidelberg in Germany45.
Total and particulate lead (Pb) concentrations in the (A) main stem of the Athabasca River, (B) tributary streams and (C) groundwater. The bars in the graphs show the average Pb concentrations of triplicate samples and error bars are the standard deviations. The dashed lines in the graphs show the average dissolved Pb and dotted lines show the one standard deviation. The dissolved Pb in the main stem AR and tributaries were reported previously (Shotyk et al.45), but the dissolved Pb in groundwater is generated in this study. The results show that almost all the Pb is in particulate form. Note: while the concentrations of total and dissolved Pb in groundwater presented here represent accurate and precise measurements, the abundance of particulate matter in the groundwater samples is partly a reflection of the procedure employed (drive point wells) to collect them. The values presented here should not be viewed as the final word on the topic, rather the starting point: research on these groundwaters is ongoing.
To determine the possible impact of ABS mining and upgrading activities, total Pb concentrations were compared between the sites located on the AR adjacent to the tailing ponds, mining activities and bitumen upgraders (sites A18 to A9; Fig. 1) and the sites far from industry (A8 and A5; Fig. 1). No significant difference (P = 0.16; two tailed Student’s t.test) was found between the total Pb concentration in the industrial zone (105 ± 67 ng L−1; n = 8) and in the area far from these activities (181 ± 8 ng L−1; n = 2). Total Pb in the main stem of the AR (127 ± 58 ng L−1; n = 13, Fig. 2A) and the five tributary streams (119 ± 112 ng L−1; n = 5, Fig. 2B) show no significant differences (P = 0.85; two tailed Student’s t.test). Although it has been suggested that there are significant inputs of Pb to the AR from the industrial development of the bituminous sands46, there is no evidence of this in the total Pb concentrations which is the fraction upon which the CCME guideline for the protection of aquatic life is based. Greater concentrations of total Pb were found in the groundwater samples (0.2 to 2.5 μg L−1; Fig. 2C), but this is a reflection of the greater abundance of particles in groundwater compared to the surface waters (Figs S2 and S3).
To distinguish natural from anthropogenic Pb in atmospheric aerosols and other environmental monitoring media such as snow, ice, moss and peat, Pb can be normalized to a conservative lithophile element such as thorium (Th) or scandium (Sc)10,49,50. The normalization provides an indication of the extent to which industrial activities have contaminated the sample, relative to the corresponding natural background (crustal) ratio in soil-derived dust particles. The same approach can be used with bulk water samples, to account for the contribution of suspended mineral matter to total concentrations. The ratio of Pb to Th in the bulk surface water samples from the main stem of the AR (2.5 ± 0.3; n = 13) are similar to those of groundwater (2.2 ± 0.3); these are remarkably similar to the Pb to Th ratio in the Wye River (2.4 ± 0.7; n = 3), a small farm stream sampled near Elmvale, Ontario43. The total Pb concentration in the Wye River (74 ± 5 ng L−1; n = 3) is lower than in the AR, but this most likely reflects the differences in flow rates: 623 m3 sec−1 on average, in Fort McMurray compared to ~1 m3 sec−1 in the Wye River (Environment Canada). Simple Pearson Correlation between total Pb and total Th in the surface water of the AR main stem, its tributaries and groundwater (r = 0.93, 0.98 and 0.94, respectively) reveals that overall the Pb distribution in water follows Th remarkably well, which suggests that Pb concentration in the river are almost exclusively a reflection of the abundance of mineral particles.
Particulate (>0.45 μm), dissolved (<0.45 μm), colloidal (1 kDa to 0.45 μm) and mainly ionic Pb (<1 kDa)
Dissolved Pb is very low in both the main stem of the AR (20 ± 7 ng L−1: n = 13, Fig. 2A) as well as the tributary streams (25 ± 30 ng L−1; n = 5, Fig. 2B). Based on these values and the total concentrations presented here, it is clear that almost all of the Pb in the AR is associated with particulate fraction (Fig. 2A,B). Over 90% of the total Pb measured in the Wye river water was also found in the particulate form (data not shown). Here we also report the first reliable measurements of dissolved Pb (123 ± 82 ng L−1; n = 5) in the groundwaters which feed the AR (Fig. 2C): even in the groundwaters, almost all of the Pb is in the particulate fraction.
Dissolved Pb in the AR is very low and fractionating the dissolved Pb into colloidal (1 kDa to 0.45 μm) and mainly ionic (<1 kDa) forms reveals that on average ~60% of the dissolved Pb (~20 ng L−1) is bound with organic and inorganic colloids which means that the bioaccessible Pb concentration is on the order of 8 ng L−1 (Figs 3 and 4). This is similar to the proportion of colloidal Pb measured in an estuary using crossflow ultrafiltration (64 ± 9%; Wen et al.51, and to the proportion measured in stream water during snowmelt using AF4-ICP-MS with a 1 kDa membrane (59%; Stolpe et al.52). Curiously, Neubauer et al.53 found that 116 and 70% of dissolved (<0.2 μm) Pb was colloidal in two peat bog drainage samples using AF4-ICP-MS with the 0.3 kDa membrane, whereas 89 and 96% was respectively found to be colloidal in the same samples using a stirred ultrafiltration cell. Comparing results across studies is challenging given disparities in analytical methods, DOM sources, physicochemical conditions, and filter pore sizes. The distribution of Pb amongst different size fractions also depends upon its source (e.g. wind erosion of soils vs high temperature combustion vs reprocessed colloids), so that the useful comparison of size distributions across studies requires more thorough analysis of trace element properties.
DOM and Fe peaks at ~0.98 and 1.7 kDa, and peak at >20 kDa (middle panel) show the primarily organic and primarily inorganic colloids, respectively. Fractionation of dissolved Pb (upper panel) shows Pb association with both organic and inorganic colloids. Data is baseline corrected and Pb data is smoothed (5 point running average). Here we define colloidal Pb = Pb associated with organic colloids + Pb associated with inorganic colloids, and mainly ionic Pb = Dissolved Pb - Colloidal Pb. The crossflow was shut off to release large material at a retention time corresponding to a molecular weight of ~104. Thus, the specific molecular weight of material that is larger than this cannot be determined.
Characterizing particulate Pb
We developed a precise mechanical procedure to recover each polytetrafluoroethylene (PTFE) filter membrane from its plastic housing (Fig. S4) along with an effective protocol to digest the particles recovered (SI). The Pb concentrations in these suspended solids (Figs S5A and B), whether they were recovered from the main stem of the AR (8.6 ± 5.6 μg g−1, n = 13) or the tributary streams (4.7 ± 2.8 μg g−1, n = 5), are similar to the values for sedimentary rocks20 which constitute the Western Canadian Sedimentary Basin54 and are similar to the average Pb content of sandstones and limestones (7 μg g−1 Pb)55. These Pb values in the suspended solids in the AR are similar to the Pb concentrations in the suspended particles (2.5 μg g−1) reported in Siberian rivers draining basalts56. Among the different samples, Pb concentrations in the suspended solids at site AR at Beaver Creek (~24 μg g−1 Pb) was about 3 times higher than the average Pb in the other sites. Comparing the TSS results showed that this site (AR at Beaver Creek) contained more TSS (0.7 g/L) than the average TSS (0.3 g/L) at other sites. The groundwater samples have slightly greater Pb concentrations in the particulate fraction, (15.1 ± 3.1 μg g−1, n = 5; Fig. S5C), but these still resemble the abundance of Pb in the Upper Continental Crust which is 17 μg g−1 according to both Wedepohl57 and Rudnick and Gao58. We suspect that the difference in Pb concentrations of the particulate fraction in groundwater versus surface water may be a particle size effect. A linear regression of Pb against Th in the suspended solids of the AR (Fig. 5A) shows a remarkable correlation ([Pb] = 1.6 × [Th] + 0.0; R2 = 0.99). The slope of the regression line is identical to the Pb/Th ratio (1.6) for the Upper Continental Crust57,58. Taken together, the results show that Pb in the AR is almost exclusively in the form of mineral particles derived from physical weathering and erosion within in the watershed, with a tiny contribution to the Pb inventory from the dissolved fraction which must be supplied by chemical weathering. In contrast to the AR, the average Pb concentration reported in the suspended solids of world rivers (25–100 μg g−1) is far higher59,60,61.
Scatter plots between lead (Pb) and thorium (Th) concentrations in (A) the suspended solids in the main stem of the Athabasca River (AR) and (B) the dust particles in snow samples collected from five peat bogs in the study area. The slopes of the regression lines (1.6) between Pb and Th in both graphs are similar to the ratio in the Upper Continental Crust57,58. aThe Pb/Th ratio in the water samples studied here is also compared with the Pb/Th ratio in groundwaters collected from the HDPE well of the Elmvale Groundwater Observatory. The HDPE tubes and screen used to construct this well (known as EGO-2) are from Rotek (Denmark); they had been shipped to the University of Heidelberg, Germany, for cleaning in HNO3. After rinsing, drying in metal-free, laminar flow clean air cabinets, and packing in sealed PE bags, the well materials were shipped from Germany to Canada and installed near (3 m) but hydrologically upgradient of EGO-1, a flowing, artesian groundwater sampling well constructed entirely of stainless steel. Water samples are collected from the EGO-2 (HDPE) well via a purpose-built, acid-cleaned Teflon valve, within a metal-free, laminar flow clean air cabinet. Both of these flowing, artesian wells near the Village of Elmvale, Ontario, emanate from an aquifer at a depth of 13 m: together they are known as the Elmvale Groundwater Observatory and are dedicated to the study of trace metals in water. The water samples were collected in March of 2011 and all measurements performed at the University of Heidelberg, Germany using an Element 2 ICP-SMS (Shotyk and Krachler, unpublished data). The data presented here from EGO-2 represents water samples (n = 10) that were collected using acid-cleaned HDPE bottles provided by Dr. Jiancheng (James) Zheng of the Geological Survey of Canada, Natural Resources Canada, Ottawa. In fact, these are the same types of bottles, prepared in the same manner, used in the study of Pb in the ice core from Devon Island, Nunavut, in the Canadian Arctic4,44. The Pb/Th ratio in the particulate and dissolved fractions of the AR is remarkably similar to the Pb/Th ratio found in this groundwater. bPb/Th ratio in the Upper Continental Crust/10.
Lead in snow
Snow pack sampling has been used to suggest that atmospheric deposition is an important source of industrial Pb to the AR46. In March 2015, we collected snow samples from peat bogs in several areas close to the ABS mining and upgrading activities, and measured total as well as dissolved (<0.45 μm) Pb (Fig. S1 and Table S1). The details of the sample locations, snow sampling, handling and Pb determinations are given in the Supporting Information. The average concentration of dissolved (<0.45 μm) Pb in the snow samples was 4.2 ± 0.7 ng L−1 (n = 5; Fig. 6A). The dissolved Pb concentrations in both the snow samples and the AR main stem surface water are low, furthermore, the dissolved Pb in the snow collected from the peat bogs closest to the industrial activity is ten times lower than the average dissolved Pb in the AR main stem surface water. To put the dissolved Pb concentration in the snow into perspective, Pb concentrations in contemporary snow (1994 to 2004) from the Arctic are ten times greater4. In fact, the average concentration of Pb from the “cleanest” section of the ice core collected on Devon Island, Nunavut, representing snow accumulation from ca. 5,000 to 8,000 years ago, is 5.1 ± 1.4 ng/L44. Dissolved Pb in the snow samples follows the distribution of dissolved Th (n = 5; Fig. 6B). Average total Pb concentrations in the snow was 749 ± 421 μg L−1 (n = 5; Fig. 6C) compared with total Th (347 ± 294 ng L−1, n = 5; Fig. 6D); Pb showed a significant positive correlation (r = 0.99, P < 0.01) with total Th. Moreover, the ratio of total/dissolved Pb in the snow (180) matches remarkably well the total/dissolved Th (189). These results show that dust particles (primarily mineral matter) in the snow are the main factor controlling Pb concentrations. Comparing the Pb and Th concentrations in the dust particles in snow, a significant positive correlation was found ([Pb] = 1.6 × [Th] + 0.1; R2 = 0.93, Fig. 5B). Similar to the suspended solids in the AR, the slope of the regression line is identical to the Pb/Th ratio (1.6) for the Upper Continental Crust57,58. If mining and upgrading of ABS was a significant source of Pb to the air, the Pb concentrations in the snow from this region should be enriched, relative to the abundance of Th in snow. Clearly, that is not the case and the Pb/Th ratios fail to identify a detectable anthropogenic contribution.
Concentrations of (A) dissolved (<0.45 μm) lead, (B) dissolved thorium, (C) total lead and (D) total Th in snow samples collected from five peat bogs in the study area, the locations of bogs are shown in Fig. S1. The bars in the graph show average Pb or Th concentrations of triplicate samples collected from every site and the error bars represent one standard deviation.
Ecological significance of Pb in the AR?
The Peace-Athabasca Delta (PAD) is one of the largest freshwater deltas in the world, and of tremendous ecological significance. There are legitimate concerns about a range of environmental impacts on the delta, including contaminants received from the AR watershed62, one of them being Pb. It was suggested long ago14,59 that more than 90% of the Pb in rivers should be in the particulate fraction. Although many studies have found that Pb in most rivers is dominated by the particulate fraction63,64,65,66, in the past there had been a large anthropogenic contribution which apparently dominated the dissolved fraction and contributed in a significant way to total Pb concentrations. As anthropogenic emissions of Pb to the atmosphere have declined worldwide, it has become increasingly difficult to measure dissolved Pb in natural waters. The lack of reliable measurements of Pb in the dissolved fraction has limited our understanding of the potential ecological significance of Pb in aquatic systems: this fraction is the key to bioaccessibility and bioavailability of Pb. In the AR, using the metal-free ultraclean procedures and protocols developed for polar snow and ice, we see that Pb in the dissolved fraction is almost inconsequential. Further fractionation of dissolved Pb into colloidal (1 kDa to 0.45 μm) and mainly ionic (<1 kDa) forms reveals that a significant proportion (~60%) of the traditionally defined as “dissolved” Pb fraction is, in fact, bound with colloids, and these are expected to have limited bioavailability.
To bring these results into a broader prospective and understand the overall Pb inputs to the PAD, based on dissolved Pb reported previously (Shotyk et al.45), and Pb in the suspended solids presented here, we find that almost the entire Pb load to the PAD is in the particulate fraction, in the form of mineral particles, whereas dissolved Pb accounts for no more than 1–2% of total Pb. Moreover, within the “dissolved” fraction, approximately 60% of the Pb is bound with colloidal particles (Fig. 4). Again, with the hope of bringing perspective to the data, consider that air Pb emissions from all industrial sources in Alberta was 1757 kg in 201467. The AR watershed (150,000 km2) represents approximately 23% of the surface area of the Province of Alberta. If 23% of all annual industrial Pb emissions in Alberta were to become dissolved in the river, it would only increase the concentration of dissolved Pb i.e., the <0.45 μm fraction (20 ng L−1) by a factor of ~2. If the same mass of industrial Pb was exclusively in the form of particulates, it would contribute less than 2% to the Pb inventory in the suspended solids.
Summary
International efforts over decades to reduce anthropogenic emissions of Pb to the environment have successfully reduced air Pb concentrations worldwide6, and benefits to aquatic ecosystems such as the AR are now becoming apparent as well. Lead in the AR is clearly dominated by natural inputs, mainly erosion, to such an extent that an anthropogenic component is now difficult to discern.
Experimental
From October 7 to 17, 2014 using acid-cleaned polypropylene (PP) bottles, raw surface water samples were collected from thirteen sites along the main stem Athabasca River (AR) starting upstream of Fort McMurray and travelling downstream a distance of approximately 125 km. The physicochemical properties of the main stem AR water samples are provided in Table 1. Along this route, water was also collected from five tributary streams draining into the AR and groundwater from five sites (Fig. 1). The sampling sites were selected to represent the upstream locations (WWTP, A20 and A19 sites), the industrial zone (from site A18 to A9 on the river, sites that are adjacent to tailing ponds and bitumen upgraders) as well as the sites far from industry (A8 and A5; Fig. 1). All the sample collection, handling and measurements were carefully performed employing the protocols and procedures developed at the University of Heidelberg, Germany for the determination of Pb and stable Pb isotopes in Arctic ice cores4,44. The raw water samples (2 mL) were digested with 5 mL of double distilled concentrated nitric acid (HNO3) using high pressure microwave digestion (Ultraclave, MLS Leutkirch, Germany) and Pb determined using inductively-coupled plasma mass spectrometry (ICP-MS; iCAP Qc). The details of the determination of Pb in the water samples are provided in the Supporting Information (SI).
Particulate Pb in the main stem AR, tributary streams, and groundwater samples was calculated by subtracting the dissolved Pb from total Pb:

Dissolved Pb concentrations for the main stem and tributaries from this sampling campaign have been presented elsewhere (Shotyk et al.45). Dissolved Pb in the groundwater samples was measured in this study.
The distribution of Pb into colloidal (1 kDa to 0.45 μm) and mainly ionic (<1 kDa) forms within the dissolved fraction (<0.45 μm) was determined using asymmetrical flow field-flow fractionation (AF4) equipped with an auto injector (AF2000 MF and PN5300, respectively, Postnova Analytics, Salt Lake City, Utah, USA), coupled to a UV-Visible absorbance detector (G4212 DAD, Agilent Technologies, Santa Clara, California, USA) and ICP-MS (iCAP Qc, Thermo Fisher). The use of the term “mainly ionic Pb” assumes that Pb in this size fraction is mainly in an ionic form such as Pb2+, an inorganic complex such as PbCl+ or PbSO40, a low molecular weight organic complex, or some combination of these. The fractionation program and peak deconvolution method were adapted from earlier work68,69, and are discussed in detail in the SI.


In the field, water samples for dissolved Pb were filtered through acid-cleaned 0.45 μm PTFE filters into bottles containing HNO3. The filters containing the suspended particles (>0.45 μm) were stored, and preserved for later study. An excellent mechanical setup was devised to cut and open the filter housing to acquire the filter membranes without any loss of suspended sediment (Fig. S4). The suspended sediments were digested in double distilled concentrated HNO3 (6 mL) and HBF4 (0.2 mL) using high pressure microwave digestion (Ultraclave, MLS) and Pb determined using ICP-MS (iCAP Qc). The details of the sample handling, preparation and measurement is provided in the SI. The micromorphology of the suspended solids were study using scanning electron microscope following Javed et al.70, the details of the method are provided in SI.
In addition to water and suspended solids, snow samples were also collected from the Athabasca Bituminous Sands (ABS) mining and upgrading area (Fig. S1). The details of the snow sampling, handling and measurements for dissolved and total Pb and Th concentrations, and Pb and Th concentrations in the dust particles in snow are provided in the SI. Briefly, snow samples collected in acid cleaned PP bottles (1 L) were thawed in the metal free, laminar flow clean air cabinets in the SWAMP laboratory, filtered through acid cleaned 0.45 μm PTFE filters and dissolved Pb and Th concentrations determined using ICP-MS (iCAP Qc).
Additional Information
How to cite this article: Javed, M. B. et al. Size-resolved Pb distribution in the Athabasca River shows snowmelt in the bituminous sands region an insignificant source of dissolved Pb. Sci. Rep. 7, 43622; doi: 10.1038/srep43622 (2017).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Boutron, C. F., Candelone, J. P. & Hong, S. Past and recent changes in the largescale tropospheric cycles of lead and other heavy metals as documented in Antarctic and Greenland snow and ice: a review. Geochim. Cosmochim. Acta. 58, 3217–3225 (1994).
Ng, A. & Patterson, C. C. Natural concentration of lead in ancient Arctic and Antarctic ice. Geochim. Cosmochim. Acta. 45, 2109–2121 (1981).
Nriagu, J. O. Biogeochemistry of Lead in the Environment (Elsevier/North Holland Biomedical Press, Amsterdam, 1, pp. 422 (1978).
Shotyk, W. et al. Predominance of industrial Pb in recent snow (1994–2004) and ice (1842–1996) from Devon Island, Arctic Canada. Geophysical Research Letters 32, L21814, doi: 10.1029/2005GL023860 (2005).
Boutron, C. F., Görlach, U., Candelone, J.-P., Bolshov, M. A. & Delmas, R. J. Decrease in anthropogenic lead, cadmium and zinc in Greenland snows since the late 1960s. Nature. 353, 153–156 (1991).
Petit, D., Véron, A., Flament, P., Deboudt, K. & Poirier, A. Review of pollutant lead decline in urban air and human blood: A case study from northwestern Europe. Comptes Rendus Geosci. 347(5–6), 247–256 (2015).
Amaya, M. A., Jolly, K. W. & Pingitore, N. E. Blood lead in the 21st Century: The sub-microgram challenge. Journal of Blood Medicine. 1, 71–78 (2010).
Shotyk, W. & Krachler, M. The isotopic evolution of atmospheric Pb in central Ontario since AD 1800, and its impacts on the soils, waters, and sediments of a forested watershed, Kawagama Lake. Geochimica et Cosmochimica Acta. 74(7), 1963–1981 (2010).
Shotyk, W. et al. Peat bogs in northern Alberta, Canada reveal decades of declining atmospheric Pb contamination. Geophys. Res. Lett. 43, 9964–9974 (2016).
Shotyk, W. et al. History of atmospheric lead deposition since 12,370 14C yr BP from a peat bog, Jura Mountains, Switzerland. Science. 281, 1635–1640 (1998).
Patterson, C. C. & Settle, D. M. Review of data on eolian fluxes of industrial and natural lead to the lands and seas in remote regions on a global scale. Marine Chemistry. 229(2), 137–162 (1987).
Boyle, E. A. et al. Anthropogenic lead emissions in the ocean: The evolving global experiment. Oceanography. 27(1), 69–75 (2014).
Bridgestock, L. et al. Return of naturally sourced Pb to Atlantic surface waters. Nat. Commun. 7, 12921, doi: 10.1038/ncomms12921 (2016).
Hem, J. D. & Durum, W. H. Solubility and occurrence of lead in surface water. J. Am. Water Work. Assoc. 65, 562–568 (1973).
Nriagu, J. O. The rise and fall of leaded gasoline. Sci, Tot. Environ. 92, 13–28 (1990).
Pokrovsky, O. S. & Shirokova, L. S. Diurnal variations of dissolved and colloidal organic carbon and trace metals in a boreal lake during summer bloom. Wat. Res. 47(2), 922–932 (2013).
Pokrovsky, O. S., Schott, J. & Dupré, B. Trace element fractionation and transport in boreal rivers and soil porewaters of permafrost-dominated basaltic terrain in Central Siberia, Geochim. et Cosmochim. Ac. 70(13), 3239–3260 (2006).
Ilina, S. M. et al. Speciation, size fractionation and transport of trace elements in the continuum soil water–mire–humic lake–river–large oligotrophic lake of a Subarctic watershed. Aquat. Geochem. 22(1), 65–95 (2016).
Gélabert, A., Pokrovsky, O. S., Schott, J., Boudou, A. & Feurtet-Mazel, A. Cadmium and lead interaction with diatom surfaces: a combined thermodynamic and kinetic approach. Geochim. Cosmochim. Acta. 71, 3698–3716 (2007).
Hem, J. D. Study and Interpretation the Chemical of Natural of Characteristics Water. U. S Geol. Surv. Water-Supply Pap. 2254 (1985).
Hirao, Y. & Patterson, C. C. Lead aerosol pollution in the High Sierra overrides. Natural mechanisms which exclude lead from a food chain. Science 184(4140), 989–992 (1974).
Settle, D. M. & Patterson, C. C. Lead in albacore: guide to lead pollution in Americans. Science 207(4436), 1167–1176 (1980).
Boutron, C. F. A clean laboratory for ultralow concentration heavy metal analysis. Fresenius J. of Anal Chem. 337, 482–491 (1990).
Nriagu, J. O., Lawson, G., Wong, H. K. T. & Azcue, J. M. A protocol for minimizing contamination in the analysis of trace metals in Great Lakes waters. J. of Great Lakes Research. 19(1), 175–182 (1993).
Benoit, G. et al. Partitioning of Cu, Pb, Ag, Zn, Fe, Al, and Mn between filter-retained particles, colloids, and solution in six Texas estuaries. Marine Chem. 45, 307–336 (1994).
Benoit, G., Hunter, K. S. & Rozan, T. F. Sources of trace metal contamination artifacts during collection, handling, and analysis of freshwaters. Anal Chem. 69(6), 1006–1011 (1997).
Papadakis, I., Taylor, P. D. P. & De Bievre, P. CCQM-K2 key comparison: cadmium and lead content in natural water. Metrologia 38, 543–547 (2001).
LaZerte, B., Evans, D. & Grauds, P. Deposition and transport of trace metals in an acidified catchment of Central Ontario. Sci of the Tot Environ 87/88, 209–221 (1989).
Erel, Y., Patterson, C. C., Scott, M. J. & Morgan, J. J. Transport of industrial lead in snow through soil to stream water and groundwater. Chem. Geol. 85, 383–392 (1990).
Erel, Y., Morgan, J. J. & Patterson, C. C. Natural levels of lead and cadmium in a remote mountain stream. Geochimica et Cosmochimica Acta. 55, 707–719 (1991).
Erel, Y. & Patterson, C. C. The relationships between rock-derived lead and iron in natural waters. Geochimica et Cosmochimica Acta. 56, 4157–4167 (1992).
Erel, Y. & Patterson, C. C. Leakage of industrial Pb into the hydrocycle. Geochimica et Cosmochimica Acta. 58, 3289–3296 (1994).
Tarvainen, T., Lahermo, P. & Mannio, J. Sources of trace metals in streams and headwater lakes in Finland. Water Air and Soil Pollution. 94(1), 1–32 (1997).
Outridge, P. M. Lead biogeochemistry in the littoral zones of south-central Ontario lakes, Canada, after the elimination of gasoline lead additives. Water Air and Soil Pollution. 118(1–2), 179–201 (2000).
Emmanuel, S. & Y. Erel . Implications from concentrations and isotopic data for Pb partitioning processes in soils. Geochimica et Cosmochimica Acta. 66(14), 2517–2527 (2002).
Vinogradoff, S. I. et al. Investigation of the concentration and isotopic composition of inputs and outputs of Pb in waters at an upland catchment in NE Scotland. J. of Environ Monit. 7(5), 431–444 (2005).
Graham, M. C. et al. Using size fractionation and Pb iIsotopes to study Pb transport in the waters of an organic-rich upland catchment. Environ Sci Technol. 40(4), 1250–1256 (2006).
Klaminder, J. et al. Flux rates of atmospheric lead pollution within soils from a small catchment in northern Sweden and their implication for future stream water quality. Environ Sci Technol. 40, 4639–4645 (2006).
Bindler, R., Renberg, I., Rydberg, J. & Andren T. Widespread waterborne pollution in central Swedish lakes and the Baltic Sea from pre-industrial mining and metallurgy. Environ Pollut. 157, 2132–2141 (2008).
Bur, T., Probst, J. L., N’guessan, M. & Probst, A. Distribution and origin of lead in stream sediments from small agricultural catchments draining Miocene molassic deposits (SW France). App Geochem. 24(7), 1324–1338 (2009).
Landre, A. L., Watmough, S. A. & Dillon, P. J. The effects of dissolved organic carbon, acidity and seasonality on metal geochemistry within a forested catchment on the Precambrian Shield, central Ontario, Canada. Biogeochem. 93(3), 271–289 (2009).
Field, M. P. & Sherrell, R. M. Direct determination of ultra-trace levels of metals in fresh water using desolvating micronebulization and HR-ICP-MS: application to Lake Superior waters. J. of Anal At Spectrometry 18, 254–259 (2003).
Shotyk, W., Krachler, M., Aeschbach-Hertig, W., Hillier, S. & Zheng, J. Trace elements in recent groundwater of an artesian flow system and comparison with snow: enrichments, depletions, and chemical evolution of the water. J. Environ. Monit. 12, 208–217 (2010).
Zheng, J., Shotyk, W., Krachler, M. & Fisher, D. 15,800 years of atmospheric lead deposition on Devon Ice Cap, Nunavut, Canada: Natural and anthropogenic enrichments, isotopic composition, and predominant sources. Global Biogeochemical Cycles. 21, GB2027, doi: 10.1029/2006GB002897 (2007).
Shotyk, W. et al. Trace metals in the dissolved fraction (<0.45 μm) of the lower Athabasca River: Analytical challenges and environmental implications. Sci of the Tot Environ, 10.1016/j.scitotenv.2016.12.012 (2016).
Kelly, E. N. et al. Oil sands development contributes elements toxic at low concentrations to the AR and its tributaries. Proc. Natl. Acad. Sci. USA. 107, 16178–16183 (2010).
CCME: Canadian Environmental Quality Guidelines; Canadian Council of Ministers of the Environment. Winnipeg, MB (2015).
Guéguen, C., Clarisse, O., Perroud, A. & McDonald, A. Chemical speciation and partitioning of trace metals (Cd, Co, Cu, Ni, Pb) in the lower Athabasca river and its tributaries (Alberta, Canada). J. Environ. Monit. 13, 2865–2872 (2011).
Shotyk, W. et al. Sphagnum mosses from 21 ombrotrophic bogs in the Athabasca Bituminous Sands region show no significant atmospheric contamination of “heavy metals”. Environ. Sci. Technol. 48, 12603–12611 (2014).
Krachler, M. & Shotyk, W. Natural and anthropogenic enrichments of molybdenum, thorium, and uranium in a complete peat bog profile, Jura Mountains, Switzerland. J. Environ. Monit. 6, 418–426 (2004).
Wen, L.-S., Santschi, P., Gill, G. & Paternostro, C. Estuarine trace metal distributions in Galveston Bay: importance of colloidal forms in the speciation of the dissolved phase. Mar. Chem. 63, 185–212 (1999).
Stolpe, B., Hassellöv, M., Andersson, K. & Turner, D. R. High resolution ICPMS as an on-line detector for flow field-flow fractionation; multi-element determination of colloidal size distributions in a natural water sample. Anal. Chim. Acta 535, 109–121 (2005).
Neubauer, E., v.d. Kammer, F. & Hofmann, T. Using FLOWFFF and HPSEC to determine trace metal-colloid associations in wetland runoff. Wat. Res. 47, 2757–2769 (2013).
Wright, G. N., McMechan, M. E. & Potter, D. E. G. Structure and architecture of the western Canada sedimentary basin. Canadian Society of Petroleum Geologists. Geological Atlas of the Western Canada Sedimentary Basin. Chapter 3. 1994).
Aubert, H. & Pinta, M. Trace elements in soils. Development in Soil Science 7. Elsevier scientific publishing Co. Amsterdam-Oxford-New York. 395 pp (1977).
Pokrovsky, O. S., Schott, J. & Dupré, B. Trace element fractionation and transport in boreal rivers and soil porewaters of permafrost-dominated basaltic terrain in Central Siberia. Geochim. Cosmochim. Acta. 70, 3239–3260 (2006).
Wedepohl, K. H. The composition of the continental crust. Geochim. Cosmochim. Acta. 59, 1217–1232 (1995).
Rudnick, R. L. & Gao, S. The Composition of the Continental Crust, pp. 1–51, In The Crust (2014), R. L. Rudnick, editor, Treatise on Geochemistry, Second Edition, Volume 4, (eds H. D. Holland & K. K. Turekian ), Elsevier-Pergamon, Oxford 805 p (2014).
Martin, J. M. & Meybeck, M. Elemental mass balance of material carried by major world rivers. Mar Chem. 7, 173–206 (1979).
Savenko, V. S. Chemical composition of world river’s suspended matter, GEOS, Moscow, Russia, 175 pp., ISBN 5-89118-345-5 (2006).
Viers, J., Dupré, B. & Gaillardet, J. Chemical composition of suspended sediments in world rivers: New insights from a new database. Sci. Total Environ. 407, 853–868 (2009).
Timoney, K. P. The Peace-Athabasca Delta: Portrait of a Dynamic Ecosystem. University of Alberta Press, (Date of access:31/10/2016) (2013).
Hart, B. T. & Davies, S. H. R. Trace metal speciation in the fresh-water and estuarine regions of the Yarra River, Victoria. Estuarine, Coastal and Shelf Science. 12, 353–374 (1981).
McDuffie, B., EI-Barbary, I., Hollod, G. J. & Tiberio, R. D. Trace metals in rivers-speciation, transport, and role of sediments. In Proc. Univ. Missouri Annu. Conf. Trace Subst. Environ. Health, Vol. X. D.D. Hemphill (Ed), P. 85–95 (1976).
DeGroot, A. J., Salmons, W. & Allersma, E. Processes affecting heavy metals in estuarine sediments. In. Burton, J. D. & P. S. Liss (Eds.), Estuarine Chemistry. Acdemic Press, London. P. 131–197 (1976).
Yang, Z. et al. Dissolved and particulate partitioning of trace elements and their spatial–temporal distribution in the Changjiang River. J. Geochemical Exploration. 145, 114–123 (2014).
Environment and Climate Change Canada. Air Pollutant Emissions for Alberta, National Pollutant Release Inventory. prepared by Environment Canada, Ottawa, Ontario. http://ec.gc.ca/inrp-npri/donnees-data/index.cfm?do=results&process=true&lang=en&opt_report_year=2014&opt_facility_name=&opt_npri_id=&opt_cas_name=NA+-+08&opt_cas_num=&opt_province=AB&opt_postal_code=&opt_urban_center=&opt_province_comm=&opt_naics6=&opt_naics3=&opt_naics4=&opt_nai6code=&opt_csic=&opt_media=all&submit=Submit (Date of access:31/10/2016).
Guéguen, C. & Cuss, C. W. Characterization of aquatic dissolved organic matter by asymmetrical flow field-flow fractionation coupled to UV-Visible diode array and excitation-emission matrix fluorescence. J. Chrom. A. 1218, 4188–4198 (2011).
Cuss, C. W. & Guéguen, C. Determination of relative molecular weights of fluorescent components in dissolved organic matter using asymmetrical flow field-flow fractionation and parallel factor analysis. Anal. Chim. Acta. 733, 98–102 (2012).
Javed, M. B., Kachanoski, G. & Siddique, T. A modified sequential extraction method for arsenic fractionation in sediments. Anal Chim Acta 787, 102–110 (2013).
Gibson, J. J. et al. Isotopic and Geochemical Tracers for Fingerprinting Process-Affected Waters in the Oils Sands Industry: A Pilot Study. Oil Sands Research and Information Network (OSRIN), University of Alberta, School of Energy and Environment, Edmonton, Alberta, Canada., pp 109 (2011).
Acknowledgements
Funding for this project was provided jointly by AB Innovates (AI), with special thanks to John Zhou and Brett Purdy, and the Canadian Oil Sands Innovation Alliance (COSIA), with special thanks to John Brogly. Thanks to Tommy Noernberg (Mechanical Engineering Technologist, University of Alberta) for devising the precision cutting of filters and for outstanding field support. We are grateful to Mark Donner for field assistance. Thanks to Rick Pelletier (University of Alberta) for preparing the maps for Figures 1 and S1, and to Tracy Gartner and Karen Lund for providing administrative support. Independent confirmation of Pb and Th concentrations in selected samples by Dr. Beatriz Bicalho using ICP-SFMS is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
M.B.J. performed all the experimental work, C.W.C. and I.G.W. performed the dissolved lead fractionation. M.B.J. wrote the manuscript and prepared the figures and tables, C.W.C. provided the AF4-ICP-MS methodology and prepared Figure 3. W.S. designed the study, collected the waters, supervised the experiments and reviewed the manuscript. All authors discussed the results and reviewed the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Javed, M., Cuss, C., Grant-Weaver, I. et al. Size-resolved Pb distribution in the Athabasca River shows snowmelt in the bituminous sands region an insignificant source of dissolved Pb. Sci Rep 7, 43622 (2017). https://doi.org/10.1038/srep43622
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep43622
This article is cited by
-
Monsoon climate controls metal loading in global hotspot region of transboundary air pollution
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.