Abstract
The appearance and expansion of C4 plants in the Late Cenozoic was a dramatic example of terrestrial ecological change. The fire hypothesis, which suggests fire as a major cause of C4 grassland is gaining support, yet a more detailed relationship between fire and vegetation-type change remains unresolved. We report the content and stable carbon isotope record of black carbon (BC) in a sediment core retrieved from the northeastern equatorial Pacific that covers the past 14.3 million years. The content record of BC suggests the development process of a flammable ecosystem. The stable carbon isotope record of BC reveals the existence of the Late Miocene C4 expansion, the ‘C4 maximum period of burned biomass’ during the Pliocene to Early Pleistocene, and the collapse of the C4 in the Late Pleistocene. Records showing the initial expansion of C4 plants after large fire support the role of fire as a destructive agent of C3-dominated forest, yet the weak relationships between fire and vegetation after initial expansion suggest that environmental advantages for C4 plants were necessary to maintain the development of C4 plants during the late Neogene. Among the various environmental factors, aridity is likely most influential in C4 expansion.
Similar content being viewed by others
Introduction
Various lines of evidence such as stable carbon isotope data1,2, phylogenetic structure3, fossil phytoliths4, and macrofossils5 show an abrupt expansion of C4 plants during the Late Neogene, yet the cause of this ecological event is controversial. Decreased atmospheric CO2 concentration1,6,7, climate drying4,8,9,10, and fire11,12 have been suggested as possible causes. Among these, the fire hypothesis is gaining support from African vegetation modeling13 and sharp increase in charcoal flux in ocean sediment cores from the North Pacific14 and Atlantic15,16 Oceans during the Late Miocene. These studies focused mainly on matching the timing of fire with the initial C4 grassland expansion event, but they did not provide further comparison between the two events.
Black carbon (BC), defined as a carbon continuum formed by incomplete combustion of fossil fuels and plant materials, includes many different carbon compounds such as soot, charcoal, and other pyrogenic carbons17. Soot has the lowest reactivity and a very long tracer range of up to 1000 s of km18, making it an important fraction of marine sedimentary organic carbon19,20. The BC in deep-sea sediment is not affected by coastal or shelf processes or other disturbances. It is assumed that BC of pre-industrial periods was formed mainly by fire events of natural vegetation and that the amount of BC can provide the scale of fire that occurred in the continents upwind. Additionally, the stable carbon isotope composition of BC preserves the characteristics of the original plant biomass21,22. Thus, the BC record in deep-sea sediments has the potential to provide continuous information about long-time interactions between fire and vegetation.
In this study, we use BC in deep-sea sediments retrieved from the northeastern equatorial Pacific Ocean (Fig. 1) as a proxy to reconstruct the terrestrial vegetation and fire history for the past 14.3 million years. First, we present the content of BC, which is related to the scale of fire events. Then, we present the carbon isotope composition of BC to reconstruct paleovegetation history. The studied BC record can be divided into two time intervals (14.3–7.6 Ma and post −7.6 Ma) with different source regions (the Southern Hemisphere and the Northern Hemisphere, respectively23,24,25,26), revealing interactions between fire and vegetation. With this information, it is possible to evaluate the role of fire in late Cenozoic C4 grassland development and expansion.
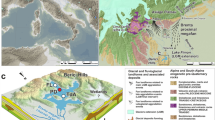
Location of core site KODOS 02-01-02.
Open symbols represent the backtrack path of the study site with increments of 5 Myr. Paleo-locations of the ITCZ are shown as horizontal lines: for Late Miocene23 and for present26, respectively. Blue and red arrows represent modern main ocean currents. Green bold line shows the wind trajectory from East Asia to the study area (March 20, 1984)36. Modified by PaintTool SAI from Global Multi-Resolution Topography (GMRT) MapTool71.
Results
BC content record
The content of BC in pre-industrial deep-sea sediments reflects both the quantity of burned biomass and the wind transport mechanism. The quantity of burned biomass is affected by several factors. The productivity of vegetation provides the potential supply of fuel, and aspects of the fire events, such as frequency, size, intensity, type, and severity27, determine the amount of BC produced. The type of vegetation burned would also affect BC production, because forest fire has a higher emission factor than savanna or grass fire28. However, the low age resolution and broad source areas of deep-sea sediment do not help constrain the relative influence of these factors.
After being produced, BC is transported from continent to ocean as aerosol. It is reported that natural fire events occur intensively in a season with dry conditions29 or frequent lightning30. Thus, if seasonal winds from continent to ocean during fire seasons strengthen, the content of BC would increase compared to those of other eolian sediments. This case was reported from the South China Sea31, where the intensification of the East Asian winter monsoon in the Pleistocene was recorded in the corresponding BC content. Our study area is far from continents and thus is relatively free from local, seasonal winds, such as the Santa Ana in western North America and the Asian Monsoon. Accordingly, we may assume that the effect of seasonal winds would be relatively insignificant.
Down-core BC content ranges from 0.00% to 0.23%, with an average of 0.05% (Dataset 1; Fig. 2A). Prior to 7.6 Ma, when the sediment was mainly delivered from Southern Hemisphere continents, Central and South America23,24,32, the BC content averages 0.04% (range of 0.00–0.12%). Two periods with high BC content occurring at 14.3–12.7 Ma and 11.1–10.2 Ma are noticeable, and the BC content decreases gradually with time from the high values. These two periods coincide with arid periods in tropical South America33 (Fig. 2B), which indicates a potential relationship between tropical South American aridity and an increase in the quantity of burned biomass, because the arid climate results in low fuel moisture content and easy spread of fire. The gradual decrease of BC content following a peak can be explained by shortage of fuel due to frequent fires.
(A) BC content from deep-sea core sediment obtained from the northeastern equatorial Pacific. (B) Goethite/(goethite + hematite), G/(G + H) value at ODP Site 926 to represent aridity of Amazon and proto-Amazon lowland source areas. Lower G/(G + H) value suggests more arid condition (modified from Harris and Mix33). (C) Mass accumulation rate of eolian dust at ODP Site 885/886 (modified from Rea et al.39). (D) BC mass accumulation rate (MAR), (E) BC δ13C value, and (F) calculated proportion of C4 plants from deep-sea core sediment obtained from the northeastern equatorial Pacific. Black bold line across the center represents the time interval when the source area changed from Southern Hemisphere to Northern Hemisphere. Grey areas represent arid time intervals. Blue area represents error range assuming extreme environmental condition(closed-forest to steppe). Dotted lines in (E) and (F) define five time intervals (1–5) showing noticeable characteristics in δ13C value (see text for details).
After 7.6 Ma, the eolian components are interpreted to have been sourced mainly from the continents in Northern Hemisphere, East Asia or North America34,35,36. The BC content ranges from 0.01% to 0.23% with an average of 0.05%. The BC content decreases gradually from 6 Ma to 4 Ma, then increases from 4 Ma to present. As open-habitat grassland became ecologically dominant in both North America37 and China38 before the Middle Miocene and maintained its dominance till date, it seems that the abrupt BC content change related with the burning of large forest would not have occurred and affected the BC content significantly. Thus, it is reasonable to regard low fire activity as the main cause of the decreasing BC content around 4 Ma. A humid time interval from about 6 Ma to 3.8 Ma in Asia39 (Fig. 2C) supports this interpretation, as the humid conditions may have suppressed fire events and decreased the quantity of burned biomass. After 4 Ma, increasing BC content suggests development of a new flammable ecosystem continues to exist till date, with increasing aridity in Asia due to uplift of the Tibetan Plateau (i.e. refs 39 and 40). Unlike the Southern Hemispheric record showing an abrupt increase of BC content, the Northern Hemispheric record shows a gradual increase of BC content from 4 Ma. This suggests that the flammable ecosystem in Northern Hemisphere continents developed gradually, with an increase in fuel through time.
From the observation that the BC content increases during arid conditions of source area (14.3–12.7 Ma and 11.1–10.2 Ma, Central and South America) and decreases during humid conditions of source area (around 5 Ma, Asia), it seems that the long-term scale of natural fires (quantity of burned biomass) varies mainly with the degree of aridity. In addition, the remarkably high BC contents at 6.8 Ma, 4.4 Ma, and 2.2–2.1 Ma are notable. These may indicate short-term, large-scale fire events, although the exact causes of such events are not clear at present.
Down-core BC mass accumulation rate (MAR) ranges from 0.33 mg/cm2/1000 yrs to 47.47 mg/cm2/1000 yrs (Fig. 2D). A sharp increase of MAR across 7.6 Ma is noticeable (3.70 mg/cm2/1000 yrs to 9.48 mg/cm2/1000 yrs on average). This is likely due to the increasing linear sedimentation rate23,24. Considering the changes in eolian source from Southern Hemisphere to Northern Hemisphere, the large difference in BC MAR across 7.6 Ma can be accounted for by the larger amount of landmass in the Northern Hemisphere than in the Southern Hemisphere, which increased the average production amount of eolian sediments including BC.
Carbon isotope record
The δ13C value of BC ranges between −29.3‰ and −20.5‰ (Dataset 2; Fig. 2E). The δ13C values of BC can be divided into five time intervals (Dataset 3). (1) From the beginning of the core record at 14.3 Ma to 10.5 Ma, BC shows depleted δ13C values (−26.5 ~ −29.3‰), with the most depleted value from 11 Ma to 10.5 Ma. (2) From 10.5 Ma to 7.6 Ma, δ13C values are generally increased (−24.7 ~ −27.1‰) compared with the previous interval. (3) From 7.6 Ma to 5.3 Ma, δ13C values are more increased than values of the immediately preceding interval. The amplitude of δ13C fluctuation is also increased visibly (−23.1 ~ −29.1‰). (4) The interval with the highest δ13C values (−20.5 ~ −28.6‰) is the interval from 5.3 Ma to 1.2 Ma. (5) After 1.2 Ma, the δ13C value decreases gradually, until reaching a value near −29‰ (−23.3 ~ −28.8‰).
By correcting the carbon isotope fractionation factor for the burning of vegetation and the isotopic composition of atmospheric CO2, the approximate composition of C3 and C4 vegetation of BC can be estimated. The five time intervals representing the carbon isotope composition variations can be understood in the context of vegetation development (Fig. 2F). From the beginning of the core record, 14.3 Ma to 10.5 Ma, C3 plants in Central and South America occupied nearly 100% of BC, and there is no isotopic evidence for the existence of C4 vegetation. The first recognizable presence of burned C4 plants occurs at 10 Ma, which is about 2 million years earlier than the timing of the major C4 expansion. This represents the existence of patches of C4 vegetation at that time, which agrees with the stable carbon isotope records of rodent teeth in Argentina41. After this time, the contribution of C4 plants to BC maintains a value up to 10%, indicating that C4 vegetation in Central and South America continued to exist from 10 Ma, although it did not become the major terrestrial vegetation.
After 7.6 Ma, the contribution of C4 plants reaches up to 20% of the burned biomass, suggesting the possible existence of open-canopy C4 grassland environment in the source areas. As 7.6 Ma is close to the timing of global expansion of C4 vegetation (~8 Ma), the estimation of C4 vegetation proportion using this method is consistent with other available information (i.e. ref. 3). The increasing contribution of C4 vegetation continued until reaching its highest proportion (up to 50%) of the BC in the fourth time interval, forming the ‘C4 maximum period of burned biomass’ during the Pliocene to Early Pleistocene in the Northern Hemisphere. The beginning of the C4 maximum proportion in the terrestrial ecosystem in the Northern Hemisphere seems largely coincident with the expansion of C4 vegetation in East Asia (Chinese Loess Plateau42 and central Inner Mongolia43, China). After 1.1 Ma, C3 plants gradually regained their dominance, reaching more than 80% of burned vegetation. The diminishment of C4 plants in the Late Pleistocene can be observed in other data from China31,43,44,45 and North America4 (Fig. 3).
(A) δ13C value of BC from deep-sea core sediment obtained from the northeastern equatorial Pacific (this study). (B) δ13C value of tooth enamel form central Inner Mongolia, China (modified from Zhang et al.43). (C) Portion of C4 plant of fossil phytoliths from the Great Plains, North America (modified from Strömberg and McInerney4). Note the decrease of δ13C value or portion of C4 plant (black arrows) after the development of C4 ecosystem (grey areas).
Discussion
The BC content represents the quantity of burned biomass, whereas the carbon isotope composition of BC provides information about the type of vegetation that was burned. The combination of these two parameters therefore indicates a linked relationship between fire activity and type of vegetation burned in the ecosystem.
The relationship between fire and vegetation in Central and South America can be explained easily. Large-scale fire events in South America (11.1–10.2 Ma) are associated with the most depleted δ13C values, indicative of large-scale forest burning, which was followed by, although minor, the first recognizable presence of C4 plants. A similar observation can be made from South China Sea data, in which a large-scale fire event near 3.5 Ma was followed by an abrupt increased contribution of C4 vegetation to burned vegetation31. These observations suggest that fire served as a trigger for early C4 expansion by burning C3 forests and eventually providing open habitats for C4 grasses which are more adaptable to an environment frequently prone to fire46.
The cases of South America (this study) and China31,45 both suggest C4 expansion events triggered by breakout of fire. However, the proportion changes of burned C4 vegetation after the fire event is clearly different. In South America, the C4 vegetation just formed patches in the ecosystem after the fire event from 11.1–10.2 Ma (0–10%), while the C4 vegetation in South China shows significantly increased contribution after the fire event from 4–3 Ma (20–40%, calculated by the method of this study). This variation in C4 development process between each case seems to be caused by the difference of circumstance. The South American case occurred prior to a steep decline in atmospheric CO2 that occurred at ~7 Ma7, whereas the Chinese event occurred after that. Thus, it is reasonable to guess that lower atmospheric CO2 provided conditions more favorable to C4 vegetation in the case of China, resulting in the more dramatic expansion of C4 vegetation.
After C4 vegetation occupied certain portion of the ecosystem, short-term large fire events (6.8 Ma, 4.4 Ma, and 2.2–2.1 Ma) do not show a clear correlation with a dramatic increase of C4 vegetation. Instead, when the contribution of C4 vegetation is over 20%, which indicates the existence of open-canopy condition, carbon isotope value and BC content show weak negative correlation (coefficient of correlation = 0.33). It shows that increased fire events occur with low carbon isotope value, which indicates C3 vegetation burning, whereas decreased BC production coincides with increased contribution of C4 vegetation. This relationship resembles, although minor, the fire-triggered C4 development event which can be characterized by high BC content–low δ13C value due to the burning of C3 vegetation followed by low BC content–high δ13C value which represents C4 vegetation development. Thus, the role of fire after the establishment of open-canopy environment seems to maintain C4-dominated environment, rather than causing a major event of C4 vegetation. Also, low coefficient of correlation between carbon isotope value and BC content suggests not only fire but also other environmental factors, such as CO2, temperature, seasonality, precipitation8,47,48, or edaphic ghettos49, would have played roles in controlling the C3–C4 ratio of the ecosystem, as shown from the case of modern vegetation50.
The effect of environmental factors rather than fire on the contribution of C4 vegetation to the ecosystem can be observed more clearly in the Late Pleistocene, when the long-term increase of BC content (4 Ma to present) co-exists with the latter gradual ceasing of C4 plant expansion (1.2 Ma to present). This event may have been affected by decreased sea-surface temperatures after the mid-Pleistocene transition51, which controlled the atmospheric moisture content and consequentially the aridity52, or the decreasing temperature conditions from the Middle Miocene onward53 may have reached a threshold value for C4 plant survival. The latter is more likely to be the main cause of decreasing C4 plants, as the known records of C4 decrease during Pliocene to Pleistocene are from high-latitude regions, and most records from low-latitude regions do not indicate such a decrease in C4 plants (Fig. 3).
To summarize, a drastic expansion of C4 vegetation triggered by fire breakout requires the following conditions: (1) an original landscape composed of closed habitat, so fire events can provide high light conditions for C4 grasses; (2) environmental conditions that are favorable to C4 vegetation, so C4 grasses can continue developing in competition with C3 grasses like lower atmospheric CO2; and (3) environmental conditions favorable to fire. After the open-canopy environment is established, the influence of fire as a driver of drastic ecosystem development wanes, yet fire still plays a role in maintaining C4-dominated ecosystem as one of the environmental conditions.
Considering these hypotheses, we suggest aridity as the most important factor that induced C4 expansion, as aridity can provide both opportunity for fire breakout and environmental conditions favorable to C4 vegetation growth and sustainment. Under general arid conditions, clearance of forest by fire events would result in changes to the hydrological cycle through reductions of evapotranspiration and cloud formation, thus enhancing the arid conditions. In North Africa, aridification of the Sahara Desert caused by the shrinkage of the Tethys Sea during the Late Miocene preceded the 8 Ma C4 expansion event54 and higher concentrations of charred particles16, suggesting that the aridification strengthened the fire activity and eventually led to C4 expansion. In South Asia, the 8 Ma C4 expansion event8 matches with the timing of Asian drying caused by the Tibetan uplift39, and the later C4 expansion event in East Asia seems to have been affected by the evolution and change in the monsoon climate that increased the seasonal aridity31,55,56. As the origination of C4 lineages is interpreted to have begun at 35 Ma and major diversification began at around 15 Ma57, aridity events after 15 Ma might have reasonably provided favorable environmental conditions coupled with open habitat formed by fire events, resulting in the expansion of C4 grassland. The long C4 maximum period of burned biomass from the Pliocene to the Late Pleistocene may reflect a time interval having increased territory with water stress favorable to C4 vegetation, until the low temperature effect surpassed the advantages provided by aridity. The latter effect resulted in the eventual C4 vegetation decrease of the Late Pleistocene.
Methods
Sample description
The sediment samples used in this study are from a 328-cm-long piston core (KODOS 02-01-02) collected at 16°12′N, 125°59′W at a water depth of 4550 m in the Clarion-Clipperton fracture zone of the northeastern equatorial Pacific (Fig. 1). The chronology and mineralogical and geochemical characteristics of the studied core were reported by Hyeong et al.23,24 and are summarized briefly below. The coring site is located more than 2000 km from the East Pacific Rise and the nearest land mass (North and Central America) and thus is composed mostly of pelagic sediments. The core sediments consist of slightly bioturbated homogeneous siliceous red clays and are divided into two intervals based on a distinct color change; dark brown upper interval above 250 cm from the core top and yellowish brown lower interval below 250 cm. The inorganic fraction of the bulk pelagic sediments is considered to be eolian dust. The eolian sediment of the lower interval below 250 cm (15.5–7.6 Ma) is composed of smectite- and phillipsite-rich minerals with a very uniform mass flux of 5 ± 1 mg/cm2/103 yr, whereas that in the upper interval above 250 cm (~7.6 Ma) is characterized by a quartz- and illite-rich mineralogy with a mass flux over 2 times higher than that of the lower interval at 12 ± 1 mg/cm2/103 yr. The eolian components in the lower interval are interpreted to have been sourced from Central and South America, whereas those in the upper interval are from Northern Hemisphere continents. It was interpreted that the distinctive differences in the eolian dust source between the lower and upper intervals of the studied core are due to a change in the paleolatitude of the Intertropical Convergence Zone which moved southwards through the coring site at ~7.6 Ma (Fig. 1).
Analytical methods
In total, 189 samples collected at a 1 cm interval from the split core were used to determine BC concentration. There are four main techniques to determine the BC content of sediment: thermal, chemical, optical, and molecular marked58. Each of these procedures measures a different region within the combustion continuum17. Among these, the thermal and chemical methods are known to detect the highly condensed BC18. These two methods do not detect charcoal, but as charcoal is scarce in the deep-sea environment, the problem of limited detection of charcoal can be ignored. The thermal method produces artifacts by the charring of samples, which may result in overestimation of BC59. The chemical method using dichromate acid requires different mass yield corrections for each type of sample, yet it still allows precise, reproducible isotope measurements60. Because isotopic analysis was essential to reconstruct vegetation history, we followed the chemical oxidation method of Lim and Cachier61 for BC extraction.
Carbon content was obtained using an elemental analyzer (FlashEA 1112). The error range was ±3.3% of the reported value. By applying the total carbon value and weight of sample after chemical oxidation, the BC content in total sediment was calculated. Content of BC was analyzed using a CN analyzer (NA Series 2, CE Instruments, Italy). Carbon isotopic values of BC were analyzed by a stable isotope ratio mass spectrometer (IsoPrime-EA, Micromass, UK) interfaced with a CN analyzer at the National Instrumentation Center for Environmental Management, Seoul National University. Carbon isotopic values were presented in ‰ deviation from the Peedee belemnite (PDB). The analytical precision based on repeated measurements of the laboratory standard was better than ±0.1‰ (i.e. ref. 62).
Converting δ13C value into distribution of vegetation type
To use stable carbon isotope data of BC as a proxy to reconstruct vegetation history, estimation of C3 and C4 plant δ13C composition was made by correcting the stable carbon isotope composition of paleoatmospheric CO2 in the studied time interval and the carbon isotope fractionation factor for vegetation burning. The isotope composition of paleoatmospheric CO2 has been obtained from contemporary marine carbonate. Following Ekart et al.63, we assumed that the difference in isotope composition between surface ocean carbonates and atmospheric carbon dioxide is 8‰ based on the isotope composition of pre-industrial carbon dioxide trapped in glacial ice64 and contemporaneous surface ocean carbonates65,66. A similar apparent isotope enrichment value, 7.9 ± 1.1‰, was suggested by Passey et al.67 using δ13C of planktonic foraminifera calcite tests as a proxy for δ13C of atmospheric CO2. The geologic record of Miocene–Pleistocene open ocean carbonates of Shackleton et al.68 was used to calculate δ13C of the paleoatmospheric carbon dioxide (δ13CpCO2) during the studied time interval, which can be calculated by subtracting 8‰ from δ13C of planktonic foraminifera (δ13CFORAM).
Laboratory burning of C3 and C4 vegetation has revealed that there exists significant fractionation of carbon isotopes between the parent material and the corresponding aerosol (soot) produced during burning21,22,69. Aerosol derived from burning of C3 vegetation is enriched in 13C by 0.5‰, whereas aerosol from burning of C4 vegetation is depleted in 13C by 3.5‰. The average δ13C values of modern C3 plant, −28.5‰70, and of modern C4 plant, −12.5‰1, are used as end-member values for estimation of the C3–C4 biomass ratios present in the ecosystem. Compared to C4 plant, the δ13C range of C3 plant is large, depending on the environmental conditions, and values lighter than −28.5‰ are considered to represent environments receiving mean annual precipitation higher than the threshold precipitation for sustaining C3 forest (1500 mm/year70). We assumed that the δ13C of BC produced from modern C3 plant burning (δ13CmBC−C3) is −28‰ and that the δ13C from modern C4 plant burning (δ13CmBC-C4) is −16‰.
From these assumptions, the approximate shifts in δ13C of burned C3 and C4 vegetation through the time interval can be estimated as follows, considering the difference between δ13C of modern atmospheric carbon dioxide (δ13CmCO2) and δ13CpCO2:

where δ13CBC-C3 and δ13CBC-C4 are the carbon isotope compositions of BC from C3 plant and BC from C4 plant, respectively. If we assume that δ13CmCO2 is about −8‰, and δ13CpCO2 can be substituted as (δ13CFORAM −8‰), this fomular can be simplified like below:


Using these values, the approximate composition of C3 and C4 vegetation burned can be estimated by the following simple mass-balance relationship:

which can be changed as

where f is the fraction of BC from C3 plant, (1−f) is the fraction of BC from C4 plant.
In this calculation, we assumed δ13CmBC−C3 and δ13CmBC−C4 as constant value by fixing carbon isotope fractionation during combustion, and δ13C values of modern plants. However, δ13C value of BC varies by diverse factors under real conditions, which makes the f value as a rough estimate. δ13C value of C3 plant changes by the climate region. If we assume the whole C3 plants burned were from closed-forest canopy (δ13C = −32‰), f value will decrease <28% from the original value. If we assume the whole C3 plants burned were from semi-arid steppe environment (δ13C = −26%), f value will increase <30% (Fig. 2F). Also, both C3 (−0.6 to 1.8%) and C4 (−0.9 to −7.0%) vegetation has a certain range of carbon isotope fractionation during burning22, which increases a possible error range. This error range of f increases with (δ13CFORAM−δ13CBC) value. In our study, the maximum margin of error can be found at 7.2 Ma (δ13CFORAM−δ13CBC = 30.90%), where f ranges from 0.91 to 2.43 with the average of 1.24, while the minimum margin of error can be found at 4.57 Ma (δ13CFORAM−δ13CBC = 21.89%), where f ranges from 0.18 to 0.79 with the average of 0.49. On average (δ13CFORAM−δ13CBC = 26.60%), f ranges from 0.91 to 2.43 with the average of 1.24.
Additional Information
How to cite this article: Kim, D. et al. Terrestrial biome distribution in the Late Neogene inferred from a black carbon record in the northeastern equatorial Pacific. Sci. Rep. 6, 32847; doi: 10.1038/srep32847 (2016).
References
Cerling, T. E. et al. Global vegetation change through the Miocene/Pliocene boundary. Nature 389, 153–158 (1997).
Cerling, T. E. The stable isotopic composition of modern soil carbonate and its relationship to climate. Earth Planet Sc Lett 71, 229–240 (1984).
Edwards, E. J., Osborne, C. P., Strömberg, C. A. & Smith, S. A. The origins of C4 grasslands: integrating evolutionary and ecosystem science. Science 328, 587–591 (2010).
Strömberg, C. A. & McInerney, F. A. The Neogene transition from C3 to C4 grasslands in North America: assemblage analysis of fossil phytoliths. Paleobiology 37, 50–71 (2011).
Tidwell, W. D. & Nambudiri, E. M. V. Tomlisonia thomassonii, gen. et sp. nov., a permineralized grass from the upper Miocene Ricardo Formation, California. Rev Palaeobot Palyno 60, 165–177 (1989).
Singh, S., Awasthi, A., Parkash, B. & Kumar, S. Tectonics or climate: What drove the Miocene global expansion of C4 grasslands? Int J Earth Sci 102, 2019–2031, doi: 10.1007/s00531-013-0893-5 (2013).
Bolton, C. T. & Stoll, H. M. Late Miocene threshold response of marine algae to carbon dioxide limitation. Nature 500, 558–562 (2013).
Edwards, E. J. & Smith, S. A. Phylogenetic analyses reveal the shady history of C4 grasses. P Natl Acad Sci USA 107, 2532–2537 (2010).
Huang, Y., Clemens, S. C., Liu, W., Wang, Y. & Prell, W. L. Large-scale hydrological change drove the late Miocene C4 plant expansion in the Himalayan foreland and Arabian Peninsula. Geology 35, 531–534 (2007).
Levin, N. E., Quade, J., Simpson, S. W., Semaw, S. & Rogers, M. Isotopic evidence for Plio–Pleistocene environmental change at Gona, Ethiopia. Earth Planet Sc Lett 219, 93–110 (2004).
Bond, W., Woodward, F. & Midgley, G. The global distribution of ecosystems in a world without fire. New Phytol 165, 525–538 (2005).
Keeley, J. E. & Rundel, P. W. Fire and the Miocene expansion of C4 grasslands. Ecol Lett 8, 683–690 (2005).
Scheiter, S. et al. Fire and fire‐adapted vegetation promoted C4 expansion in the Late Miocene. New Phytol 195, 653–666 (2012).
Herring, J. R. Charcoal fluxes into sediments of the North Pacific Ocean: the Cenozoic record of burning. The Carbon Cycle and Atmospheric CO2: Natural Variations Archean to Present. (Ed. E. T. Sundquist & W. Broecker ), American Geophysical Union: Geophysical Monograph Series 32, 419–442 (1985).
Morley, R. & Richards, K. Gramineae cuticle: a key indicator of Late Cenozoic climatic change in the Niger Delta. Rev Palaeobot Palyno 77, 119–127 (1993).
Hoetzel, S., Dupont, L., Schefuß, E., Rommerskirchen, F. & Wefer, G. The role of fire in Miocene to Pliocene C4 grassland and ecosystem evolution. Nat Geosci 6, 1027–1030 (2013).
Goldberg, E. D. Black carbon in the environment: properties and distribution. (John Wiley and Sons, New York, 1985).
Masiello, C. New directions in black carbon organic geochemistry. Mar Chem 92, 201–213 (2004).
Masiello, C. & Druffel, E. Black carbon in deep-sea sediments. Science 280, 1911–1913 (1998).
Middelburg, J. J., Nieuwenhuize, J. & van Breugel, P. Black carbon in marine sediments. Mar Chem 65, 245–252 (1999).
Das, O., Wang, Y. & Hsieh, Y.-P. Chemical and carbon isotopic characteristics of ash and smoke derived from burning of C3 and C4 grasses. Org Geochem 41, 263–269 (2010).
Turekian, V. C., Macko, S., Ballentine, D., Swap, R. J. & Garstang, M. Causes of bulk carbon and nitrogen isotopic fractionations in the products of vegetation burns: laboratory studies. Chem Geol 152, 181–192 (1998).
Hyeong, K., Park, S.-H., Yoo, C.-M. & Kim, K.-H. Mineralogical and geochemical compositions of the eolian dust from the northeast equatorial Pacific and their implications on paleolocation of the Intertropical Convergence Zone, Paleoceanography, 20, PA1010, doi: 10.1029/2004PA001053.
Hyeong, K., Yoo, C.-M., Kim, J., Chi, S.-B. & Kim, K.-H. Flux and grain size variation of eolian dust as a proxy tool for the paleo-position of the Intertropical Convergence Zone in the northeast Pacific. Palaeogeogr Palaeoclimatol Palaeoecol 241, 214–223 (2006).
Kim, J. et al. Southward shift of the Intertropical Convergence Zone in the western Pacific during the late Tertiary: Evidence from ferromanganese crusts on seamounts west of the Marshall Islands. Paleoceanography 21, PA4218, doi: 10.1029/2006PA001291.
Pettke, T., Halliday, A. N. & Rea, D. K. Cenozoic evolution of Asian climate and sources of Pacific seawater Pb and Nd derived from eolian dust of sediment core LL44‐GPC3. Paleoceanography 17, 3-1-3-13 (2002).
Flannigan, M., Stocks, B. J. & Wotton, B. Climate change and forest fires. Sci Total Environ 262, 221–229 (2000).
Janhäll, S., Andreae, M. O. & Pöschl, U. Biomass burning aerosol emissions from vegetation fires: particle number and mass emission factors and size distributions. Atmos Chem Phys 10, 1427–1439 (2010).
Westerling, A. L., Hidalgo, H. G., Cayan, D. R. & Swetnam, T. W. Warming and earlier spring increase western US forest wildfire activity. Science 313, 940–943 (2006).
Stocks, B. et al. Large forest fires in Canada, 1959–1997. J Geophys Res-Atmos (1984–2012) 107, FFR 5-1-FFR 5-12 (2002).
Jia, G., Peng, P. a., Zhao, Q. & Jian, Z. Changes in terrestrial ecosystem since 30 Ma in East Asia: Stable isotope evidence from black carbon in the South China Sea. Geology 31, 1093–1096 (2003).
Olivarez, A. M., Owen, R. M. & Rea, D. K. Geochemistry of eolian dust in Pacific pelagic sediments: Implications for paleoclimatic interpretations. Geochim Cosmochim Ac 55, 2147–2158 (1991).
Harris, S. E. & Mix, A. C. Climate and tectonic influences on continental erosion of tropical South America, 0–13 Ma. Geology 30, 447–450 (2002).
Nakai, S., Halliday, A. N. & Rea, D. K. Provenance of dust in the Pacific Ocean. Earth Planet Sc Lett 119, 143–157 (1993).
Jones, C. E., Halliday, A. N., Rea, D. K. & Owen, R. M. Neodymium isotopic variations in North Pacific modern silicate sediment and the insignificance of detrital REE contributions to seawater. Earth Planet Sc Lett 127, 55–66 (1994).
Merrill, J. T., Uematsu, M. & Bleck, R. Meteorological analysis of long range transport of mineral aerosols over the North. J Geophys Res 94, 8584–8598 (1989).
Strömberg, C. A. Decoupled taxonomic radiation and ecological expansion of open-habitat grasses in the Cenozoic of North America. P Natl Acad Sci USA 102, 11980–11984 (2005).
Xingxue, L. et al. Fossil floras of China through the geological ages. (Guangdong Science and Technology Press, 1995).
Rea, D. K., Snoeckx, H. & Joseph, L. H. Late Cenozoic eolian deposition in the North Pacific: Asian drying, Tibetan uplift, and cooling of the northern hemisphere. Paleoceanography 13, 215–224 (1998).
Li, J. et al. Late Miocene–Quaternary rapid stepwise uplift of the NE Tibetan Plateau and its effects on climatic and environmental changes. Quaternary Res 81, 400–423 (2014).
Hynek, S. A. et al. Small mammal carbon isotope ecology across the Miocene–Pliocene boundary, northwestern Argentina. Earth Planet Sc Lett 321, 177–188 (2012).
Passey, B. H. et al. Strengthened East Asian summer monsoons during a period of high-latitude warmth? Isotopic evidence from Mio-Pliocene fossil mammals and soil carbonates from northern China. Earth Planet Sc Lett 277, 443–452 (2009).
Zhang, C. et al. C4 expansion in the central Inner Mongolia during the latest Miocene and early Pliocene. Earth Planet Sc Lett 287, 311–319 (2009).
Ding, Z. & Yang, S. C3/C4 vegetation evolution over the last 7.0 Myr in the Chinese Loess Plateau: evidence from pedogenic carbonate δ13C. Palaeogeogr Palaeoclimatol Palaeoecol 160, 291–299 (2000).
Zhou, B. et al. Late Pliocene–Pleistocene expansion of C4 vegetation in semiarid East Asia linked to increased burning. Geology 42, 1067–1070 (2014).
Ripley, B., Donald, G., Osborne, C. P., Abraham, T. & Martin, T. Experimental investigation of fire ecology in the C3 and C4 subspecies of Alloteropsis semialata. J Ecol 98, 1196–1203 (2010).
Ehleringer, J. R., Cerling, T. E. & Helliker, B. R. C4 photosynthesis, atmospheric CO2, and climate. Oecologia 112, 285–299 (1997).
Osborne, C. P. Atmosphere, ecology and evolution: what drove the Miocene expansion of C4 grasslands? J Ecol 96, 35–45 (2008).
Bond, W. J. Fires in the Cenozoic: a late flowering of flammable ecosystems. Front Plant Sci 5, (2014).
Griffith, D. M. et al. Biogeographically distinct controls on C3 and C4 grass distributions: merging community and physiological ecology. Global Ecol Biogeogr 24, 304–313 (2015).
Medina-Elizalde, M. & Lea, D. W. The mid-Pleistocene transition in the Tropical Pacific. Science 310, 1009–1012 (2005).
Schefuß, E., Schouten, S., Jansen, J. F. & Damsté, J. S. S. African vegetation controlled by tropical sea surface temperatures in the mid-Pleistocene period. Nature 422, 418–421 (2003).
Zachos, J., Pagani, M., Sloan, L., Thomas, E. & Billups, K. Trends, rhythms, and aberrations in global climate 65 Ma to present. Science 292, 686–693 (2001).
Zhang, Z. et al. Aridification of the Sahara desert caused by Tethys Sea shrinkage during the Late Miocene. Nature 513, 401–404 (2014).
Sun, J., Lü, T., Zhang, Z., Wang, X. & Liu, W. Stepwise expansions of C4 biomass and enhanced seasonal precipitation and regional aridity during the Quaternary on the southern Chinese Loess Plateau. Quaternary Sci Rev 34, 57–65 (2012).
An, Z. et al. Multiple expansions of C4 plant biomass in East Asia since 7 Ma coupled with strengthened monsoon circulation. Geology 33, 705–708 (2005).
Bouchenak-Khelladi, Y., Slingsby, J. A., Verboom, G. A. & Bond, W. J. Diversification of C4 grasses (Poaceae) does not coincide with their ecological dominance. Am J Bot 101, 300–307 (2014).
Schmidt, M. W. & Noack, A. G. Black carbon in soils and sediments: analysis, distribution, implications, and current challenges. Global Biogeochem Cy 14, 777–793 (2000).
Simpson, M. J. & Hatcher, P. G. Overestimates of black carbon in soils and sediments. Naturwissenschaften 91, 436–440 (2004).
Masiello, C. A., Druffel, E. R. M. & Currie, L. A. Radiocarbon measurements of black carbon in aerosols and ocean sediments. Geochim Cosmochim Ac 66, 1025–1036, doi: 10.1016/S0016-7037(01)00831-6 (2002).
Lim, B. & Cachier, H. Determination of black carbon by chemical oxidation and thermal treatment in recent marine and lake sediments and Cretaceous-Tertiary clays. Chem Geol 131, 143–154 (1996).
Hong, S. K. & Lee, Y. I. Contributions of soot to δ13C of organic matter in Cretaceous lacustrine deposits, Gyeongsang Basin, Korea: Implication for paleoenvironmental reconstructions. Palaeogeogr Palaeoclimatol Palaeoecol 371, 54–61 (2013).
Ekart, D. D., Cerling, T. E., Montanez, I. P. & Tabor, N. J. A 400 million year carbon isotope record of pedogenic carbonate: implications for paleoatmospheric carbon dioxide. Am J Sci 299, 805–827 (1999).
Friedli, H., Lötscher, H., Oeschger, H., Siegenthaler, U. & Stauffer, B. Ice core record of the 13C/12C ratio of atmospheric CO2 in the past two centuries. Nature 324, 237–238 (1986).
Shackleton, N., Hall, M., Line, J. & Shuxi, C. Carbon isotope data in core V19-30 confirm reduced carbon dioxide concentration in the ice age atmosphere. Nature 306, 319–322 (1983).
Veizer, J. et al. 87Sr/86Sr, δ13C and δ18O evolution of Phanerozoic seawater. Chem Geol 161, 59–88 (1999).
Passey, B. H. et al. Environmental change in the Great Plains: an isotopic record from fossil horses. J Geol 110, 123–140 (2002).
Shackleton, N. J., Hall, M. A. & Boersma, A. Oxygen and carbon isotope data from Leg 74 foraminifers. 599–612 (U.S. Government Printing Office, Washington, 1984).
Cachier, H., Buat-Menard, P., Fontugne, M. & Rancher, J. Source terms and source strengths of the carbonaceous aerosol in the tropics. J Atmos Chem 3, 469–489 (1985).
Kohn, M. J. Carbon isotope compositions of terrestrial C3 plants as indicators of (paleo) ecology and (paleo) climate. P Natl Acad Sci USA 107, 19691–19695 (2010).
Ryan, W. B. F. et al. Global Multi-Resolution Topography synthesis, Geochem Geophys Geosyst 10, Q03014, doi: 10.1029/2008GC002332.
Acknowledgements
This work was supported by grants from the Korea-Research Foundation (2014 R1A2A2A01005404), the Ministry of Oceans and Fisheries (PM 57380) and the SNU-SEES BK 21 Program. We thank Drs H.S. Lim and M.K. Lee for helping measurement of BC contents.
Author information
Authors and Affiliations
Contributions
Y.I.L. developed the conceptual idea for this study. D.K. conducted sample and data analyses with some help by K.H. and C.M.Y., D.K. and Y.I.L. wrote the manuscript and all authors contributed to discussion and reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Kim, D., Lee, Y., Hyeong, K. et al. Terrestrial biome distribution in the Late Neogene inferred from a black carbon record in the northeastern equatorial Pacific. Sci Rep 6, 32847 (2016). https://doi.org/10.1038/srep32847
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep32847
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.