Abstract
The aim of this study was to determine the clinical differences between early and conventional loading protocols for dental implants. A comprehensive search of the Medline, Embase and OVID databases for studies published through January 10, 2015 was conducted. Fourteen studies were included in our analysis. We found that early loading imposed a significantly higher risk of implant failure than did conventional loading (risk ratio = 2.09, 95% confidence interval [CI] [1.18, 3.69], P = 0.01), while no significant differences between the methods were found with regards to the marginal bone loss (weighted mean differences [WMD] = 0.11, 95% CI [−0.07, 0.28], P = 0.23), periotest value (WMD = 0.02, 95% CI [−0.83, 0.87], P = 0.96), or implant stability quotient (WMD = 0.79, 95% CI [−0.03, 1.62], P = 0.06). As for the health status of the peri-implant tissue, conventionally loaded implants demonstrated better performance than did early loaded implants. Subgroup analyses demonstrated that the sample size, time of publication, loading definition, implant position, extent and restoration type influenced the results. Although early implant loading is convenient and comfortable for patients, this method still cannot achieve the same clinical outcomes as the conventional loading method.
Similar content being viewed by others
Introduction
Since Branemark introduced the osseointegration system in 19771, a healing period of at least 3–4 months without loading has been advocated to achieve osseointegration of dental implants2. Any micromotion of the implant may disturb the healing process causing fibrous scar tissue to form that separates the implant and bone rather than promoting bone apposition3,4. When the micromotion reaches a certain threshold, it can eventually lead to failure of the implant5.
However, this nonloading period is usually troublesome and is associated with functional and aesthetic disturbances, especially in completely edentulous patients due to the need to use removable dentures6,7. With the evolution of dentistry over the last few decades, this nonloading period has become shorter. Branemark’s protocols have been reevaluated and modified significantly by the development of non-submerged healing for two-stage implants, immediate or early implant placement after tooth extraction and immediate or early loading. Moreover, with the improvement of implant surface technology, which has shortened the loading waiting period from 12–24 weeks to 6–8 weeks without reducing the success rate, faster and steadier osseointegration can be attained8.
A recent meta-analysis corroborated the findings of previous studies showing that immediate loading is associated with a significantly higher implant failure rate, lower marginal bone loss and a higher implant stability quotient (ISQ) compared to conventional loading9. However, to the best of our knowledge, no study has reported the clinical differences between early and conventional loading. Therefore, the aim of this study was to determine whether or not early loading resulted in different clinical outcomes than conventional loading in terms of the implant failure rate, marginal bone loss, implant stability, peri-implant parameters and complications.
Methods
Focused question
We hoped to answer the following question by developing a protocol: does early implant loading produce different results than conventional loading in terms of the implant failure rate, marginal bone loss, implant stability, health of the peri-implant soft tissue and complications. This question was developed by using the following PICOs definitions:
-
Population: patients who need at least one implant, older than 18 years, having adequate oral hygiene, having adequate alveolar bone volume to place implants and in good general health to permit implant surgery.
-
Intervention: early loading of implants (test group) was defined as implants loaded between 1 week and 2 months after insertion.
-
Comparison: conventional loading of implants (control group) was defined as implants loaded more than 2 months after insertion; also referred to as delayed loading10.
-
Outcomes: (1) the primary outcome was the implant failure rate, defined as the rate of mobility and removal of implants11; and (2) the secondary outcomes were: (a) marginal bone loss (mm): measured as the distance from the implant shoulder to the first osseointegration of each implant by radiographic analysis; (b) implant stability: measured as the ISQ and periotest value (PTV); (c) peri-implant tissue health status, which included the relative attachment level (AL), probing pocket depth (PD), modified bleeding index (BI), modified gingival index (GI) and plaque index (PI); and (d) advent of complications (technical and biological): technical complications were defined as mechanical damage of the implant components including fractures and loosening of the screws or abutments, while biological complications were defined as events that directly concerned the implants, varying from numbness of the lower lip to peri-implant infection.
-
Study design: randomized controlled trials, prospective clinical comparative studies and retrospective clinical comparative studies were included.
Search strategies
An electronic search limited to English publications with abstracts was conducted using the Medline, Embase and OVID databases and included all studies published through January 10, 2015.
The following search terms and strategies were used: (dental implant OR oral implant) AND (early loading OR early restoration OR early prostheses OR early overdenture) AND (conventional loading OR conventional restoration OR conventional prostheses OR conventional overdenture) AND (delayed loading OR delayed restoration OR delayed prostheses OR delayed overdenture).
The reference lists of identified studies and relevant reviews on the same subject were also searched for other potential studies.
Inclusion and exclusion criteria
The inclusion criteria were as follows: (1) human studies, (2) clinical comparative studies, (3) studies comparing the implant failure rates in any group of patients who received early and conventionally loaded implants, (4) studies providing data regarding the relevant clinical outcomes, (5) studies with follow-up periods of at least 6 months after loading and (6) studies published in English.
Furthermore, we used the following exclusion criteria: (1) in vitro or laboratory studies, (2) animal studies, (3) reviews, (4) case reports, (5) craniofacial implant studies and (6) studies only focusing on the different clinical efficacy between implants loaded after less than 1 week (immediately loaded implants) and implants loaded conventionally.
Study selection
Two reviewers (Y. Zhu and Y. Xu) screened the titles and abstracts independently for relevant subjects. Studies that could not be excluded definitively based on the information from the abstracts were searched using the full manuscript to determine whether they should be included. If an agreement could not be reached regarding inclusion/exclusion between the two reviewers, a discussion was held with a third reviewer until an agreement was reached (X. Qu).
Data extraction
Two reviewers (Y. Zhu and Y. Xu) used a specially designed extraction form to extract data independently. Any disagreement about the data extraction was discussed with a third reviewer (X. Qu). The percentage of agreement and Kappa analyses were calculated to evaluate the inter-reviewer reliability of data extraction. The most complete data were extracted when the results of a study were published more than once or presented in different publications. The following information was extracted: first author’s name, country, year of publication, study design, follow-up period, number and age of patients, loading protocol, number and placement position of implants (maxilla or mandibular), extent of restoration (full or single implant), type of restoration (fixed or removable), type of implant system and data on dental implant failure, marginal bone loss, ISQs, PTVs and peri-implant parameters (AL, PD, BI, GI, PI).
Quality assessment
The recommendations by Cochrane12 were used to assess the risk of bias. Studies were classified as having low risk of bias if they met the following criteria: (1) random allocation, (2) clear selection criteria for subjects, (3) blinding of the patients and examiners, (4) detailed reporting of the outcome data, (5) selection of representative population groups and (6) use of identical treatments between two groups except for the intervention. If a study missed one of these criteria, it was considered to have a moderate risk of bias. Missing two or more of these criteria led to a high risk of bias13.
Data analysis
For dichotomous variables (implant failure rates), the risk ratio (RR) and 95% confidence interval [CI] were calculated to compare the selected studies. For continuous variables (marginal bone loss, ISQ, PTV and peri-implant parameters), the weighted mean difference (WMD) and 95% CI were used.
Heterogeneity among studies was tested through Q-tests and I2 statistics (I2 ≤ 25%: low, 25% < I2 < 50%: moderate, I2 ≥ 75%: high)14.
A fixed-effects model was used as a common measure for study-specific estimates, while a random-effects model was considered if significant heterogeneity was found15. The level of statistically significant heterogeneity was set at P < 0.1. In addition, subgroup analyses were conducted whenever possible to identify associations between the risk of implant failure, marginal bone loss and other relevant elements as possible sources of heterogeneity. The subgroup analyses used the following explanatory variables: (1) length of follow-up, (2) sample size (implants), (3) design of study, (4) geographical location, (5) time of publication, (6) definition of early loading, (7) definition of conventional loading, (8) extent of restoration, (9) implant placement position and (10) type of restoration.
Visualization of funnel plots, Begg’s test and Egger’s regression tests were measured to assess publication biases16. Forest plots were drawn to demonstrate the effects in the meta-analyses. Stata version 11 (StataCorp, College Station, TX) and Review Manager 5.2 (The Cochrane Collaboration, Copenhagen, Denmark) were used to conduct the statistical analyses.
Results
Literature Search
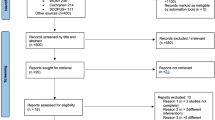
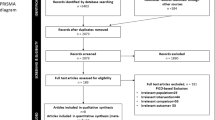
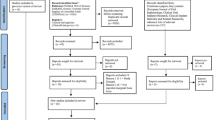
Figure 1 provides a flow chart of the literature search process. The search yielded a total of 1474 primary papers. Of these, 1360 papers were discarded by the two reviewers, leaving 114 papers after the titles and abstracts were evaluated (inter-rater agreement = 86%, kappa = 0.87). An additional 17 papers were included after searching the references of relevant studies, resulting in 137 papers that required full-text evaluation. After the full-text evaluation, 23 papers that reported data from 14 investigations remained. The data from four papers reporting on three investigations were repeated17,18,19,20,21,22,23 and five papers from two investigations reported different results on the same material in different years24,25,26,27,28,29,30. Therefore, nine papers were excluded, leaving 14 papers in the final analyses. One of the included papers was a three-armed study that utilized two experimental groups based on the different definitions of early loading31. Another of the included papers performed two controlled experiments depending on the different types of implant systems32. These two papers were divided into two parts when calculated.
Study characteristics
The detailed study characteristics are presented in Table 1. In the 14 studies, 1619 implants were completely inserted, while 1265 implants remained after years of follow-up (681 implants in the test group, 584 implants in the control group). The number of implants in each study ranged from 2033 to 49020. The earliest study was published in 200034, whereas the latest was published in 201230. The follow-up period of the studies ranged from a minimum of 6 months33 to a maximum of 10 years31. As for the study design, six studies enrolled patients prospectively18,33,34,35,36,37, seven were randomized controlled trials (RCTs)23,26,30,31,32,38,39 and one was a retrospective study20. In terms of the geographic locations, four studies were conducted in Asia23,30,33,37, seven in Europe18,20,31,34,35,36,39 and three in Oceania26,32,38.
The group definitions and prosthetic characteristics are presented in Supplementary Table S1. In both groups, different definitions were used. Ten studies defined loading within 3 weeks as early loading18,20,23,26,30,31,33,34,35,36, while the other five studies placed the prostheses within 6 weeks31,32,37,38,39. One study had two experimental groups: one group had the implants loaded within 3 weeks and the other group had the implants loaded within 6 weeks31. The majority of studies in the conventional group loaded implants 3–4 months after insertion, except one, which loaded the implant after 6 months37.
Two studies inserted implants in the maxilla26,37, while 11 studies inserted implants in the mandible18,20,23,30,31,32,33,34,35,36,38. One study placed implants both in the maxilla and in the mandible39. For completely edentulous patients, overdentures were used in eight studies23,30,31,32,33,35,36,38 and fixed full-arch dental prostheses were used in four studies18,20,26,34. Fixed single implants were used in two studies37,39.
Study Outcomes
Implant failure
In seven of the 14 included studies, no implant failure occurred in the study groups23,30,31,33,34,35,38. In all of the studies except one39, the reported implant failure rate was higher in the early loading group (0–29.2%) than it was in the conventional loading group (0–20%). The results showed that early loading imposed a significantly higher risk of implant failure compared to conventional loading (RR = 2.09, 95% CI [1.18, 3.69], P = 0.01). There was low heterogeneity among the included studies (P = 0.63, I2 = 0%). The results from the meta-analysis are presented in Fig. 2.
Forrest plots of the individual studies for the implant failure rate.
Note that Tawse-Smith (2002) (test 1) and (test 2) indicate that in the same study, two controlled experiments were performed. Ma (2010) (test 1) and (test 2) indicate that in the same study, two different test groups were compared to the same control group.
The subgroup analyses showed a higher risk of implant failure for early loaded implants restored with fixed prostheses (RR = 1.91, 95% CI [1.00, 3.64], P = 0.05) compared to conventionally loaded implants restored with fixed prostheses and for implants in the mandibular region (RR = 2.67, 95% CI [1.36, 5.24], P = 0.004) compared to implants in the maxilla . Patients who were over 50 years old (RR = 2.13, 95% CI [1.08, 4.19], P = 0.03) compared to those who were less than 50 and patients who were completely edentulous (RR = 2.37, 95% CI [1.28, 4.39], P = 0.006) compared to those who were singly edentulous had a higher risk of implant failure in the early loading groups. Similar outcomes occurred in the studies published before 2007 (RR = 2.57, 95% CI [1.11, 5.94], P = 0.03) compared to the studies published after 2007, in the studies that defined early implant loading as implants loaded 6 weeks after placement (RR = 2.60, 95% CI [1.17, 5.79], P = 0.02) compared to implants loaded 1 week or 3 weeks after placement and in the studies that defined conventional implant loading as implants loaded less than 3 months after placement (RR = 2.78, 95% CI [1.07, 7.22], P = 0.04) compared to implants loaded more than 3 months after placement. The results from the subgroup analyses are presented in Table 2.
Marginal bone loss
The marginal bone level in the early loading group ranged from 0.17 to 2.55 mm at baseline, from 0.97 to 2.87 mm at 1 year after loading, from 1.25 to 2.86 mm at 3 years and from 1.61 to 2.98 mm at 5 years after loading; in the conventional loading group, the marginal bone level ranged from 0.11 to 3.5 mm at baseline, from 0.90 to 3.30 mm at 1 year after loading, from 1.26 to 3.5 mm at 3 years and from 1.57 to 3.70 mm at 5 years after loading.
The results of the meta-analysis regarding the overall marginal bone loss are presented in Fig. 3. Eleven studies that compared early and conventionally loaded implants reported marginal bone loss. The results illustrated that no significant difference was found in the overall marginal bone loss between these two loading protocols (WMD = 0.11, 95% CI [−0.07, 0.28], P = 0.23). Seven studies included marginal bone loss at 1 year after loading18,20,23,31,32,36,38. The results illustrated that early loaded implants had more marginal bone loss than did conventionally loaded implants after 1 year of loading (WMD = 0.18, 95% CI [0.00, 0.35], P = 0.05). Five studies included marginal bone loss at 2–3 years after loading30,31,34,36,39. No significant difference was found between the two loading types after 2–3 years of loading (WMD = 0.26, 95% CI [−0.15, 0.68], P = 0.21). Three studies reported the marginal bone loss at over 5 years after loading26,30,31. Again, no significant difference was found between the two groups after over 5 years of loading (WMD = 0.10, 95% CI [−0.10, 0.29], P = 0.33).
Subgroup analyses showed that the early loaded implants in the maxilla had a higher risk of marginal bone loss than did the implants in the mandibular region, while other elements had little significant influence on the marginal bone loss. The results from the subgroup analyses and meta-analysis on cumulative marginal bone loss are presented in Table 3.
Implant stability
Clinical implant stability was measured by two methods: the Ostellt device, which measured the resonance frequency of a small transductor that was attached to the implant abutment and that was stimulated using a wide range of frequencies (ISQ); and the Periotest device, which measured the temporal contact of the tip of the instrument during repetitive percussions on the implant (PTV)40. Two studies were excluded since they only reported medians and ranges35,38. Therefore, PTVs were collected from four studies32,33,34,36. The mean values varied from −4.9 to −1.1 in the early loading group and from −4.52 to −3 in the conventional group. No statistically significant difference was found between the two groups (WMD = 0.02, 95% CI [−0.83, 0.87], P = 0.96).
In the four studies that reported ISQs23,26,30,37, the mean values varied from 66.6 to 81.9 in the test group and from 65 to 81.2 in the control group. Two studies were excluded because they lacked standard deviations38,39. The results showed a greater change in ISQ in the test group compared to in the control group, but this difference was not statistically significant (WMD = 0.79, 95% CI [−0.03, 1.62], P = 0.06). The forest plots of the PTVs and ISQs are presented in Figs 4 and 5 respectively.
Peri-implant tissue health status
The peri-implant parameters were obtained from three studies30,32,38. Five different indexes were recorded to estimate the peri-implant tissue health status: AL, PD, BI, GI and PI. The results showed that conventionally loaded implants had a better peri-implant tissue health status than did the early loaded implants in terms of their AL (WMD = 0.43, 95% CI [0.01, 0.85], P = 0.04) and PD (WMD = 0.26, 95% CI [0.11, 0.45], P = 0.001), while no statistically significant differences were observed between their BI (WMD = 0.12, 95% CI [−0.04, 0.29], P = 0.14), GI (WMD = 0.05, 95% CI [−0.11, 0.22], P = 0.52) and PI (WMD = 0.07, 95% CI [−0.17, 0.31], P = 0.58). However, there was an increase in these outcomes in the test group. The forest plots of these results are presented in Supplementary Figures S1–S5.
Complications
Four studies reported technical complications20,23,26,37. The most common complications were fractures of the prostheses and loosening of the abutment screws. Technical complications occurred more often in the test groups according to the records from the included studies. All of these complications were solved by adjusting the implants or prostheses without affecting the outcomes. One study noted that denture contouring was the most common adjustment. The number of patients needing denture contouring adjustments was higher in the test group(15 people) than it was in the control group (12 people). This could be explained by the secondary gingival healing after surgery in the early loading group, which may result in space around the abutments, while relining impressions performed in the conventional loading group after a period of healing avoided this space23.
Biological complications were reported in three studies20,26,33. The most common complication was peri-implant inflammation, which eventually returned to normal.
Two studies reported no complications in either group32,34. None of the remaining studies mentioned any complications.
Quality assessments
The results of the quality assessments of the included studies are presented in Supplementary Table S2. Of the 14 studies, one was considered to have a low risk of bias39, while three had a moderate risk of bias31,32,38. The remainder of the studies had a high risk of bias.
Publication Bias
Publication bias was determined by Begg’s test, Egger’s regression test and funnel plot visualization. With the exception of marginal bone loss (Begg’s test [B]: P = 0.093; Egger’s test [E]: P = 0.007), no evidence of publication bias was found for the failure rate (B: 0.453; E: 0.411), PTV (B: 1.00; E: 0.779), ISQ (B: 0.174; E: 0.087), PD (B: 0.117; E: 0.115), AL (B: 0.317), PI (B: 0.117; E: 0.042), GI (B: 0.317), or BI (B: 0.317). Given the limited number of included studies, only the publication bias of the implant failure rate was determined by funnel plot visualization. The funnel plot is presented in Supplementary Figure S6.
Discussion
Immediate loading, early loading and conventional loading are the three main loading protocols in implantology. A recent meta-analysis has already comprehensively compared immediately loaded implants to conventionally loaded implants9. The results from that study demonstrated that immediate loading may impose a higher risk for implant failure compared to conventional loading. While early loading protocols (implants loaded 1 week to 2 months after insertion) permit a longer healing time than immediate loading protocols (implants loaded less than 1 week after insertion) and the efficacy difference between early loading and conventional loading protocols is uncertain, so the present meta-analysis, which utilized 14 different studies that compared early vs. conventional loading protocols with data from 1619 implants, aimed to establish whether early loading can achieve the same clinical outcomes as conventional loading with shorter time.
The results of this meta-analysis indicate that early loading has a higher risk of failure compared with conventional loading, which is not totally in agreement with the conclusions published recently in similar systematic reviews. The results from two recent meta-analyses demonstrated that the data were insufficient to determine whether or not there was a clinically important difference in implant failure or marginal bone loss between the two groups based on the limited number of available studies10,41. The results from two other meta-analyses did not show any significant differences between early and delayed loading42,43.
These discrepancies can be explained by the relatively small number of available studies together with their small sample sizes, which are not sufficient to establish definitive conclusions. Moreover, studies with a follow-up period of 4–12 months were included in a recent Cochrane systematic review, which was considered as not long enough to assess implant failure10. Therefore, the present meta-analysis included studies with a follow-up period of 6 months to 7 years.
Although recent systematic reviews have also studied the different failure rates between early and conventional loading10,41,42,43, the limitation of these studies was that insufficient data were available to perform comparative analyses of prosthodontic and peri-implant outcomes with reliable clinical results. In the present study, we enlarged the search scale and updated the search time frame. Thus, to our knowledge, the present study is the most comprehensive and newest meta-analysis estimating the clinical difference between early and conventional loading protocols in terms of implant failure, marginal bone loss, ISQ, PTV and peri-implant parameters. The possible influence of the outcomes was also assessed through subgroup analyses involving the length of follow-up, sample size (implants), design of the study, geographical location, time of publication, definition of early loading, definition of conventional loading, extent of restoration, implant placement position and type of restoration. The current meta-analysis also calculated the cumulative marginal bone loss in years, which gave a continuous view of the differences these two loading protocols caused over time, while other recent systematic reviews only presented the results of implant failure and marginal bone loss10,41,42,43.
Despite the strengths above, this meta-analysis still has several limitations. First, it was difficult to completely eliminate the confounding factors inherent in the included studies that may have resulted in a bias to the outcomes20. Second, the I2 estimates of marginal bone loss (I2 = 97%; P < 0.001) and PTV (I2 = 93%, P < 0.001) were evaluated as high. Heterogeneity introduced by methodological differences between the included studies could not be avoided. Therefore, subgroup analyses were conducted on studies of marginal bone loss, which indicated that the selected elements had no influence on the outcomes. The data on the PTV, however, were insufficient for a subgroup analysis. Third, the literature search was limited to studies in English and only covered three electronic databases, which might have resulted in an election bias to the outcomes. Considering the limitations mentioned above, the results of this meta-analysis should be interpreted with caution.
Conclusions
The primary outcome of this study was that early loading had a higher risk of implant failure than did conventional loading. The secondary outcomes revealed that the two loading protocols were not significantly different in terms of the associated marginal bone loss, changes in implant stability and health status of the peri-implant tissues (except for the AL and PD), which indicated that these two loading protocols behaved similarly once osseointegration occurred. Although early implant loading is advantageous with regards to convenience and comfort for patients, this method still cannot achieve the same clinical outcomes as the conventional loading. Careful selection of patients and surgery indications is still required. These findings may provide dentists with additional insights when developing treatment strategies and making prognoses. Meanwhile, based on this review, more high-quality and well-designed RCTs with large sample sizes are needed.
Additional Information
How to cite this article: Zhu, Y. et al. Clinical efficacy of early loading versus conventional loading of dental implants. Sci. Rep. 5, 15995; doi: 10.1038/srep15995 (2015).
References
Branemark, P. I. et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl 16, 1–132 (1977).
Branemark, P. I. et al. Osseointegrated titanium fixtures in the treatment of edentulousness. Biomaterials 4, 25–8 (1983).
Schatzker, J., Horne, J. G. & Sumner-Smith, G. The effect of movement on the holding power of screws in bone. Clin Orthop Relat Res 111, 257–62 (1975).
Roberts, W. E., Smith, R. K., Zilberman, Y., Mozsary, P. G. & Smith, R. S. Osseous adaptation to continuous loading of rigid endosseous implants. Am J Orthod 86, 95–111 (1984).
Gao, S. S., Zhang, Y. R., Zhu, Z. L. & Yu, H. Y. Micromotions and combined damages at the dental implant/bone interface. Int J Oral Sci 4, 182–8 (2012).
Borges Tde, F. et al. Mandibular overdentures with immediate loading: satisfaction and quality of life. Int J Prosthodont 24, 534–9 (2011).
Erkapers, M., Ekstrand, K., Baer, R. A., Toljanic, J. A. & Thor, A. Patient satisfaction following dental implant treatment with immediate loading in the edentulous atrophic maxilla. Int J Oral Maxillofac Implants 26, 356–64 (2011).
Cochran, D. L., Morton, D. & Weber, H. P. Consensus statements and recommended clinical procedures regarding loading protocols for endosseous dental implants. Int J Oral Maxillofac Implants 19 Suppl, 109–13 (2004).
Sanz-Sanchez, I., Sanz-Martin, I., Figuero, E. & Sanz, M. Clinical efficacy of immediate implant loading protocols compared to conventional loading depending on the type of the restoration: a systematic review. Clin Oral Implants Res 26, 964–82 (2015).
Esposito, M., Grusovin, M. G., Maghaireh, H. & Worthington, H. V. Interventions for replacing missing teeth: different times for loading dental implants. Cochrane Database Syst Rev 3, CD003878 (2013).
Capelli, M. et al. A 5-year report from a multicentre randomised clinical trial: immediate non-occlusal versus early loading of dental implants in partially edentulous patients. Eur J Oral Implantol 3, 209–19 (2010).
Higgins, J. P., Thompson, S. G. & Spiegelhalter, D. J. A re-evaluation of random-effects meta-analysis. J R Stat Soc Ser A Stat Soc 172, 137–159 (2009).
Ten Heggeler, J. M., Slot, D. E. & Van der Weijden, G. A. Effect of socket preservation therapies following tooth extraction in non-molar regions in humans: a systematic review. Clin Oral Implants Res 22, 779–88 (2011).
Higgins, J. P., Thompson, S. G., Deeks, J. J. & Altman, D. G. Measuring inconsistency in meta-analyses. BMJ 327, 557–60 (2003).
DerSimonian, R. & Laird, N. Meta-analysis in clinical trials. Control Clin Trials 7, 177–88 (1986).
Egger, M., Davey Smith, G., Schneider, M. & Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–34 (1997).
Engquist, B. et al. Simplified methods of implant treatment in the edentulous lower jaw. Part II: Early loading. Clin Implant Dent Relat Res 6, 90–100 (2004).
Engquist, B. et al. Simplified methods of implant treatment in the edentulous lower jaw: a 3-year follow-up report of a controlled prospective study of one-stage versus two-stage surgery and early loading. Clin Implant Dent Relat Res 7, 95–104 (2005).
Eliasson, A. On the role of number of fixtures, surgical technique and timing of loading. Swed Dent J Suppl, 3–95 (2008).
Eliasson, A., Blomqvist, F., Wennerberg, A. & Johansson, A. A retrospective analysis of early and delayed loading of full-arch mandibular prostheses using three different implant systems: clinical results with up to 5 years of loading. Clin Implant Dent Relat Res 11, 134–48 (2009).
Turkyilmaz, I. Clinical and radiological results of patients treated with two loading protocols for mandibular overdentures on Branemark implants. J Clin Periodontol 33, 233–8 (2006c).
Turkyilmaz, I., Tumer, C., Avci, M., Hersek, N. & Celik-Bagci, E. A short-term clinical trial on selected outcomes for immediately loaded implant-supported mandibular overdentures. Int J Prosthodont 19, 515–9 (2006d).
Turkyilmaz, I. & Tumer, C. Early versus late loading of unsplinted TiUnite surface implants supporting mandibular overdentures: a 2-year report from a prospective study. J Oral Rehabil 34, 773–80 (2007b).
Fischer, K. & Stenberg, T. Early loading of ITI implants supporting a maxillary full-arch prosthesis: 1-year data of a prospective, randomized study. Int J Oral Maxillofac Implants 19, 374–81 (2004).
Fischer, K. & Stenberg, T. Three-year data from a randomizd, controlled study of early loading of single-stage dental implants supporting maxillary full-arch prostheses. Int J Oral Maxillofac Implants 21, 245–52 (2006).
Fischer, K., Stenberg, T., Hedin, M. & Sennerby, L. Five-year results from a randomized, controlled trial on early and delayed loading of implants supporting full-arch prosthesis in the edentulous maxilla. Clin Oral Implants Res 19, 433–41 (2008).
Turkyilmaz, I., Tozum, T. F., Tumer, C. & Ozbek, E. N. A 2-year clinical report of patients treated with two loading protocols for mandibular overdentures: early versus conventional loading. J Periodontol 77, 1998–2004 (2006a).
Turkyilmaz, I., Sennerby, L., Tumer, C., Yenigul, M. & Avci, M. Stability and marginal bone level measurements of unsplinted implants used for mandibular overdentures: a 1-year randomized prospective clinical study comparing early and conventional loading protocols. Clin Oral Implants Res 17, 501–5 (2006b).
Turkyilmaz, I., Tozum, T. F. & Tumer, C. Early versus delayed loading of mandibular implant-supported overdentures: 5-year results. Clin Implant Dent Relat Res 12 Suppl 1, e39–46 (2010).
Turkyilmaz, I., Tozum, T. F., Fuhrmann, D. M. & Tumer, C. Seven-year follow-up results of TiUnite implants supporting mandibular overdentures: early versus delayed loading. Clin Implant Dent Relat Res 14 Suppl 1, e83–90 (2012).
Ma, S., Tawse-Smith, A., Thomson, W. M. & Payne, A. G. Marginal bone loss with mandibular two-implant overdentures using different loading protocols and attachment systems: 10-year outcomes. Int J Prosthodont 23, 321–32 (2010).
Tawse-Smith, A., Payne, A. G., Kumara, R. & Thomson, W. M. Early loading of unsplinted implants supporting mandibular overdentures using a one-stage operative procedure with two different implant systems: a 2-year report. Clin Implant Dent Relat Res 4, 33–42 (2002).
Anil Kumar, S., Sahoo, N., Radhakrishnan, V. & Sandhu, H. S. Clinical evaluation of early loaded and unloaded implants in edentulous mandible. J Maxillofac Oral Surg 11, 21–8 (2012).
Ericsson, I., Randow, K., Nilner, K. & Peterson, A. Early functional loading of Branemark dental implants: 5-year clinical follow-up study. Clin Implant Dent Relat Res 2, 70–7 (2000).
Roynesdal, A. K., Amundrud, B. & Hannaes, H. R. A comparative clinical investigation of 2 early loaded ITI dental implants supporting an overdenture in the mandible. Int J Oral Maxillofac Implants 16, 246–51 (2001).
De Smet, E., Duyck, J., Vander Sloten, J., Jacobs, R. & Naert, I. Timing of loading—immediate, early, or delayed—in the outcome of implants in the edentulous mandible: a prospective clinical trial. Int J Oral Maxillofac Implants 22, 580–94 (2007).
Turkyilmaz, I., Avci, M., Kuran, S. & Ozbek, E. N. A 4-year prospective clinical and radiological study of maxillary dental implants supporting single-tooth crowns using early and delayed loading protocols. Clin Implant Dent Relat Res 9, 222–7 (2007a).
Payne, A. G., Tawse-Smith, A., Duncan, W. D. & Kumara, R. Conventional and early loading of unsplinted ITI implants supporting mandibular overdentures. Clin Oral Implants Res 13, 603–9 (2002).
Barewal, R. M., Stanford, C. & Weesner, T. C. A randomized controlled clinical trial comparing the effects of three loading protocols on dental implant stability. Int J Oral Maxillofac Implants 27, 945–56 (2012).
Meredith, N., Book, K., Friberg, B., Jemt, T. & Sennerby, L. Resonance frequency measurements of implant stability in vivo. A cross-sectional and longitudinal study of resonance frequency measurements on implants in the edentulous and partially dentate maxilla. Clin Oral Implants Res 8, 226–33 (1997).
Stafford, G. L. Different loading times for dental implants - no clinically important differences? Evid Based Dent 14, 109–10 (2013).
Alsabeeha, N., Atieh, M. & Payne, A. G. Loading protocols for mandibular implant overdentures: a systematic review with meta-analysis. Clin Implant Dent Relat Res 12 Suppl 1, e28–38 (2010).
Su, M. et al. Comparison of implant success rates with different loading protocols: a meta-analysis. Int J Oral Maxillofac Implants 29, 344–52 (2014).
Acknowledgements
We would like to thank Editage (http://www.editage.cn) for English language editing.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Y.Z., X.Q., E.L. and M.Z. Performed the experiments: Y.Z., X.Z., G.Z. and Y.X. Analyzed the data: Y.Z., X.Z. and G.Z. Contributed materials/analysis tools: Y.Z. and X.Q. Wrote the paper: Y.Z., X.Q., E.L. and M.Z. All authors reviewed the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhu, Y., Zheng, X., Zeng, G. et al. Clinical efficacy of early loading versus conventional loading of dental implants. Sci Rep 5, 15995 (2015). https://doi.org/10.1038/srep15995
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep15995
This article is cited by
-
Immediate and early loading of hydrothermally treated, hydroxyapatite-coated dental implants: a 7-year prospective randomized clinical study
International Journal of Implant Dentistry (2021)
-
Accentuated osseointegration in osteogenic nanofibrous coated titanium implants
Scientific Reports (2019)
-
A 5-year prospective clinical trial on short implants (6 mm) for single tooth replacement in the posterior maxilla: immediate versus delayed loading
Odontology (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.