Abstract
The resource-driven concept, which is an important school for investigating ecosystem production, has been applied for decades. However, the regulatory mechanisms of production by multiple resources remain unclear. We formulated a new algorithm model that integrates multiple resource uses to study ecosystem production and tested its applications on a water-availability gradient in semi-arid grassland. The result of our experiment showed that changes in water availability significantly affected the resources of light and nitrogen, and altered the relationships among multiple resource absorption rate (ε), multiple resource use efficiency (mRUE), and available resource (Ravail). The increased water availability suppressed ecosystem mRUE (i.e., “declining marginal returns”); The changes in mRUE had a negative effect on ε (i.e., “inverse feedback”). These two processes jointly regulated that the stimulated single resource availability would promote ecosystem production rather than suppress it, even when mRUE was reduced. This study illustrated the use of the mRUE model in exploring the coherent relationships among the key parameters on regulating the ecosystem production for future modeling, and evaluated the sensitivity of this conceptual model under different dataset properties. However, this model needs extensive validation by the ecological community before it can extrapolate this method to other ecosystems in the future.
Similar content being viewed by others
Introduction
Biophysical regulations and resource-driven investigations are the two primary approaches for modeling the magnitudes and dynamics of ecosystem production and understanding its mechanisms1. In general, ecosystem production is influenced by the complex interactions of biology, environment and disturbances2 (i.e., biophysical regulation). If based on economic principles, ecosystem production can be also interpreted as an output of carbohydrates and other organic compounds synthesized from the input source of water, CO2, and nutrients3 (i.e., resource-driven concept). To continue the analogy, various available resources are treated as “investments”, gross primary production (GPP) as “revenue” and the net primary production (NEP) as “profit”. In practice, one perused a maximized profit would tend to get more production under the limited multiple resources4. However, in recent decades, ecosystem studies have been predominantly conducted with empirical, theoretical, or modeling endeavors based on biophysical regulations from various processes, whereas resource-driven investigations lag significantly behind.
Extensive research has demonstrated that resource use efficiency varies by resource type (e.g., nutrient, light, and water), ecosystem type, physical condition (e.g., soil and climate) and location, with uneven consequences on ecosystem production5,6,7. In general, a highly available resource associated with more production is expected to possess either a high or near constant resource use efficiency. A previous study across biomes indeed verified that high rainfall (i.e., high water availability) contributes to greater dry matter given a certain amount of absorbed resources5. For example, forests generally have higher water-use efficiency (WUE) than grasslands8. This phenomenon has also been reported in many ecosystem-scale studies, with most focusing on available light and nitrogen in forests. Light-use efficiency (LUE) is highly correlated with photosynthetically active radiation (PAR)9. Plants in fertile soils show stronger responses to nutrient addition (i.e., nutrient availability) than those in infertile soils10, resulting in a higher nitrogen-use efficiency (NUE)11 and consequently promoting ecosystem production12,13. However, other studies have reported an inverse response of resource use efficiency (RUE) to available resources—a phenomenon known as “declining marginal returns” that describes that the increased rate of ecosystem production (i.e., RUE), due to the addition of a single resource, will progressively decline compared to the previous rate. For example, elevated precipitation (i.e., water availability) can substantially lower the WUE in grasslands14 and wheat15, and the enhanced nitrogen supply can significantly reduce NUE in a forest plantation16. In tropical montane forests, instantaneous phosphorus-use efficiency (PUE) decreases with the gradual increase in phosphorus17. Clearly, the ecosystem production is often regulated by multiple resources, and the single resource use process was generally constrainted by the other resources. Therefore, the availability and efficiency of an individual resource may not reflect the overall RUE for any ecosystem.
Multiple resources usually change synchronously4; the changes in one resource will likely induce changes in other resource uses at leaf, species, and community levels and at different temporal scales. Using the experimental data from Acacia auriculiformis, Phillips and Riha18 demonstrated that drought stress can reduce absorbed photosynthetically-active radiation (aPAR) and LUE, but can enhance WUE, implying that, at the annual scale, the shift in one resource availability would cause disproportionate consequences on other resource use efficiency. Doubled CO2 concentration can increase annual LUE and NUE with different magnitudes, the LUE being higher in the later than early stage of canopy, while NUE being greater in early than later stage at community scale19. At shoot scale, great transpiration tends to promote NUE, but reduce WUE and LUE in a mixed coniferous stand20. Similarly, elevated temperature can increase LUE by 8–9% but reduce WUE by 19–34% in a pure Scots pine (Pinus sylvestris) stand21. These conditional relationships among multiple resources also exist at species level. Field et al.22 reported an inverse relationship between photosynthesis NUE and photosynthesis WUE in evergreen vegetation; and Reich et al.23 found significantly negative relationships between photosynthesis NUE and WUE in an elm stand. Nevertheless, these studies fail to systematically explore the influence paths or mechanisms about how one resource, use efficiency, affect the others by including intermediate variables (e.g., absorbed light, soil water or nitrogen), when the ecosystem was directly triggered by one resource change.
The economic principle in ecosystem production emphasizes a higher production return with a relatively low investment. A greater multiple RUE (mRUE) does not always warrant high production. For example, declined water availability in a semi-arid grassland generally promote WUE, but ecosystem production does not increase as expected14. As the result, we infer that the magnitudes and dynamics of production probably depend on the independent and interactive effects of multiple resource absorption rate (ε) and mRUE. From the viewpoint of plant physiology, an increase in water availability in soil would probably enhance the water uptake and soluble materials (e.g., nitrogen) of the plant through transpiration pull, leading to a higher ε. Meanwhile, the mRUE may be reduced if the amount of absorbed resources surpasses the demand of plants24, suggesting that the increased ε combined with the decreased mRUE may still enhance plant production. Previous studies have documented that the increased ε of water25 and nitrogen26 would stimulate mRUE, suggesting that ε and mRUE may not independently regulate ecosystem production. Unfortunately, few studies have examined the complex causal relationships between ε and mRUE in mediating ecosystem production. Distinguishing the independent and interactive effects of ε and mRUE in regulating ecosystem production will help to fill these knowledge gaps.
The objective of this study was to establish a new conceptual model by integrating multiple resources on ecosystem production, quantify the dynamics of available resources, ε, and mRUE, and promote the potential applications of mRUE in ecosystem ecology. In this scientific adventure for understanding the relations between mRUE and ecosystem production, the field data from a water-availability gradient with increased and decreased precipitation in a semi-arid grassland on the Mongolia Plateau were used to illustrate the model applications. The specific questions of this study were: (i) how does water availability affect other resources and contribute to the variations of ε and mRUE; and (ii) is there a causal relation between ε and mRUE in regulating the production?
Developing the multiple-resource use efficiency (mRUE) model
Environmental resources have long been recognized as one of the important proxies regulating ecosystem production. The LUE model utilizes the principle of fixed carbohydrate as output energy from solar radiation in estimating gross primary production27:

where GPP is gross primary production, fPAR is the fraction of absorbed PAR (MJ m−2) by vegetation, PAR is incident photosynthetically active radiation (MJ m−2) during a certain time period, εmax is potential LUE (gC m−2 MJ−1) that can be scaled to a range of 0–1 using plant caloric values, and f is a scalar limiting εmax from other environmental drivers.
Based on the Liebig Law, which states that ecosystem production is regulated by limited cues, light resource is not expected to be the sole driving force in some ecosystem types. Binkley et al.25 synthesized published results from a Eucalyptus plantation, extending the concept of RUE to include water, nutrients and light use as:

These two schools of thought, although expressed differently, have many similarities and can be united under RUEi when individual resource is considered. RUEi as the accumulated dry matter per unit of absorbed multiple resources. Production is often expressed as the dry matter, such as GPP, net primary production (NPP), or aboveground net primary production (ANPP). Using annual ANPP as an example, RUEi can be modeled as:

where Ri is the amount of absorbed resources within a year. RUEi can be applied to specific terms, such as water (WUE = ANPP:Tr, where Tr is the transpiration), nitrogen (NUE = ANPP:Nuptake, where Nuptake is the absorbed nitrogen amount), and light (LUE = ANPP:aPAR, where aPAR is the absorbed PAR).
The absorbed resources (Ri) can be further expressed as two quantitative variables, Ravail and εi as follow:

Combining Eq. (4) with Eq. (3), we have:

where ANPP is regulated by an individual resource, such as light, water and nitrogen:



where PAR, Ms and Ns are accumulated available resources at the annual scale. Ms is the accumulated soil water content. Ns is the accumulated available nitrogen in soil (e.g., NO3−, NH4+), and this variable was substituted by soil total nitrogen content in this study. εL, εw, and εN are ratios of absorbed light, water, and nitrogen, respectively. RUEi and εi are the ratios with a range of 0–1. The units of PAR and aPAR can be converted to g m−2, if divided by plant caloric values. The units of other variables, except RUEi and εi, are also in g m−2. In this study, we used 19.38 kJ g−1 as the caloric value in a semi-arid grassland28 (see the supplemental materials for details).
When more resources are involved, ANPP is regulated as:

where εi = Tr:Ms = Nuptake:Ns = aPAR:PAR. In Eq. (9), the integrated term of multiple RUEi (i.e., (RUE1 × RUE2 × …RUEn)1/n) refers to mRUE, and that of multiple εi (i.e., (ε1 × ε2 × …εn)1/n) refers to ε. Compared with Binkley’s model, our multiple resource model (Eq. (9)) included the interactions among the drivers and constraints of one resource on another. Another advantage of this model is that these resources may interactively determine ANPP, although one or some resources may dominate the magnitudes and directions of others as these resources are limiting factors (Fig. 1). We used the data collected in a field experiment with precipitation manipulation in semi-arid temperate grassland to test the potential use of this new mRUE model (Eq. 9). The major acronyms were shown in Table 1.
The conceptual framework of this study.
Within the matrix of the bio-physical environment (microclimate and disturbance), the magnitude of aboveground net primary productivity (ANPP) is determined by the resource use matrix of [ε, mRUE, Ravail] and their complex interactions. For each type of resource, there exists complex interactions among [ε, mRUE, Ravail] at various temporal scales. Alteration of any element of the resource use matrix will trigger changes in other elements. This study will examine the feedbacks among the elements, with a focus on water, light, and nitrogen.
Coherent relationships among the key parameters
To examine the complex interactions among the elements of multiple resources (i.e., ε, mRUE, Ravail), a non-recursive structural equation model (SEM) for our experimental data in the semi-arid grassland (Fig. 4a without coefficients) was constructed to test four specific hypotheses:
-
1
In a water-limited environment, increased available water significantly stimulates absorbed light (Ms → aPAR)29. Associated with the process of water uptake by roots, the mass flow of mineral nutrient simultaneously transplant to different organs in plants (Tr → Nuptake). The absorbed nutrient usually acts as catalyst (at the scale of leaves and seconds) and maintains nutrient demand for plant growth (at individuals and community levels), leading to more photosynthetic products, which will indirectly increase the absorbed PAR (Nuptake → aPAR)12.
-
2
ε is a function of absorbed resources and available resources and, thus, is significantly affected by available soil water (Ms → ε) and by absorbed water, light, and nitrogen (Tr → ε; aPAR → ε; Nuptake → ε)25. At the ecosystem level, ε is expected to have significant effects on mRUE (ε → mRUE).1
-
3
The economic principles of “declining marginal returns”, which describes the decline of productivity or dry matter per unit of absorbed (mRUE) when the supply of single resource increases, can be applied for explaining mRUE (Ms → mRUE; aPAR → mRUE; Nuptake → mRUE)25,30,31.
-
4
There may exist a feedback between RUE and ε, which is responsible for the changes in production (mRUE → ε).
The left panel was the non-recursive SEM model. All the variables are observed indicators (in boxes).
Dash line referred to insignificant correlation between these two variables, “NS” meant insignificance. A set of model performance indexes, such as λ = 2.412 (P = 0.299, df = 2), GFI = 0.994, AGFI = 0.993, NFI = 0.997, RMSEA = 0.041, and stability index = 0.025, suggested that this model was fitted well with our data. The determination coefficient (R2) represented the overall explanation on this dependent variable, and the other coefficients were correlation coefficients (r). We should note that these coefficients were calculated based on the experimental data (after log-transformed), so the spurious correlations were unavoidable. Thus, the right panel pointed out these spurious determination coefficients when the X and Y variables are non-independent.
Resource use efficiency among ecosystems
Our case study in the semi-arid grassland of the Mongolia Plateau showed that there were lower use efficiencies of water, light, and nitrogen than those found in most ecosystems, with the mean WUE, LUE and NUE values in the control plots of 0.05%, 1.2%, and 62.47, respectively (Table 2). The WUE in this system was significantly lower than that in a forest at the annual scale32 and in some grasslands at daily scales33, but it was close to the maximum WUE of 0.042% in the driest years for arid ecosystems34. In other words, the annual mean WUE of this arid grassland was close to the potential maximum WUE of arid biomes, further supporting the consensus on water sensitivity (i.e., WUE) in these water-limited ecosystems. However, light did not appear to be limiting production in this semiarid grassland. In addition, there appeared a lower LUE than that in forests5,29, croplands3, and in other grasslands35. The NUE in our system was substantially higher than that in crops, likely because the overcropped and unsuitable management practices in crops might have reduced available nutrients and aggravated leaching processes36. The NUE in this grassland was also lower than those in a forest31 and in a semi-arid grassland on the Mongolia Plateau37.
Complex interactions among the elements of mRUE
Water availability was responsible for the large changes in resource use and efficiency in the semi-arid grassland, including ε, mRUE, and Ravail in this study. Experimental manipulation with both increased and decreased precipitation constituted for a wide spectrum of water availability, ranging from 70 mm to 345 mm prior to measuring ANPP, producing significant changes in water, light, and nitrogen resources. Among these available or absorbed resources, Pearson correlations revealed that Nuptake had a linear relationship with Tr (Fig. 2g, R2 = 0.09). aPAR showed significantly positive correlations with Nuptake (Fig. 2h, R2 = 0.21) and Ms (Fig. 2i, R2 = 0.45), with a stronger effect on Ms than Nuptake, suggesting a greater response of aPAR to water than nitrogen resource. These correlations indicated positive relationships among these three resources. However, this conclusion was inconsistent with some previous studies, where one resource was reported to have opposite correlations with other resources22. As for ε and mRUE, ε was linearly correlated with the absorbed light (aPAR; Fig. 2a, R2 = 0.55, Fig. 4b, spurious R2 = 0.16), nitrogen (Nuptake; Fig. 2b, R2 = 0.69, Fig. 4b, spurious R2 = 0.32) and water (Ms: Fig. 2c, R2 = 0.22, Fig. 4b, spurious R2 = 0.04; Tr: Fig. 2d, R2 = 0.52, Fig. 4b, spurious R2 = 0.10) by plants. mRUE also showed linear dependence upon the absorbed nitrogen (Nuptake; Fig. 2e, R2 = 0.44, Fig. 4b, spurious R2 = 0.038) and water (Ms; Fig. 2f, R2 = 0.07).
When distinguishing the relative contributions of ε and mRUE to ANPP, we found that ε and mRUE had dominant roles in regulating ANPP under all the treatments, with greater contribution from mRUE than that from ε (Fig. 3a). WUE, LUE, and NUE accounted for, on average, 47.55%, 37.90%, and 14.55% of the variations of mRUE across the treatments, respectively (Fig. 3b). These results suggest that the resource absorption ratio and efficiency were critical factors in determining ecosystem production, and that the driving factor (i.e., water resource) always produce a main effect on multiple RUE.
Tradeoff effects of ε and mRUE on production
The water-driven changes in other two resources (i.e., light and nitrogen) significantly affected ε (Fig. 4, Table 3). Plant transpiration directly promoted ε (β = 0.43, r = 0.71, spurious R2 = 0.1 × 28% = 0.028) but was greatly offset by the direct effect of available water (β = −0.15, r = 0.44, spurious R2 = 0.04 × 5% = 0.002), contributing to a total effect of 0.28 (Table 3: β = 0.43 + (−0.15) = 0.28, spurious R2 = 0.03). In addition to the direct effects, water resource indirectly stimulated ε via regulating absorbed light and nitrogen (Table 3: β = 0.20 + 0.25 = 0.42, spurious R2 = 0.062). Therefore, there was a greater contribution from water-induced changes in light and nitrogen than that from the water resource directly. In addition, water, light and nitrogen resources collectively explained 75% (96–19% (spurious R2) = 77%, Fig. 4b) of the variations of ε. Our results are consistent with previous studies that water addition may significantly enhance leaf area index of forest canopy13 and aPAR29. However, these studies emphasize the ε responses to individual resources only, which would lead to inconsistent results from multiple resources. In conclusion, understanding the influence paths and processes of the multi-resource absorption rate would be necessary to comprehensively characterize and explore the change dynamics and control mechanisms over ε.
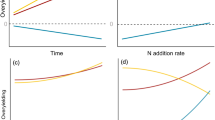
We confirmed our third hypothesis regarding the application of “declining marginal returns” in modeling ecosystem production from multiple resource use efficiency (Fig. 4). This concept was initially proposed for modeling the instantaneous photosynthesis process at leaf level by Ögren and Evans38, who hypothesized that the increase in the photosynthesis rate would decrease with incident light exceeding the demand (i.e., decreases on instantaneous energy-use efficiency, i.e. LUE). In our study, we found that available water directly reduced mRUE (Table 3: β = −0.49, Fig. 4a: r = −0.28) and transpiration indirectly promoted mRUE via increasing plant nitrogen content (i.e., Nuptake, β = 0.32×0.66 = 0.21, spurious R2 = 0.013). The total effects of water resource eventually reduced mRUE by 28% (Table 3: β = −0.49 + 0.21 = −0.28), jointly contributing to about 50% of the total variations when we excluded the spurious contribution (mean spurious R2 = 0.042, Fig. 4b). Our results demonstrated a response from mRUE to an individual resource that was similar to the responses of individual RUEs found in literature. Nevertheless, our conclusion may not be extrapolated to all ecosystem types, particularly for forests. For example, Binkley et al.39 reported that a more productive site had higher absorption rate and resource use efficiency in an Eucalyptus plantation. Also, Balster and Marshall12 reported that fertilization promoted the ratio of converted stemwood in Douglas-fir stands (Pseudotsuga mensizii). Thus, our mRUE model—an alternative for characterizing the relationships between resource use efficiency and available resources—would be ecosystem specific, because of ecosystem-specific regulations and the complex interactions with other biophysical drivers.
The ε and mRUE presented different distributions along the water availability gradient. The ε increased significantly along the water availability gradient; but over three years, it showed distinct levels (Fig. 5a). mRUE did not show a similar trend with ε, but had different probability distributions (i.e., density) created by different ranges of water-availability (Fig. 5b). Moreover, mRUE had a negative correlation with ε (Fig. 5c).
This negative correlation did not imply that water-induced changes in ε had significant influences on mRUE; but instead, mRUE indeed produced a negative effect on ε (i.e., one-way causality) (Fig. 4a). This phenomenon is in agreement with the classical economic principle of “inverse feedback”40. In our study, the semi-arid ecosystem encountered intense stress due to water availability, the mRUE, as one of the regulatory factors of ecosystem production, would represent a negative feedback to other biological cues (e.g., ε) to alleviate its fluctuation on ecosystem production. Specifically, water resource had a significantly positive influence on ε (Table 3: β = (−0.15) + 0.20 + 0.43 + 0.25 = 0.73, spurious R2 = 0.092) and a negative effect on mRUE (Table 3: β = (−0.49) + 0.21 = −0.28, spurious R2 = 0.013), suggesting that the increase in water resource (i.e., Ms and Tr) can reduce mRUE dramatically, but enhance ε. In addition, ε was further elevated by the negative effects from mRUE (mRUE → ε, β = −0.11) to ease the negative changes of mRUE on production and ultimately promoted the production under the increased water availability. These tradeoff effects between ε and mRUE determined that the increased single resource availability would promote ecosystem production rather than decrease it, even when the mRUE reduced. Thus, the processes of “declining marginal returns” and “inverse feedback” jointly explain this phenomenon in nature. For example, forests are always more productive than grasslands, and fertile soil is always more productive than infertile soil for a given ecosystem.
Sensitivity analysis on mRUE model
Quantifying the potential spurious correlations among the elements of mRUE will increase the effectiveness of our mRUE model. In this study, we calculated the range of spurious determination coefficients of a manipulative experiment (Fig. 6a). We hypothesized some situations for the dataset that mRUE model will encounter in future modelling (Fig. 6b–d). The standard deviation (SD) of common (A) and independent variables (B) were both needed for calculating ε and mRUE in all analyses of our Monte Carlo test. The spurious correlations about our manipulative experiment had embedded in the above description (Fig. 6a). As for the possible situations of dataset in the future modelling, we found that there were no significant influences among the different SDs of both A and B (SD = 1, 2, 5) on the relationships between the common variable (A) and ε (Fig. 6b left panel). There was a significant effect between SD of 5 and the other SDs (1, 2) on the relationships between the common variable (A) and mRUE (Fig. 6b right panel). When the SD of B was two, five or ten times that of the SD of A, there were significant differences in the range of the spurious determination coefficients among T2, T5 and T10 situations (Fig. 6c). When holding the SD of A and B as constant with random sample sizes of 500–10,000, we found that the sample size produced identical means while yielding a lower variability of spurious determination coefficients along the sample size gradient (Fig. 6d). Thus, the bivariate correlations involving spurious correlations should not be arbitrarily considered meaningless, but should be dealt with case by case.
The spurious determination coefficient (R2) in the experiment (a) and several possible situations of SEM model (b–d) using stochastic simulations. All simulations ran 1000 times. Box-and-whisker plots represented the median (horizontal bar), mean (triangle), the 25th and 75th percentiles (box range), 10th and 90th percentiles (whisker), and the maximum and minimum outliers (point) of their distributions. The different lower case letters meant significant differences, and the same one or two letters meant insignificant difference between them. A variable was set as the common variable in ε or mRUE, B was set as the independent variable for calculating ε or mRUE. In subplot (a) we used normal random numbers, which have an identical mean, standard deviations (SD) and sample size (n = 126) as our experiment. In subplots (b,c), we used the normal random number with the sample size of 200 and mean of 20. Additionally, we set the initial SD of A as 1. In subplot (b), the varied ranges of spurious R2 were produced when the standard deviations of both A and B variables were multiple times of initial SD(A). For example, T2 means both SD of A and B were 2 in (b) subplot. In subplot (c), the SD of B was multiple times that of the initial SD (A). In subplot (d), varied sample size from 500 to 10,000 with an SD of A and B were 1. For example, 1000 means 1000 samples.
Take-Home Message
This study was designed to refine a fundamental concept regarding production in ecosystem studies. Following a newly proposed multiple resource use efficiency model (mRUE), we used data from a manipulative experiment to assess the performance of key variables by addressing several key issues. With the experimental data, we found that the water-driven changes on water, light, and nitrogen resources had profound impacts on ecosystem production. The increased available water suppressed ecosystem mRUE (i.e., “declining marginal returns”). The variations of mRUE caused significant negative effects on ɛ (i.e., “inverse feedback”). This mRUE model will facilitate the explanation of the resource uses and ecosystem production in semi-arid grasslands, providing us with promising evidences for broader applications of the mRUE in modelling ecosystem production.
The test-bed and data
The experiment was located in the Duolun Restoration Ecology Station of Inner Mongolia (42°02′N, 116°17′E, 1324 m a.s.l.). This region has a continental monsoon climate. The annual mean temperature is 2.2 °C. The average annual precipitation was about 380 mm, with 67% of annual precipitation occurring in the growing season (May–October). There are enough light, but lacked water and nutrient in soil.
The water-availability gradient experiment was conducted in 2010. This experiment was a randomized block design with seven levels of precipitation treatments: −60% (P−6), −40% (P−4), −20% (P−2), ambient precipitation as a control (CK), +20% (P + 2), +40% (P + 4), and +60% (P + 6), with six replicates for each treatment. The treatments were applied to a total of 42 plots (seven levels and six replications), with 4 × 4 m2 for each plot. The intercepted rainfall by rainout shelters was collected to water to the increased-precipitation plots. More details about the measurements were referred to the “Supplementary Information” section.
The relative importance analyses (function “calc.relimp” in “relaimpo” package in R) were performed to test the relative importance values on mRUE, ε and ANPP. Prior to our SEM analysis, all variables were calculated for their growing season accumulations (i.e., annual accumulations, see “Supplementary Information”). Non-recursive SEM was constructed to test how the water-driven variables of Ms and Tr affected light and nitrogen, and synthetically affected the multiple resource ratio and efficiency. Prior to the modeling, all variables were log-transformed to assure the homogeneity of variance. In addition, univariate and multivariate normality analysis were performed to meet the model assumptions before SEM analysis using R functions (“skewness” and “kurtosis” in the “moments” package, and “mardia” in the “psych” package, Supplementary Table S1). The maximum likelihood procedure was used to estimate the regression coefficients, variances, covariance and correlations in our SEM. Final model selections were made by the comprehensive results of λ2 (P > 0.05), GFI (>0.9), AGFI (>0.9) and RMSEA (<0.05). The package of AMOS 17.0 in SPSS was used for our SEM analysis.
Our mRUE model was developed to evaluate ecosystem productivity by involving multiple resources (e.g., light, potassium and phosphor) in an effort of exploring the complex interactions among elements. However, some bivariate analyses would have spurious correlations due to the shared elements (e.g., aPAR and ε), which would magnify the true relationships among them. Consequently, one needs to quantify the contributions of spurious correlations and separate them from the total41. In our study, Monte Carlo simulations were performed to quantify the spurious correlations. All the simulations were conducted 1,000 times. In our SEM model, we generated the normal random numbers by using a random number generator (function “rnorm” in R) with a sample size of 126 for mean ± SD of aPAR (10 ± 0.3), PAR (11 ± 0.04), Tr (13 ± 0.3), Ms (15 ± 0.2), Nuptake (1.2 ± 0.05), Navail (3.7 ± 0.17) and ANPP (5.3 ± 0.4) in our experiment. Pearson correlations were performed to calculate the range of the spurious determination coefficient (spurious R2). In addition, the relative contributions of each common variable on ε or mRUE were calculated by the relative importance analyses (function “calc.relimp” in “relaimpo” package in R) to calculate the total spurious contributions of all common variables on ε or mRUE. We also performed stochastic simulations to assess the sensitivity of mRUE to spurious correlation by changing: (i) both SD of the common variable (A) and the independent variables (B); (ii) the SD of (B) as the multiple times of SD (A); and (iii) sample sizes of 500, 1000, 2000 and 10000 with a constant SD on A and B.
Additional Information
How to cite this article: Han, J. et al. Multiple Resource Use Efficiency (mRUE): A New Concept for Ecosystem Production. Sci. Rep. 6, 37453; doi: 10.1038/srep37453 (2016).
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Waring, R. H. & Running, S. W. Forest ecosystems: analysis at multiple scales. Second edition. 1–16 (1998).
Chen, J. et al. Biophysical controls of carbon flows in three successional Douglas-fir stands based on eddy-covariance measurements. Tree Physiol. 22, 169–177 (2002).
Monteith, J. L. Climate and efficiency of crop production in Britain. Phil Trans Roy Soc London B. 281, 277–294 (1977).
Bloom, A. J., Chapin, F. S. & Mooney, H. A. Resource limitation in plants - an economic analogy Annu Rev Ecol Syst. 16, 363–392 (1985).
Garbulsky, M. F. et al. Patterns and controls of the variability of radiation use efficiency and primary productivity across terrestrial ecosystems. Global Ecol Biogeogr. 19, 253–267 (2010).
Niu, S. et al. Water‐use efficiency in response to climate change: from leaf to ecosystem in a temperate steppe. Glob Change Biol. 17, 1073–1082 (2011).
Brummer, C. et al. How climate and vegetation type influence evapotranspiration and water use efficiency in Canadian forest, peatland and grassland ecosystems. Agr Forest Meteorol. 153, 14–30 (2012).
Xiao, J. et al. Carbon fluxes, evapotranspiration, and water use efficiency of terrestrial ecosystems in China. Agr Forest Meteorol. 182, 76–90 (2013).
Yebra, M., Van Dijk, A. I. J. M., Leuning, R. & Guerschman, J. P. Global vegetation gross primary production estimation using satellite-derived light-use efficiency and canopy conductance. Remote Sens Environ. 163 (2015).
Chapin III, F. S., Vitousek, P. M. & Van Cleve, K. The nature of nutrient limitation in plant communities. Amer Naturalist. 48–58 (1986).
Hiremath, A. J., Ewel, J. J. & Cole, T. G. Nutrient use efficiency in three fast-growing tropical trees. For. Sci. 48, 662–672 (2002).
Balster, N. J. & Marshall, J. D. Eight-year responses of light interception, effective leaf area index, and stemwood production in fertilized stands of interior Douglas-fir (Pseudotsuga menziesii var. glauca). Can J Forest Res. 30 (2000).
Albaugh, T. J., Allen, H. L., Dougherty, P. M. & Johnsen, K. H. Long term growth responses of loblolly pine to optimal nutrient and water resource availability. Forest Ecol Manag. 192, 3–19 (2004).
Bai, Y. F. et al. Primary production and rain use efficiency across a precipitation gradient on the Mongolia plateau. Ecology 89, 2140–2153 (2008).
Lauenroth, W. K., Burke, I. C. & Paruelo, J. M. Patterns of production and precipitation-use efficiency of winter wheat and native grasslands in the central Great Plains of the United States. Ecosystems 3, 344–351 (2000).
Aerts, R. & Decaluwe, H. Nitrogen use efficiency of carex species in relation to nitrogen supply. Ecology 75, 2362–2372 (1994).
Hidaka, A. & Kitayama, K. Divergent patterns of photosynthetic phosphorus-use efficiency versus nitrogen-use efficiency of tree leaves along nutrient-availability gradients. J Ecol. 97, 984–991 (2009).
Phillips, J. G. & Riha, S. J. Canopy development and solar conversion efficiency in acacia-auriculiformis under drought stress. Tree Physiol. 12, 137–149 (1993).
Hirose, T. & Bazzaz, F. Trade-off between light-and nitrogen-use efficiency in canopy photosynthesis. Ann Bot. 82, 195–202 (1998).
Tarvainen, L., Raentfors, M. & Wallin, G. Seasonal and within-canopy variation in shoot-scale resource-use efficiency trade-offs in a Norway spruce stand. Plant Cell Environ. 38, 2487–2496 (2015).
Wang, K., Kellomäki, S., Li, C. & Zha, T. Light and Water‐use Efficiencies of Pine Shoots Exposed to Elevated Carbon Dioxide and Temperature. Ann Bot. 92, 53–64 (2003).
Field, C., Merino, J. & Mooney, H. A. Compromises between water-use efficiency and nitrogen-use efficiency in five species of California evergreens. Oecologia 60, 384–389 (1983).
Reich, P. B., Walters, M. B. & Tabone, T. J. Response of Ulmus americana seedlings to varying nitrogen and water status. 2 Water and nitrogen use efficiency in photosynthesis. Tree Physiol. 5, 173–184 (1989).
Chapin, F. S., Vitousek, P. M. & Vancleve, K. The nature of nutrient limitation in plant-communities. Amer Naturalist. 127, 48–58 (1986).
Binkley, D., Stape, J. L. & Ryan, M. G. Thinking about efficiency of resource use in forests. Forest Ecol Manag. 193, 5–16 (2004).
Stape, J. L., Binkley, D. & Ryan, M. G. Eucalyptus production and the supply, use and efficiency of use of water, light and nitrogen across a geographic gradient in Brazil. Forest Ecol Manag. 193, 17–31 (2004).
Landsberg, J. J. & Wright, L. L. Comparisons among populus clones and intensive culture conditions, using an energy-conversion model. Forest Ecol Manag 27, 129–147 (1989).
Chen, Z. & Zhang, H. The caloric values of the steppe plants in the typical steppe zone of Inner Mongolia. Chinese Bulletin of Botany, 26–27 (1993).
Allen, C. B., Will, R. E., McGarvey, R. C., Coyle, D. R. & Coleman, M. D. Radiation-use efficiency and gas exchange responses to water and nutrient availability in irrigated and fertilized stands of sweetgum and sycamore. Tree Physiol. 25, 191–200 (2005).
Birk, E. M. & Vitousek, P. M. Nitrogen availability and nitrogen use efficiency in loblolly pine stands. Ecology 67, 69–79 (1986).
Harrington, R. A., Fownes, J. H. & Vitousek, P. M. Production and resource use efficiencies in N- and P-limited tropical forests: A comparison of responses to long-term fertilization. Ecosystems 4, 646–657 (2001).
Stape, J. L. Production ecology of clonal Eucalyptus plantations in northeastern Brazil, Colorado State University (2002).
Hu, Z. et al. Effects of vegetation control on ecosystem water use efficiency within and among four grassland ecosystems in China. Glob Change Biol. 14, 1609–1619 (2008).
Huxman, T. E. et al. Convergence across biomes to a common rain-use efficiency. Nature 429, 651–654 (2004).
Propastin, P. A., Kappas, M. W., Herrmann, S. M. & Tucker, C. J. Modified light use efficiency model for assessment of carbon sequestration in grasslands of Kazakhstan: combining ground biomass data and remote-sensing. International Journal of Remote Sensing 33, 1465–1487 (2012).
Berendse, F. & Aerts, R. Nitrogen-use-efficiency: a biologically meaningful definition? Funct Ecol. 1, 293–296 (1987).
Yuan, Z. Y. et al. Nitrogen response efficiency increased monotonically with decreasing soil resource availability: a case study from a semiarid grassland in northern China. Oecologia 148, 564–572 (2006).
Ögren, E. & Evans, J. Photosynthetic light-response curves. Planta 189, 182–190 (1993).
Binkley, D., Campoe, O. C., Gspaltl, M. & Forrester, D. I. Light absorption and use efficiency in forests: Why patterns differ for trees and stands. Forest Ecol Manag. 288, 5–13 (2011).
Turrigiano, G. Homeostatic signaling: the positive side of negative feedback. Curr Opin Neurobiol. 17, 318–324 (2007).
Brett, M. T. When is a correlation between non‐independent variables “spurious”? Oikos 105, 647–656 (2004).
Running, S. W., Thornton, P. E., Nemani, R. & Glassy, J. M. Global terrestrial gross and net primary productivity from the earth observing system. Methods in Ecosystem Science. 44–57 (2000).
Cassman, K. G. et al. Opportunities for increased nitrogen-use efficiency from improved resource management in irrigated rice systems. Field Crop Res. 56, 7–39 (1998).
Hiremath, A. J. & Ewel, J. J. Ecosystem nutrient use efficiency, productivity, and nutrient accrual in model tropical communities. Ecosystems 4, 669–682 (2001).
Acknowledgements
This study were partially supported by the Ministry of Science and Technology of China (2013CB956300), the US Department of Energy’s Great Lakes Bioenergy Research Center (DOE Office of Science, DE-FC02-07ER64494), the “Dynamics of Coupled Natural and Human Systems (CNH)” Program of the NSF (#1313761), and the Natural Science Foundation of China (31500403). We thank Shiping Chen for providing climatic data from an eddy covariance tower near our experiment and Gabriela Shirkey for editing the language of the manuscript.
Author information
Authors and Affiliations
Contributions
S.W. designed the experiment, Y.W. made the experiment, J.C. developed the mRUE model, J.H. proposed the scientific questions, analyzed the data and wrote the manuscript. S.W., J.C. and J.H. revised this manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Han, J., Chen, J., Miao, Y. et al. Multiple Resource Use Efficiency (mRUE): A New Concept for Ecosystem Production. Sci Rep 6, 37453 (2016). https://doi.org/10.1038/srep37453
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep37453
This article is cited by
-
The Shifting Role of mRUE for Regulating Ecosystem Production
Ecosystems (2020)
-
Enhanced peak growth of global vegetation and its key mechanisms
Nature Ecology & Evolution (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.