Abstract
Clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated9 (Cas9) endonuclease system is a powerful RNA-guided genome editing tool. CRISPR/Cas9 has been well studied in model plant species for targeted genome editing. However, few studies have been reported on plant species without whole genome sequence information. Currently, no study has been performed to manipulate metabolic pathways using CRISPR/Cas9. In this study, the type II CRISPR/SpCas9 system was used to knock out, via nonhomologous end-joining genome repair, the 4′OMT2 in opium poppy (Papaver somniferum L.), a gene which regulates the biosythesis of benzylisoquinoline alkaloids (BIAs). For sgRNA transcription, viral-based TRV and synthetic binary plasmids were designed and delivered into plant cells with a Cas9 encoding-synthetic vector by Agrobacterium-mediated transformation. InDels formed by CRISPR/Cas9 were detected by sequence analysis. Our results showed that the biosynthesis of BIAs (e.g. morphine, thebaine) was significantly reduced in the transgenic plants suggesting that 4′OMT2 was efficiently knocked-out by our CRISPR-Cas9 genome editing approach. In addition, a novel uncharacterized alkaloid was observed only in CRISPR/Cas9 edited plants. Thus, the applicabilitiy of the CRISPR/Cas9 system was demonstrated for the first time for medicinal aromatic plants by sgRNAs transcribed from both synthetic and viral vectors to regulate BIA metabolism and biosynthesis.
Similar content being viewed by others
Introduction
Plants provide many nutrients and metabolites that are either directly assimilated by humans or used as raw material in different industries. Recently, new plant breeding technologies have been employed in order to obtain a multitude of beneficial traits like increased metabolite and nutrient production and resistance to harsh stresses.
Protein based editing tools, including zinc-finger nucleases (ZFNs) and transciption activator-like endunucleases (TALENs), have been powerful tools for manipulating genomes at the transcriptional level in the past few years1. ZFN and TALEN technologies employ a protein-based endonuclease called Fok-I to knock-out the gene of interest by introducing double-strand breaks (DSBs) in the target DNA2. Targeting desired sequences in the genome can be troublesome with these systems as they require designing two different protein hybrids that can recognize rarely occurring regions flanking the target site3,4. The efficiencies of these systems are challenged by the presence of those DNA sequences along with the ability of both proteins to orient correctly and dimerize with the provided spacer length5,6. Some of those difficulties were overcome with the discovery of the CRISPR/Cas9 system that does not require protein dimerization7. The system promotes gene editing by utilizing well-designed guide RNAs that recognizes a 3 base-pair PAM sequence downstream of the target site7,8,9,10,11. Since the first implementation of CRISPR system in plants, it has been applied by many researchers in several plant species including Arabidopsis thaliana12, Brassica oleracea13, Citrus sinensis14, Cucumis sativus15, Glycine max16, Hordeum vulgare13, Lactuva sativa17, Marchantia polymorpha18, Medicago truncatula19, Nicotiana attenuate17, Nicotiana benthamiana12,20, Nicotiana tabacum21, Oriza sativa22, Populus tomentosa23, Populus tremula24, Solanum lycopersicum25, Solanum tuberosum26, Sorghum bicolor27, Triticum aestivum28 and Zea mays29.
P. somniferum (2n = 22), from the family of Papaveraceae, is one of the two opium poppy species known to biosynthesize morphine30,31. Despite of its importance as an oil plant, opium poppy also produces many benzylisoquinoline alkaloids (BIAs) used in biomedicine, like antitussive codeine and noscapine, anticancer noscapine, antispasmodic papaverine and analgesic morphine. BIA biosynthesis starts with the condensation of dopamine and the 4-hydroxyphenylacetaldehyde (4-HPAA) to yield (S)-norcoclaurine32. After a series of methylation and hydroxylation steps, (S)-norcoclaurine is converted to central intermediate S-reticuline33,34. This step is catalysed by 3′-hydroxy-N-methylcoclaurine 4′-O-methyltransferase (4′OMT)35 (Fig. 1). Several end products such as morphine, noscapine and papaverine are derived from S-reticuline via different BIA pathways. The epimerization of (S)- reticuline to (R)-reticuline results in the production of morphine (morphinian)36. Moreover, the conversion of (S)-reticuline to (S)-scoulerine is catalyzed by Berberine Bridge Enzyme (BBE) and this is the first committed step in the biosynthesis of sanguinarine (benzophenanthridine) and noscapine (phthalideisoquinoline) alkaloids37.
Morphine, thebaine and codeine biosynthesis in opium poppy is catalyzed by several essential enzymes including 4′OMT2.
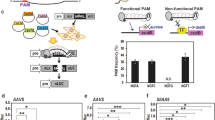
By delivering CRISPR/Cas9 system components of sgRNA and Cas9 inside plant tissues with agroinoculation, it’s possible to knock-out the desired gene segment to manipulate BIA production flux for engineered biosynthesis. This figure shows the target for CRISPR/Cas9.
Recently, it has been reported that BIA production in opium poppy can be manipulated by altering the expression of some particular genes in the BIA pathway. Over-expression and TRV-mediated gene silencing studies in opium poppy revealed that the amount of alkaloid biosynthesis could be manipulated in a tissue-specific manner38,39,40,41. Both the over-expression and the silencing of (R, S)-reticuline 7-O-methyltransferase (7OMT) and 3′-hydroxyl-N-methylcoclaurine 4′-O-methyltransferase (4′OMT2) genes revealed their regulatory roles in BIA production in different tissues42. Nevertheless, the roles of these genes cannot be completely understood using the aforementioned strategies. In particular, the previous approaches impacted gene expression at the post-transcriptional level. This results in a significant decrease in gene expression but does not abolish the gene function. This is important, as the biosynthetic pathways do not cease and the production of metabolites continues, which could mask the overall phenotypes regulated by the targeted genes. Therefore the application of gene knock-out strategies, like CRISPR/Cas9, can help address these challenges and increase our understanding of the roles of the genes of interest.
Synthetic vectors are usually used to express CRISPR system components like sgRNAs and Cas9 endonuclease in plant and animal systems. However, synthetic vectors can be expressed in the transfected or transformed cells only and not the neighbouring cells. Additionally, viral vectors are not suitable for nucleic acid constructs like the one encoding SpCas9 (Streptococcus pyogenes Cas9)21. The efficiency of the CRISPR/Cas9 system could be increased by using viral vector backbones for the expression of guide RNAs in plant systems. In this study, we used the CRISPR/Cas9 system to knock out 4′OMT (isoform-2) which is a critical regulatory gene involved in the biosynthesis of some bioactive compounds produced in the medicinal aromatic plant, opium poppy (Papaver somniferum L.) using both synthetic and viral-based delivery systems.
Materials and Methods
Plant materials
P. somniferum (cv. Ofis-95) seeds were obtained from Toprak Mahsulleri Ofisi (TMO, Ankara, Turkey) and grown in a mixture of 50% perlite and 50% peat in an environment of 24/20 °C with 16/8 hours light/dark and 60% humidity in a plant growth cabinet.
Vectors
Vectors of 46965, 46966, 46968 and 48002 were purchased from Addgene (www.addgene.org, MA, US) while pTRV2::AtU6p::sgRNA_4OMT2 (Supplementary Fig. S1), pICH47751::AtU6p::sgRNA_4OMT2 (Supplementary Fig. S2) and pTRV1 were synthesized in our lab. Plasmid isolations were carried out using GeneJET Plasmid Miniprep Kit (Thermo Scientific, Massachusetts, US) according to the manufacturer’s protocol. The sgRNA delivery constructs were specifically designed to target and edit the 4′OMT2 sequence (GenBank ID: AY217334.1) (Supplementary Fig. S3).
Construction of synthetic vectors
4′OMT2 targeting sgRNA was synthesized by classical PCR with 4OMT2sgRNA_1F and 4OMT2sgRNA_1R primers (Table S1) using approximately 100 ng of plasmid 46966 as a template. Then, PCR products were purified using High Pure PCR Product Purification Kit (Roche, Manheim, Germany). The AtU6p promotor and synthesized sgRNA were assembled by MoClo method43. The single-step reaction was done by the proper combination of 40 fmoles of each vector (46968: an AtU6p encoding L0 plasmid, plasmid 48002: empty backbone plasmid used as MoClo L1 destination vector) and 100 ng purified sgRNA_4OMT2 amplicon with 2.5 Weiss units of Bsa I (New England Biolabs, MA, US) along with 200 CEU of T4 Ligase (Thermo Scientific, Massachusetts, US), 1.5 μL of 10 × T4 ligase buffer and 1.5 μL 10 × BSA to the final volume of 15 μL. The reaction mixture was incubated in a thermal-cycler for 1 min at 37 °C, followed by 30 cycles alternating between 3 min at 37 °C and 4 min at 16 °C and one cycle of 5 min 50 °C and 5 min at 80 °C, respectively. The end-product was chemically transferred into competent E. coli (strain DH10B) cells by heat-shock for blue-white colony screening.
Construction of viral vectors
sgRNApaIF and sgRNApaIR primers were used to link Apa I recognition sites to the flanking sites of AtU6p::sgRNA_4OMT2 insert by PCR (Table S1). The PCR product was purified using High Pure PCR Product Purification Kit (Roche, Manheim, Germany) and was then ligated into pGEM®-T (Promega, Wisconsin, US), T-A cloning vector using T4 DNA ligase. Ligation mixture was transfected to chemically competent E. coli cells by heat-shock and positive clones were then confirmed by colony-PCR using sgRNApaIF and sgRNApaIR primers. Plasmids were further verified with Sanger dideoxy sequencing (RefGen, Ankara, Turkey). Verified inserts were digested and ligated into pTRV expression vectors for in-planta expression using T4 DNA ligase. Ligation products were transfected into chemically competent E. coli cells and grown in an appropriate medium for colony selection with PCR utilizing sgRNApaIF and sgRNApaIR primers (Table S1). Chemically competent cells were prepared with RbCl buffers according to a previous report44.
Agro-infiltration of the constructs into plant cells
Plasmids, containing 35S::hCas9 and AtU6p:sgRNA_4OMT2 inserts were transferred into the electro-competent A. tumefaciens (strain EHA105 and GV3101) cells by using an electroporator (Invitrogen, Neon Transfection System, Massachusetts, US). Alternatively, chemically competent cells were prepared by using CaCl2 according to Holsters’s protocol45. Plasmids were harvested from bacteria grown overnight(OD600: 2-2.5) and re-suspended in a final concentration of OD600: 0.8 in an induction buffer containing 10 mM MgCl2, with 100 μM acetosyringone and 1 mM MES at pH 5.6 46. The prepared mixture was incubated overnight at room temperature and infiltrated into leaves by a needleless syringe (Fig. 1). Inoculations of the synthetic plasmids were performed using 1:1 ratio of mixed bacteria containing each sgRNA and hCas9 expressor plasmids. Twenty 8–10 weeks old plants were co-infiltrated for each treatment with vectors of pK7WGF2::hCas9 (Addgene plasmid #46965) and pICH47751:: AtU6p::sgRNA_4OMT2. For TRV-mediated CRISPR/Cas9 implementations, TRV1, TRV2 (pTRV2::AtU6p::sgRNA_4OMT2) and hCas9 encoding pK7WGF2::hCas9 (Addgene #46965) plasmids were infiltrated in 1:1:1 ratio to precisely maintain the expression of Cas9 inside plant tissues. Two days after the initial infiltration, this process was repeated one more time to enhance sgRNA/Cas9 effectivity.
gDNA isolation from sgRNA_Cas9 treated tissues
After 4 days of agro-infiltration, leaves were harvested for gDNA isolation. gDNA from both control group and treated plants were extracted by using standardized CTAB & sucrose plant DNA extraction protocol. 100 mg of frozen leaves were mechanically homogenized using TissueLyser II system (Qiagen, Hilden, Germany) and each sample was treated with RNase A (Qiagen). Isolated gDNA samples were enriched by enzymatic digestion assay performed by digesting gDNAs with Age I (Jena Biosciences, Jena, Germany) for 4′OMT2 targeted sequences.
Analysis of InDels with DNA sequencing
100 ng of the enriched gDNA was used to clone the targeted regions of the 4′OMT2 gene for each experimental group. Targeted DNA segments were amplified with PCR by using High Fidelity DNA polymerases (Jena Biosciences, Jena, Germany). For the amplification of 4′OMT2 region, primers of 4OMT2_AgeIF and 4OMT2_AgeIR were used. In order to eliminate non-digested DNA, amplicons were further enriched by a second digestion assay and run on a 3% agarose gel for AFLP analyses. Identified DNA segments with potential insertion and deletions (InDels) were isolated from gel by using a GeneJET Gel Extraction Kit following the manufacturer’s protocol and then cloned into pGEM®-T vector by T4 DNA ligase reaction. The end product was transfected to E. coli DH5α cells for blue-white colony screening. After verifying positive colonies with M13 colony PCR, plasmids containing 4′OMT2 gene segments were isolated using a GeneJET Plasmid Miniprep Kit (Thermo Scientific, Massachusetts, US) as described in the manufacturer’s protocol and further analysed with Sanger dideoxy sequencing. 20-nt of the 4′OMT2 targeting segment in the sgRNA sequence was used for a search in the BLASTN program of NCBI. In particular, the seed sequence of sgRNA (N)12, was examined to look for potential possible off-targets using our CRISPR/Cas9 system.
Metabolite Analyses
To determine the products of the BIA pathway, the opium poppy leaves were collected after 4 days of agro-infiltration and dried at 28 °C for 2–3 days according to previous reports41,47. Then, 0.1 g of dried samples were soaked in methanol (HPLC grade, Merck, Darmstadt, Germany) at room temperature and incubated for 24 hours. Four biological replicates were used for each control and transgenic line. For separation, marc filtration was performed and the samples were evaporated. The extracts were dissolved to a concentration of 2,000 ppm and diluted to 1/200 ppm from stock. The extracts were filtered through 0.45 μm membranes for HPLC-TOF/MS analysis. Morphine, codeine, thebaine, papaverine, noscapine and laudanosine alkaloids were analysed using their respective standards. Alkaloids were quantified in an Agilent 1260 Infinity HPLC system (Agilent, Palo Alto, CA) coupled with Agilent 6210 TOF-MS detector and Agilent EC 250/4 Nucleosil 100-5 (HPLC Column, Nucleosil C18, 100A, 5um, 4 × 250 mm). The column temperature was adjusted to 35 °C and the injection volume was set to 10 μL. Mobile phases A and B are water 1 mg/L and acetonitrile (in 0.1% formic acid) respectively. The elution program was: 0–6 min, 40% B; 6–10 min, 50% B; 10–15 min, 90% B; 15-16 min, 90% B; 16–25 min, 40% B. The statistical analysis was performed using one-way ANOVA test.
Results
Knock-out of 4′OMT2 with CRISPR/Cas9 system
In order to examine the efficiency of sgRNA mediated CRISPR/Cas9 genome editing in opium poppy. We targeted the poppy 4′OMT2 gene by introducing double strand brakes (DSBs) and observed InDel mutations directed by Non-Homologous End Joining (NHEJ) repair mechanism. A 20-bp sequence in the 5′ region of the 4′OMT2 locus included a 3 bp protospacer adjacent motif (PAM) was selected for targeting by sgRNA transcribed from both synthetic and viral vectors. The U6 promoter of A. thaliana (AtU6p) and sgRNA_4OMT2 (with 38.8% GC content) were assembled in a single cut-ligation MoClo L1 (Modular Cloning Level-1) reaction. The assembled sequence of AtU6p_sgRNA_4OMT2 was cloned into the ptv:00 (pTRV2) vector. These two cassettes were used as binary vectors with an hCas9 encoding plasmid (Addgene #46965) to transform the CRISPR/Cas9 system into plant cells. pTRV1 was also used during the transformation of the TRV-mediated CRISPR/Cas9 system for the effective expression of pTRV2 in plant tissues. Simultaneous expression of sgRNA and Cas9 in opium poppy resulted in the induction of frame-shift mutation in desired segments of the genome.
Identification of mutated gene segments in 4′OMT2
Constructed plasmids of 46965 (pK7WGF2::hCas9), pICH47751::AtU6p::sgRNA_4OMT2 and pTRV2::AtU6p::sgRNA_4OMT2 were transformed into opium poppy by Agrobacterium-mediated transformation to induce the expression of sgRNA and Cas9 in leaf tissues. To identify InDels in the target region, an isolated gDNA sample was enriched by using AgeI restriction enzyme to eliminate non-targeted 4′OMT2 gene segments and was then amplified by PCR and subjected to Sanger sequencing. A total of 48 clones were sequenced and used for InDel analyses. InDel sequence analysis revealed that 16 out of 20 (80%) randomly sequenced clones showed deletions from the synthesized -sgRNA and 24 out of 28 (85%) clones showed deletions as a result of viral-based sgRNA-mediated endonuclease activity of Cas9 (Fig. 2). The majority of the identified InDels were located at the region targeted by the seed sequence of sgRNA. The consequential mutations ranged from 1 to 4 bp insertion/deletions in the targeted segment. These results support the idea that CRISPR/Cas9 system is a precise and reliable tool to knock-out the gene of interest in the opium poppy genome with a huge genome size of nearly 3.3 Gb48.
After induction of mutagenesis in the Ps4′OMT gene region by CRISPR/Cas9 system, targeted sites were amplified from outside by PCR and InDels were identified by Sanger dideoxy sequencing.
Both results show that genome knockout lines were generated and the majority of gene knockout deleted 1 to 4 nt from the genome sequences.
To identify the presence of any potential off-targets, we used the online BLASTN tool of NCBI. The 20-nt sgRNA sequence was used to search for off-target candidate sites in the P. somniferum genome49. We identified a single candidate gene, 4′OMT1 (isoform-1), which had 17 bp matches out of 20 bp and 94% identity with a single base difference 9 bp upstream of the 3′ end.
The impact of CRISPR/Cas9 mediated gene knock out on plant BIA biosynthesis
To determine the efficiency of CRISPR/Cas9-mediated gene knockout, we employed HPLC-ToF/MS to measure the expression levels of several alkaloids (morphine, codeine, S-reticuline, noscapine, thebaine, laudanosine and papaverine) regulated by the 4′OMT2 gene. HPLC-ToF/MS data showed that the production of these detected alkaloids was significantly reduced in gene knockout plants (P < 0.005) (Figs 3 and 4; Supplementary Table S2). The level of total alkaloid accumulation was reduced in both synthetic and viral-based CRISPR-knockout plants. Interestingly, the decrease of total alkaloid in synthetic-based CRISPR-mediated plant knockout was higher than viral-based applications. In addition, S-reticuline was significantly decreased in the opium poppy leaves of the knockout plants and the most reduced alkaloid was found to be thebaine (Fig. 3). Also, the biosynthesis of the end-products morphine, noscapine and papaverine was reduced.
Discussion
This study demonstrates that successful application of the CRISPR/Cas9 system in opium poppy is possible by both synthetic and TRV-mediated systems. Previously, the TRV-based post-transcriptional gene silencing approach was successfully applied in opium poppy50,51. We took advantage of the efficiency of the system and used the TRV vector to express sgRNAs to improve the CRISPR/Cas9 system. TRV, as a viral plasmid, can systemically distribute sgRNAs from the transfected cell to its neighbouring plant cells. Additionally, there was a published report about the usage of TRV vectors for sgRNA transcription in Cas9 encoding tissues of N. benthamiana52.
In this study, we successfully showed the effectiveness of CRISPR/Cas9 system in P. somniferum, using synthetic and viral plasmid backbones to knock-out the 4′OMT2 gene which has a role in the regulation of the biosynthesis of many valuable medicinal aromatic metabolites including noscapine and morphine. The delivery method of the sgRNA/Cas9 system into plant cells is a limiting factor for highly efficient CRISPR/Cas9-mediated gene editing. Conventional Agrobacterium-mediated gene transformation requires more time to see results. With this in mind, we employed Agrobacterium-mediated leaf infiltration to deliver the Cas9 and sgRNAs into plant cells and investigate the gene editing efficiency. The 4′OMT2 gene was knocked-out using sgRNAs transcribed from both viral (TRV) and synthetic backbone vectors. No significant differences in efficiency were observed between those different applications. This could be due to using synthetic vectors for hCas9 in both strategies, which could have masked potential differences. When applying CRISPR/Cas9 in plants, it is not possible to express both Cas9 and sgRNA from a single viral vector. This is due to the low-cargo capacity of plant viral vectors. Hypothetically, some strategies could be used to overcome these obstacles. For example, the Cas9 sequence could be split and transferred into organisms. Then, it could be triggered to re-assemble into a functional protein form in vivo53. Another possibility is to use a smaller Staphylococcus aureus Cas9 protein which is one third the size of SpCas9 53,54. Such approaches can facilitate the expression of Cas9 endonuclease in plant tissues by using virus-based vectors. The TRV system works systemically in plant cells and it’s preferred for the delivery of sgRNA and human-codon optimized Cas9 (hCas9) coding sequences into the plant leaves52. It was thus used to express sgRNA_4OMT2 and sgRNA_PDS under an AtU6 promoter.
Analysis of off-target effects with the web-based NCBI database was limited due to lack of genome sequence information for the opium poppy. The homology between 4′OMT2 and 4′OMT1 was not considered problematic due to the recognition requirements for Cas9 endonuclease activity on its targets, which is based on the presence of an upstream PAM sequence. Mismatches at the 3′ end adjacent to the PAM sequence are not tolerated by the CRISPR/Cas9 system55. Therefore, this system can distinguish between 4′OMT2 and 4′OMT1 despite their high homology.
We also showed that the metabolite accumulation level in opium poppy was influenced by CRISPR/Cas9 based gene-editing (Fig. 3). As expected, the total alkaloid content was reduced in the case of gene editing. The reduction in the alkaloid might due to the low production of downstream compounds of BIA pathway. Consistent with previous results, the silencing of 4′OMT2 resulted in a considerable decrease in thebaine, codeine, noscapine and papaverine levels in stem41. Additionally, we detected a dramatic reduction in the S-reticuline and laudanosine content which are products of 4′OMT2 (Fig. 4). In accordance with our previous report, 4′OMT2 silencing led to the decrease of S-reticuline levels, indicating its possible role in morphine and noscapine biosynthesis47. We also detected a slight reduction in thebaine levels which is likely mediated by the RNA-based silencing of 4′OMT2 in leaf47. This might be explained by the high thebaine accumulation in control plants.
Consequently, our study demonstrated that CRISPR/Cas9 system is applicable in P. somniferum which can be used as a framework in future studies to address some probable questions regarding the BIA biosynthesis pathway (Fig. 1). By applying CRISPR/Cas9 in Papaver species, opium poppy and related species can be converted into Biofactories for the mass-production of BIAs simply by introducing breaks in related gene sequences. The CRISPR/Cas9 system provides a wide range of applications, with high accuracy and precision in comparison to other genome editing tools. Therefore, it will be a commonly used technique for plant metabolic engineering in the near future.
Additional Information
How to cite this article: Alagoz, Y. et al. Manipulating the Biosynthesis of Bioactive Compound Alkaloids for Next-Generation Metabolic Engineering in Opium Poppy Using CRISPR-Cas 9 Genome Editing Technology. Sci. Rep. 6, 30910; doi: 10.1038/srep30910 (2016).
References
Chen, K. & Gao, C. Targeted Gene Mutation in Plants. in Somatic Genome Manipulation (eds Li, X. Q. et al.), 253–272 (Springer, 2015).
Gaj, T., Gersbach, C. A. & Barbas, C. F. ZFN, TALEN and CRISPR/Cas-based methods for genome engineering. Trends in biotechnology 31, 397–405 (2013).
Li, T. et al. TAL nucleases (TALNs): hybrid proteins composed of TAL effectors and FokI DNA-cleavage domain. Nucleic acids research 39, 359–372 (2011).
Kim, Y.-G., Cha, J. & Chandrasegaran, S. Hybrid restriction enzymes: zinc finger fusions to Fok I cleavage domain. Proceedings of the National Academy of Sciences 93, 1156–1160 (1996).
Händel, E.-M., Alwin, S. & Cathomen, T. Expanding or restricting the target site repertoire of zinc-finger nucleases: the inter-domain linker as a major determinant of target site selectivity. Molecular Therapy 17, 104–111 (2009).
Urnov, F. D., Rebar, E. J., Holmes, M. C., Zhang, H. S. & Gregory, P. D. Genome editing with engineered zinc finger nucleases. Nature Reviews Genetics 11, 636–646 (2010).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Jinek, M. et al. RNA-programmed genome editing in human cells. Elife 2, e00471 (2013).
Jiang, W., Bikard, D., Cox, D., Zhang, F. & Marraffini, L. A. RNA-guided editing of bacterial genomes using CRISPR-Cas systems. Nature biotechnology 31, 233–239 (2013).
Hwang, W. Y. et al. Efficient genome editing in zebrafish using a CRISPR-Cas system. Nature biotechnology 31, 227–229 (2013).
Cho, S. W., Kim, S., Kim, J. M. & Kim, J.-S. Targeted genome engineering in human cells with the Cas9 RNA-guided endonuclease. Nature biotechnology 31, 230–232 (2013).
Li, J.-F. et al. Multiplex and homologous recombination-mediated genome editing in Arabidopsis and Nicotiana benthamiana using guide RNA and Cas9. Nat. Biotechnol. 31, 688–691 (2013).
Lawrenson, T. et al. Induction of targeted, heritable mutations in barley and Brassica oleracea using RNA-guided Cas9 nuclease. Genome Biol. 16, 1 (2015).
Jia, H. & Wang, N. Targeted genome editing of sweet orange using Cas9/sgRNA. PLoS One 9, e93806 (2014).
Chandrasekaran, J. et al. Development of broad virus resistance in non-transgenic cucumber using CRISPR/Cas9 technology. Molecular plant pathology (in press), doi: 10.1111/mpp.12375 (2016).
Jacobs, T. B., LaFayette, P. R., Schmitz, R. J. & Parrott, W. A. Targeted genome modifications in soybean with CRISPR/Cas9. BMC Biotechnol. 15, 1 (2015).
Woo, J. W. et al. DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins. Nat. Biotechnol. (2015).
Sugano, S. S. et al. CRISPR/Cas9 Mediated Targeted Mutagenesis in the Liverwort Marchantia polymorpha L. Plant Cell Physiol. 55, 475–481 (2014).
Michno, J.-M. et al. CRISPR/Cas mutagenesis of soybean and Medicago truncatula using a new web-tool and a modified Cas9 enzyme. GM crops & food 6, 243–252 (2015).
Nekrasov, V., Staskawicz, B., Weigel, D., Jones, J. D. & Kamoun, S. Targeted mutagenesis in the model plant Nicotiana benthamiana using Cas9 RNA-guided endonuclease. Nat. Biotechnol. 31, 691–693 (2013).
Baltes, N. J., Gil-Humanes, J., Cermak, T., Atkins, P. A. & Voytas, D. F. DNA replicons for plant genome engineering. The Plant Cell 26, 151–163 (2014).
Xie, K. & Yang, Y. RNA-guided genome editing in plants using a CRISPR–Cas system. Molecular plant 6, 1975–1983 (2013).
Fan, D. et al. Efficient CRISPR/Cas9-mediated targeted mutagenesis in Populus in the first generation. Sci. Rep 5 (2015).
Zhou, X., Jacobs, T. B., Xue, L. J., Harding, S. A. & Tsai, C. J. Exploiting SNPs for biallelic CRISPR mutations in the outcrossing woody perennial Populus reveals 4-coumarate: CoA ligase specificity and redundancy. New Phytologist 208, 298–301 (2015).
Ron, M. et al. Hairy root transformation using Agrobacterium rhizogenes as a tool for exploring cell type-specific gene expression and function using tomato as a model. Plant Physiol. 166, 455–469 (2014).
Wang, S. et al. Efficient targeted mutagenesis in potato by the CRISPR/Cas9 system. Plant Cell Rep 34, 1473–1476 (2015).
Jiang, W. et al. Demonstration of CRISPR/Cas9/sgRNA-mediated targeted gene modification in Arabidopsis, tobacco, sorghum and rice. Nucleic Acids Res. 41, e188 (2013).
Shan, Q. et al. Targeted genome modification of crop plants using a CRISPR-Cas system. Nat. Biotechnol. 31, 686–688 (2013).
Liang, Z., Zhang, K., Chen, K. & Gao, C. Targeted mutagenesis in Zea mays using TALENs and the CRISPR/Cas system. Journal of Genetics and Genomics 41, 63–68 (2014).
Fairbairn, J. & Wassel, G. The alkaloids of Papaver somniferum L.—I.: Evidence for a rapid turnover of the major alkaloids. Phytochemistry 3, 253–258 (1964).
La Valva, V., Sabato, S. & Gigliano, G. S. Morphology and alkaloid chemistry of Papaver setigerum DC.(Papaveraceae). Taxon 34, 191–196 (1985).
Ilari, A. et al. Structural basis of enzymatic (S)-norcoclaurine biosynthesis. Journal of biological chemistry 284, 897–904 (2009).
Choi, K.-B., Morishige, T., Shitan, N., Yazaki, K. & Sato, F. Molecular cloning and characterization of coclaurinen-methyltransferase from cultured cells of Coptis japonica. Journal of Biological Chemistry 277, 830–835 (2002).
Ziegler, J., Diaz-Chávez, M. L., Kramell, R., Ammer, C. & Kutchan, T. M. Comparative macroarray analysis of morphine containing Papaver somniferum and eight morphine free Papaver species identifies an O-methyltransferase involved in benzylisoquinoline biosynthesis. Planta 222, 458–471 (2005).
Morishige, T., Tsujita, T., Yamada, Y. & Sato, F. Molecular Characterization of theS-Adenosyl-l-methionine: 3′-Hydroxy-N-methylcoclaurine 4′-O-Methyltransferase Involved in Isoquinoline Alkaloid Biosynthesis in Coptis japonica. Journal of Biological Chemistry 275, 23398–23405 (2000).
De-Eknamkul, W. & Zenk, M. H. Purification and properties of 1, 2-dehydroreticuline reductase from Papaver somniferum seedlings. Phytochemistry 31, 813–821 (1992).
Facchini, P. J. et al. Opium poppy: blueprint for an alkaloid factory. Phytochemistry Reviews 6, 97–124 (2007).
Frick, S. et al. Transformation of opium poppy (Papaver somniferumL.) with antisense berberine bridge enzyme gene (anti-bbe) via somatic embryogenesis results in an altered ratio of alkaloids in latex but not in roots. Transgenic research 13, 607–613 (2004).
Apuya, N. R. et al. Enhancement of alkaloid production in opium and California poppy by transactivation using heterologous regulatory factors. Plant biotechnology journal 6, 160–175 (2008).
Hosseini, B. et al. Transient Expression of cor Gene in Papaver somniferum. BioImpacts: BI 1, 229 (2011).
Desgagné-Penix, I. & Facchini, P. J. Systematic silencing of benzylisoquinoline alkaloid biosynthetic genes reveals the major route to papaverine in opium poppy. The Plant Journal 72, 331–344 (2012).
Gurkok, T. et al. Functional Characterization of 4′OMT and 7OMT Genes in BIA Biosynthesis. Frontiers in Plant Science 7, doi: 10.3389/fpls.2016.00098 (2016).
Weber, E., Engler, C., Gruetzner, R., Werner, S. & Marillonnet, S. A modular cloning system for standardized assembly of multigene constructs. PLoS One 6, e16765 (2011).
Hanahan, D. Techniques for transformation of E. coli. In DNA cloning: a practical approach, ed Glover D. M. (I. R. L. Press, Oxford, United Kingdom), 1, 109–135 (1985).
Holsters, M. et al. Transfection and transformation of Agrobacterium tumefaciens. Molecular and General Genetics MGG 163, 181–187 (1978).
Lu, R., Martin-Hernandez, A. M., Peart, J. R., Malcuit, I. & Baulcombe, D. C. Virus-induced gene silencing in plants. Methods 30, 296–303 (2003).
Gurkok, T. et al. Functional Characterization of 4′ OMT and 7OMT Genes in BIA Biosynthesis. Frontiers in plant science 7 (2016).
Ziegler, J. & Facchini, P. J. Alkaloid biosynthesis: metabolism and trafficking. Annu. Rev. Plant Biol. 59, 735–769 (2008).
Gao, J. et al. CRISPR/Cas9-mediated targeted mutagenesis in Nicotiana tabacum. Plant molecular biology 87, 99–110 (2015).
Hileman, L. C., Drea, S., Martino, G., Litt, A. & Irish, V. F. Virus‐induced gene silencing is an effective tool for assaying gene function in the basal eudicot species Papaver somniferum (opium poppy). The Plant Journal 44, 334–341 (2005).
Wijekoon, C. P. & Facchini, P. J. Systematic knockdown of morphine pathway enzymes in opium poppy using virus‐induced gene silencing. The Plant Journal 69, 1052–1063 (2012).
Ali, Z., Abul-faraj, A., Piatek, M. & Mahfouz, M. M. Activity and Specificity of TRV-Mediated Gene Editing in Plants. Plant signaling & behavior 10, e1044191 (2015).
Zetsche, B., Volz, S. E. & Zhang, F. A split-Cas9 architecture for inducible genome editing and transcription modulation. Nat. Biotechnol. 33, 139–142 (2015).
Ran, F. A. et al. In vivo genome editing using Staphylococcus aureus Cas9. Nature 520, 186–191 (2015).
Sternberg, S. H., Redding, S., Jinek, M., Greene, E. C. & Doudna, J. A. DNA interrogation by the CRISPR RNA-guided endonuclease Cas9. Nature 507, 62–67 (2014).
Acknowledgements
The authors would like to acknowledge TUBİTAK with grant no: 111O036, Cankiri Karatekin University BAP office and Turkish Academy of Science for GEBIP support. We greatly appreciate Ms. Faten Taki and Mr. Kevin Adams for their carefully proofread this manuscript.
Author information
Authors and Affiliations
Contributions
B.Z. and T.U. designed the experiment. Y.A. and T.G. performed the experiments. Y.A., T.G., B.Z. and T.U. did data analysis and wrote the manuscript. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Alagoz, Y., Gurkok, T., Zhang, B. et al. Manipulating the Biosynthesis of Bioactive Compound Alkaloids for Next-Generation Metabolic Engineering in Opium Poppy Using CRISPR-Cas 9 Genome Editing Technology. Sci Rep 6, 30910 (2016). https://doi.org/10.1038/srep30910
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep30910
This article is cited by
-
A high-efficient protoplast transient system for screening gene editing elements in Salvia miltiorrhiza
Plant Cell Reports (2024)
-
Agronomical Strategies to Improve Growth, Physio-Biochemistry, Yield and Quality Attributes of Mint Plants Under the Varied Environmental Conditions: a Review
Journal of Soil Science and Plant Nutrition (2023)
-
Biotechnology based strategies for secondary metabolites enhancement: a review
Vegetos (2023)
-
Low phytate soybean: next generation metabolic engineering using CRISPR-Cas 9 genome editing technology
Journal of Plant Biochemistry and Biotechnology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.