Abstract
Confiscated and displaced mammals are often taken to sanctuaries, where the explicit goal may be reintroduction to the wild. By inadvertently collecting animals from different source populations, however, such efforts risk reintroducing individuals that have not been in genetic contact for significant periods of time. Using genetic analyses and 44 years of data from Camp Leakey, an orang-utan rehabilitation site on Borneo, we determined the minimum extent to which orang-utans representing non-native, geographically and reproductively isolated taxa were reintroduced into the surrounding wild population. We found two reintroduced females were from a non-native subspecies and have since produced at least 22 hybridized and introgressed descendants to date, of which at least 15 are living. Given that Bornean orang-utan subspecies are thought to have diverged from a common ancestor around 176,000 years ago, with marked differentiation over the last 80,000 years, we highlight the need for further evaluation of the effects of hybridizing orang-utans of different taxa — particularly in light of the ~1500 displaced orang-utans awaiting urgent reintroduction. As endangered mammals are increasing in number in sanctuaries worldwide, we stress the need for re-examination of historical reintroductions, to assess the extent and effects of de facto translocations in the past.
Similar content being viewed by others
Introduction
Populations of large and endangered mammals are increasingly impacted by humans and may live – even in wild or semi-wild settings – under intensive human management (e.g. black-footed ferrets, Mustela nigripes1; golden lion tamarins, Leontopithecus rosalia2; northern white rhinos, Ceratotherium simum cottoni3; giant Galápagos tortoises, Chelonoidis hoodensis4). Though law enforcement and community engagement have traditionally proven effective for conventional custodial management, more invasive measures are now increasingly needed to ensure the long-term viability of threatened populations5,6. For example, in Rwanda’s Virunga Massif, close monitoring of individual animals – facilitating the detection and treatment of medical conditions – has led to an annual 4.1%±0.09% increase in the number of mountain gorillas (Gorilla beringei beringei) in groups habituated to human observation, versus an annual 0.7%±0.06% decline in the number of unhabituated conspecifics7. Proactive captive-breeding of giant pandas, meanwhile, has resulted in the birth of numerous offspring through artificial insemination, producing offspring both for exhibition in zoos and for introduction to the wild – effectively reversing the decline of a conservation flagship species8. Though direct intervention may be necessary for the effective conservation of wildlife, unintended effects may ultimately harm populations. For instance, the death of all African wild dogs (Lycaon pictus) under study in the Serengeti Ecosystem has been attributed to a rabies vaccination programme, which may have inadvertently compromised dogs’ immune responses through their immobilization, vaccination and radio collaring9, though the ill effects of this programme continue to be debated10.
Another form of human intervention is placement of endangered animals in sanctuaries. As a result of declining suitable habitat and illegal capture, endangered large mammals are often displaced entirely from wild populations and may be relocated to sanctuaries for short- or long-term care11,12. By inadvertently collecting animals from various and potentially unknown points of origin, however, these centres may unintentionally amalgamate individuals from populations that have not been in genetic contact for substantial periods of time. If sanctuaries are in a position to offer lifetime care, with little or no inter-breeding, such mixed genetic origins may not prove to be of importance. Often, however, reintroduction into natural populations is a primary objective of sanctuaries, assuming that concerns regarding disease and suitable habitat are adequately met12,13,14. Given that this can lead to de facto translocation, it is important to investigate possible outcomes by looking at deliberate or inadvertent translocations of mammals in the past.
The hybridization of genetically distant individuals may, under some circumstances, increase population fitness15,16,17. For example, the introduction of 8 female panthers from Texas (Puma concolor stanleyana) to a remnant population of 50 Florida panthers (P. c. coryi) approximately doubled the estimated genetic diversity of the population and was associated with a marked reduction in the frequency of deleterious traits associated with the breeding of related individuals, such as kinked tails, cryptorchidism and poor sperm quality18,19,20. Measures of fitness and survival improved and the population tripled in size over 15 years21. In grey wolves (Canis lupus), the arrival of a single wolf to a bottlenecked Scandinavian population originating from only two individuals was sufficient to dramatically increase genetic diversity (heterozygosity) and exponentially increase population growth22. Indeed, the introduction of novel genetic variants into small or genetically depauperate wild populations has been shown, over multiple generations, to augment genetic diversity, reverse indications of inbreeding depression and increase population size in a range of other animal taxa23,24,25.
Despite this, the hybridization and introgression of representatives of genetically divergent lineages may have negative effects on a population’s overall fitness, as exemplified by the lack of fertility common in offspring resulting from the inter-breeding of different species26,27,28. Reduced fitness, termed outbreeding depression, may also be observed following the hybridization of subspecies, or of other populations that may have independently diverged26, particularly if they occupy different habitat types or if no genetic exchange has occurred within the last 500 years15. Outbreeding depression can occur via the disruption of gene complexes adapted to local conditions, potentially even giving rise to harmful combinations and/or mutations28,29. Though there is less empirical evidence for outbreeding depression as compared to inbreeding depression, its consequences are thought to be of a similar magnitude in and after the second generation of offspring29. This delay can be attributed to recombination: while first-generation hybrids inherit intact sets of chromosomes from each parental lineage, these chromosomes recombine in subsequent generations, disrupting positive epistatic interactions among parental alleles28. Developmental, genetic and other abnormalities have been documented in a wide range of outbred animal taxa, including fish30,31, invertebrates32,33, birds34 and mammals35,36 and are further reviewed by Edmands29. In some cases, particularly when a population is small, such hybridization and introgression can lead to ‘hybrid swarms’, in which introgressed individuals ‘swamp’ the original population, thus leading to the latter’s extinction26. Given that our understanding of outbreeding depression has advanced substantially in recent decades, there is cause for concern that historical reintroductions may have proven deleterious to the fitness of wild populations.
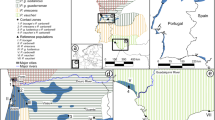
More than 2,650 great apes are confined in sanctuaries across the ranges of all four species: c. 963 chimpanzees (Pan troglodytes), c. 72 bonobos (P. paniscus), c. 106 gorillas (Gorilla gorilla) (Pan African Sanctuary Alliance, pers. comm., 17 January 2016) and c. 1516 orang-utans (Pongo spp.)14. For many sanctuaries, reintroduction is an ultimate goal. The situation is especially pressing for orang-utans, however, given the Indonesian government’s declaration that all sanctuaries should be swiftly emptied through reintroductions, having missed a deadline for doing so by the end of 201537. On the basis of genetic and morphological studies, orang-utans are now known to comprise two species on the islands of Borneo (Pongo pygmaeus) and Sumatra (Pongo abelii), with three geographically and reproductively isolated subspecies on Borneo: P. p. wurmbii in Central and southern West Kalimantan, P. p. morio in North and East Kalimantan and Sabah and P. p. pygmaeus in northern West Kalimantan and Sarawak. Each Bornean subspecies is thought to have shared a common ancestor around 176,000 years ago, before radiating to different refugia following climatic fluctuations in the Pleistocene38. Particularly marked population differentiation has occurred over the last 80,000 years39, with orang-utans currently separated by largely insurmountable river and mountain barriers38,40,41 (Fig. 1). Though not always reciprocally monophyletic38,42, potentially as the result of human intervention through translocations42, subspecies can typically be observed on different clades in a phylogenetic tree of the maternally inherited mitochondrial DNA molecule43.
Distribution of orang-utan subspecies on Borneo, in relation to Tanjung Puting National Park.
Three subspecies diverged from a common ancestor during climatic fluctuations around 176,000 years ago38, with marked population differentiation evolving over the last 80,000 years39. Pongo pygmaeus pygmaeus is restricted to northern West Kalimantan and Sarawak and is primarily isolated by the Kapuas River from P. p. wurmbii, in southern West and Central Kalimantan. P. p. morio is found in East and North Kalimantan and in Sabah and is isolated by rivers, mountains and geographic distance from the other two subspecies. Figure (including base map) drawn in Adobe Illustrator CC 2015 (http://www.adobe.com/illustrator); orang-utan distribution follows that previously described77.
In 1971, however – when Galdikas and Brindamour established their pioneering research and reintroduction site, Camp Leakey, in Tanjung Puting National Park – all orang-utans were classified as a single species, P. pygmaeus. Over 14 years, Galdikas and Brindamour rehabilitated and released more than 90 ex-captive or displaced orang-utans into the wild at Camp Leakey, on the southernmost promontory of Central Kalimantan44,45. Most were confiscated from illegal captivity in the pet trade. Though many were translocated from areas close to the National Park, others came from further afield: though none were thought to have been taken from Sumatra, several were transferred from the provincial capital of Palangka Raya, c. 470 kilometres from Tanjung Puting and others from captivity in Java. In spite of this, Yeager46 asserted that at least one juvenile Sumatran female was reintroduced at Camp Leakey prior to 198146, though Galdikas counters that she appeared Bornean in morphology, did not reproduce and was reported dead by 1994. Further, Galdikas claims to have rejected orang-utans that appeared Sumatran in origin44, though she acknowledges that morphology alone is insufficient to diagnose species. Yeager46 also suggested that orang-utans from both eastern and western Borneo were probably reintroduced at Camp Leakey and have potentially inter-bred with the wild population. In spite of this, it is assumed by Galdikas – on the basis of her notes from the 1970s and 1980s regarding individuals’ provenance – that most orang-utans she reintroduced would have been local to Tanjung Puting National Park and the surrounding areas.
Camp Leakey’s contemporary population comprises approximately 60 individuals, including 8 reintroduced female orang-utans. Two further reintroduced females have recently died. All 10 females reproduced; in one case, offspring are now into their fourth generation. As some paternities are known47 and as female reproductive success and maternities are well documented as a consequence of four decades of behavioural observation by Galdikas, we determined the taxonomic composition of the orang-utans at Camp Leakey on the basis of mitochondrial DNA. We hypothesised that orang-utans of multiple species or subspecies were translocated and reintroduced and have since inter-bred with the wild population.
Methods
Faecal samples were collected from orang-utans in and around the Camp Leakey study area and genomic DNA extracted, as previously described47. Variation in mitochondrial DNA sequences is typically concordant with orang-utan geographic origin and so sequence analysis of individuals of unknown provenance can aid in inferring their likely origin and taxonomic affiliation43. We amplified 397 bp of mitochondrial DNA, primarily spanning a primary stretch of variation in the control region, with the primers Pp-5′ (5′-GCACTTAACTTCACCATC-3′) and Pp-3′ (5′-AAACAAGGGACCACTAAC-3′)41. Each 25 μl reaction volume comprised 2.76 μl 10x PCR Buffer (Bioline), 1.03 μl 50 mM MgCl2, 1.38 μl 10 mM dNTP mix, 0.83 μl 10 mg/mL BSA, 0.134 μl each primer, 0.28 μl Taq polymerase (Life Technologies), 15.7 μl ddH2O and 2.75 μl DNA. Thermal cycling conditions were as follows: initial denaturation at 94 °C for 12 minutes; 40 cycles of 94 °C for 40 seconds, 61 °C for 30 seconds and 72 °C for 1 minute; and a final extension at 72 °C for 10 minutes. To avoid amplification of nuclear mitochondrial insertions (‘numts’), which have proved problematic in prior studies of hominoid mtDNA taxa48, we also amplified ~1500 bp of mitochondrial DNA in a single PCR – spanning the complete mitochondrial DNA control region – to ensure the resulting longer sequences matched the shorter segments amplified (Banes & Galdikas, in prep.). In all cases, the sequences were concordant and no numts were detected.
Sequences were manually corrected in MEGA 6.0649 and aligned using MUSCLE50 with published sequences from 20 Bornean and 4 Sumatran orang-utans of known geographic origin (Warren et al., 200143, some updated by Arora et al., 201038). Accession codes for these published sequences are detailed in Supplementary Table S1. Though reportedly generated from amplicons of 278 bp, some published sequences were found to be as short as 164 bp in length, while updated versions were up to 452 bp in length. Consequently, in order to reliably infer a phylogenetic tree, we cut our alignment to 235 bp to encompass the majority of the published sequence data. Phylogenetic trees were inferred using a Bayesian algorithm in MrBAYES 3.2.451 with the HKY + I model of nucleotide substitution, as selected under the Bayesian Information Criterion (BIC) in jModelTest 2.1.652. We applied four independent runs at the default temperature of 0.2, with 10 million generations from a randomly generated tree, sampling every 100 generations. The first 25% of trees generated before convergence were discarded. Sequences that grouped into clades with those of orang-utans of known geographic origin were deemed to have descended ancestrally from those regions and thus be of the same taxon. This information was combined with parentage data from our prior study47 to assess the extent to which reintroduced orang-utans had inter-bred with the wild population.
Results
Mitochondrial DNA control region sequences were generated from 10 presumed unrelated adult orang-utans (1 male, 9 females) that were reintroduced to the wild at Camp Leakey in the 1970s and 1980s. An additional sequence was obtained from the first-generation offspring (Siswi) of a now-deceased reintroduced female (Siswoyo) and assumed to represent this female given that mtDNA is near-clonally maternally inherited. Of the 11 founders represented, five haplotypes – labelled A to E – were observed in the alignment. Of these, two (A and B) were identical to previously published sequences (GenBank accession codes AJ391121.1 and AJ391108.2, respectively), while three (C, D and E) were novel (GenBank accession codes KU523975-KU523977). Haplotype C featured an insertion that was confirmed by sequencing an additional PCR product amplified from the same DNA extract. Haplotypes D and E each featured a transition, at different bases, when compared with published sequences. These transitions were consistent in all sequences generated from both PCR protocols.
Phylogenetic analysis placed all five observed haplotypes into clades with Bornean orang-utans of known geographic origin (Fig. 2; Supplementary Table S2). If originally derived from populations in the broad region encompassing Camp Leakey, the five observed haplotypes would be expected to group on the tree with the clade of Bornean orang-utans of P. p. wurmbii. We found that nine orang-utan mtDNAs, comprising four haplotypes (B: N = 5, C: N = 1, D: N = 1 and E: N = 2) did indeed fall in this clade. In addition, two females, Rani and Siswoyo, were found to share a haplotype (A) that clustered in the tree with mtDNAs derived from the subspecies P. p. pygmaeus from Northern West Kalimantan or Sarawak (Fig. 1).
Evolutionary relationships for a 235 bp alignment of 27 unique orang-utan DNA sequences, comprising a primary stretch of variation within the mitochondrial DNA control region that is diagnostic of subspecies.
Five haplotypes are represented from Camp Leakey, along with published haplotype sequences from 20 Bornean and 4 Sumatran orang-utans of known geographic origin38,43.
Both Rani and Siswoyo have bred with native males at Camp Leakey, giving birth to hybridized offspring that – on the basis of paternity and long-term population monitoring – are known to have since introgressed (Fig. 3). Rani (b. ~1969) was reintroduced prior to sexual maturation, having been confiscated in Java. Rani’s descendants include seven first-generation offspring, one of which died, five known second-generation offspring and two known third-generation offspring, one of which died. With the exception of the two deceased offspring, who both died in infancy, all of Rani’s known descendants are presumed to be alive and none are known to have required veterinary interventions. Siswoyo (b. ~1962, d. 1991) was an adolescent female at the time of her reintroduction, having also been confiscated on Java. Her descendants are comparatively few, with only five first-generation and three second-generation offspring. Two of Siswoyo’s own offspring died in infancy; infection following the latter pregnancy resulted in Siswoyo’s own death ten days post parturition. Her only daughter, Siswi, produced a stillborn offspring, a daughter that died in infancy and a son that required frequent medical interventions, having been blinded in his right eye after an accident caused by humans in 1993. He was last seen at the age of 16 before disappearing into the forest (Galdikas, unpublished data) and potentially has died. Though in poor condition, Sampson was able to achieve at least one paternity as an unflanged male prior to his departure from the study area47. Siswi was given life-saving surgery in 1997 to repair a perforated intestine and has since been medically unable to conceive (Galdikas, unpublished data).
Inter-breeding between Rani (a) and Siswoyo (b) with native males is known to have resulted in hybridized offspring.
To illustrate subsequent introgression, this diagram is based on the assumption that 50% of alleles native to each subspecies are always uniformly inherited: thus, by example, ‘Rimba’ is three-quarters Pongo pygmaeus wurmbii and one quarter P. p. pygmaeus. The figure illustrates examples of the extent of admixture known to have occurred within and between these lineages over multiple generations. In practice, as detailed in the text, Rani’s direct descendants number(ed) at least 14 hybridized and/or introgressed individuals over 3 generations; Siswoyo’s number(ed) at least 8 over 2 generations. At least 9 of their combined descendants are males likely capable of fathering offspring. Their reproductive output is unknown, but has inevitably resulted in further admixture in other lineages.
Discussion
Our findings provide genetic evidence that individuals of a non-native orang-utan subspecies were translocated and introduced into a wild orang-utan population and have since hybridized and introgressed over multiple generations. The 22 known descendants of Rani and Siswoyo – of which at least 15 are still alive – can be assumed to carry a ‘cocktail’ of genes that would not normally occur in the wild, comprising maternally inherited mitochondrial DNA specific to P. p. pygmaeus, Y-chromosomes inherited from P. p. wurmbii fathers and a mixture of autosomal genes deriving from each subspecies. Such offspring are unlikely to be localised to the Camp Leakey study area: though females are thought to be philopatric, males disperse over vast home ranges that may be larger than 2500 ha53. Of Rani’s descendants, at least six are males that could be sexually mature; of Siswoyo’s, at least three might be able to father offspring. It is therefore highly probable that inter-breeding and the production of introgressed offspring is occurring elsewhere in Tanjung Puting National Park, potentially far from Camp Leakey itself, as a consequence of Rani and Siswoyo’s introduction.
Given the size and scale of the reintroduction programme, however, Rani and Siswoyo may simply prove the tip of the iceberg. Galdikas estimates that, of the 90 orang-utans she reintroduced to Camp Leakey, at least 80 were probably alive by the time the programme there ceased, although a number had disappeared into the forest45. These include two males and one female that were confiscated from the same owner as Siswoyo, though acquired by him at different times and thus potentially from different sources. These orang-utans might plausibly be P. p. pygmaeus or another subspecies entirely. At least 15 reintroduced females at Camp Leakey are known, cumulatively, to have produced at least 78 descendants over up to four generations – of which at least 61 were presumed or known to be living by the end of 2011. With the exception of one adolescent male, all of the deceased offspring were infants, of which at least one was stillborn. It is therefore unclear to what extent the natural population has experienced the introduction of other non-native alleles, which might even incorporate those of Sumatran orang-utans46. Yeager’s earlier testimony of Sumatran orang-utans in Tanjung Puting is compounded by the fact that animals are known to have been translocated between the two islands through the pet trade: by genetic testing, two privately owned Bornean individuals were identified in Sumatra in 2006 and 2009 and were consequently repatriated in 2011 for reintroduction into Borneo’s Lamandau Wildlife Reserve (A E Leiman, pers. comm., 1 May 2015). The issue is further confounded by conflicting reports on the extent to which orang-utans were reintroduced into Tanjung Puting. The Indonesian government is known to have released ex-captive orang-utans at two other sites in the National Park throughout the 1980s and 1990s. Galdikas herself reports that, though she ceased reintroduction at Camp Leakey in 1985, she released more than 200 orang-utans into the wider National Park up until 1995, when she ceased all such reintroductions in accordance with changing Indonesian law. Despite this, Yeager46 reported that more than 180 orang-utans were released into the wider National Park between 1977 and 199746. Similar claims were made by Spalding, who concurred that many releases took place after 1995, both at Camp Leakey and elsewhere in the National Park54,55. In the case of all these reintroductions — including those by Galdikas — few records are published or publicly available to specify the exact number of animals released, or when or where those releases occurred. Few attempts have been made to document their outcomes45,56.
The effects of inter-breeding different orang-utan taxa remain unclear, particularly in reference to their health and reproductive viability. By 1985, such concerns had led zoos to manage their orang-utans as two separate, species-distinct populations57, though the potential for outbreeding among Bornean subspecies has yet to receive significant attention. It is noteworthy that Siswoyo and her descendants have experienced poor reproductive success, poor reproductive health, high infant mortality and overall ill health – incidents that might characterise outbreeding depression. By comparison, however, Rani and her descendants have enjoyed one of the greatest reproductive successes of any matriline at the site. Our data are too few, therefore, to determine the extent to which these factors might be linked to their introgression – and, given the slow reproductive rate of orang-utans, which typically reproduce only once every eight years – too few generations of offspring are available to study and potentially correlate these effects. Nonetheless, there is a general consensus in the literature that orang-utan subspecies should not be hybridized in the wild. Arguments against hybridization have ranged from morals and ethics58,59,60 to fears of reduced fertility61 and concerns that differing characteristics must be independently preserved62,63. The genetic effects of outbreeding are of particular concern, however, in light of the substantial differences that have evolved in each subspecies, in morphology, habitat, diet and behavioural ecology64 in their tens of thousands of years of independent evolution39, all of which may contribute to outbreeding depression15.
In spite of this, there is evidence in other primate taxa that hybridization occurs naturally along the borders of different species’ and subspecies’ ranges. Gonder et al. (2011) observed a high number of shared private alleles between chimpanzees from the Gulf of Guinea (Pan troglodytes ellioti) and from Southern Cameroon (P. t. verus), suggesting recent introgression within a purported hybrid zone65. Charpentier et al. (2012) identified natural hybridization of anubis (Papio anubis) and yellow (P. cynocephalus) baboons in the Amboseli basin in southern Kenya. This hybridization was notably asymmetric: more anubis baboons dispersed into and were reproductively successful, within yellow baboon populations than vice-versa66. Such asymmetric rates of gene flow have also been observed from rhesus (Macaca mulatta) into long-tailed (M. fascicularis) macaques67 and, ancestrally, from western (Pan troglodytes verus) into central (P. t. troglodytes) and eastern (P. t. schweinfurthii) chimpanzees68. Though behavioural explanations for ancestral admixture are speculative, more recent hybridization is thought to suggest an adaptive advantage: baboons with more anubis ancestry might therefore be better adapted for survival among unadmixed yellow baboons, for example66. However, in most of the aforementioned cases, the long-term effects of recent hybridization events are unknown and apparent hybrid vigour may yet give way to outbreeding depression. Furthermore, most hybridization events occurred in the absence of obstructive physical barriers, occurring instead between directly adjacent populations or those separated by an expanse of land that could, in theory, be traversed within an individual’s lifetime. Indeed, some researchers have questioned the validity of the taxonomic units of central (Pan troglodytes troglodytes) and eastern (P. t. schweinfurthii) chimpanzees, pointing out that the variation in these chimpanzees can be better described as following a pattern of increasing genetic differentiation with increasing distance69. In contrast, orang-utan subspecies are separated by largely insurmountable riverine and mountain barriers that limit the potential for large-scale introgression and have ensured independent evolution of the Bornean subspecies for approximately 80,000 years38,39.
In the decades following the reintroductions at Camp Leakey, extensive guidelines have emerged to better facilitate wildlife reintroductions. Over time, these guidelines have placed more explicit focus on genetic considerations. In their Guidelines for the Reintroduction of Animals Born or Held in Captivity, the Association of Zoos and Aquariums suggested that the genotype of reintroduction candidates “should be the closest possible match” to that of the destination population, “so that subspecific distinctions can be maintained”70. More recent ‘best practice’ guidelines from the IUCN — including those for the placement of all species of confiscated animals11, in addition to those specific to apes71,72— go substantially further in mandating that genetic testing be performed prior to reintroductions. In the IUCN’s Guidelines for Reintroductions and Other Conservation Translocations, both pre- and post-release genetic monitoring is encouraged, plus the use and banking of non-invasive samples to help measure reintroduction success12. Recommendations made to the Indonesian government in 199173 – many of which helped form the basis of the aforementioned guidelines – have also led to significant changes in Indonesian law. In 1995, the Indonesian government mandated that orang-utans can only be reintroduced into areas housing their same subspecies and that ex-captives cannot be released into forest that is home to extant wild populations (Decree No. 280/KPTS-II/1995).
In practice, however, these guidelines and legislation are challenging to follow or enforce. In many cases, it is simply too expensive to perform genetic testing: rescuing and accommodating displaced animals takes priority in the face of extremely limited funds. The origin of displaced animals might therefore be inferred as the region from which they were rescued (e.g. Tutin et al.74). A lack of quality laboratory facilities – notably in developing range countries – has also been shown to prove problematic, leading Goossens et al.75 to call for the development of simpler, cheaper molecular methods that can be employed in basic laboratories by inexperienced personnel75. For orang-utans, few rehabilitation centres have performed the required genetic testing in accordance with Indonesian law – those that have done so are thought to have relied solely on mitochondrial DNA, which is uniparentally inherited and thus insufficient to diagnose hybrids – avoiding the costly amplification and use of a geo-referenced panel of autosomal and Y-chromosomal markers (Anonymous 1, pers. comm., 2015). In some cases, orang-utans are known to have been reintroduced into viable wild populations in contravention of the 1995 decree (Anonymous 2, pers. comm., 2015). It is important to acknowledge, however, that efforts to genetically test orang-utans are largely hampered by restrictive Indonesian and Malaysian legislation on the mere collection of biomaterials from endangered species. Further, Indonesia’s domestic legislation on CITES has regulated that faeces be CITES controlled: as a consequence, export of non-invasively collected samples to well-equipped and well-funded laboratories abroad may be time-consuming or impractical. Even with genetic testing, securing protected habitat within the range of each subspecies is proving to be difficult and cost-prohibitive.
Our study demonstrates that reintroduction from sanctuaries can lead – and has led – to de facto translocation, which may ultimately have serious consequences for the health and viability of threatened wild populations. As a consequence, we strongly advise adherence to established international guidelines in future reintroductions, given that the effects of such introgression can be applied to a wide range of endangered animal taxa. For orang-utans, however, the issue may require more pressing attention: having failed to meet the terms of an earlier declaration to close all sanctuaries by the end of 2015, policymakers are now exploring their options to urgently meet this objective. Warren’s76 suggestion of developing isolated, ‘mixed’ populations of rehabilitant individuals may be an attractive course of action, but would only prove a responsible solution if inter-breeding different orang-utan subspecies can be shown to have no detrimental effects.
Additional Information
Accession codes: Novel sequences can be found in GenBank under the accession codes KU523975-KU523977. http://www.nature.com/srep
How to cite this article: Banes, G. L. et al. Reintroduction of confiscated and displaced mammals risks outbreeding and introgression in natural populations, as evidenced by orang-utans of divergent subspecies. Sci. Rep. 6, 22026; doi: 10.1038/srep22026 (2016).
References
Lockhart, J. M., Thorne, E. T. & Gober, D. R. In Recovery of the black-footed ferret: progress and continuing challenges (eds Roelle, J. E., Miller, B. J., Godbey, J. L. & Biggins, D. E. ) 6–19 (US Geological Survey Scientific Investigations Report 2005-5293, 2005).
Kierulff, M. C. M. et al. The golden lion tamarin Leontopithecus rosalia: a conservation success story. Int. Zoo. Yearb. 46, 36–45 (2012).
Platt, J. R. Eyes in the sky: crowdfunded drones could help protect Kenyan rhinos. Sci. Am. 308, 32–32 (2013).
Gibbs, J. P., Hunter, E. A., Shoemaker, K. T., Tapia, W. H. & Cayot, L. J. Demographic outcomes and ecosystem implications of giant tortoise reintroduction to Española Island, Galapagos. PLoS ONE 9, e110742, doi: 10.1371/journal.pone.0110742 (2014).
Keane, A., Jones, J. P. G., Edwards-Jones, G. & Milner-Gulland, E. J. The sleeping policeman: understanding issues of enforcement and compliance in conservation. Anim. Conserv. 11, 75–82 (2008).
Challender, D. W. S. & MacMillan, D. C. Poaching is more than an enforcement problem. Conserv. Lett. 7, 484–494 (2014).
Robbins, M. M. et al. Extreme conservation leads to recovery of the Virunga mountain gorillas. PLoS ONE 6, e19788, doi: 10.1371/journal.pone.0019788 (2011).
Shan, L. et al. Large-scale genetic survey provides insights into the captive management and reintroduction of giant pandas. Mol. Biol. Evol. 31, 2663–2671 (2014).
Burrows, R., Hofer, H. & East, M. L. Demography, extinction and intervention in a small population: the case of the Serengeti wild dogs. Proc. R. Soc. B. 256, 281–292 (1994).
Woodroffe, R. Assessing the risks of intervention: immobilization, radio-collaring and vaccination of African wild dogs. Oryx 35, 234–244 (2001).
IUCN/SSC. IUCN Guidelines for the Placement of Confiscated Animals (IUCN Species Survival Commission, Gland, Switzerland, 2000).
IUCN/SSC. Guidelines for Reintroductions and other Conservation Translocations (IUCN Species Survival Commission, Gland, Switzerland, 2013).
Unwin, S. et al. Does confirmed pathogen transfer between sanctuary workers and great apes mean that reintroduction should not occur? Am. J. Primatol. 74, 1076–1083 (2012).
Trayford, H. R. & Farmer, K. H. Putting the spotlight on internally displaced animals (IDAs): a survey of primate sanctuaries in Africa, Asia and the Americas. Am. J. Primatol. 75, 116–134 (2012).
Frankham, R. et al. Predicting the probability of outbreeding depression. Conserv. Biol. 25, 465–475 (2011).
Frankham, R. Genetic rescue of small inbred populations: meta-analysis reveals large and consistent benefits of gene flow. Mol. Ecol. 24, 2610–2618 (2015).
Waller, D. M. Genetic rescue: a safe or risky bet? Mol. Ecol. 24, 2595–2597 (2015).
Hedrick, P. W. Gene flow and genetic restoration: the Florida panther as a case study. Conserv. Biol. 9, 996–1007 (1995).
Land, E. D. & Lacy, R. C. Introgression level achieved through Florida panther genetic restoration. Endang. Sp. Update, 7(5), 100–105 (2000).
Mansfield, K. G. & Land, E. D. Cryptorchidism in Florida panthers: prevalence, features and influence of genetic restoration. J. Wildl. Dis. 38, 693–698 (2002).
Johnson, W. E. et al. Genetic restoration of the Florida panther. Science 329, 1641–1645 (2010).
Vilà, C. et al. Rescue of a severely bottlenecked wolf (Canis lupus) population by a single immigrant. Proc. R. Soc. B. 270, 91–97 (2003).
Westemeier, R. L. et al. Tracking the long-term decline and recovery of an isolated population. Science 282, 1695–1698 (1998).
Madsen, T., Shine, R., Olsson, M. & Wittzell, H. Conservation biology: restoration of an inbred adder population. Nature 402, 34–35 (1999).
Arrendal, J., Walker, C. W., Sundqvist, A.-K., Hellborg, L. & Vilà, C. Genetic evaluation of an otter translocation program. Conserv. Genet. 5, 79–88 (2004).
Rhymer, J. M. & Simberloff, D. Extinction by hybridization and introgression. Annu. Rev. Ecol. Syst. 27, 83–109 (1996).
Allendorf, F. W., Leary, R. F., Spruell, P. & Wenburg, J. K. The problems with hybrids: setting conservation guidelines. Trends Ecol. Evol. 16, 613–622 (2001).
Tallmon, D., Luikart, G. & Waples, R. The alluring simplicity and complex reality of genetic rescue. Trends Ecol. Evol. 19, 489–496 (2004).
Edmands, S. Between a rock and a hard place: evaluating the relative risks of inbreeding and outbreeding for conservation and management. Mol. Ecol. 16, 463–475 (2007).
Gharrett, A. J., Smoker, W. W., Reisenbichler, R. R. & Taylor, S. G. Outbreeding depression in hybrids between odd- and even-broodyear pink salmon. Aquaculture 173, 117–129 (1999).
Huff, D. D., Miller, L. M., Chizinski, C. J. & Vondracek, B. Mixed-source reintroductions lead to outbreeding depression in second-generation descendants of a native North American fish. Mol. Ecol. 20, 4246–4258 (2011).
Alstad, D. N. & Edmunds, G. F. Selection, outbreeding depression and the sex ratio of scale insects. Science 220, 93–95 (1983).
Edmands, S. Heterosis and outbreeding depression in interpopulation crosses spanning a wide range of divergence. Evolution 53, 1757 (1999).
Marr, A. B., Keller, L. F. & Arcese, P. Heterosis and outbreeding depression in descendants of natural immigrants to an inbred population of song sparrows (Melospiza melodia). Evolution, 56, 131–42 (2002).
Turček, F. J. & Hickey, J. J. Effect of introductions on two game populations in Czechoslovakia. Jour. Wild. Mgmt. 15, 113 (1951).
Lacy, R. C., Petric, A. & Warneke, M. In The Natural History of Inbreeding and Outbreeding (ed Thornhill, N. W. ) 352–374 (University of Chicago Press, 1993).
Soehartono, T. et al. Strategi dan rencana aksi konservasi orangutan Indonesia 2007–2017 (Direktorat Jenderal Perlindungan Hutan dan Konservasi Alam, Departemen Kehutanan, Jakarta, 2007).
Arora, N. et al. Effects of Pleistocene glaciations and rivers on the population structure of Bornean orangutans (Pongo pygmaeus). PNAS 107, 21376–21381 (2010).
Nater, A. et al. Reconstructing the demographic history of orang-utans using Approximate Bayesian Computation. Mol. Ecol. 24, 310–327 (2015).
Goossens, B. et al. Patterns of genetic diversity and migration in increasingly fragmented and declining orang-utan (Pongo pygmaeus) populations from Sabah, Malaysia. Mol. Ecol. 14, 441–456 (2005).
Jalil, M. F. et al. Riverine effects on mitochondrial structure of Bornean orang-utans (Pongo pygmaeus) at two spatial scales. Mol. Ecol. 17, 2898–2909 (2008).
Kanthaswamy, S. & Smith, D. G. Population subdivision and gene flow among wild orangutans. Primates 43, 315–327 (2002).
Warren, K. S. et al. Speciation and intrasubspecific variation of Bornean orangutans, Pongo pygmaeus pygmaeus. Mol. Biol. Evol. 18, 472–480 (2001).
Galdikas, B. M. F. Reflections of Eden. (Little, Brown and Co., New York, 1995).
Galdikas, B. M. & Ashbury, A. Reproductive parameters of female orangutans (Pongo pygmaeus wurmbii) 1971–2011, a 40-year study at Tanjung Puting National Park, Central Kalimantan, Indonesia. Primates, 54, 61–72 (2012).
Yeager, C. P. Orangutan rehabilitation in Tanjung Puting National Park, Indonesia. Conserv. Biol. 11, 802–805 (1997).
Banes, G. L., Galdikas, B. M. F. & Vigilant, L. Male orang-utan bimaturism and reproductive success at Camp Leakey in Tanjung Puting National Park, Indonesia. Behav. Ecol. Sociobiol. 69, 1785–1794 (2015).
Thalmann, O., Hebler, J., Poinar, H. N., Paabo, S. & Vigilant, L. Unreliable mtDNA data due to nuclear insertions: a cautionary tale from analysis of humans and other great apes. Mol. Ecol. 13, 321–335 (2004).
Tamura, K., Stecher, G., Peterson, D., Filipski, A. & Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 30, 2725–2729 (2013).
Edgar, R. C. MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 32, 1792–1797 (2004).
Ronquist, F. et al. MrBayes 3.2: Efficient Bayesian phylogenetic inference and model choice across a large model space. Syst. Biol. 61, 539–542 (2012).
Darriba, D., Taboada, G. L., Doallo, R. & Posada, D. jModelTest 2: more models, new heuristics and parallel computing. Nature Methods 9, 772–772 (2012).
Utami-Atmoko, S. S., Singleton, I., van Noordwijk, M. A., van Schaik, C. P. & Mitra-Setia, T. In Orangutans: Geographic Variation in Behavioral Ecology and Conservation (eds Wich, S. A., Utami Atmoko, S. S., Mitra Setia, T. & van Schaik, C. P. ) 225–234 (Oxford University Press, 2008).
Spalding, L. The jungle took her. Outside, 23(5), 78–90 & 189-191 (1998).
Spalding, L. The Follow. (Key Porter Books, Toronto, 1998).
Kuze, N. et al. Factors affecting reproduction in rehabilitant female orangutans: young age at first birth and short inter-birth interval. Primates 53, 181–192 (2012).
Perkins, L. A. & Maple, T. L. North American orangutan species survival plan: current status and progress in the 1980s. Zoo Biol. 9, 135–139 (1990).
van Bemmel, A. C. V. Contribution to the knowledge of the geographical races of Pongo pygmaeus (Hoppius). Bijdr. Dierkd. 38, 13–15 (1968).
Jones, M. L. In Recent Advances in Primatology (ed. Hofer, H. O. ) 217–223 (Karger, Basel, 1969).
Mallinson, J. “Cocktail” orangutans and the need to preserve pure-bred stock. Dodo. 15, 69–77 (1978).
Seuanez, H. N., Evans, H. J., Martin, D. E. & Fletcher, J. An inversion of chromosome 2 that distinguishes between Bornean and Sumatran orangutans. Cytogenet. Cell Genet. 23, 137–140 (1979).
Xu, X. & Arnason, U. The mitochondrial DNA molecule of sumatran orangutan and a molecular proposal for two (Bornean and Sumatran) species of orangutan. J. Mol. Evol. 43, 431–437 (1996).
Zhang, Y.-W., Ryder, O. A. & Zhang, Y.-P. Genetic divergence of orangutan subspecies (Pongo pygmaeus). J. Mol. Evol. 52, 516–526 (2014).
van Schaik, C. P., Marshall, A. J. & Wich, S. A. In Orangutans: Geographic Variation in Behavioral Ecology and Conservation (eds Wich, S. A., Utami Atmoko, S. S., Mitra Setia, T. & van Schaik, C. P. ) 351–362 (Oxford University Press, 2008).
Gonder, M. K. et al. Evidence from Cameroon reveals differences in the genetic structure and histories of chimpanzee populations. PNAS 108, 4766–4771 (2011).
Charpentier, M. J. E. et al. Genetic structure in a dynamic baboon hybrid zone corroborates behavioural observations in a hybrid population. Mol. Ecol. 21, 715–731 (2011).
Stevison, L. S. & Kohn, M. H. Divergence population genetic analysis of hybridization between rhesus and cynomolgus macaques. Mol. Ecol. 18, 2457–2475 (2009).
Hey, J. The divergence of chimpanzee species and subspecies as revealed in multipopulation isolation-with-migration analyses. Mol. Biol. Evol. 27, 921–933 (2010).
Fünfstück, T. et al. The genetic population structure of wild western lowland gorillas (Gorilla gorilla gorilla) living in continuous rain forest. Am. J. Primatol. 76, 868–878 (2014).
Association of Zoos and Aquariums. Guidelines for reintroduction of animals born or held in captivity (Association of Zoos and Aquariums, Silver Spring, 1992).
Beck, B. et al. Best practice guidelines for the re-introduction of great apes: occasional paper of the IUCN Species Survival Commission No. 35. 1–56 (SSC Primate Specialist Group of the World Conservation Union, Gland, Switzerland, 2007).
Campbell, C. O., Cheyne, S. M. & Rawson, B. M. Best practice guidelines for the rehabilitation and translocation of gibbons. (IUCN SSC Primate Specialist Group, Gland, Switzerland, 2015).
Sajuthi, D. et al. Recommendations to the Department of Forestry of the Republic of Indonesia on the medical quarantine of orangutans intended for reintroduction. Proceedings of the Great Apes Conference, 132–136 (Indonesian Department of Forestry and Department of Tourism, Jakarta, 1991).
Tutin, C. E. G. et al. Conservation biology framework for the release of wild-born orphaned chimpanzees into the Conkouati Reserve, Congo. Conserv. Biol. 15, 1247–1257 (2001).
Goossens, B. et al. Measuring genetic diversity in translocation programmes: principles and application to a chimpanzee release project. Anim. Conserv. 5, 225–236 (2002).
Warren, K. S. Orang-utan conservation-epidemiological aspects of health management and population genetics. Ph.D. thesis, Murdoch University, Division of Veterinary and Biomedical Sciences, March (2001).
Wich, S. A. et al. Understanding the impacts of land-use policies on a threatened species: is there a future for the Bornean orang-utan? PLoS ONE 7(11), e49142, doi: 10.1371/journal.pone.0049142 (2012).
Acknowledgements
We thank the Indonesian State Ministry of Research and Technology (RISTEK), the Indonesian State Ministry of Forestry (PHKA), the Indonesian Institute of Sciences (LIPI), the Indonesian Agency for Natural Resource Conservation (BKSDA) and the Tanjung Puting National Park Office (BTNTP) for granting their permission to undertake this research and to our formal counterpart, S. H. Limin of CIMTROP at Universitas Palangka Raya, for his generous support of this work. We are indebted to A. E. Leiman OBE, S. Brend, P. A. Racey and L. Howe who were instrumental to facilitating this study, to D. Rachmawan and the Indonesian assistants who assisted with sample collection, to R. J. Turton for laboratory assistance and to V. Städele for her comments on an earlier version of this manuscript. This research was generously funded by the University of Aberdeen (UoA) Expedition Fund; UoA Small Grants Fund; UoA Alumni Annual Fund; the John Reid Trust; the Royal Geographical Society with Institute of British Geographers; the Royal Scottish Geographical Society; the Gilchrist Educational Trust; the Orangutan Foundation UK; the North of England Zoological Society at Chester Zoo; Darwin College, Cambridge; the Primate Society of Great Britain, through a Charles A Lockwood Memorial Grant; the ARCUS Foundation; the Leverhulme Centre for Human Evolutionary Studies, University of Cambridge; the Ridgeway-Venn Studentship; the Miss Millie Foundation; the American Association of Zoo Keepers Los Angeles Chapter; Henry Vilas Zoo; The Orang-utan Conservation Genetics Trust; the Max Planck Society; the Chinese Academy of Sciences and the National Natural Science Foundation of China (grant numbers 31450110070 and 91331203). GLB was supported by the Orang Utan Republik Foundation through the 2012 LP Jenkins Memorial Fellowship and by the German Academic Exchange Service (DAAD). The long-term research conducted at Camp Leakey, which facilitated this genetic study, was primarily funded by the Orangutan Foundation International. Samples were exported from the Republic of Indonesia to the European Union, via the United Kingdom, under permits 14459/IV/SATS-LN/2008 and 00459/IV/SATS-LN/2011 from the Convention on International Trade in Endangered Species of Wild Fauna and Flora (CITES).
Author information
Authors and Affiliations
Contributions
G.L.B. and B.M.F.G. collected the samples, G.L.B. performed the analyses and prepared the figures, G.L.B. and L.V. designed the study and wrote the manuscript, which all authors reviewed and revised.
Ethics declarations
Competing interests
There are potential competing financial interests. BMFG continues to manage Camp Leakey and – as director of a nearby orang-utan rehabilitation centre – remains actively involved in the rehabilitation, reintroduction and translocation of these species. BMFG is paid a salary for her work by the Orangutan Foundation International.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Banes, G., Galdikas, B. & Vigilant, L. Reintroduction of confiscated and displaced mammals risks outbreeding and introgression in natural populations, as evidenced by orang-utans of divergent subspecies. Sci Rep 6, 22026 (2016). https://doi.org/10.1038/srep22026
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep22026
This article is cited by
-
Population history of chimpanzees introduced to Lake Victoria’s Rubondo Island
Primates (2021)
-
Prioritising source populations for supplementing genetic diversity of reintroduced southern brown bandicoots Isoodon obesulus obesulus
Conservation Genetics (2021)
-
Genomic targets for high-resolution inference of kinship, ancestry and disease susceptibility in orang-utans (genus: Pongo)
BMC Genomics (2020)
-
Genetic structure of regional water vole populations and footprints of reintroductions: a case study from southeast England
Conservation Genetics (2020)
-
Phenotypic, Genetic, and Cytogenetic Evidence of Hybridization Between Species of Trans-Andean Tamarins (Genus Saguinus)
International Journal of Primatology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.