Abstract
Carbon–carbon bond formation by metal-free cross-coupling of two reactants with low reactivity represents a challenge in organic synthesis. Secondary amides and alkenes are two classes of bench-stable compounds. The low electrophilicity of the former and low nucleophilicity of the latter make the direct coupling of these two partners challenging yet highly desirable. We report herein an unprecedented intermolecular reaction of secondary amides with alkenes to afford α,β-unsaturated ketimines or enones, which are versatile intermediates for organic synthesis and are prevalent in bioactive compounds and functional materials. Our strategy relies on the chemoselective activation of the secondary amide with trifluoromethanesulfonic anhydride (Tf2O)/2-fluoropyridine to generate a highly reactive nitrilium intermediate, which reacts efficiently with alkenes. This metal-free synthesis is characterized by its mild reaction conditions, excellent functional group tolerance and chemoselectivity, allowing the preparation of multi-functionalized compounds without using protecting groups.
Similar content being viewed by others
Introduction
Organic chemistry is the chemistry of carbon compounds. Thus carbon–carbon (C–C) bond-forming reactions occupy the central position in organic synthesis. Most of these reactions are polar1 by nature and involve the reaction of a nucleophile with an electrophile. The direct reaction of a weak nucleophile with a weak electrophile (Nuw-Elw) is difficult under conventional reaction conditions. Alkenes are a class of stable and easily available weak π-nucleophiles, which can only undergo transition metal-mediated C–C bond forming reactions, or react with reactive electrophiles such as acyl chlorides (Friedel-Crafts acylation of alkenes)2 or in situ generated highly electrophilic intermediates, such as nitrilium ions [the extended Bischler–Napieralski (B–N) reaction3,4,5,6,7], iminium8/N-acyliminium ions9, and acid-activated aldehydes/ketones (Prins reaction10). On the other hand, although nitrilium ions are key intermediates in several classical reactions such as Houben-Hoesch11, Ritter12,13, von Braun13, Bischler-Napieralski13, Beckmann13, Schmidt13, and Ugi reactions14, their participation in synthetically useful intermolecular reactions with alkenes is unknown. Amides are another class of bench-stable compounds with low electrophilicity15,16 due to the strong resonance between the π* orbital of the carbonyl group and the nitrogen lone pair. It is thus challenging to couple alkenes with amides, especially secondary amides because of the acidic proton on the N-atom. As a result, only isolated examples of intramolecular coupling reactions under harsh conditions are known (Fig. 1a). An efficient intermolecular cross-coupling reaction of alkenes with secondary amides remains elusive (Fig. 1b). However, such a transformation would be highly useful considering the widespread use of secondary amides as intermediates in organic synthesis and the requisite conversion of these species into other classes of compounds16,17 at lower oxidation states15,18,19,20,21,22,23,24 as well as the versatility of α,β-unsaturated ketimines (enimines)25 and α,β-unsaturated ketones (enones) in organic synthesis, medicinal chemistry26, and molecular switches27.
In response to this challenge, we report herein a metal-free intermolecular coupling reaction of secondary amides with alkenes to afford multi-functionalized α,β-unsaturated ketimines and enones (Fig. 1b). Importantly, with the use of trifluoromethanesulfonic (triflic) anhydride (Tf2O) as a powerful yet chemoselective amide-activating reagent, the reactions are conducted under mild conditions and tolerate a host of sensitive functional groups in both the nucleophilic (alkenes) and electrophilic (secondary amides) reaction partners.
Results and Discussion
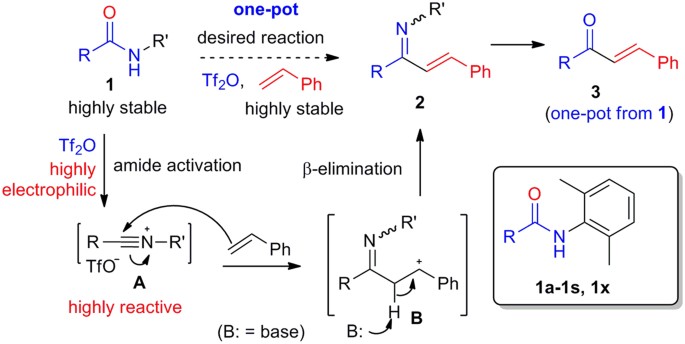
Reaction design
To realize the direct cross-coupling of an alkene with a secondary amide, it is necessary to activate one of the reaction partners. Inspired by the B–N reaction, we opted for the in situ activation of the amide group. Considering the low efficiency of the classical amide activators such as P2O5 and POCl33,4,10, highly electrophilic trifluoromethanesulfonic (triflic) anhydride (Tf2O)28 was selected for our purpose. Tf2O in combination with a base such as 2,6-di-tert-butyl-4-methylpyridine (DTBMP)29, Hünig base30, 2-chloropyridine31, 2-fluoropyridine32,33,34,35,36, 2-iodopyridine37, 2,4,6-collidine38,39,40,41, and 3-cyanopyridine42 had been employed for the activation of amides in various C–C bond-forming reactions. A secondary amide 1, once treated with Tf2O, would generate a highly reactive nitrilium intermediate A32,34,35,36,43 (Fig. 2). The latter could then be captured by an alkene to give an enimine 2 after C–C bond formation and deprotonation. Hydrolysis of the enimine 2 would afford enone 3 in one-pot from amide 1.
Optimization of reaction conditions
To avoid possible side reactions such as 1,5-hydride migration reaction36, the amides 1 bearing a N-2,6-dimethylphenyl group were designed as substrates for the investigation (Fig. 3). At the outset of our studies, base-free amide activation protocol was attempted. To our delight, successive treatment of a solution of secondary amide 1a (1.0 equiv) in CH2Cl2 (0.25 M) with Tf2O (1.1 equiv) at 0 °C for 10 min and then styrene (3.0 equiv) at room temperature for 2 h produced the desired α,β-unsaturated ketimine 2a in 51% yield as a mixture of E/Z isomers in a ratio of 5.5:1 (entry 1). The stereochemistry of major geometric isomer was determined as E by NOESY technique (cf. Supplementary Figure 53). Note that Tf2O failed to promote the B–N cyclization reaction in the absence of a base unless highly electron-rich substrates were used44. Encouraged by this result, the effects of base were surveyed. Among the bases screened, 2-F-pyridine was found to be the best (entries 3–9). Under these conditions, the amount of styrene could be reduced to 1.2 equiv without affecting the reaction efficiency (entries 10 and 11). The optimal conditions were thus defined as successive treatment of a solution of secondary amide 1a (1.0 equiv) and 2-fluoropyridine (1.2 equiv) in CH2Cl2 (0.25 M) with Tf2O (1.1 equiv) at 0 °C for 10 min, and then with styrene (1.2 equiv) at room temperature or 40 °C for 2 h. The reaction mixture was concentrated without work-up and subjected to flash chromatographic purification to give α,β-unsaturated ketimine 2a.
Substrate scope of the direct C–H alkyliminylation
With optimized conditions in hand, the coupling reactions of a series of N-(2,6-dimethylphenyl)benzamides 1 with a number of alkenes were investigated (Fig. 4). Styrene bearing electron-donating groups (Me, OMe) and electron-withdrawing halogens (Br, Cl, F) reacted smoothly to give the corresponding enimines in excellent yields (2b–2f, 88–99% yields), demonstrating superior reactivity compared with reported methods. α-Methylstyrene and α-phenylstyrene were also competent substrates (2g, 2h). Gratifyingly, the reaction was also compatible with the use of di- and trisubstituted aliphatic alkenes and 1,3-dienes (2i–2l). The reaction of 2-methyl-2-butene produced non-conjugated β,γ-unsaturated ketimine 2k. Further investigation revealed that the reaction was rather insensitive to the electronic properties of the benzamide derivatives and tolerated electron-donating groups such as methyl group (2m) and methoxy group (2n), as well as the highly electron-withdrawing nitro group (2p, 2q).
aReaction conditions: Amide (1.0 equiv), 2-F-Pyr. (1.2 equiv), CH2Cl2 (0.25 M), then 0 °C, Tf2O (1.1 equiv), 10 min. Alkene (1.2 equiv), 2 h. bIsolated yield. cReaction ran at room temperature (rt). dReaction ran at 40 °C. eThe E/Z ratio of imines was determined by 1H NMR. fThe structure was determined by X-Ray analysis (cf. Supplementary Figure 54). g2.5 equiv of amide 1f and 1.0 equiv of 1,4-divinylbenzene were used. Ts = 4-toluenesulfonyl, TBS = tert-butyldimethylsilyl.
The current reaction is characterized by its broad tolerance of sensitive functional groups including bromo (2o), nitro (2p, 2q), ester (2r, 2w), ketone (2s, 2z), aldehyde (2t), cyano (2u), azido (2x), tertiary amide (2v, 2y), sulfonamide (2aa), phenol (2ab) and silyl ether groups (2ac), many of which are not compatible with organometallic reagents. The highly functionalized products were all obtained in good to excellent yields, demonstrating great potential for the Tf2O-promoted method in the synthesis of complex structures. Interestingly, p-vinylstyrene could react selectively at one end giving 2ad in 87% yield, or at both alkenes leading to 2ae in 85% yield. Finally, the reaction could be scaled up to 20 mmol-scale without yield loss as demonstrated by the reaction of 1a with styrene (2a, yield: 95%, 5.91 g, Fig. 4).
Substrates scope of the direct C–H acylation
We then turned our attention to the synthesis of enones by in situ hydrolysis of the ketimine products. After the Tf2O/2-fluoropyridine-mediated dehydracoupling, the reaction mixture was concentrated and heated to reflux in a mixture of ethanol and 3 M HCl (1:1, v/v) to afford the desired enones (Fig. 5). Functionalized chalcones 3c–3h were synthesized in good to excellent yields by employing styrenyl alkenes and benzamide derivatives as substrates. Aliphatic and α,β-unsaturated amides were also excellent substrates (3i–3k, 3m). N-Alkyl amides are valuable directing group for both classical lithiation-functionalization45 and modern C–H functionalization reactions46,47,48,49. As a result, the transformation of the functionalized amide products obtained in these reactions into other classes of compounds are imperative. To demonstrate the value of our method in this context, the N-methyl amides 1t45, 1u48 and 1v49, which were previously obtained through transition-metal-catalyzed C–H activation, were converted into the corresponding enones 3n–3p in 60–73% yields.
Synthetic applications
To demonstrate the synthetic potential of the enone synthesis, the coupling reaction of styrene with (S)-N-methyl-tetrahydro-5-oxo-2-furaneamide (1w), readily available in 99% ee from L-glutamic acid50, was undertaken (Fig. 6a). To our delight, the desired enone (S)-3q was obtained in 70% yield without racemization (cf. Supplementary Figure 55). Multi-functionalized lactone-enones like 3q are versatile building blocks for the synthesis of bioactive natural products51.
The synthetic utility was further demonstrated by the synthesis of okanin (4) (Fig. 6b), a natural product that has been found in various folk medications used in China and Korea for treating inflammation, malaria, hypertension, diabetes, snake bite and smallpox52. The amide 1x, prepared in one step from commercially available 2,3,4-trimethoxybenzoic acid by amidation using Ye’s coupling reagent53 (cf. Supplementary Figure 60), reacted smoothly with 3,4-dimethoxystyrene to afford enone 3r in 86% yield. Exhaustive demethylation using BBr3 furnished okanin (4) in 84% yield.
Mechanistic investigation
To provide some experimental proofs for the presumed intermediacy of a highly electrophilic nitrilium ion, a series of NMR experiments were carried out. Secondary amide 1p was chosen for the mechanistic studies and base-free amide activation with Tf2O was first investigated (Fig. 7a). After addition of Tf2O into a solution of amide 1p, the formation of iminium salt Cp (as a 3.4:1 mixture of two geometric isomers) and nitrilium ion Ap in a ratio of Cp:Ap = 37:63 (1H NMR, Fig. 8a) was observed. The presence of nitrilium ion Ap was manifested by the characteristic triplet resonance and the coupling constant J13C-14N of a nitrilium which appeared at δC2 = 123.4 (t, J13C-14N = 45.6 Hz54), as well as the nitrilium N-α aromatic carbon at δC9 = 121.9 (t, J13C-14N = 13.5 Hz54) (13C NMR, Fig. 8b). Besides, the formation of TfOH was also observed by 1H and 13C NMR spectra. The same reaction by bench chemistry produced the enimine 2af in 51% yield along with the recovered starting 1p in 31% yield (Fig. 7a). Hence, the results obtained from the NMR experiments (Cp:Ap = 37:63) and those from the bench reaction (2af:1p = 38:62) suggested that nitrilium ion Ap was probably the only competent intermediate that reacted with styrene to produce enimine 2af. The less reactive iminium salt Cp was inert to styrene addition and hydrolyzed upon work-up to regenerate the starting material 1p. These results also implicated that addition of a base would facilitate the conversion of iminium salt Cp to nitrilium ion Ap, and thus improve the yield of enimine 2af. Experimentally, the addition of 1.2 equiv of 2-fluoropyridine boosted the yield of 2af to 97%. In addition, treating a mixture of amide 1p and 2-fluoropyridine in CD2Cl2 with Tf2O at 0 °C resulted in quantitative formation of nitrilium ion intermediate Ap (Fig. 7b) along with 2-fluoropyridinium salt D within 10 min (cf. Supplementary Figure 57 for NMR spectrum). These results confirmed that 2-fluoropyridine promoted the transformation of the iminium salt Cp to nitrilium ion intermediate Ap (Fig. 7b). Moreover, the in situ IR monitoring showed the formation of iminium salt Cp (1663 cm−1) and nitrilium ion Ap (2310 cm−1) upon treatment of amide 1p with Tf2O. The former was converted completely into the latter by action of 2-fluoropyridine. A strong absorption of 2-F-pyridinium trifluoromethanesulfonate32 (1635 cm−1) was observed, while no absorption corresponding to pyridinium ion Ep was observed (cf. Supplementary Figure 58 for in situ IR spectra).
Conclusion
In summary, we have developed a general method for the metal-free intermolecular C–H functionalization of alkenes with secondary amides. This method provides a direct and high-yielding access to α,β-unsaturated ketimines and enones from two classes of readily available and stable starting materials. The one-pot reaction exhibits excellent functional group tolerance for both alkenes and amides allowing convenient and efficient synthesis of a variety of functionalized α,β-unsaturated ketimines and enones. The present method could find wide applications in organic synthesis especially considering the remarkable chemoselectivity.
Methods
General procedure for the direct C–H alkyliminylation and acylation of alkenes with secondary amides to give α,β-unsaturated ketimines (enimines) 2 and α,β-unsaturated ketones 3 (enones)
Into a dry 10-mL round-bottom flask equipped with a magnetic stirring bar were added successively a secondary amide (0.5 mmol, 1.0 equiv), 2 mL of anhydrous CH2Cl2 and 2-fluoropyridine (0.6 mmol, 1.2 equiv) under an argon atmosphere. After being cooled to 0 °C, trifluoromethanesulfonic anhydride (Tf2O) (0.55 mmol, 1.1 equiv) was added dropwise via a syringe and the reaction was stirred for 10 min. To the resulting mixture, an alkene (0.6 mmol, 1.2 equiv) was added dropwise at 0 °C. The mixture was allowed to warm-up to room temperature (or 40 °C) and stirred for 2 h. The reaction mixture was concentrated under reduced pressure, and the residue was purified by flash column chromatography (FC) on silica gel (pre-neutralized with 2% Et3N in n-hexane) to afford the desired α,β-unsaturated ketimine 2.
Alternately, to the resulting residue were added 5 mL of EtOH and 5 mL of an aqueous solution of HCl (3.0 M). The resulting mixture was heated to reflux until completion of the reaction as monitored by TLC analysis (2–12 h). After being cooled to room temperature, 10 mL of CH2Cl2 was added, and the mixture extracted with CH2Cl2 (3 × 10 mL). The combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated under reduced pressure. The residue was purified by flash column chromatography on silica gel to afford the desired α,β-unsaturated ketone 3.
Data availability
The X-ray crystallographic coordinates for structures reported in this study have been deposited at the Cambridge Crystallographic Date Center (CCDC), under deposition number CCDC 1438540 (for 2w). These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Additional Information
How to cite this article: Huang, P.-Q. et al. Metal-Free C–H Alkyliminylation and Acylation of Alkenes with Secondary Amides. Sci. Rep. 6, 28801; doi: 10.1038/srep28801 (2016).
References
Ho, T.-L. Polarity control for synthesis. (Wiley & Sons, Ltd -August 1991).
Snider, B. B. & Jackson, A. C. Use of ethylaluminum dichloride as a catalyst for the Friedel-Crafts acylation of alkenes. J. Org. Chem. 47, 5393‒5395 (1982).
Bischler, A. & Napieralski, B. Zur Kenntniss einer neuen Isochinolinsynthese. Ber. 26, 1903–1908 (1893).
Schnider, O. & Hellerbach, J. Synthese von Morphinanen. (2. Mitteilung). Helv. Chim. Acta 33, 1437‒1448 (1950).
Sugasawa, S. & Ushioda, S. Extension of the Bischler-Napieralski reaction‒II synthesis of pyrroline derivatives. Tetrahedron 5, 48‒52 (1959).
Gawley, R. E. & Chemburkar, S. Generation and cyclization of nitrilium ions from amides. Asymmetric synthesis of fused azabicyclics. Tetrahedron Lett. 27, 2071–2074 (1986).
Marquart, A. L. et al. Synthesis of chiral hydroxylated quinolizidines via vinylogous Bischler-Napieralski nitrilium ion cyclizations. J. Org. Chem. 59, 2092–2100 (1994).
Royer, J., Bonin, M. & Micouin, L. Chiral heterocycles by iminium ion cyclization. Chem. Rev. 104, 2311–2352 (2004).
Maryanoff, B. E., Zhang, H.-C., Cohen, J. H., Turchi, I. J. & Maryanoff, C. A. Cyclizations of N-acyliminium ions. Chem. Rev. 104, 1431–1628 (2004).
Snider, B. B. 2.03 Prins reactions and carbonyl, imine, and thiocarbonyl Ene. Reactions in Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, from Comprehensive Organic Synthesis II (Second Edition). 2, 148–191 (2014).
Wang, Z. Houben-Hoesch reaction, In Comprehensive Organic Name Reactions and Reagents, Chapt. 334, pp. 1496–1500 (John Wiley & Sons, Inc. 2010).
Bishop, R. 6.07 Ritter-type reactions, in Reference Module in Chemistry, Molecular Sciences and Chemical Engineering, from Comprehensive Organic Synthesis II (Second Edition). 6, 239–295 (2014).
Fodor, G. & Nagubandi, S. Correlation of the von Braun, Ritter, Bischler-Napieralski, Beckmann and Schmidt reactions via nitrilium salt intermediates. Tetrahedron 36, 1279–1300 (1980).
Kaim, L. E. & Grimaud, L. Beyond the Ugi reaction: less conventional interactions between isocyanides and iminium species. Tetrahedron 65, 2153–2171 (2009).
Pace, V., Holzer, W. & Olofsson, B. Increasing the reactivity of amides towards organometallic reagents: an overview. Adv. Synth. Catal. 356, 3697–3736 (2014).
Ruider, S. A. & Maulide, N. Strong bonds made weak: towards the general utility of amides as synthetic modules. Angew. Chem., Int. Ed. 54, 13856–13858 (2015).
Hie, L. et al. Conversion of amides to esters by the nickel-catalysed activation of amide C–N bonds. Nature 524, 79–83 (2015).
Weires, N. A., Baker, E. L. & Garg, N. K. Nickel-catalysed Suzuki–Miyaura coupling of amides. Nat. Chem. 8, 75–79 (2016).
Wu, X. et al. Cobalt-catalysed site-selective intra- and intermolecular dehydrogenative amination of unactivated sp3 carbons. Nat. Commun. 6, 6462–6471 (2015).
Gregory, A. W., Chambers, A., Hawkins, A., Jakubec, P. & Dixon, D. J. Iridium-catalyzed reductive nitro-Mannich cyclization. Chem. Eur. J. 21, 111–114 (2015).
Chen, Z.-H., Tu, Y.-Q., Zhang, S.-Y. & Zhang, F.-M. Development of the intramolecular Prins cyclization/Schmidt reaction for the construction of the azaspiro[4,4]nonane: Application to the formal synthesis of (±)-Stemonamine. Org. Lett. 13, 724–727 (2011).
Li, B.-S. et al. Carbon–carbon bond activation of cyclobutenones enabled by the addition of chiral organocatalyst to ketone. Nat. Commun. 6, 6207–6211 (2015).
Shirokane, K. et al. Total synthesis of (±)-Gephyrotoxin by amide-selective reductive nucleophilic addition. Angew. Chem., Int. Ed. 53, 512–516 (2014).
Oda, Y., Sato, T. & Chida, N. Direct chemoselective allylation of inert amide carbonyls. Org. Lett. 14, 950–953 (2012).
Duttwyler, S., Lu, C., Rheingold, A. L., Bergman, R. G. & Ellman, J. A. Highly diastereoselective synthesis of tetrahydropyridines by a C–H activation–cyclization–reduction cascade. J. Am. Chem. Soc. 134, 4064–4067 (2012).
Thebtaranonth, C. & Thebtaranonth, Y. The Chemistry of Enones, vol. 29 (Eds.: Patai, S. & Rappoport, Z. ), Wiley, New York, 199–280 (1989).
Lumento, F. et al. Quantum chemical modeling and preparation of a biomimetic photochemical switch. Angew. Chem. Int. Ed. 46, 414–420 (2007).
Baraznenok, I. L., Nenajdenko, V. G. & Balenkova, E. S. Chemical transformations induced by triflic anhydride. Tetrahedron 56, 3077‒3119 (2000).
Chen, L.-Y. & Ghosez, L. Study of chiral auxiliaries for the intramolecular [2 + 2] cycloaddition of a keteniminium salt to an olefinic double bond. A new asymmetric synthesis of cyclobutanones. Tetrahedron Lett. 31, 4467‒4470 (1990).
Sisti, N. J., Fowler, F. W. & Grierson, D. S. N-phenyl-2-cyano-1-azadienes: New versatile heterodienes in the Diels-Alder reaction. Synlett. 816–818 (1991).
Movassaghi, M. & Hill, M. D. A versatile cyclodehydration reaction for the synthesis of isoquinoline and β-carboline derivatives. Org. Lett. 10, 3485–3488 (2008).
Medley, J. W. & Movassaghi, M. Direct dehydrative N-pyridinylation of amides. J. Org. Chem. 74, 1341–1344 (2009).
Bechara, W. S., Pelletier, G. & Charette, A. B. Chemoselective synthesis of ketones and ketimines by addition of organometallic reagents to secondary amides. Nat. Chem. 4, 228–234 (2012).
Xiao, K.-J., Wang, A.-E. & Huang, P.-Q. Direct Transformation of secondary amides into secondary amines: triflic anhydride activated reductive alkylation. Angew. Chem., Int. Ed. 51, 8314–8317 (2012).
Xiao, K.-J., Wang, A.-E., Huang, Y.-H. & Huang, P.-Q. Versatile and direct transformation of secondary amides into ketones by deaminative alkylation with organocerium reagents. Asian J. Org. Chem. 1, 130‒132 (2012).
Xiao, K.-J., Huang, Y.-H. & Huang, P.-Q. General direct transformation of secondary amides to ketones via amide activation. Acta Chim. Sinica 70, 1917–1922 (2012).
Peng, B., Geerdink, D., Farès, C. & Maulide, N. Chemoselective intermolecular α-arylation of amides. Angew. Chem., Int. Ed. 53, 5462–5466 (2014).
Madelaine, C., Valerio, V. & Maulide, N. Unexpected electrophilic rearrangements of amides: a stereoselective entry to challenging substituted lactones. Angew. Chem., Int. Ed. 49, 1583–1586 (2010).
Valerio, V., Petkova, D., Madelaine, C. & Maulide, N. Direct room-temperature lactonisation of alcohols and ethers onto amides: an “amide strategy” for synthesis. Chem. Eur. J. 19, 2606–2610 (2013).
Peng, B., Huang, X., Xie, L.-G. & Maulide, N. A Brønsted acid catalyzed redox arylation. Angew. Chem., Int. Ed. 53, 8718–8721 (2014).
Padmanaban, M. et al. Investigation of cationic Claisen-type electrophilic rearrangements of amides. Tetrahedron 71, 5994–6005 (2015).
Medley, J. W. & Movassaghi, M. A concise and versatile double-cyclization strategy for the highly stereoselective synthesis and arylative dimerization of Aspidosperma alkaloids. Angew. Chem., Int. Ed. 51, 4572–4576 (2012).
Fodor, G., Gal, J. & Phillips, B. A. The mechanism of the Bischler–Napieralski reaction. Angew. Chem., Int. Ed. 11, 919–920 (1972).
Nagubandi, S. & Fodor, G. Novel condensing agents for Bischler–Napieralski type cyclodehydration. Heterocycles 15, 165–177 (1981).
Lutz, G. P., Du, H., Gallagher, D. J. & Beak, P. Synthetic applications of the β-lithiation of β-aryl secondary amides: diastereoselective and enantioselective substitutions. J. Org. Chem. 61, 4542–4554 (1996).
He, J. et al. Ligand-controlled C (sp3)-H arylation and olefination in synthesis of unnatural chiral α-amino acids. Science 343, 1216–1220 (2014).
Tang, R.-Y., Li, G. & Yu, J.-Q. Conformation-induced remote meta-C–H activation of amines. Nature 507, 215–220 (2014).
Chen, Q., Ilies, L. & Nakamura, E. Cobalt-catalyzed ortho-alkylation of secondary benzamide with alkyl chloride through directed C−H bond activation. J. Am. Chem. Soc. 133, 428–429 (2011).
Yu, D.-G., Gensch, T., Azambuja, F., de, Vásquez-Céspedes, S. & Glorius, F. Co(III)-catalyzed C–H activation/formal SN-Type reactions: Selective and efficient cyanation, halogenation, and allylation. J. Am. Chem. Soc. 136, 17722–17725 (2014).
Ruan, Y.-P. et al. Detailed studies on the enantioselective synthesis and HPLC enantioseparation of N-protected 3-hydroxyglutarimides. Chirality 17, 595–599 (2005).
Evidente, A., Sparapano, L., Fierro, O., Bruno, G. & Motta, A. Sapinofuranones A and B, two new 2(3H)-dihydrofuranones produced by Sphaeropsis sapinea, a common pathogen of conifers. J. Nat. Prod. 62, 253–256 (1999).
Kil, J.-S. et al. Okanin, a chalcone found in the genus Bidens, and 3-penten-2-one inhibit inducible nitric oxide synthase expression via heme oxygenase-1 induction in RAW264.7 macrophages activated with lipopolysaccharide. J. Clin. Biochem. Nutr. 50, 53–58 (2012).
Li, H. et al. 3-(Diethoxyphosphoryloxy)-1,2,3-benzotriazin-4(3H)-one (DEPBT): A new coupling reagent with remarkable resistance to racemization. Org. Lett. 1, 91–94 (1999).
Jochims, J. C., Hehl, S. & Herzberger, S. Preparation and Beckman rearrangement of o-(chiorooxalyl)oximes. Synthesis 1128–1133 (1990).
Acknowledgements
We are grateful for financial support from the NSF of China (21332007), the National Basic Research Program (973 Program) of China (Grant No. 2010CB833200), and the Program for Changjiang Scholars and Innovative Research Team in University (PCSIRT) of Ministry of Education, China. We think Professor Ai-Wen Lei (WHU) and Professor Shi-Gang Sun (XMU) for providing their in situ IR facilities, and Mr. Hua-Min Wang (WHU) and Mr. Jian-Yu Ye (XMU) for the help during the IR measurements. We thank Professor Hai-Chao Xu (XMU) for helpful discussion. In memory of the late Professor Wei-Yuan Huang.
Author information
Authors and Affiliations
Contributions
P.-Q.H. conceived, initiated and directed the project, and wrote the manuscript. Y.-H.H. contributed to the conception of the project, carried out the experimental work, and analyzed the data. H.G. contributed, in part, to the experimental work and data analysis. J.-L.Y. contributed to the in situ NMR analysis and analysis of single crystal X-ray data. All authors commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Huang, PQ., Huang, YH., Geng, H. et al. Metal-Free C–H Alkyliminylation and Acylation of Alkenes with Secondary Amides. Sci Rep 6, 28801 (2016). https://doi.org/10.1038/srep28801
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep28801
This article is cited by
-
Direct alkylation of N,N-dialkyl benzamides with methyl sulfides under transition metal-free conditions
Communications Chemistry (2021)
-
Benzylic aroylation of toluenes with unactivated tertiary benzamides promoted by directed ortho-lithiation
Science China Chemistry (2021)
-
A redox-neutral synthesis of ketones by coupling of alkenes and amides
Nature Communications (2019)
-
Metal-free synthesis of quinolines by direct condensation of amides with alkynes: revelation of N-aryl nitrilium intermediates by 2D NMR techniques
Science China Chemistry (2018)
-
Tf2O-TMDS combination for the direct reductive transformation of secondary amides to aldimines, aldehydes, and/or amines
Science China Chemistry (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.