Abstract
Intergroup competition has an important impact on the survival and fitness of individuals in group-living species. However, factors influencing the probability of winning an encounter are not fully understood. We studied the influence of numerical advantage and location of the encounter on the chances of winning in eight neighboring groups of Verreaux’s sifakas (Propithecus verreauxi), in Kirindy Forest, western Madagascar. Intergroup encounters were inferred from spatial data collected via GPS loggers over a period of two years. Location, i.e., the proximity to the respective core area, rather than the numerical advantage of a group in a given encounter, influenced the probability of winning. Accordingly, the high value that resident groups attribute to exclusive and intensively used areas increased their motivation in defending these locations against intruders. Moreover, losers used the encounter area less often than winners within a month after the encounter, suggesting that losing also entails long-term costs. Thus, our results suggest that in gregarious animals the particular circumstances of each encounter, such as the location, can outweigh group characteristics and predict the chances of winning an intergroup encounter.
Similar content being viewed by others
Introduction
Intergroup competition is a crucial aspect in the life of group-living animals because it mediates access to important resources such as food and/or mating opportunities. Therefore, it has an important impact on the survival and fitness of individuals. In addition, direct competition during group encounters can involve high levels of aggression, which has potential fitness implications for all individuals in the group1. After a decided encounter, winners will enjoy the benefits of accessing the contested resource, whereas losers will suffer the costs of defeat2,3. Potential costs of losing an intergroup encounter can range from alteration of travelling parameters (distance, speed and sinuosity), increasing energetic demands for losers2, to the permanent loss of access to resources, which can lead to group dissolution in the long-term4,5.
Disputes over access to resources are often decided based on asymmetries between the contestants6. These asymmetries have been described as the “Pay-off asymmetry” and the “Asymmetry in fighting ability”6. The first refers to differences among individuals in their interest in defending resources, while the second is related to differences in the power in defending resources, known as the resource holding power (RHP)6. In group-living animals ranging from insects to primates, encounters tend to be decided based on asymmetries between contestants that can be present on the individual level, with stronger individuals tending to defeat weaker ones, but also on the group level, with an effect of numerical advantage, where large groups defeat smaller ones3,7,8,9,10,11,12,13,14,15,16,17,18,19. Numerical advantage is therefore an important general predictor of the outcome of intergroup encounters.
Since resources are rarely uniformly distributed, the economic value of areas varies with the availability of resources. Accordingly, animals should show variable levels of motivation in defending different areas within their home range10,11,17,20,21. Areas of intensive use can have a high value because, among other factors, residents are familiar with the distribution and availability of resources in this part of the home range21. This phenomenon, called the residence effect, suggests that due to differences in the economic value attributed to the area, residents have higher motivation to defend the area and as a consequence, higher chances of winning encounters than intruders21. In fact, in several species the location of encounters seems to be a better predictor for the outcome of a conflict than the numerical advantage between groups11,17,20,22. However, it is still not well known how these two factors or their interaction determine the outcome of group encounters.
We set out to study the influence of asymmetries in RHP, i.e. differences in group size and location of group encounters on the outcome of intergroup conflicts in Verreaux’s sifakas (Propithecus verreauxi), a group-living primate from Madagascar. Verreaux’s sifakas are a suitable and interesting species to investigate these questions because they live in relatively small groups of about 6 ± 2 individuals, but the largest groups can be up to five times bigger than the smallest ones23,24,25. Additionally, they exhibit an interesting pattern of territorial behavior, characterized by partial home range overlap and core areas for exclusive use23,26. In a field study on eight neighboring groups of Verreaux’s sifakas, we tested the influence of numerical advantage and the location of encounters on the outcome of group encounters. Moreover, we investigated the costs of losing group encounters related to alteration in traveling patterns and access to encounter areas after defeat.
Methods
Study site and species
The study was conducted from March 2012 to May 2014 in Kirindy Forest, western Madagascar (44°39′E, 20°03′S). We inferred intergroup encounters among eight neighboring groups from spatial data obtained from GPS loggers. One individual per group was equipped with a GPS logger (E-OBS Digital Telemetry GmbH, Gruenwald, Germany), recording locations continuously over a period of 3–4 months every 15 min from 4:30 h to 20:30 h (Table 1); the time settings were chosen based on the diurnal pattern of activities of sifakas27. We equipped subjects with GPS collars during brief anesthesia after blow-pipe darting28. Anesthetized individuals recovered within two hours and were returned to their social groups. Since sifakas are cohesive in their movements29, one GPS logger per group was sufficient to infer movements and the occurrence of intergroup encounters. In addition, we could include group size in the analyses because during directly observed encounters between the study groups all members were present. Group size in the study population varied during the course of the study period between three to eight individuals.
Group encounters
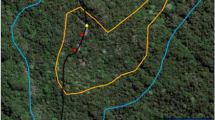
The distance between groups used to infer intergroup encounters from the spatial data was based on the direct observation of 71 encounters in the field. We first calculated the mean duration of the observed encounters (23 ± 22 min). Next, we tested different possible encounter distances ranging from 15 to 150 m. For each proposed distance, we calculated the duration of encounters that would have occurred by counting the number of GPS relocations that were separated by the proposed encounter distance or less. Finally, we selected the encounter distance that resulted from the smallest difference (in absolute terms) between the calculated and the observed average encounter duration, which was 42 m (Supplementary Fig. S1). We therefore defined the beginning of an encounter when two groups were 42 m or less apart from each other and the end when they were again at a distance of more than 42 m from each other for more than one hour. We set one hour as a conservative time limit based on the average duration of observed encounters. We defined the winner of the encounter as the group that stayed in the encounter area, i.e., the group that had the higher number of relocations (GPS points) within the encounter area within the hour following the end of the encounter. The loser of the encounter was defined as the group that left the encounter area. To analyze the data we randomly assigned the groups as being either a focal group or an opponent group.
For each encounter, we mapped an encounter area, which was defined as a buffer around all relocations during an encounter for both participating groups. The buffer width was set as the encounter distance 42 m + 1 m. Additionally we estimated kernel density home ranges (reference bandwidth30) with relocations that were recorded one month prior to the encounter, using R (CRAN) and package rhr (version 1.2.905)31. We then calculated the overlap between core areas (50% isopleth) and the encounter area.
Location
We estimated total home range size (95% isopleth), core areas (50% isopleth) with kernel density home ranges (reference bandwidth30) based on relocations that were recorded one month prior to the encounter using the package rhr in R (version 3.1.2)32. Kernel density estimation is a non-parametrical statistical method for obtaining probability density surfaces from telemetry data and it is commonly used to investigate ranging patterns of wild animals33,34. To examine whether the location of an encounter had an effect on its outcome we examined two aspects: first, we calculated the relative proportion of overlap between the encounter area and the core area of each group (proportion of overlap of focal group minus proportion of overlap of opponent group) during the month before the encounter, which we called proportion of overlap. Therefore, this measurement referred to the relative proportion of GPS relocations of the encounter area that were within the core area of each group. Accordingly, a higher proportion of overlap of one group reflects a higher overlap between the encounter area and the core area compared to their opponent. Since sifakas changed the location of their core areas (intensively and exclusively used areas) within their home ranges, each 2–4 weeks (Supplementary Fig. S2), we used the relocations from a month before the encounter to calculate the core area. Second, we calculated the distance between the encounter and the center of the home range of each group. For that we calculated the distance between the first encounter point and the centroid of the home range of each group based on the entire dataset, since the location of the center of the home range was stable over the study period (Supplementary Fig. S3).
Costs of defeat
To infer short-term costs of defeat we compared travelling parameters (distance, speed, sinuosity) between winners and losers from each group within 1, 2 and 3 h after an encounter. The distance travelled was inferred through the sum of consecutive step lengths within 1, 2 or 3 h after the encounter; speed was calculated by dividing distance by time; and sinuosity by dividing distance by the straight line distance between the first and last relocation within the time frame. To assess long-term costs of losing an encounter, we compared the intensity of use (relative density of relocations) of the encounter area between winners and losers from each group for one month following an encounter, which we called the post-encounter effect. The relative density of relocations was based on the fraction of relocations in the encounter area within the month after the conflict.
Statistical analyses
We used a binomial test to investigate whether one group of a given dyad won encounters more often than expected by chance. Binomial Generalized Linear Mixed Models (GLMM) from the package lmer435 R version 3.1.2 were used to investigate the predictors for outcome of group encounters. We tested two different measurements for the location of an encounter: the proportion of overlap and the distance from the encounter area to the center of the home range of each group. In the first model, we included relative group size (group size of the focal group minus group size of the opponent group) to estimate the numerical advantage and the proportion of overlap between the encounter area and the core area of each group involved in the encounter (arcsine square root transformed). In the second model, we included the relative group size and the distance from the encounter area to the center of the home range of each group (Table 2). In both models dyadic identity of the groups involved in the encounter was included as random factor. We controlled for interaction effects between predictor variables in both models but did not report them because they were not significant. We used maximum likelihood ratio tests to verify whether fixed factors explained a significant amount of the variance and to test the final model with fixed factors against the null model including only the random factors36. Wilcoxon signed-rank tests were used to investigate potential costs of losing an encounter (Table 2). Comparisons of potential costs of losing an encounter after 1, 2 and 3 h after the encounters were Bonferroni corrected, resulting in a p-value of 0.02 for these comparisons. All statistical analyses were performed in R version 3.1.232.
Ethical approval and informed consent
This study is in accordance with the German and Malagasy (Commission Tripartite CAFF) legal and ethical requirements of appropriate animal procedures. Consultation of the Animal Welfare Body of the German Primate Center is documented (No. 4–15). Research protocols and capture procedures were approved by the Ministry for the Environment, Water and Forests of Madagascar (MINEEF).
Results
We recorded 759 encounters among eight neighboring groups, of which 624 were decided according to our definition of winning and losing. Sifakas did not exhibit clear and stable intergroup dominance relationships, because the frequency of winning encounters differed significantly only between 2 out of 13 dyads (Table 3). The probability of winning a group contest was not influenced by the relative group size of the opponents, indicating that the numerical advantage of larger over smaller groups did not influence the chances of winning an encounter (Table 4). Instead, the location of encounters, i.e. proportion of overlap, was crucial, since only the proportion of overlap of intensively used areas predicted the probability of winning an encounter (Table 4, Fig. 1). Moreover, the distance between the encounter area and the center of the home ranges of the groups involved in the encounter did not influence the probability of winning the intergroup encounter (Table 5).
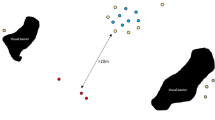
The loser of an encounter travelled longer distances (Wilcoxon test: V = 36, p = 0.008), straighter (Wilcoxon test: V = 1, p = 0.01) and with higher speed (Wilcoxon test: V = 36, p = 0.008) than the winner (Fig. 2), but only within the first hour after the encounter (Fig. 2). Losers used the encounter area less often than winners within a month after the encounter, suggesting that losing also entails long-term costs (Wilcoxon test: V = 0, p = 0.007, Fig. 3).
Discussion
Our results revealed that sifakas do not exhibit clear intergroup dominance relationships because in the majority of dyads no group consistently dominated an opponent group. The given numerical advantage did not influence the outcome of intergroup encounters. Instead, the location, the proportion of overlap, but not the distance to the respective home range center, was the main predictor for the probability of winning an encounter. Thus, the current resource value of an area mainly influenced sifakas’ motivation to fight, irrespective of the numerical disadvantage. Sifakas also did not pay heavily in terms of additional travelling costs after losing an encounter, but losers used the encounter area less intensively than winners within a month after the encounter. This post-encounter effect suggests that sifakas face long-term costs of losing a group encounter.
When the potential benefits of group encounters are not equally shared among all group members, some individuals may free-ride37. In sifakas, resources are not shared equally within groups. Dominant individuals of both sexes enjoy increased access to resources and the presence of free-riders among subordinates is common23,38,39,40,41. Hence, the lack of a numerical advantage effect observed in this study might be due to collective action problems41. Therefore, a better predictor of the RHP of groups and their chances of winning is probably the number of actual participants in each encounter, rather than differences in total group size, consistent with what Crofoot and collaborators proposed11.
In several other species, the location of the encounter also influenced the outcome of encounters in favor of residents3,11,17,20. Indeed, residents are more familiar with the distribution and availability of resources in the disputed area, which creates an asymmetry in RHP between contestants in favor of the residents21. Additionally, due to the high attributed value to the disputed area, residents face higher costs from losing an encounter than intruders. They are therefore expected to be more motivated to defend the area and, hence, to free-ride less often than intruders6. In contrast, the high potential for free-riding in intruder groups may decrease their RHP22,41,42,43,44,45,46. In sifakas, the residence effect was the main predictor of winning an encounter, suggesting that resident groups were able to overcome other asymmetries in RHP, such as a numerical disadvantage.
Potential costs of losing an intergroup encounter have so far been rarely studied. In white-faced capuchins (Cebus capucinus), losers had higher travel costs than winners and the underlying change in travel patterns was still present on the next day2. Sifakas also showed alteration in travelling parameters after losing an encounter; however, the effect was present only within the first hour after the encounter. In that first hour, losers travelled on average 15 m more than winners, a distance that is still within the general average hourly activity range (mean 71 ± 43 m). Thus, the variation in travelling parameters observed probably does not represent a major absolute cost and is most likely explained by the retreat of the losers to their core area.
Another potential cost of losing an encounter is the inability of using the resources within the encounter area after an encounter. For instance, in yellow baboons (Papio cynocephalus), losers used the encounter area less often than winners3. Sifakas also showed a post-encounter effect, with losers using the encounter area less often than winners in the month following an encounter. In contrast to other species that are highly motivated to defend space irrespective of the actual level of use, such as white-faced capuchins11, sifakas are highly motivated to defend intensively used areas, likely due the availability of specific food resources.
In conclusion, our results support the assumption that the particular circumstances of group encounters, such as the location of the conflict, are crucial predictors for the outcome of encounters3,11,17,20,22,41. Since the particular conditions of each encounter ought to impede the establishment of stable dominance relationships between groups, our data support the notion that the dilemma between cooperation and competition in gregarious animals occurs at the individual level3,11,41,46. Hence, future studies should investigate individual characteristics and detailed environmental conditions to achieve a better understanding of the dynamics in intergroup competition. Finally, the direct costs of losing conflicts are rarely taken into account in studies of intergroup encounters and further research may also help to elucidate the potential fitness consequences of defeat.
Additional Information
How to cite this article: Koch, F. et al. The role of the residence-effect on the outcome of intergroup encounters in Verreaux’s sifakas. Sci. Rep. 6, 28457; doi: 10.1038/srep28457 (2016).
References
Cheney, D. L. & Seyfarth, R. M. The influence of intergroup competition on the survival and reproduction of female vervet monkeys. Behav. Ecol. Sociobiol. 21, 375–386 (1987).
Crofoot, M. C. The cost of defeat: capuchin groups travel further, faster and later after losing conflicts with neighbors. Am. J. Phys. Anthropol. 152, 79–85 (2013).
Markham, A., Alberts, S. & Altmann, J. Intergroup conflict: ecological predictors of winning and consequences of defeat in a wild primate population. Anim. Behav. 82, 399–403 (2012).
Goodall, J. The Chimpanzees of Gombe: Patterns of Behavior. (Belknap Press of Harvard University, 1986).
Robinson, J. G. Group size in wedge-capped capuchin monkeys Cebus olivaceus and the reproductive success of males and females. Behav. Ecol. Sociobiol. 23, 187–197 (1988).
Maynard Smith, J. & Parker, G. A. The logic of asymmetric contests. Anim. Behav. 24, 159–175 (1976).
Cheney, D. L. In Primate Societies (eds Smuts, B. B., Cheney, D. L., Seyfarth, R. M., Wrangham, R. W. & Struhsaker, T. T. ) Ch. 22, 267–281 (University of Chicago Press, 1987).
Kitchen, D. & Beehner, J. Factors affecting individual participation in group-level aggression among non-human primates. Behaviour 144, 1551–1581 (2007).
Cant, M. A., Otali, E. & Mwanguhya, F. Fighting and mating between groups in a cooperatively breeding mammal, the banded mongoose. Ethology 108, 541–555 (2002).
Cassidy, K. A., MacNulty, D. R., Stahler, D. R., Smith, D. W. & Mech, D. L. Group composition effects on aggressive interpack interactions of gray wolves in Yellowstone National Park. Behav. Ecol. 26, 1352–1360 (2015).
Crofoot, M. C., Gilby, I. C., Wikelski, M. C. & Kays, R. W. Interaction location outweighs the competitive advantage of numerical superiority in Cebus capucinus intergroup contests. Proc. Natl. Acad. Sci. USA 105, 577–81 (2008).
Wilson, M. L., Hauser, M. D. & Wrangham, R. W. Does participation in intergroup conflict depend on numerical assessment, range location, or rank for wild chimpanzees? Anim. Behav. 61, 1203–1216 (2001).
Benson-Amram, S., Heinen, V. K., Dryer, S. L. & Holekamp, K. E. Numerical assessment and individual call discrimination by wild spotted hyaenas. Crocuta crocuta. Anim. Behav. 82, 743–752 (2011).
McComb, K., Packer, C. & Pusey, A. Roaring and numerical assessment in contests between groups of female lions. Panthera leo. Anim. Behav. 47, 379–387 (1994).
Harris, T. R. Multiple resource values and fighting ability measures influence intergroup conflict in guerezas (Colobus guereza). Anim. Behav. 79, 89–98 (2010).
Scarry, C. J. Between-group contest competition among tufted capuchin monkeys, Sapajus nigritus and the role of male resource defence. Anim. Behav. 85, 931–939 (2013).
Roth, A. M. & Cords, M. Effects of group size and contest location on the outcome and intensity of intergroup contests in wild blue monkeys. Anim. Behav. 113, 49–58 (2016).
Black, J. M. & Owen, M. Agonistic behaviour in barnacle goose flocks: assessment, investment and reproductive success. Anim. Behav. 37, 199–209 (1989).
Tanner, C. J. Numerical assessment affects aggression and competitive ability: a team-fighting strategy for the ant Formica xerophila. Proc. R. Soc. Lond. B. 273, 2737–2742 (2006).
Furrer, R. D., Kyabulima, S., Willems, E. P., Cant, M. A. & Manser, M. B. Location and group size influence decisions in simulated intergroup encounters in banded mongooses. Behav. Ecol. 22, 493–500 (2011).
Haley, M. P. Resource-holding power asymmetries, the prior residence effect and reproductive payoffs in male northern elephant seal fights. Behav. Ecol. Sociobiol. 34, 427–434 (1994).
Pride, R. E., Felantsoa, D. & Randriamboavonjy, R. In Ringtailed Lemur Biology (eds Jolly, A., Sussman, R. W., Koyama, N. & Rasamimanana, H. ) Ch. 13, 208–232 (Springer US Press, 2006).
Jolly, A. Lemur Behavior. (Chicago Press, 1966).
Richard, A. F., Dewar, R. E., Schwartz, M. & Ratsirarson, J. Mass change, environmental variability and female fertility in wild Propithecus verreauxi. J. Hum. Evol. 39, 381–91 (2000).
Kappeler, P. M. & Fichtel, C. In Long-Term Field Studies of Primates (eds Kappeler, P. M. & Watts, D. P. ) Ch. 5, 21–45 (Springer, 2012).
Benadi, G., Fichtel, C. & Kappeler, P. M. Intergroup relations and home range use in Verreaux’s sifaka (Propithecus verreauxi). Am. J. Primatol. 70, 956–65 (2008).
Erkert, H. G. & Kappeler, P. M. Arrived in the light: diel and seasonal activity patterns in wild Verreaux’s sifakas (Propithecus v. verreauxi; Primates: Indriidae). Behav. Ecol. Sociobiol. 57, 174–186 (2004).
Springer, A., Razafimanantsoa, L., Fichtel, C. & Kappeler, P. M. Comparison of three short-term immobilization regimes in wild Verreaux’s sifakas (Propithecus verreauxi): ketamine-xylazine, ketamine-xylazine-atropine and tiletamine-zolazepam. J. Zoo Wildl. Med. 46, 482–490 (2015).
Trillmich, J., Fichtel, C. & Kappeler, P. M. Coordination of group movements in wild Verreaux’s sifakas (Propithecus verreauxi). Behaviour 141, 1103–1120 (2004).
Worton, B. J. Kernel methods for estimating the utilization distribution in home-range studies. Ecology 70, 164–168 (1989).
Signer, J. & Balkenhol, N. Reproducible home ranges (rhr): a new user-friendly R package for analyses of wildlife telemetry data. Wildl. Rep. Bull. 39, 358–363 (2015).
R Core Team R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL http://www.R-project.org/ (2013).
Laver, P. N. & Kelly, M. J. A critical review of home range studies. J. Wildl. Manage. 72, 290–298 (2008).
Signer, J., Balkenhol, N., Ditmer, M. & Fieberg, J. Does estimator choice influence our ability to detect changes in home-range size? Anim. Biotelemetry 3, 16 (2015).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. arXiv:1406.5823 (2014). (Date of access:01/06/2015).
Faraway, J. J. Extending the Linear Model with R: Generalized Linear Mixed Effects and Nonparametric Regression Models, 1. (Chapman & Hall Press, 2006).
Nunn, C. L. & Deaner, R. O. Patterns of participation and free riding in territorial conflicts among ringtailed lemurs (Lemur catta). Behav. Ecol. Sociobiol. 57, 50–61 (2004).
Kappeler, P. M. & Schäffler, L. The lemur syndrome unresolved: extreme male reproductive skew in sifakas (Propithecus verreauxi), a sexually monomorphic primate with female dominance. Int. J. Primatol. 62, 1007–1015 (2008).
Kappeler, P. M., Mass, V. & Port, M. Even adult sex ratios in lemurs: potential Costs and Benefits of Subordinate Males in Verreaux’s sifaka (Propithecus verreauxi) in the Kirindy Forest. Am. J. Phys. Anthropol. 497, 487–497 (2009).
Port, M., Johnstone, R. A. & Kappeler, P. M. The evolution of multimale groups in Verreaux’s sifaka, or how to test an evolutionary demographic model. Behav. Ecol. 23, 889–897 (2012).
Koch, F., Signer, J., Kappeler, P. M. & Fichtel, C. Intergroup encounters in Verreaux’s sifakas (Propithecus verreauxi): who fights and why? Behav. Ecol. Sociobiol. 70, 797–808 (2016).
Watts, D. P. & Mitani, J. C. Boundary patrols and integroup encounters in wild chimpanzees. Behaviour 138, 299–327 (2001).
Nunn, C. L. In Primate Males (ed. Kappeler, P. M. ) Ch. 17, 192–204 (Cambridge University Press, 2000).
Bonanni, R., Valsecchi, P. & Natoli, E. Pattern of individual participation and cheating in conflicts between groups of free-ranging dogs. Anim. Behav. 79, 957–968 (2010).
Willems, E. P., Hellriegel, B. & van Schaik, C. P. The collective action problem in primate territory economics. Proc. R. Soc. Lond. B. 280, 20130081 (2013).
Crofoot, M. C. & Gilby, I. C. Cheating monkeys undermine group strength in enemy territory. Proc. Natl. Acad. Sci. USA 109, 501–5 (2012).
Acknowledgements
We thank the Commission Tripartite de Direction des Eaux et Forêts and the CNFEREF Morondava for their authorization and support for this study. We are grateful to the team of field assistants in Kirindy Forest for their support.
Author information
Authors and Affiliations
Contributions
F.K. and C.F. designed the study. F.K. and J.S. performed the analyses. F.K., C.F. and P.M.K. wrote the paper. All authors gave final approval for publication.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Koch, F., Signer, J., Kappeler, P. et al. The role of the residence-effect on the outcome of intergroup encounters in Verreaux’s sifakas. Sci Rep 6, 28457 (2016). https://doi.org/10.1038/srep28457
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep28457
This article is cited by
-
Factors Affecting the Outcome and Intensity of Intergroup Encounters in Crested Macaques (Macaca nigra)
International Journal of Primatology (2023)
-
Exploring Strategic Functions of Sleeping Sites in Crested Macaques (Macaca nigra): Evidence from Intergroup Encounters
International Journal of Primatology (2023)
-
Non-aggressive inter-group interactions in wild Northern Gray gibbons (Hylobates funereus)
acta ethologica (2023)
-
Drivers of gut microbiome variation within and between groups of a wild Malagasy primate
Microbiome (2022)
-
Interrelationship among spatial cohesion, aggression rate, counter-aggression and female dominance in three lemur species
Behavioral Ecology and Sociobiology (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.