Abstract
Previous studies have suggested that Klotho provides reno-protection against unilateral ureteral obstruction (UUO)-induced renal tubulointerstitial fibrosis (RTF). Because the existing studies are mainly performed using heterozygous Klotho mutant (HT) mice, we focused on the effect of UUO on homozygous Klotho mutant (kl/kl) mice. UUO kidneys from HT mice showed a significantly higher level of RTF and TGF-β/Smad3 signaling than wild-type (WT) mice, whereas both were greatly suppressed in kl/kl mice. Primary proximal tubular epithelial culture cells isolated from kl/kl mice showed no suppression in TGF-β1-induced epithelial mesenchymal transition (EMT) compared to those from HT mice. In the renal epithelial cell line NRK52E, a large amount of inorganic phosphate (Pi), FGF23, or calcitriol was added to the medium to mimic the in vivo homeostasis of kl/kl mice. Neither Pi nor FGF23 antagonized TGF-β1-induced EMT. In contrast, calcitriol ameliorated TGF-β1-induced EMT in a dose dependent manner. A vitamin D3-deficient diet normalized the serum 1,25 (OH)2 vitamin D3 level in kl/kl mice and enhanced UUO-induced RTF and TGF-β/Smad3 signaling. In conclusion, the alleviation of UUO-induced RTF in kl/kl mice was due to the TGF-β1 signaling suppression caused by an elevated serum 1, 25(OH)2 vitamin D3.
Similar content being viewed by others
Introduction
Klotho was first identified in close association with a premature onset of aging phenotype in mice. Homozygous Klotho mutant mice (kl/kl) exhibit a shortened life span, infertility, ectopic calcification, skin atrophy, pulmonary emphysema and osteoporosis, which resemble an aged state in humans1. Mouse Klotho is predominantly expressed in the kidneys and slightly in the brain2. Two forms of Klotho have been identified: membrane Klotho and secreted Klotho. Membrane Klotho functions as a co-receptor of the FGF23 receptor3,4. FGF23, a member of the fibroblast growth factor (FGF) family, is a bone-derived hormone that exerts its function in the kidneys, to help maintain homeostasis in phosphate and vitamin D metabolism by regulating the sodium phosphate co-transporter and the key enzymes for vitamin D metabolism CYP27B1 and CYP24A15,6,7,8. Similar to FGF23-deficient mice, kl/kl mice also exhibit hyperphosphatemia and an increased serum 1,25 (OH)2 vitamin D3 level1, which are suspected to be the primary cause of premature aging because restricting vitamin D intake improves the kl/kl phenotype8,9. Secreted Klotho is formed via either alternative splicing of the gene or shedding of the extracellular domain into the extracellular space and subsequently into the circulation2,10.
Renal tubulointerstitial fibrosis (RTF) is the final common pathological condition of chronic kidney disease (CKD) regardless of the underlying cause11. RTF is characterized by an excess accumulation of extracellular matrix (ECM), fibroblast activation and a loss of functioning nephrons12. Recent studies have suggested that Klotho plays an important role in RTF. Kidney injury, such as ischemia-reperfusion, peritoneal cisplatin injection, or angiotensin II administration, induce Klotho deficiency, whereas exogenous Klotho protein attenuates kidney injury13,14,15,16,17. In a mouse model of CKD, unilateral ureteral obstruction (UUO)-induced RTF is exaggerated in heterozygous Klotho mutant mice (HT) and is ameliorated in Klotho over-expressing mice. Secreted Klotho has been proven to interfere with TGF-β18 and Wnt/β-catenin19 signaling to suppress UUO-induced RTF.
Kl/kl mice exhibit phenotypes that are very different from wild-type or HT mice. Hyperphosphatemia and high 1, 25(OH)2 vitamin D3 and/or FGF23 are the most noticeable serologic features. ECM deposition is increased in kl/kl mice even without UUO and is thought to be due to Klotho deficiency20. However, an activated form of vitamin D and its analogues have been shown to protect the kidneys from fibrosis due to various kidney injuries21,22. Also, 1, 25(OH)2 vitamin D3 reduces gene expression related to TGF-β-induced fibrosis in human uterine leiomyoma cells23. An active form of the vitamin D3 analogue maxacalcitol recruits a PPM1A/VDR complex to phosphorylated Smad3 to accelerate its dephosphorylation24. Thus, the outcome of UUO-induced RTF becomes uncertain because of the opposing effect of an elevated vitamin D and Klotho deficiency. Because all of the existing studies on the effect of Klotho on UUO-induced RTF were performed using HT mice, whether the disrupted homeostasis in kl/kl mice has an effect is not known.
In this study, we compared the degree of UUO-induced RTF among wild-type, HT and kl/kl mice and analyzed the underlying mechanisms.
Results
UUO-induced RTF is the mildest in kl/kl mice
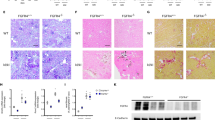
Six week-old wild-type (WT), heterozygous Klotho mutant (HT) and kl/kl mice were subjected to UUO for 3 days or 1 week. The expression of RTF markers collagen I and αSMA was assessed by immunohistochemistry and quantitative real-time PCR. RTF developed after the UUO operation in a time dependent manner in WT and HT mice compared to the sham-operated control groups. The expression of RTF markers was much higher in HT mice than WT mice, which is corroborated by previous reports18,20. In the sham-operated groups, the expression of RTF markers was comparable between the WT and HT mice, but was increased more than 2-fold in the kl/kl mice. Conversely, kl/kl mice showed only a slight increase in the expression of fibrosis markers after UUO (Fig. 1).
Kl/kl kidneys showed the mildest UUO-induced renal tubulointerstitial fibrosis.
(a, b): Representative pictures for immunohistochemistry of kidney sections from WT, HT and kl/kl mice for collagen I (a) and αSMA (b). Scale bar: 100 μm (c, d) Quantification of positive staining areas for collagen I (c) and αSMA (d). (e, f) Real-time PCR for Col1a1 (e) and αSMA (f). Note that UUO-induced collagen I deposition and fibroblast activation were the most severe in HT mice at the protein and mRNA levels and were largely abolished in kl/kl mice. Data are presented as the means ± S.D. *P < 0.05 (n = 5).
TGF-β signaling is suppressed in kl/kl mice
Previous studies showed that Klotho protein interferes with TGF-β1 binding to its receptor, resulting in suppressed TGF-β signaling18. As a result, UUO-induced RTF is exaggerated in HT mice20. To examine whether the suppressed expression of RTF markers in kl/kl mice is a result of inhibited TGF-β signaling, the phosphorylation of Smad3 and TGF-β1 expression was assayed. In parallel with RTF markers, after UUO, phosphorylated Smad3 (pSmad3) and TGF-β1 expression were the highest in HT mice and were significantly suppressed in kl/kl mice. Interestingly, in the sham-operated groups, the kl/kl mice had a higher percentage of pSmad3 positive staining cells than the other two groups, whereas this group had the lowest TGF-β1 content, suggesting that Smad3 phosphorylation in the sham-operated kl/kl mice was not a result of increased TGF-β1 (Fig. 2).
UUO-induced TGF-β1/Smad3 signaling is ameliorated in kl/kl mice.
(a) Immunofluorescence for phosphorylated (p)-Smad3. Scale bar: 100 μm (b) Comparison of the number of pSmad3-positive nuclei among WT, HT and kl/kl kidneys. (c) Western blot for pSmad3 in UUO-treated kidneys. Full-length blots are included in the supplementary information. (d) Comparison by quantification of pSmad3 in western blot among WT, HT and kl/kl kidneys. (e) The amount of TGF-β1 protein in WT, HT and kl/kl kidneys as measured by ELISA. (f) TGF-β1 mRNA level as measured by real-time PCR. Note that UUO-induced TGF-β signaling was enhanced in HT kidneys compared to WT kidneys and was ameliorated in kl/kl kidneys. Data are presented as the means ± SD. *P < 0.05 (n = 5).
No suppression of the TGF-β1-induced epithelial-mesenchymal transition is observed in primary cultured proximal tubular epithelial culture cells isolated from kl/kl mice
Because kl/kl mice have disrupted phosphate and calcium (Pi/Ca) homeostasis, determining whether the suppressed TGF-β/Smad3 signaling is a result of complete depletion of Klotho or disrupted Pi/Ca homeostasis, most noticeably high phosphate, high FGF23 and high 1, 25(OH)2 vitamin D3, is difficult. To address the underlying molecular mechanism, we compared the expression of molecules involved in epithelial-mesenchymal transition (EMT) using primary cultured proximal tubular epithelial cells (PTECs) from WT, HT and kl/kl mice. TGF-β1-induced EMT was enhanced in HT and kl/kl cells compared to WT cells. The EMT markers showed higher αSMA and lower E-cadherin mRNA and protein expression at the indicated time point (Fig. 3). Smad3 phosphorylation was also enhanced in HT and kl/kl cells. Unlike UUO kidneys, kl/kl cells showed no suppression of TGF-β1-induced EMT and Smad3 phosphorylation compared to HT cells. These results suggest that the suppressed TGF-β/Smad3 signaling observed in UUO-induced RTF in kl/kl kidneys was a result of disrupted Pi/Ca homeostasis and not complete Klotho depletion (Fig. 3).
TGF-β1-induced EMT is enhanced in proximal tubular cells isolated from HT and kl/kl kidneys compared to WT kidneys.
(a) Representative western blots for pSmad3, Smad3, E-cadherin (E-cad), αSMA and β-actin. Full-length blots are included in the supplementary information. (b–d) Comparison by quantification of western blots for pSmad3 (b), E-cadherin (c) and αSMA (d). (e–g) Real-time PCR for E-cadherin (e), Col1a1 (f) and αSMA (g). Note that unlike in vivo, no difference in EMT markers was detected between HT and kl/kl cultured proximal tubular cells. Data are presented as the means ± SD. *P < 0.05 compared to the HT group at the same time point. Experiments were repeated in triplicate.
TGF-β1-induced EMT is suppressed not by high phosphate or FGF23 but by 1,25(OH)2 vitamin D3 in a dose-dependent manner
Previous studies showed that in kl/kl mice, serum phosphate and 1,25(OH)2 vitamin D3 increase sharply1 and serum FGF23 concentration increased 2,000-fold compared to WT mice4. To identify an individual factor(s) that contribute to suppressed UUO-induced RTF expression in kl/kl mice, we used the normal kidney epithelial cell line NRK52E and these cells were cultured in the presence of high phosphate (2 or 4 mM), FGF23 (0.2 or 2 μg/ml), or 1,25(OH)2 vitamin D3 (0.1 or 1 μg/ml) to evaluate their effect on TGF-β1-induced EMT. In the cells treated with TGF-β1 together with phosphate or FGF23 for 12 or 24 h, TGF-β1-induced EMT was not affected at the mRNA or protein level compared to cells treated with TGF-β1 alone. In contrast, 1,25(OH)2 vitamin D3 ameliorated TGF-β1-induced Smad3 phosphorylation, αSMA expression and E-cadherin suppression in a dose dependent manner at the mRNA and protein levels. This suppressive effect of 1,25(OH)2 vitamin D3 on TGF-β-induced EMT lasted for 72 h after TGF-β treatment (supplementary Fig. 1).
Interestingly, the cells cultured with high phosphate (4 mM) for 48 h had more Smad3 phosphorylation and higher αSMA expression compared to cells cultured in normal DMEM, although TGF-β1 was absent. These results may explain the increased fibrosis markers observed in sham-operated kl/kl mice (Figs. 4 and 5).
TGF-β1-induced EMT in NRK52E cells is not influenced by a high concentration of phosphate.
(a) Representative western blots for pSmad3, Smad3, E-cadherin (E-cad), αSMA and β-actin. Full-length blots are included in the supplementary information. (b–d) Comparison by quantification of western blots for pSmad3 (b), E-cadherin (c) and αSMA (d). (e-g) Real-time PCR for E-cadherin (e), Col1a1 (f) and αSMA (g). A high concentration of phosphate does not enhance TGF-β1-induced EMT. Note that basal Smad3 phosphorylation and αSMA protein expression are enhanced in the presence of a high phosphate level. Data are presented as the means ± SD. *P < 0.05. Experiments were repeated in triplicate.
TGF-β1-induced EMT in NRK52E cells is suppressed by calcitriol.
(a) Representative western blots for pSmad3, Smad3, E-cadherin, αSMA and β-actin. Full-length blots are included in the supplementary information. (b–d) Comparison by quantification of western blots for pSmad3 (b), E-cadherin (c) and αSMA (d). (e–j) Real-time PCR results for E-cadherin (e, h), Col1a1 (f, i) and αSMA (g, j) in the presence of either FGF23 (e, f, g) or calcitriol (h, i, j). FGF23 have no effect on TGF-β1-induced EMT at neither low nor high concentration. Note that calcitriol ameliorates TGF-β1 induced EMT in a dose-dependent manner. Data are presented as the means ± SD. *P < 0.05 versus cells treated with TGF-β1 only. Experiments were repeated in triplicate.
Vitamin D-free diet normalizes serum 1,25(OH)2 vitamin D3 level and enhances the expression of UUO-induced RTF markers in kl/kl mice
A vitamin D-free diet (D-diet) has been reported to maintain phosphate homeostasis and improve the kl/kl phenotype8,9. In this study, we fed kl/kl mice on a D-diet to determine whether the decreased vitamin D level could enhance UUO-induced RTF. In parallel with previous studies, the D-diet normalized serum 1,25(OH)2 vitamin D3 and increased the body weight of the kl/kl mice, such that they were indistinguishable from their WT littermates (Fig. 6).
After the UUO operation, based on immunohistochemistry, expression of collagen I and αSMA was enhanced by the D-diet, with 3- and 4-fold increases, respectively, compared to kl/kl mice fed a standard diet and mRNA expression showed a similar trend (Fig. 7).
A vitamin D3-free diet enhances UUO-induced RTF in kl/kl mice to the same level as HT mice.
(a) Representative immunohistochemistry of kidney sections for collagen I (a–d) and αSMA (e–h) (b, c) Comparison by quantifying the positive staining area for collagen I (b) and αSMA (c). (d, e) Real-time PCR for Col1a1 (d) and αSMA (e). A vitamin D3 free diet significantly enhances UUO-induced protein and mRNA expression of collagen I and αSMA in kl/kl mice. Data are presented as the means ± SD. *P < 0.05, #p < 0.05 vs. sham-treated kidneys, &p < 0.05 vs. D-diet kl/kl sham-treated kidneys (n = 5).
In addition, the D-diet enhanced TGF-β/Smad3 signaling in UUO-induced RTF in kl/kl mice. The percentage of pSmad3-positive cells increased about 2.5-fold as a result of vitamin D withdrawal. ELISA was performed to measure the TGF-β1 content in kidney lysates. Protein and mRNA expression of UUO-induced TGF-β1 increased about 2-fold using the D-diet. Interestingly, in the sham-operated kl/kl mice fed the standard diet, TGF-β1 protein expression was lower than the kl/kl mice fed the D-diet, while there were more pSmad3 positive cells in kl/kl mice fed the standard diet (Fig. 8).
TGF-β1 signaling in kl/kl mice is enhanced by a vitamin D3 free diet.
(a) Representative immunofluorescence for phosphorylated (p)-Smad3. (b) Western blot for pSmad3 in UUO- or sham-treated kidneys. Full-length blots are included in the supplementary information. (c) Comparison of the number of pSmad3-positive nuclei. (d) Comparison of pSmad3 to total Smad3 ratio in western blots. (e) TGF-β1 protein expression in the kidneys as measured by ELISA. (f) TGF-β1 mRNA expression as measured by real-time PCR. Amelioration of UUO-induced activation of TGF-β1 signaling in kl/kl mice was abolished by a vitamin D3 free diet. pSmad3 positive nuclei and TGF-β1 expression were increased at the protein and mRNA levels. Data are presented as the means ± SD. *P < 0.05 (n = 5), #p < 0.05 vs. sham treated kidneys. &P < 0.05 vs. D-diet kl/kl mice.
Note that in sham-operated kl/kl mice fed a standard diet, the expression of RTF markers and activation of TGF-β/Smad3 signaling are slightly but significantly higher than kl/kl mice fed a D-diet. The results suggest that increased RTF marker expression in sham-operated kl/kl mice was not a result of Klotho depletion but the disrupted Pi/Ca homeostasis (Figs. 7 and 8).
Discussion
In this study, we demonstrated that UUO-induced RTF was greatly suppressed in kl/kl mice compared to HT mice. Conversely, slightly but significantly more severe renal fibrosis was observed in sham-operated kl/kl kidneys than WT and HT kidneys. Because kl/kl mice exhibit a variety of abnormalities, among which phosphate retention and high serum 1, 25(OH)2 vitamin D3 are the most prominent changes, we hypothesized that disrupted Pi/Ca homeostasis in kl/kl mice may play an important role in the mild renal fibrosis in sham-operated mice and the suppression of RTF in UUO-operated mice.
We observed that HT mice exhibit more severe UUO-induced RTF compared to the WT group, which is consistent with previous reports20. Klotho has been proven to protect the kidneys from injury-induced fibrosis by counteracting TGF-β/Smad3 and Wnt signaling18,19. Klotho deficiency renders the kidneys more susceptible to acute insults, while exogenous Klotho expression attenuates renal fibrosis due to various causes13,14,18,20,25. In rodent models, renal damage caused by several different kinds of stress, such as acute inflammatory stress and sustained circulatory stress, could suppress Klotho expression26,27. In the human kidneys, decreased Klotho expression occurs as early as CKD stage 228. However, unexpectedly, UUO-induced RTF in kl/kl mice was greatly attenuated. Because TGF-β/Smad3 signaling is believed to be the main driving force toward post-UUO RTF29,we examined TGF-β/Smad3 signaling, which turned out to be significantly suppressed in kl/kl kidneys. To identify the cause of this suppression, we examined TGF-β-induced EMT using primary cultured PTECs to exclude the humoral effects. Interestingly, unlike in vivo experiments, TGF-β1-induced EMT was not suppressed in cultured kl/kl PTECs and there was an increase in Smad3 phosphorylation. These results suggest that the suppression of UUO-induced RTF in kl/kl mice might result from a disturbance in Pi/Ca homeostasis due to deficiency of Klotho. We have not investigated ERK/MAPK activation in cultured kl/kl PTECs because the increase in Smad3 phosphorylation was significant, but it might be important to examine non-canonical TGF-β signaling, which also plays an important role in EMT.
Next, because phosphate retention, high serum FGF23 and high serum 1, 25(OH)2 vitamin D3 are the most prominent changes in kl/kl mice30, we examined these factors individually using in vitro cultures. NRK52E cells were challenged with TGF-β1 in the presence of high phosphate, FGF23, or calcitriol (1, 25(OH)2 vitamin D3). TGF-β1-induced EMT was not affected by the presence of high phosphate or FGF23, but was inhibited by calcitriol in a dose dependent manner. These results suggest that a high level of 1, 25(OH)2 vitamin D3 but not phosphate or FGF23 may be the cause of the suppressed UUO-induced RTF in kl/kl mice.
To confirm this mechanism, we sought to eliminate the effect of vitamin D in kl/kl mice by feeding a D-diet and determining the degree of UUO-induced RTF. The D-diet normalized 1, 25(OH)2 vitamin D3 and phosphate levels in the blood of kl/kl mice and completely rescued the kl/kl phenotype, which is in agreement with previous reports8. As we predicted, the D-diet enhanced UUO-induced RTF in kl/kl mice to the same levels as HT mice. In addition, TGF-β signaling was enhanced by eliminating vitamin D. Paricalcitol, a synthetic vitamin D analogue, has been reported to attenuate renal interstitial fibrosis in obstructive nephropathy via repressing expression of TGF-β1 and its type I receptor31. Moreover, vitamin D analogs are reported to ameliorate proteinuria and kidney injury by blocking Wnt/β-catenin signaling32. We therefore conclude that an elevated level of 1, 25(OH)2 vitamin D3 in kl/kl mice suppresses TGF-β/Smad3 signaling, resulting in amelioration of UUO-induced RTF.
In sham-operated mice, there was no significant difference in the expression of RTF markers between WT and HT kidneys, whereas expression was significantly albeit mildly increased in kl/kl kidneys compared to WT and HT kidneys. Sugiura, H et al20 suggested that this result may be due to failed TGF-β/Smad3 inhibition related to the absence of Klotho. However, our results did not support this hypothesis because the kl/kl PTECs expressed the same levels of EMT markers as WT and HT PTECs treated with TGF-β1. In addition, kl/kl mice fed a D-diet did not have any RTF onset in the sham-operated group. Interestingly, NRK52E cells cultured for 48 h in the medium containing a high concentration of phosphate (4 mM) showed Smad3 phosphorylation and increased αSMA expression compared to the control medium (Pi 1 mM), suggesting that an increased expression of RTF markers in sham-operated kl/kl mice may be a result of a disrupted Pi/Ca homeostasis, that is, a high concentration of phosphate and not a result of failed TGF-β/Smad3 inhibition by Klotho. However, at this time, we cannot explain how a high concentration of phosphate phosphorylates Smad3.
In conclusion, we are the first to report that UUO-induced TGF-β/Smad3 signaling is suppressed in kl/kl mice by an increased level of 1, 25(OH)2 vitamin D3, resulting in amelioration of UUO-induced RTF. We identified 1, 25(OH)2 vitamin D3 to be a powerful suppressor of TGF-β/Smad3 signaling and protect the kidneys from RTF.
Methods
Animals, diets, UUO animal models and tissue preparation
All experimental protocols were approved by the Animal Studies Committee of Wakayama Medical University. The methods were carried out in accordance with the approved guideline. This study used klotho/Jcl mice, which were purchased from CLEA Japan and described elsewhere1. Mice were raised in a standard facility with free access to food and water.
Animals were fed a standard diet or vitamin D3 free diet (D-diet). In the D-diet groups, heterozygous klotho female mice were maintained on a vitamin D3 free diet (PMI nutrition international, Inc.) after pregnancy, in which the composition was identical to the standard diet except for missing vitamin D3. The offspring of D-diet heterozygous klotho females were also fed a D-diet throughout the experiments.
Six- to eight-week-old male wild-type (WT), heterozygous (HT) and kl/kl mice from the standard diet and D-diet groups were used to perform UUO. UUO surgery was performed as described elsewhere33,34. In short, male mice from each group (n = 5) were anesthetized by an intraperitoneal injection of ketamine (100 mg/kg body weight) and xylazine (5 mg/kg body weight). A left-flank incision was made to expose the left ureter and then ligation was done with a 5-0 silk suture. The sham groups (n = 3) received the same procedure except for ureteral ligation.
At the time of sacrifice, the ligated kidneys (or sham kidneys) were removed and cut transversely. Tissues were fixed with 4% paraformaldehyde for histopathological examination or lysed by denaturing lysis buffer (50 mM Tris-HCl, pH 7.4, 1% triton X, 150 mM NaCl, 1 mM EDTA) with addition of a phosphatase inhibitor (2.5 mM sodium orthovanadate, 10 mM NaF and 10 mM β-glycerol phosphate) and proteinase inhibitor (Roche). Lysed samples were stored in −80°C until use for western blot analysis or ELISA.
Immunohistochemistry and immunofluorescent staining
Immunohistochemistry was performed on paraffin sections using a microwave-based antigen retrieval technique. The antibodies used in this study include collagen I (Southern Biotechnology, Birmingham, AL), pSmad3 (Cell Signaling, Danvers, MA), αSMA (Sigma) and Fsp1 (Abcam). After incubation with the primary antibodies, sections were treated with a Vectastain ABC kit (Vector Laboratories, Birlingame, CA) according to the manufacturer's instructions. For immunofluorescent staining, a Cy3-anti-rabbit IgG (Sigma Aldrich) was used as a secondary antibody.
Quantitative real-time polymerase chain reaction (qRT-PCR)
SYBR Green PCR Master Mix (Takara Bio, Inc., Japan) was used to analyze mRNAs. Each sample was measured in duplicate and gene expression levels were normalized to glyceraldehyde 3-phosphate dehydrogenase (Gapdh). Sequences of the primers used for qRT-PCR are as follows: mouse-Col1a1 forward 5′-CAA CCT GGA CGC CAT CAA G-3′ and reverse 5′-CAG ACG GCT GAG TAG GGA ACA-3′; rat-Col1a1 forward 5′-AGT GCA TTC AAC CTT ACC AA-3′ and reverse 5′-TCA AGC AAG AGG ACC AAG C-3′; mouse and rat-Fsp1 forward 5′-GAA GTG AAG ACT CCT CAG ATG-3′ and reverse 5′-CAC TAT GCT CAC AGC CAA C-3′; mouse-TGF-β1 forward 5′-AAC TAT TGC TTC AGC TCC AGA GAG A-3′ and reverse 5′-AGT TGG ATG GTA GCC CTT G-3′; rat-TGF-β1 forward 5′-TGG AGC AAC ACG TAG AAC TCT-3′ and reverse 5′-AGT TGG ATG GTA GCC CTT G-3′; mouse-αSMA forward 5′-AGA TCA CAG CCC TCG CA-3′ and reverse 5′-AGA GTA CTT GCG TTC TGG AG-3′; rat-αSMA forward 5′-AGA TCA CAG CCC TCG CT-3′ and reverse 5′-AGA ATA TTT GCG TTC TGG AG-3′; rat-αSMA forward 5′- ATG ATG AAG AGG GAG GTG G-3′ and reverse 5′- CAT CAT TTC GAA TCA CTT CC-3′; mouse-Gapdh forward 5′-CCC ATG TTT GTG ATG GGT GT-3′ and reverse 5′-GTG ATG GCA TGG ACT GTG GT-3′35.
Western Blot Analysis
Tissue or cell lysate (20 μg) was separated by SDS-PAGE and blotted on polyvinylidene difluoride membranes (Millipore, Billerica, MA). The primary antibodies were rabbit anti-pSmad3 (Acris Antibodies, Herford, Germany), rat anti-E-cadherin (Sigma Aldrich), mouse anti-αSMA (Sigma Aldrich), rabbit anti-Smad3 (Abcam), mouse anti-β-actin (Sigma Aldrich) and goat anti-GAPDH(Santa Cruz). The signals were developed with the ECL Plus detection system (Amersham Bioscience, Buckinghamshire, United Kingdom).
TGF-β1 ELISA assay
Kidney cortices were dissected and homogenized in denaturing lysis buffer containing 100 mM NaCl, 1% Triton X-100 and 0.05 M Tris-HCl, pH7.4. Protein concentrations were measured using a BCA method according to the manufacturer's instruction. Total TGF-β1 was quantified using a TGF-β1 ELISA kit (Invitrogen). Values were expressed as pg/mg protein for the protein extracts.
Measurement of active serum 1, 25(OH)2 vitamin D3 in mice
Six-week-old male mice were anesthetized with ketamine and xylazine; blood was collected from the inferior vena cava and the serum was separated by centrifugation at 7,500 g for 10 min at 4°C (n = 4). The serum level of 1, 25(OH)2 vitamin D3 was measured by RIA (SRL Inc., Tokyo, Japan).
Cell culture
Primary proximal tubular epithelial cells were generated from the kidneys of WT, HT and kl/kl mice using a method described previously33 with minor modification. Briefly, kidney cortices (6 wk male mice) were dissected, sliced, minced and digested in 300 U/ml type II collagenase (Worthington Bio Corp) in FBS free DMEM in a shaking incubator at 37°C for 30 min. Collagenase was neutralized with 10% FBS/DMEM containing 100 U/ml penicillin and 0.1 mg/ml streptomycin. The suspension was triturated by pipetting and was passed through a 70 μm cell strainer (Becton Dickinson Labware, Franklin Lakes, NJ). The samples were centrifuged (500 rpm, 5 minutes) to pellet the tubules, washed with 10 ml FBS free-DMEM, centrifuged and washed twice more. The final pellet, consisting mostly of renal tubules, was resuspended in culture medium (REBM bullet kit, Clonetics) and incubated at 37°C in a 5% CO2 incubator with medium changes every 2 days until approximately 80% confluent.
NRK52E cells were obtained from ATCC and cultured in DMEM with 5% FBS (Gibco) without antibiotic supplementation.
For TGF-β1 stimulation, cells underwent 24 h serum starvation and then were exposed to 5 ng/ml TGF-β1 (Cell signaling) for 12 or 24 h in the presence or absence of 0.2 or 2 μg/ml murine FGF23 (Peprotech) or 0.1 or1 μg/ml Calcitriol (Cayman).
To make high phosphate growth medium, an equal volume of 100 mM NaH2PO4 and 100 mM Na2HPO4 were mixed to make 100 mM Pi stocks; 2 mM and 4 mM Pi medium were made by dilution of Pi stocks with DMEM (Pi concentration in DMEM is approximately 1 mM). NRK52E cells were grown in normal DMEM with 5% FBS or high Pi DMEM with 5% FBS until 80% confluent. TGF-β1 stimulation was performed with serum free normal DMEM or high Pi DMEM.
Quantitative Analysis
To quantify activated Smad3, the positive stained nuclei were counted in 20 consecutive high power fields (400×) and divided by the total number of nuclei. The number of collagen and αSMA positive cells in the cortical interstitium was performed as described36. Band intensity in the western blots was measured using Image J software.
Statistical Analysis
The data were analyzed with Student's t-test and one-way ANOVA with a Student–Newman–Keuls test (SPSS, 13.0) and expressed as means ± SD. For the data without Gaussian distribution, Kruskal-Wallis with post-hoc Mann-Whitney was used. P < 0.05 was considered to be statistically significant.
References
Kuro-o, M. et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390, 45–51 (1997).
Shiraki-Iida, T. et al. Structure of the mouse klotho gene and its two transcripts encoding membrane and secreted protein. FEBS Lett. 424, 6–10 (1998).
Kurosu, H. et al. Regulation of fibroblast growth factor-23 signaling by klotho. J. Biol. Chem. 281, 6120–6123 (2006).
Urakawa, I. et al. Klotho converts canonical FGF receptor into a specific receptor for FGF23. Nature 444, 770–774 (2006).
Riminucci, M. et al. FGF-23 in fibrous dysplasia of bone and its relationship to renal phosphate wasting. J. Clin. Invest. 112, 683–692 (2003).
Murer, H., Hernando, N., Forster, I. & Biber, J. Proximal tubular phosphate reabsorption: molecular mechanisms. Physiol. Rev. 80, 1373–1409 (2000).
Wikvall, K. Cytochrome P450 enzymes in the bioactivation of vitamin D to its hormonal form (review). Int. J. Mol. Med. 7, 201–209 (2001).
Tsujikawa, H. Klotho, a Gene Related to a Syndrome Resembling Human Premature Aging, Functions in a Negative Regulatory Circuit of Vitamin D Endocrine System. Mol. Endocrinol. 17, 2393–2403 (2003).
Anour, R., Andrukhova, O., Ritter, E., Zeitz, U. & Erben, R. G. Klotho lacks a vitamin D independent physiological role in glucose homeostasis, bone turnover and steady-state PTH secretion in vivo. PLoS One 7, e31376 (2012).
Chen, C. D., Podvin, S., Gillespie, E., Leeman, S. E. & Abraham, C. R. Insulin stimulates the cleavage and release of the extracellular domain of Klotho by ADAM10 and ADAM17. Proc. Natl. Acad. Sci. U. S. A. 104, 19796–19801 (2007).
Harris, R. C. & Neilson, E. G. Toward a unified theory of renal progression. Annu. Rev. Med. 57, 365–380 (2006).
Zeisberg, M., Strutz, F. & Muller, G. A. Renal fibrosis: an update. Curr. Opin. Nephrol. Hypertens. 10, 315–320 (2001).
Haruna, Y. et al. Amelioration of progressive renal injury by genetic manipulation of Klotho gene. Proc. Natl. Acad. Sci. U. S. A. 104, 2331–2336 (2007).
Shroff, R. C. et al. Chronic mineral dysregulation promotes vascular smooth muscle cell adaptation and extracellular matrix calcification. J Am Soc Nephrol 21, 103–112 (2010).
Mitani, H. In Vivo klotho Gene Transfer Ameliorates Angiotensin II-Induced Renal Damage. Hypertension 39, 838–843 (2002).
Panesso, M. C. et al. Klotho has dual protective effects on cisplatin-induced acute kidney injury. Kidney Int. 85, 855–870 (2014).
Sugiura, H. et al. Klotho reduces apoptosis in experimental ischaemic acute renal failure. Nephrol. Dial. Transplant. 20, 2636–2645 (2005).
Doi, S. et al. Klotho inhibits transforming growth factor-beta1 (TGF-beta1) signaling and suppresses renal fibrosis and cancer metastasis in mice. J. Biol. Chem. 286, 8655–8665 (2011).
Satoh, M. et al. Klotho protects against mouse renal fibrosis by inhibiting Wnt signaling. Am J Physiol Renal Physiol 303, 51 (2012).
Sugiura, H. et al. Reduced Klotho expression level in kidney aggravates renal interstitial fibrosis. Am J Physiol Renal Physiol 302, F1252–1264 (2012).
Zhang, Z. et al. Renoprotective role of the vitamin D receptor in diabetic nephropathy. Kidney Int. 73, 163–171 (2008).
Tan, X., Li, Y. & Liu, Y. Paricalcitol attenuates renal interstitial fibrosis in obstructive nephropathy. J. Am. Soc. Nephrol. 17, 3382–3393 (2006).
Halder, S., Goodwin, J. & Al-Hendy, A. 1,25-Dihydroxyvitamin D3 reduces TGF-beta3-induced fibrosis-related gene expression in human uterine leiomyoma cells. The Journal of clinical endocrinology and metabolism 96, 62 (2011).
Inoue, K. et al. Maxacalcitol ameliorates tubulointerstitial fibrosis in obstructed kidneys by recruiting PPM1A/VDR complex to pSmad3. Lab. Invest. 92, 1686–1697 (2012).
Wang, Y. & Sun, Z. Klotho gene delivery prevents the progression of spontaneous hypertension and renal damage. Hypertension 54, 810–817 (2009).
Aizawa, H. et al. Downregulation of the Klotho gene in the kidney under sustained circulatory stress in rats. Biochem. Biophys. Res. Commun. 249, 865–871 (1998).
Ohyama, Y. et al. Molecular cloning of rat klotho cDNA: markedly decreased expression of klotho by acute inflammatory stress. Biochem. Biophys. Res. Commun. 251, 920–925 (1998).
John, G. B., Cheng, C. Y. & Kuro-o, M. Role of Klotho in aging, phosphate metabolism and CKD. Am. J. Kidney Dis. 58, 127–134 (2011).
Schnaper, H. W. et al. TGF-beta signal transduction in chronic kidney disease. Front Biosci (Landmark Ed) 14, 2448–2465 (2009).
Kuro-o, M. Klotho and βKlotho. Adv. Exp. Med. Biol. 728, 25–40 (2012).
Tan, X., Li, Y. & Liu, Y. Paricalcitol attenuates renal interstitial fibrosis in obstructive nephropathy. Journal of the American Society of Nephrology: JASN 17, 3382–3393 (2006).
He, W., Kang, Y. S., Dai, C. & Liu, Y. Blockade of Wnt/beta-catenin signaling by paricalcitol ameliorates proteinuria and kidney injury. J. Am. Soc. Nephrol. 22, 90–103 (2011).
Gai, Z. et al. Trps1 haploinsufficiency promotes renal fibrosis by increasing Arkadia expression. J. Am. Soc. Nephrol. 21, 1468–1476 (2010).
Sato, M., Muragaki, Y., Saika, S., Roberts, A. B. & Ooshima, A. Targeted disruption of TGF-beta1/Smad3 signaling protects against renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction. J. Clin. Invest. 112, 1486–1494 (2003).
Choi, D. E. et al. Aliskiren ameliorates renal inflammation and fibrosis induced by unilateral ureteral obstruction in mice. J. Urol. 186, 694–701 (2011).
Cochrane, A. L. et al. Renal structural and functional repair in a mouse model of reversal of ureteral obstruction. J. Am. Soc. Nephrol. 16, 3623–3630 (2005).
Acknowledgements
We thank Ms. Mayumi Akira for her technical assistance. This work was supported in part by a Grant-in-Aid for Scientific Research (24590488) from the Ministry of Education, Science, Sports and Culture of Japan (to Y.M.).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: Y.M., Y.S. Performed the experiments: Y.S., T.G., A.S. Analyzed the data: Y.S., G.Z., K.O., M.N., F.S., Y.M. Wrote the paper: Y.S., Y.M.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
Supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Sun, Y., Zhou, G., Gui, T. et al. Elevated serum 1,25(OH)2-vitamin D3 level attenuates renal tubulointerstitial fibrosis induced by unilateral ureteral obstruction in kl/kl mice. Sci Rep 4, 6563 (2014). https://doi.org/10.1038/srep06563
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep06563
This article is cited by
-
Effect and mechanism of vitamin D activation disorder on liver fibrosis in biliary atresia
Scientific Reports (2021)
-
TGF-β1 promotes expression of fibrosis-related genes through the induction of histone variant H3.3 and histone chaperone HIRA
Scientific Reports (2018)
-
FTO modulates fibrogenic responses in obstructive nephropathy
Scientific Reports (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.