Abstract
Rising levels of atmospheric CO2 have been implicated in changes in the nitrogen (N) and phosphorus (P) content of terrestrial vegetation; however, questions remain over the role of C, N and P interactions in driving plant nutrient stoichiometry, particularly whether N and P additions alter vegetation responses to CO2 enrichment singly. Here we use meta-analysis of 46 published studies to investigate the response of plant N and P to elevated CO2 alone and in combination with nutrient (N and P) additions across temperate vs. tropical biomes. Elevated CO2 reduces plant N concentrations more than plant P concentrations in total biomass pools, resulting in a significant decline in vegetation N/P. However, elevated CO2 treatments in combination with N additions increase plant P concentrations, whereas P additions have no statistical effect on plant N concentrations under CO2 enrichment. These results point to compensatory but asymmetrical interactions between N, P and CO2; that changes in N rapidly alter the availability of P, but not the converse, in response to increased CO2. Our finding implies widespread N limitation with increasing atmospheric CO2 concentrations alone. We also suggest that increased anthropogenic N deposition inputs could enhance plant N and P in a progressively CO2-enriched biosphere.
Similar content being viewed by others
Introduction
Atmospheric carbon dioxide concentrations (CO2) have grown considerably due to human actions, with an anticipated peak concentration of greater than 700 μmol mol−1 by the end of this century1. The modern rise in CO2 is thought to stimulate terrestrial productivity2, resulting in negative feedback on atmospheric CO2 levels and reductions in the pace of climate warming; however, the magnitude of this feedback is thought to be constrained by growth-limiting nutrients, especially nitrogen (N) and phosphorus (P) availability3,4,5. Here we use meta-analysis to examine changes in N, P and N/P in plants in response to elevated CO2 and nutrient fertilization across a range of sites and conditions.
The stoichiometry of plant N and P concentrations has provided insight in patterns of N versus P limitation across terrestrial ecosystems6,7,8, including nutrient limitation responses to elevated CO29. Past work has demonstrated that elevated CO2 reduces plant N concentrations generally10,11, largely as a result of the carbohydrate dilution12 and the inhibition of nitrate assimilation within plants13. The effect of CO2 enrichment on plant P concentrations has been more variable than for N, with evidence for decreased14, increased15 or neutral effects on plant P16 in individual study systems. Using meta-analysis, Duval et al.17 showed that plant P responses to elevated CO2 varied among plant functional groups. More recently, Yuan and Chen18 showed that elevated CO2 on average decreases plant N/P across an array of sites; however, this study did not examine tropical plant N/P responses, despite tropical systems having different nutrient conditions than many temperate ones3. Further, past syntheses have not addressed the response of plant N/P to CO2 in combination with other N, P or N plus P additions. Questions remain over whether N or P will become progressively more limiting under elevated CO2 and how such limitations will be affected by changes in P and N inputs across temperate vs. tropical ecosystems. This is critical given that C, N and P are among the most anthropogenically altered biogeochemical cycles on Earth19.
It is increasingly important to recognize that plant responses to CO2 are acting in combination with other global changes, particularly changes in anthropogenic N deposition and P inputs through cropland fertilization, which can also affect patterns of nutrient cycling and limitation on land. Nitrogen inputs can increase plant productivity and lead to over-enrichment with N20 and can even result in what has been termed “anthropogenic P limitation”3–the progressive occurrence of P limitation to plant productivity via chronic N inputs from human sources. Indeed, past evidence indicates increased plant N/P with increased N inputs18,21. On the other hand, the increasing use of P fertilizer is improving its availability in ecosystems4. The response of plant N/P to altered P availability can be expected, showing a decrease in plant N/P under P fertilization18.
Yuan and Chen18 suggested that multiple global change treatments including elevated CO2, precipitation, warming and N deposition result in additive effects on plant N/P ratios. However, they did not study the effects of elevated CO2 with P additions or with N and P additions combination. Evidence for widespread nutrient co-limitation suggests that the cycling of one nutrient can influence the availability of another22. Past work in Hawaii has shown that N fertilization increased plant-root and soil phosphatase levels23, a finding that was later demonstrated across a wide variety of terrestrial ecosystems24. P fertilization was found to enhance N uptake by plants and make plant roots competitive for N against free-living microbes in forest ecosystems25. These interactions between N and P have also been shown to play a role in global patterns of symbiotic N2-fixation26 and allow plants and mycorrhizal fungi to allocate biomass and energy towards the acquisition of limiting resources in general27.
Here, we analyze plant N/P in response to elevated CO2 alone and in combination with N, P and N + P additions to test the hypotheses that (1) elevated CO2 decreases plant N and P concentrations;(2) nutrient inputs prevent declines in plant nutrient contents of the fertilized nutrient under elevated CO2; and (3) inputs of N or P alters plant nutrient contents of the other nutrient under elevated CO2 due to compensatory interactions among C, N and P cycles.
Results
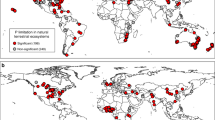
We collected 133 observations from 46 separate studies to examine CO2 treatment effects on plant N/P (see Supplementary References). The majority of the data was from the temperate regions (70%) as opposed to the (sub-)tropics (28%) (see Fig. 1 and Supplementary Table S1). Of these observations, 28 measured plant N/P in FACE; 97 measured plant N/P in chambers; and the remaining 8 were classified as others including branch bag techniques, natural CO2 springs and screen-aided CO2 control. Of these species, 86 observations were collected from woody plants and 47 from non-woody plants. There were 19 observations for legumes and 114 for non-legumes. Most studies focused on aboveground plant nutrient concentrations (102 observations) compared to 27 belowground observations and 4 whole-plant concentrations reported. Of the compiled studies, there were 34 observations for elevated CO2 with N alone, 22 observations for elevated CO2 with added P alone and 15 observations for elevated CO2 with added N and P in combination (see Supplementary Table S1).
Map of sites of the collected studies.
Figure 1 was created by R (R Core Team, 2014)54 using the maps package.
Effects of elevated CO2 on plant N, P and N/P
Plant N concentrations decreased significantly with elevated CO2 (~12%), regardless of the climatic zones, the kind of CO2 delivery, plant functional group or plant tissue examined (Fig. 2a). Across all observations in this analysis, elevated CO2 was associated with an averaged 4% decrease in plant P concentrations, but not consistently among factors as observed for decreased N concentrations (Fig. 2b). Rather, elevated CO2 decreased plant P concentration in temperate regions but increased it in (sub-)tropics. CO2-incuced declines in plant P concentrations were observed for non-woody plants and aboveground tissues, but not for woody plants and belowground tissues. Hence, elevated CO2 significantly decreased plant N/P by 11% compared to control conditions, but this response varied with climatic zones (Fig. 2c). Overall, elevated CO2 decreased plant N/P to a greater extent in (sub-)tropics (23%) than in temperate regions (6%). CO2 enrichment caused significant declines in plant N/P for chambers (14%) but not FACE experiments, for woody species (13%) but not non-woody species and for legumes (11%) but not for non-legumes.
Effects of elevated CO2 on plant N and P stoichiometry.
(a) plant N concentrations; (b) plant P concentrations; (c) plant N/P. The error bars show the 95% confidence interval of LnRR. LnRR, the natural logarithm of response ratio that is calculated as the experimental mean divided by the control mean. The asterisk (*) denotes the effect of elevated CO2 was significant. Different lowercases in the left indicate significant differences between groups. The number of observations for each category is given in the right.
Effects of elevated CO2 with nutrient fertilization on plant N, P and N/P
In the dataset of elevated CO2 with N fertilization, elevated CO2 alone (without N fertilization) significantly decreased plant N concentration (3%) and increased plant P concentration (16%), thus resulting in a decrease of 20% in plant N/P (see Supplementary Fig. S1a). However, elevated CO2 with N addition did not affect plant N/P, as it led to statistically equivalent increases in plant N concentration (20%) and in plant P concentration (16%) (Fig. 3a). The response ratios of plant N concentrations, P concentrations and N/P with CO2 enrichment and N fertilization were not related to the amount of N added (see Supplementary Fig. S2).
Effects of elevated CO2 and nutrient fertilization on plant N and P stoichiometry.
(a) elevated CO2 with N fertilization (CO2 + N); (b) elevated CO2 with P fertilization (CO2 + P); (c) elevated CO2 with N and P fertilizations (CO2 + NP). The error bars show the 95% confidence interval of LnRR. LnRR, the natural logarithm of response ratio that is calculated as the experimental mean divided by the control mean. The asterisk (*) denotes the effect of treatments was significant. The number of observations for each category is given in the right.
Elevated CO2 without P fertilization tended to negatively affect plant N concentrations, P concentrations and N/P (see Supplementary Fig. S1b). However, the patterns were altered when P was added. CO2 plus P treatments consistently decreased plant N concentrations (7%), increased plant P concentrations (24%) and decreased plant N/P ratios (26%) (Fig. 3b). The responses of plant N and P concentrations and plant N/P to elevated CO2 with P fertilization were not related to the amount of P added (see Supplementary Fig. S2).
In the dataset of elevated CO2 with N and P fertilizations, N and P fertilizations did not significantly affect plant N concentration, P concentration and N/P responses to elevated CO2 (see Supplementary Fig. S1c and Fig. 3c).
Relationships between plant N and P concentrations
The response ratios (the experimental mean divided by the control mean) of plant N concentrations to elevated CO2 were significantly positively related to those of plant P concentrations (R2 = 0.1716, P < 0.01) (Fig. 4a). There was a significant negative relationship between the response ratios of plant N and P concentration under elevated CO2 when N added (Fig. 4b), while no significant relationship was observed under elevated CO2 when P or NP added (Fig. 4c,d).
Relationships between the response ratios of plant N and P concentrations to elevated CO2 alone and elevated CO2 with nutrient fertilization.
LnRR, the natural logarithm of response ratio that is calculated as the experimental mean divided by the control mean. The circles indicate the values which were greater than 1 or smaller than −1. Linear regressions were estimated using the points excluding the circles. (a) Elevated CO2 alone (CO2); (b) elevated CO2 with N fertilization (CO2 + N); (c) elevated CO2 with P fertilization (CO2 + P); (d) elevated CO2 with N and P fertilizations (CO2 + NP).
Discussion
This cross-system analysis supports expectations for our first hypothesis – that plant N and P contents decline systematically under CO2 enrichment singly. This result was clear and systematic in the case of plant N concentrations and generally confirmed results of previous research10,11,28,29. In contrast, plant P responses to CO2 have been less-examined. Our results showed that P exhibited a more complex CO2 response than N across a broad range of terrestrial ecosystems, which resulted in some differences in plan N/P. For example, plant P was increased under elevated CO2 in (sub-)tropics rather than temperate regions, which imply that relatively high soil N availability in (sub-)tropics could help plants acquire P with high energy (C) inputs26. While aboveground P concentrations declined with CO2 enrichment, P in belowground tissues did not change with elevated CO2. This suggests that plants may be allocating more P to roots under elevated CO2, or actively mining soil P pools, perhaps via increased investment in mycorrhizal fungi and fine roots30,31. In addition, woody plants showed almost no change in P concentrations under elevated CO2, whereas P concentrations of herbaceous vegetation declined significantly, revealing strong functional-group dependencies in plant P responses. The relative stability in legume N/P under elevated CO2 indicate that N2-fixing species has the ability to balance N and P through direct access to N from atmosphere and investment in phosphatase and mycorrhizae to acquire P26,32.
Our results showed net declines in plant N/P under elevated CO2, which were in line with those reported for temperate ecosystems by Yuan and Chen18. Our study advances their analysis by revealing that this decline was driven by much larger reductions in plant N concentrations (12%) than plant P concentrations (4%), including both temperate and tropical ecosystems. In fact, the coefficient of the linear regression (slope) of the responses of plant P vs. N contents to elevated CO2 was less than unity (P < 0.01) (Fig. 4a). This means that the changes in plant N contents exceeded those of plant P across our global data synthesis. These findings are consistent with a recent study of C3 plants in which a 15% reduction in N but only 9% reduction in P occurred with enhanced CO2 concentrations33.
Generally, the decline in plant N/P we observed points to more substantial N vs. P limitation with CO2 enrichment, which can explained by factors that differentially affect plant metabolism of N vs. P11,12. Elevated CO2 may increase the efficiency of photosynthesis or metabolically down-regulate photosynthetic enzymes, thus causing increasing photosynthetic N use efficiency with decreasing N supplies34,35. Further, higher ATP requirements in response to elevated CO2 would disproportionally increase P vs. N demands in ecosystems36,37. In addition, elevated CO2 could increase plant growth rates, which requires P-rich ribosomal RNA (rRNA)38,39. Any combination of these mechanisms would lower N/P in response to elevated CO2.
Our results also support the second hypothesis – that nutrient inputs alter plant N/P response to elevated CO2 (Fig. 4). This result implies that plant nutrient responses to rising CO2 will depend on the magnitude of changes in N and P inputs in ecosystems9. This is important given that to anthropogenic alterations of N and P that have occurred in concert with atmospheric CO2. The positive responses of plant N and P to N and P inputs, respectively, have the potential to compensate for their declines induced by elevated CO2. These results contrast with Cotrufo et al.10, who found no evidence for an effect of N fertilization on tissue N concentrations beyond the declines observed with elevated CO2. This discrepancy could reflect our differing approaches; Cotrufo et al.10 divided their observations into arbitrary classes of N addition whereas we used a combination of meta-analysis and regression to examine nutrient by CO2 effects. Elevated CO2 frequently stimulates rates of photosynthesis, yet N or P supply can modulate the magnitude of the CO2-fertilization effect via its effect on the carboxylation capacity29,40,41. Wholesale reductions or longer-term acclimation of photosynthesis to elevated CO2 can be associated with declines in tissue N concentrations42,43. Our results imply that either N or P supplies can offset any down-regulation of photosynthesis in response to CO2 enrichment.
Finally, we found support for the third hypothesis – that input of N or P alters plant nutrient contents of the other nutrient– in the case of N by CO2 additions but not for P by CO2 treatments. Plant P concentrations increased under elevated CO2 with N addition, thereby stabilizing plant N/P across terrestrial ecosystems. Nitrogen additions have been shown to enhance P uptake in roots from P-deficient soluble sources under elevated CO2 by inducing a set of morphological, physiological and molecular adaptive strategies44. Numerous studies have shown that N additions can facilitate P acquisition via alterations in root development45 and rhizosphere pH46 and via organism investments in phosphatase enzymes24. However, there was no apparent trade-off in responses of plant N and P to elevated CO2 or N addition in our meta-analysis. On the contrary, we found no evidence for a compensating effect of CO2 plus P fertilization treatments on the declines in plant N concentrations under CO2. Previous analysis on the effect of P additions on plant N concentrations have been inconsistent, with no changes in plant N observed under N-rich conditions47, decreases observed in N-poor soils48 and increases under conditions of P limitation25. Our results suggest that, on average, P additions do not alter the pattern of systematically declining plant N concentrations with CO2 enrichment, consistent with several past studies from individual sites49,50.
In summary, our extensive meta-analysis shows that elevated CO2 decreases plant N/P, implying a tendency for N rather than P limitation of terrestrial productivity to arise in response to elevated CO2 alone. However, when CO2 and N increase in concert, as is occurring over much of the terrestrial biosphere, compensatory interactions among C, N and P cycles stabilize total plant N/P. These results suggest that, ecosystems exposed to elevated CO2 and N deposition will not necessarily progress rapidly into conditions of P limitation, owing to a suite of non-symmetrical interactions between N and P. Longer-term effects remain unclear, however, models used to forecast the N and P cycles in response to CO2 and climate change would be well-served to consider such compensatory interactions in determining the capacity for additional CO2 sequestration in the future
Methods
We searched ISI Web of Science, using the terms “elevated CO2”, “CO2 enrichment” and “CO2 enriched”, in combination with “nutrient”, “nitrogen” and “phosphorus”, to create a database for our meta-analysis. The data were restricted to studies performed in natural terrestrial ecosystems, i.e., excluding agricultural or other managed ecosystems. All studies included in this analysis measured N/P or N and P concentrations (from which we could calculate N/P) under ambient CO2 and elevated CO2. We also limited our data to studies where means, standard deviations of the mean and number of replicates were reported or could be calculated. For studies that measured N and P concentrations at multiple time-points, only the final value was used to maintain the statistically independence between individual observations. DataThief51 was used to acquire numbers from figures where data were not presented in tables. The units for N and P concentrations were all converted to mg g−1. All N/P are presented here on a mass basis.
We categorized the data by climatic zones, experiment types, plant characteristics and presence any kind of nutrient fertilization involving N and P. The climatic zones included arctic, temperate and (sub-)tropical zones. Research facilities used to increase CO2 concentration were classified as Free-Air Carbon dioxide Enrichment (FACE) and chambers; plants were categorized based on functional type (woody vs. non-woody plants and non-legume vs. legume) and plant tissues examined (i.e., aboveground vs. belowground).
To examine interactions between elevated CO2 and nutrient (N and P) fertilization, we also collected data on the level N and/or P additions where available, including elevated CO2 with N fertilization (from 1 g N m−2 yr−1 to 20 g N m−2 yr−1) (CO2 + N), elevated CO2 with P fertilization (from 1.5 g P m−2 yr−1 to 16 g P m−2 yr−1) (CO2 + P) and elevated CO2 with N and P fertilizations (CO2 + NP). For each of the three datasets, we made two comparisons: elevated CO2 without nutrient fertilization vs. control (ambient CO2 without nutrient fertilization) and elevated CO2 with nutrient fertilization vs. control.
The treatment effects were examined by calculating response ratios (RR) from each individual study. RR is used as an indicator of effect size and is calculated as the experimental mean divided by the control mean (ambient CO2 without nutrient fertilization)52. The natural logarithm of response ratio (LnRR) was then used to perform statistical analysis as it equally weighs the negative and positive responses52. The mean effect size was calculated using a mixed-effects model of the meta-analytical software MetaWin 2.053. The 95% confidence intervals (CI) for effect-size estimates were calculated using resampling techniques. The effects of experimental treatments of a variable were considered to be significant if the 95% CI values did not overlap with zero52. We compared the responses to experimental treatments between groups in each category, in which each group should have at least five studies52. The responses to experimental treatments among groups in each category were considered to be significantly different when the between-group heterogeneity was significant (P < 0.05)52. The differences between means of the groups were considered statistically significant if their 95% CI values did not overlap.
Additional Information
How to cite this article: Huang, W. et al. Plant stoichiometric responses to elevated CO2 vary with nitrogen and phosphorus inputs: Evidence from a global-scale meta-analysis. Sci. Rep. 5, 18225; doi: 10.1038/srep18225 (2015).
References
IPCC. Climate change 2013: the physical science basis. Contribution of Working Group I to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change. (eds Stocker, T. F. et al. ) (Cambridge University Press, Cambridge, UK & New York, NY, USA, 2013).
Norby, R. J. et al. Forest response to elevated CO2 is conserved across a broad range of productivity. Proc. Natl. Acad. Sci. UAS 102, 18052–18056 (2005).
Vitousek, P. M., Porder, S., Houlton, B. Z. & Chadwick, O. A. Terrestrial phosphorus limitation: mechanisms, implications and nitrogen phosphorus interactions. Ecol. Appl. 20, 5–15 (2010).
Peñuelas, J. et al. Human-induced nitrogen-phosphorus imbalances alter natural and managed ecosystems across the globe. Nat. Commun. 4, 2934 (2013).
Reich, P. B. & Hobbie, S. E. Decade-long soil nitrogen constraint on the CO2 fertilization of plant biomass. Nat. Clim. Chang. 3, 278–282 (2013).
Vitousek, P. M. . Litterfall, nutrient cycling and nutrient limitation in tropical forests. Ecology 65, 285–298 (1984).
McGroddy, M. E., Daufresne, T. & Hedin, L. O. Scaling of C : N : P stoichiometry in forests worldwide: implications of terrestrial redfield-type ratios. Ecology 85, 2390–2401 (2004).
Townsend, A. R., Cleveland, C. C., Asner, G. P. & Bustamante, M. M. C. Controls over foliar N:P ratios in tropical rain forests. Ecology 88, 107–118 (2007).
Güsewell, S. N. : P ratios in terrestrial plants: variation and functional significance. New Phytol. 164, 243–266 (2004).
Cotrufo, M. F., Ineson, P. & Scott, A. Elevated CO2 reduces the nitrogen concentration of plant tissues. Glob. Change Biol. 4, 43–54 (1998).
Taub, D. R. & Wang, X. Z. Why are nitrogen concentrations in plant tissues lower under elevated CO2 ? A critical examination of the hypotheses. J. Integr. Plant Biol 50, 1365–1374 (2008).
Loladze, I. Rising atmospheric CO2 and human nutrition: toward globally imbalanced plant stoichiometry ? Trends Ecol. Evol. 17, 457–461 (2002).
Bloom, A. J., Burger, M., Rubio-Asensio, J. S. & Cousins, A. B. Carbon dioxide enrichment inhibits nitrate assimilation in wheat and Arabidopsis. Science 328, 899–903 (2010).
Teng, N. J. et al. Elevated CO2 induces physiological, biochemical and structural changes in leaves of Arabidopsis thaliana. New Phytol. 172, 92–103 (2006).
Liu, J. X. et al. Nitrogen to phosphorus ratios of tree species in response to elevated carbon dioxide and nitrogen addition in subtropical forests. Glob. Change Biol. 19, 208–216 (2012).
Johnson, D. W. et al. Effects of elevated CO2 on nutrient cycling in a sweetgum plantation. Biogeochemistry 69, 379–403 (2004).
Duval, B. D., Blankinship, J. C., Dijkstra, P. & Hungate, B. A. CO2 effects on plant nutrient concentration depend on plant functional group and available nitrogen: a meta-analysis. Plant Ecol. 213, 505–521 (2012).
Yuan, Z. Y. & Chen, H. Y. H. Decoupling of nitrogen and phosphorus in terrestrial plants associated with global changes. Nat. Clim. Chang. 5, 465–469 (2015).
Falkowski, P. et al. The global carbon cycle: a test of our knowledge of earth as a system. Science 290, 291–296 (2000).
Matson, P., Lohse, K. A. & Hall, S. J. The globalization of nitrogen deposition: consequences for terrestrial ecosystems. Ambio 31, 113–119 (2002).
Sardans, J., Rivas-Ubach, A. & Peñuelas, J. The C:N:P stoichiometry of organisms and ecosystems in a changing world: a review and perspectives. Perspect. Plant Ecol. Evol. Syst. 14, 33–47 (2012).
Elser, J. J. et al. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol. Lett. 10, 1135–1142 (2007).
Treseder, K. K. & Vitousek, P. M. Effects of soil nutrient availability on investment in acquisition of N and P in Hawaiian rain forests. Ecology 82, 946–954 (2001).
Marklein, A. R. & Houlton, B. Z. Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol. 193, 696–704 (2012).
Blanes, M. C., Emmett, B. A., Vinegla, B. & Carreira, J. A. Alleviation of P limitation makes tree roots competitive for N against microbes in a N-saturated conifer forest: A test through P fertilization and 15N labelling. Soil Biol. Biochem. 48, 51–59 (2012).
Houlton, B. Z., Wang, Y. P., Vitousek, P. M. & Field, C. B. A unifying framework for dinitrogen fixation in the terrestrial biosphere. Nature 454, 327–330 (2008).
Johnson, N. C., Rowland, D. L., Corkidi, L., Egerton-Warburton, L. M. & Allen, E. B. Nitrogen enrichment alters mycorrhizal allocation at five mesic to semiarid grasslands. Ecology 84, 1895–1908 (2003).
Curtis, P. S. & Wang, X. Z. A meta-analysis of elevated CO2 effects on woody plant mass, form and physiology. Oecologia 113, 299–313 (1998).
Ainsworth, E. A. & Long, S. P. What have we learned from 15 years of free-air CO2 enrichment (FACE) ? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2 . New Phytol. 165, 351–372 (2005).
Nie, M., Lu, M., Bell, J., Raut, S. & Pendall, E. Altered root traits due to elevated CO2: a meta-analysis. Glob. Ecol. Biogeogr. 22, 1095–1105 (2013).
Treseder, K. K. A meta-analysis of mycorrhizal responses to nitrogen, phosphorus and atmospheric CO2 in field studies. New Phytol. 164, 347–355 (2004).
Nasto, M. K. et al. Interactions among nitrogen fixation and soil phosphorus acquisition strategies in lowland tropical rain forests. Ecol. Lett. 17, 1282–1289 (2014).
Loladze, I. Hidden shift of the ionome of plants exposed to elevated CO2 depletes minerals at the base of human nutrition. eLife 3, e02245 (2014).
Davey, P. A., Parsons, A. J., Atkinson, L., Wadge, K. & Long, S. P. Does photosynthetic acclimation to elevated CO2 increase photosynthetic nitrogen-use efficiency ? A study of three native UK grassland species in open-top chambers. Funct. Ecol. 13, 21–28 (1999).
Gifford, R. M., Barrett, D. J. & Lutze, J. L. The effects of elevated [CO2] on the C:N and C:P mass ratios of plant tissues. Plant Soil 224, 1–14 (2000).
Conroy, J. P., Milham, P. J. & Barlow, E. W. R. Effect of nitrogen and phosphorus availability on the growth response of Eucalyptus-grandis to high CO2 . Plant Cell Environ. 15, 843–847 (1992).
Lenka, N. K. & Lal, R. Soil-related constraints to the carbon dioxide fertilization effect. Crit. Rev. Plant Sci. 31, 342–357 (2012).
Sterner, R. W. & Elser, J. J. Ecological stoichiometry: the biology of elements from molecules to the biosphere. (Princeton University Press:, Princeton,, 2002).
Elser, J. J., Fagan, W. F., Kerkhoff, A. J., Swenson, N. G. & Enquist, B. J. Biological stoichiometry of plant production: metabolism, scaling and ecological response to global change. New Phytol. 186, 593–608 (2010).
Lewis, J. D., Griffin, K. L., Thomas, R. B. & Strain, B. R. Phosphorus supply affects the photosynthetic capacity of loblolly pine grown in elevated carbon dioxide. Tree Physiol. 14, 1229–1244 (1993).
Tissue, D. T. & Lewis, J. D. Photosynthetic responses of cottonwood seedlings grown in glacial through future atmospheric [CO2] vary with phosphorus supply. Tree Physiol. 30, 1361–1372 (2010).
Stitt, M. & Krapp, A. The interaction between elevated carbon dioxide and nitrogen nutrition: the physiological and molecular background. Plant Cell Environ. 22, 583–621 (1999).
Nowak, R. S., Ellsworth, D. S. & Smith, S. D. Functional responses of plants to elevated atmospheric CO2 – do photosynthetic and productivity data from FACE experiments support early predictions ? New Phytol. 162, 253–280 (2004).
Niu, Y. F. et al. Effect of elevated CO2 on phosphorus nutrition of phosphate-deficient Arabidopsis thaliana (L.) Heynh under different nitrogen forms. J. Exp. Bot. 64, 355–367 (2013).
Bloom, A. J., Meyerhoff, P. A., Taylor, A. R. & Rost, T. L. Root development and absorption of ammonium and nitrate from the rhizosphere. J. Plant Growth Regul. 21, 416–431 (2003).
Jin, J., Tang, C. X., Hogarth, T. W., Armstrong, R. & Sale, P. Nitrogen form but not elevated CO2 alters plant phosphorus acquisition from sparingly soluble phosphorus sources. Plant Soil 374, 109–119 (2014).
Fritz, C. et al. Nutrient additions in pristine Patagonian Sphagnum bog vegetation: can phosphorus addition alleviate (the effects of) increased nitrogen loads. Plant Biol. 14, 491–499 (2012).
Jin, J., Tang, C. X., Armstrong, R. & Sale, P. Phosphorus supply enhances the response of legumes to elevated CO2 (FACE) in a phosphorus-deficient vertisol. Plant Soil 358, 86–99 (2012).
Fleisher, D. H., Wang, Q., Timlin, D. J., Chu, J. A. & Reddy, V. R. Effects of carbon dioxide and phosphorus supply on potato dry matter allocation and canopy morphology. J. Plant Nutr. 36, 566–586 (2013).
Singh, S. K., Badgujar, G. B., Reddy, V. R., Fleisher, D. H. & Timlin, D. J. Effects of phosphorus nutrition on growth and physiology of cotton under ambient and elevated carbon dioxide. J. Agron. Crop Sci. 199, 436–448 (2013).
Tummers, B., DataThief III. (2006) Available at: http://datathief.org/. (Accessed: 20th September 2012)
Hedges, L. V., Gurevitch, J. & Curtis, P. S. The meta-analysis of response ratios in experimental ecology. Ecology 80, 1150–1156 (1999).
Rosenberg, M. S., Adams, D. C. & Gurevith, J. MetaWin: statistical software for meta-analysis. Version 2.0. (Sinauer Associates, Sunderland, Massachusetts, 2000).
R. Core Team . R: A language and environment for statistical computing. (R Foundation for Statistical Computing, Vienna, Austria, 2014).
Acknowledgements
Much of this work took place during Dr. Huang’s six-month visit to BZH at UC Davis. We thank Dr. Richard J. Norby and Dr. Melanie A. Mayes for their helpful comments on this manuscript. This study was jointly funded by the National Natural Science Foundation of China (Grant Numbers 31400382 and 41430529). Funds to BZH from the Andrew W. Mellon Foundation and the United States National Science Foundation (DEB-1150246) also supported this research.
Author information
Authors and Affiliations
Contributions
B.Z.H. and G.Z. conceived the study. W.H. and A.R.M. collected the data and conducted statistical analysis. W.H. and J.L. prepared the figures. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Huang, W., Houlton, B., Marklein, A. et al. Plant stoichiometric responses to elevated CO2 vary with nitrogen and phosphorus inputs: Evidence from a global-scale meta-analysis. Sci Rep 5, 18225 (2016). https://doi.org/10.1038/srep18225
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep18225
This article is cited by
-
N addition alters growth, non-structural carbohydrates, and C:N:P stoichiometry of Reaumuria soongorica seedlings in Northwest China
Scientific Reports (2022)
-
Alteration in biochemical constituents and nutrients partitioning of Asparagus racemosus in response to elevated atmospheric CO2 concentration
Environmental Science and Pollution Research (2022)
-
Long-term nitrogen addition alters nutrient foraging strategies of Populus davidiana and Betula platyphylla in a temperate natural secondary forest
European Journal of Forest Research (2022)
-
Effects of elevated CO2 on resistant and susceptible rice cultivar and its primary host, brown planthopper (BPH), Nilaparvata lugens (Stål)
Scientific Reports (2021)
-
Biodiversity conservation in climate change driven transient communities
Biodiversity and Conservation (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.