Key Points
-
Nasal septum perforation or palatal perforation may indicate chronic use of cocaine.
-
When (recent) use of cocaine is suspected, ask the patient in a quiet unprejudiced way and explain the potential dental consequences.
-
Dental treatment should be postponed after recent use of cocaine.
Abstract
In the UK almost one million individuals use cocaine on a regular basis, implying that dentists are likely to encounter individuals that use cocaine. Regular use of this drug may have several orofacial effects, such as perforation of the nasal septum and palate, gingival lesions and erosion of tooth surfaces. In addition, recent use of cocaine increases the risk of a medical emergency during dental treatment, especially when epinephrine-containing local anaesthetics or retraction cords are used. Therefore, dental treatment should be postponed for 6 to 24 hours after the use of cocaine.
Similar content being viewed by others
Introduction
Cocaine (benzoylmethylecgonine, C17H21NO4) is derived from the leaves of Ery-throxylon coca. For centuries, leaves of this plant have been consumed by South American Indians to induce a euphoric feeling.
In the UK, cocaine use is still increasing. Currently, approximately 910,000 people are using cocaine, resulting in Europe's second highest cocaine prevalence rate. The highest prevalence levels are reported in London (4.1%) and the lowest in Wales (1.6%) and Yorkshire (1.8%).1
The main method of administration is snorting cocaine.2 Within a few minutes after inhalation, an euphoric high feeling occurs which lasts for 20 to 90 minutes. For smoking of cocaine, the cocaine powder must first be converted into a smokeable form, eg by utilising sodium bicarbonate. This form is known as 'crack' because of the cracking sound made during heating.3 The popularity of crack has made intravenous injection of cocaine rather obsolete.2,4,5 Application of cocaine powder to the gingiva is mainly restricted to testing the purity of the cocaine.3
Systemic effects
In its non-ionised form, cocaine diffuses across the lipid membranes of neurons. In the axoplasm, it returns to the active cationic form, which binds to the sodium channels. This interaction prevents the generation of an action potential, resulting in a reversible anaesthetic effect.2 In addition, cocaine blocks the re-uptake of norepinephrine and dopamine, which prolongs and increases the effects of these neurotransmitters.6
Stimulation of the dopamine system induces a feeling of euphoria and arousal. Symptoms are light-headedness, dizziness, blurred vision, tinnitus and disorientation. Other symptoms are paranoia, hallucinations, restlessness, aggressive behaviour, delirium, vomiting, tremors, shivering, insomnia, dilated pupils, hyperthermia, hypertension, tachycardia and an increased respiration rate.2,4,5,6
With increasing doses of cocaine, these initial signs of central nervous system excitation are rapidly followed by a generalised state of central nervous system depression, resulting in a decreased respiration rate with periods of apnoea.2,6
The sympathomimetic effect of cocaine results in an increased heart rate and an increased oxygen demand of the myocardium. However, cocaine simultaneously induces vasoconstriction of the coronary arteries, decreasing the oxygen supply. This combination of increased myocardial oxygen demand and decreased coronary artery perfusion may lead to angina, myocardial infarction or cardiac dysrhythmias.2,5,6,7 Peripheral vasoconstriction increases the arterial blood pressure 15-20% above normal levels, further increasing the cardiac workload.4 Acute hypertension may lead to aortic dissection or rupture of an intracranial aneurysm.2
Intra-uterine effects
Cocaine and its metabolites cross the placenta to the foetus. Several human and animal studies indicate that use of cocaine during pregnancy reduces the occipital-frontal head circumference at birth and has a small negative effect on speech development.8,9 In mice, cocaine increases the risk of a foetus with a cleft palate, especially in combination with alcohol.10 In a cocaine-using pregnant woman, a foetus with bilateral cleft lip and palate was detected at 14 weeks of gestation.11
Nasal septum perforation
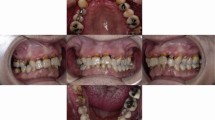
More than half of the people who snort cocaine have recurrent epistaxis, intranasal crusting, rhinitis and chronic sinusitis.12,13 Nasal septum perforation (Fig. 1) is a frequently reported complication, observed in approximately 5% of cocaine snorters.14,15 The perforation of the nasal septum reduces the nasal support and results in a broad, flat nose, the so-called saddle nose deformity12,14,15,16,17,18 (Fig. 2).
The major pathological factor seems to be the vasoconstrictive activity of cocaine, inducing local ischaemia which may lead to necrosis of the nasal septum and surrounding tissues. This process can be exacerbated by chemical irritation from adulterants in 'cut' cocaine such as quinine, caffeine, talc, plaster of Paris or amphetamines.19,20 Addition of lidocaine and procaine can cause facial numbness.20 Some patients use instruments such as pens and pencils to remove intranasal crusting, which increases the risk of nasal perforation.17,20 Finally, snorting of cocaine impairs the nasal mucociliary transport system that removes particles from the nose, thereby decreasing the nasal defence against infection.20 In a recent study, all patients with nasal septum perforation caused by cocaine abuse were nasal carriers of Staphylococcus aureus, suggesting that this micro-organism may contribute to the tissue damage.21
Palatal perforation
Chronic use of cocaine seems to have similar effects on the palate. Since 1989, at least 45 cases of cocaine-induced oronasal perforations have been reported in the literature,12,13,14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41 suggesting that this complication is much less rare than previously thought. In general, perforation of the nasal septum occurs first and is followed several months later by a slowly enlarging palatal perforation.
The reported oronasal perforations show a considerable variation in diameter: between 2 and 30 mm (Fig. 3). Except for four cases, all perforations involved the hard palate. Only in three patients was the defect limited to the soft palate.24,25,37 Another exception was a female patient with two oronasal fistulas just above the upper incisors on either side of the superior labial fraenulum.34
Intraoral view showing extensive palatal perforation in the patient depicted in Figure 2 (reproduced with permission from the Journal of Disability and Oral Health)
The majority of the patients with cocaine-induced palatal necrosis are female (72%), despite the fact that more men than women use cocaine.13 Therefore, it has been suggested that women are more susceptible to this complication of cocaine usage.14
Patients with a palatal perforation suffer from serious speech impairment. Their speech becomes hypernasal and articulation may be so poor that they can not communicate effectively.12,18,28,33,38,39 In addition, eating and drinking is difficult due to oronasal reflux of both solids and liquids.18,20,38 Some patients had resorted to obturating the defect by inserting chewing gum,17 a piece of white bread19 or a plug of tissue paper into the palatal defect.14
The initial treatment of a cocaine-induced palatal defect is complete abstinence from the drug, and the exclusion of other potential causes of midline palatal necrosis such as a tumour, Wegener's granulomatosis, trauma, chronic (fungal) infection and tertiary syphilis.40
It is advised that definitive reconstruction of the palatal defect should only be attempted after a long period of complete cocaine abstinence, at least for one year.14 The palatal defect can surgically be closed with a unilateral or bilateral pedicle flap, an anterior based tongue flap or a microvascular free flap with or without bone transfer.12,15,26,35,37,40
Surgical reconstruction of the palate failed in five out of ten reported patients.19,20,26,30,41 In three other patients, the final outcome of the surgical reconstruction was unknown because they were lost to follow up within three weeks.12,32,40 In another patient, two 1.5 mm fistulas remained after an anterior based tongue flap procedure.15 A successful one-year follow up has only been reported for one patient with two bipedicle mucoperiostous flaps.35
An alternative approach is the construction of a removable maxillary obturator. If a suprastructure is created on the obturator with the dimensions of the palatal defect, this will eliminate the oronasal reflux and restore the speech to normal18 (Fig. 4). As the palatal defect is not stable, the patient has to visit the dentist on a regular basis for small adaptations of the obturator.18,33,38
Superior surface of a maxillary obturator created for the patient depicted in Figure 2 (reproduced with permission from the Journal of Disability and Oral Health)
Long term use of a maxillary obturator for a cocaine-induced palatal defect was described by five patients as comfortable or at least acceptable.15,18,19,23,38 Considering the relatively poor outcome from surgical repair of cocaine-induced palatal defects, the non-surgical conservative approach seems the better option.
Hard tissues and restorations
Cocaine users frequently suffer from bruxism, commonly giving rise to pain in the temporomandibular joint and masticatory muscles.7 Mild attrition affecting all canines and first premolars as well as the upper lateral incisors was observed in a patient with a two-year history of regular use of cocaine and other drugs.3 Administration of cocaine to rats also induced a significant increase in attrition rate and non-functional masticatory activity.42 The increased risk of cervical abrasion is probably related to excessive vigorous tooth brushing.7
Dissolved in saliva, cocaine powder results in a substantial decrease of the salivary pH. This increases the risk of dissolving the tooth mineral calciumhydroxyapatite. In some chronic users loss of facial and occlusal tooth enamel was indeed observed, which gave the tooth surfaces a smooth and glassy appearance.43 A cocaine-induced decrease in salivary pH could also explain the rapid tarnishing of gold crowns in crack' users.44
A 14-year-old boy who rubbed cocaine on his gingivae had carious lesions on his incisors and canines.3 Another patient presented with three carious teeth which needed extraction.38 This suggests that the caries risk may be increased, due to sugar used to 'cut' pure cocaine.45
Gingivae
After oral use of cocaine, several individuals developed gingival lesions at the site of application, usually the maxillary anterior teeth. The gingivae were coated with a white slough, which could easily be removed, showing underlying ulceration and erythema. The patients reported painful, retracted gingivae.3,46,47,48 Full mouth radiographs revealed severe alveolar bone loss in one patient.45 These complications are probably related to the strong vasoconstrictive properties of cocaine.46 In addition, gingival laceration may be aggravated by excessively vigorous toothbrushing during a 'high' period.3
After abstaining from cocaine, in most subjects the gingival lesions disappeared spontaneously within two weeks to 18 months.3,46,47,48 However, in one patient a necrotic lesion of the alveolar mucosa led to exposure of the bone. Despite antibiotic treatment the lesion worsened, eventually leading to loss of both upper left incisors.48
Another patient rubbed cocaine on the gingival tissue that covered a maxillary bone graft. After three months, the bone graft was totally exposed in the oral cavity.49
Other orofacial effects
Several individuals have been described with orofacial pain compatible with cluster headache. Cocaine triggered pain in the premolar zone of the maxilla, followed by spread to the periorbital zone on the same side.48,50 Medication used to reduce symptoms of cocaine withdrawal may also have oral side effects. This medication includes antidepressants, lithium and levodopa.7 Antidepressants are a well-known cause of xerostomia. Levodopa and lithium may alter taste perception, induce a red discoloration of saliva, or induce involuntary facial movements.51
Dental treatment
Cocaine blocks nerve conduction similarly to the dental local anaesthetics lidocaine, xylocaine and articaine. The vasoconstrictive activity of cocaine enhances the response to epinephrine, frequently used as a vasoconstrictor in local anaesthetics.52 Therefore, administration of a local anaesthetic after recent use of cocaine may induce an acute increase in blood pressure. During tooth extraction, extreme variations in blood pressure were observed in patients who used cocaine or crack.53 Animal experiments have shown that a combination of cocaine and a local anaesthetic containing epinephrine can be fatal.52,54 Furthermore, lidocaine in combination with cocaine potentiates the risk of convulsions.2,4,5,6
Because of the potential medical risks during dental treatment, dentists should try to identify patients that use cocaine. Dentists should therefore be alert for signs of recent or chronic use of cocaine (eg agitation and damage to the nasal septum, respectively). Since many individuals will deny the use of cocaine, this information may be difficult to retrieve. An open conversation with the patient about the topic, based on correct information and without any prejudice, may help. In this way, the dentist can explain his professional interest in the potential use of cocaine by the patient.
As a general precaution, local anaesthetics with epinephrine and epinephrine-impregnated retraction cords should not be used when recent use of cocaine is suspected. Dental treatment should be postponed for 6 to 24 hours after the use of cocaine to allow for elimination of the drug.7,52,54
In a recent study, a small proportion of UK dental students reported using cocaine.55 Cocaine abuse by dentists has also been reported.55,56,57 In May 2000, a dentist was found guilty of misusing cocaine and other drugs in the dental surgery and was erased from the dental register.55 Therefore, all dental professionals would be wise to abstain from the use of cocaine.57
References
United Nations Office on Drugs and Crime. World drug report 2007. pp 63–94. Vienna: UNODC, 2007.
Engel J D . Cocaine: a historical and modern perspective. Nebr Med J 1991; 76: 263–270.
Parry J, Porter S, Scully C, Flint S, Parry M G . Mucosal lesions due to oral cocaine use. Br Dent J 1996; 180: 462–464.
Isaacs S O, Martin P, Willoughby J H . 'Crack' (an extra potent form of cocaine) abuse: a problem of the eighties. Oral Surg Oral Med Oral Pathol 1987; 63: 12–16.
Goldstein F J . Toxicity of cocaine. Compend Contin Educ Dent 1990; 11: 710–716.
Lee C Y, Mohammedi H, Dixons R A . Medical and dental implications of cocaine abuse. J Oral Maxillofac Surg 1991; 49: 290–293.
Friedlander A H, Gorelick D A . Dental management of the cocaine addict. Oral Surg Oral Med Oral Pathol 1988; 65: 45–48.
Cone-Wesson B . Prenatal alcohol and cocaine exposure: influences on cognition, speech, language and hearing. J Commun Disord 2005; 38: 279–302.
Morris P, Binienda Z, Gillam M P et al. The effect of chronic cocaine exposure during pregnancy on maternal and infant outcomes in the rhesus monkey. Neurotoxicol Teratol 1997; 18: 147–154.
Al-Motabagani M A, Mohamed A S . Congenital malformations in mice induced by addiction to alcohol and cocaine. East Afr Med J 2005; 82: 433–438.
Markov D, Jacquemyn Y, Leroy Y . Bilateral cleft lip and palate associated with increased translucency and maternal cocaine abuse at 14 weeks of gestation. Clin Exp Obstet Gynecol 2003; 30: 109–110.
Mattson-Gates G, Jabs A D, Hugo N E . Perforation of the hard palate associated with cocaine abuse. Ann Plast Surg 1991; 29: 466–468.
Blanksma C J, Brand H S . Cocaine abuse: orofacial manifestations and implications for dental treatment. Int Dent J 2005; 55: 365–369.
Lancaster J, Belloso A, Wilson C A, McCormick M . A rare case of naso-oral fistula with extensive osteocartilaginous necrosis secondary to cocaine abuse: review of otorhinolaryngological presentations in cocaine addicts. J Laryngol Otol 2000; 114: 630–633.
Mari A, Arranz C, Gimeno X et al. Nasal cocaine abuse and centrofacial destructive process: report of three cases including treatment. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002; 93: 435–439.
Deutsch H L, Millard D R . A new cocaine abuse complex. Involvement of nose, septum, palate, and pharynx. Arch Otolaryngol Head Neck Surg 1989; 115: 235–237.
Villa P D . Midfacial complications of prolonged cocaine snorting. J Can Dent Assoc 1999; 65: 218–223.
Brand H S, Blanksma C J, Gonggrijp S . A maxillary obturator for a cocaine-induced oronasal defect. J Disabil Oral Health 2007; 8: 37–40.
Tsoukalas N, Johnson C D, Engelmeier R L, Delattre V F . The dental management of a patient with a cocaine-induced maxillofacial defect: a case report. Spec Care Dentist 2000; 20: 139–142.
Smith J C, Kacker A, Anand V K . Midline nasal and hard palate destruction in cocaine abusers and cocaine's role in rhinologic practice. Ear Nose Throat J 2002; 81: 172–177.
Simsek S, de Vries X H, Jol J A D et al. Sino-nasal bony and cartilaginous destruction associated with cocaine abuse, S. aureus and antineutrophil cytoplasmic antibodies. Neth J Med 2006; 64: 248–251.
Kuriloff D B, Kimmelman C P . Osteocartilaginous necrosis of the sinonasal tract following cocaine abuse. Laryngoscope 1989; 99: 918–924.
Kuriloff D B . Perforation of the hard palate associated with cocaine abuse. Ann Plast Surg 1992; 28: 397.
Sousa O, Rowley S . Manifestaciones otorinolaringologicas por el abuso intranasal de cocaina. A proposito de un caso. Rev Med Panama 1994; 19: 55–60.
Armstrong M, Shikani A H . Nasal septal necrosis mimicking Wegner's granulomatosis in a cocaine abuser. Ear Nose Throat J 1996; 75: 623–626.
Helie F, Fournier J . Destructive lesions of the median line secondary to cocaine abuse. J Otolaryngol 1997; 26: 67–69.
Sastry R C, Lee D, Har-El G . Palate perforation from cocaine abuse. Otolaryngol Head Neck Surg 1997; 116: 565–566.
Gendeh B S, Ferguson B J, Johnson J T, Kapadia S . Progressive septal and palatal perforation secondary to intranasal cocaine abuse. Med J Malaysia 1998; 53: 435–438.
Sittel C, Eckel H E . Nasal cocaine abuse presenting as a central destructive granuloma. Eur Arch Otorhinolaryngol 1998; 255: 446–447.
Braverman I, Raviv E, Frienkiel S . Severe avascular necrosis of the nasal chambers secondary to cocaine abuse. J Otolaryngol 1999; 28: 351–353.
Cottrell D A, Mehra P, Malloy J C, Ghali G E . Midline palatal perforation. J Oral Maxillofac Surg 1999; 57: 990–995.
Talbott J F, Gorti G K, Kock R J . Midfacial osteomyelitis in a chronic cocaine abuser: a case report. Ear Nose Throat J 2001; 80: 738–743.
Seyer B A, Grist W, Muller S . Aggressive destructive midfacial lesion from cocaine abuse. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2002; 94: 465–470.
Vilela R J, Langford C, McCullagh L, Kass E S . Cocaine-induced oronasal fistulas with external nasal erosion but without palate involvement. Ear Nose Throat J 2002; 81: 562–563.
Monasterio L, Morovic G C . Midline palate perforation from cocaine abuse. Plast Reconstruct Surg 2003; 112: 914–915.
Trimarchi M, Nicolai P, Lombardini D et al. Sinonasal osteocartilaginous necrosis in cocaine abusers: experience in 25 patients. Am J Rhinol 2003; 17: 33–43.
Ladner T, Linker M, Gitani J . Functional repair of a major necrotic palatine defect caused by chronic cocaine inhalation. Rev Stomatol Chir Maxillofac 2004; 105: 291–293.
Bains M K, Hosseini-Ardehali M . Palatal perforations: past and present. Two case reports and a literature review. Br Dent J 2005; 199: 267–269.
Padilla-Rosas M, Jimenez-Santos C I, Garcia-Gonzalez C L . Palatine perforations induced by cocaine. Med Oral Patol Oral Cir Bucal 2006; 11: E239–E242.
Goodger N M, Wang J, Pogrel M A . Palatal and nasal necrosis resulting from cocaine misuse. Br Dent J 2005; 198: 333–334.
Harkes M L P, van der Wal K G H . A man with a headache and haemorrhagic nasal discharge. Ned Tijdschr Geneeskd 2007; 151: 597.
Gomez F M, Areso M P, Giralt M T et al. Effects of dopaminergic drugs, occlusal disharmonies, and chronic stress on non-functional masticatory activity in the rat, assessed by incisal attrition. J Dent Res 1998; 56: 323–329.
Krutchkoff D J, Eisenberg E, O'Brien J E et al. Cocaine-induced dental erosions. N Engl J Med 1990; 322: 408.
Brown R S, Johnson C D . Corrosion of dental gold restorations from inhalation of 'crack' cocaine. Gen Dent 1994; 42: 242–246.
Driscoll S E . A pattern of erosive lesions from cocaine use: a retrospective study. J Dent Res 2002; 81: A461.
Quart A M, Small C B, Klein R S . The cocaine connection. Users imperil their gingiva. J Am Dent Assoc 1991; 122: 85–87.
Kapila Y L, Kashani H . Cocaine-associated rapid gingival recession and dental erosion. A case report. J Periodontol 1997; 68: 485–488.
Gandara-Rey J M, Diniz-Freitas M, Gandara-Vila P, Blanco-Carrion A, Garcia-Garcia A . Lesions of the oral mucosa in cocaine users who apply the drug topically. Med Oral 2002; 7: 103–107.
Shibli J A, Marcantonio E, Spolidorio L C, Marcantorio E . Cocaine associated with onlay bone graft failure: a clinical and histologic report. Implant Dent 2005; 14: 248–251.
Penarrocha M, Bagan J V, Penarrocha M J, Silvestre F J . Cluster headache and cocaine use. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2000; 90: 271–274.
Vissink A, van Nieuw Amerongen A, Oremus E T H G J . The effect of drugs on the orofacial area. Ned Tijdschr Tandheelkd 1999; 106: 254–263.
Yagiela J A . Adverse drug interactions in dental practice: interactions associated with vasoconstrictors. Part V of a series. J Am Dent Assoc 1999; 130: 701–709.
Johnson C D, Lewis V A, Faught K S et al. The relationship between chronic cocaine or alcohol use and blood pressure in black men during uncomplicated tooth extraction. J Oral Maxillofac Surg 1998; 56: 323–329.
Goulet J P, Perusse R, Turcotte J Y . Contraindications to vasoconstrictors in dentistry: Part III. Pharmacologic interactions. Oral Surg Oral Med Oral Pathol 1992; 74: 692–697.
Barber M W, Fairclough A . A comparison of alcohol and drug use among dental undergraduates and a group of non-medical, professional undergraduates. Br Dent J 2006; 201: 581–584.
Giangrego E, Oberg S W . Chemical dependency, the road to recovery. J Am Dent Assoc 1987; 115: 17–27.
Sfikas P M . The private lives of professionals. J Am Dent Assoc 1999; 130: 1649–1651.
Author information
Authors and Affiliations
Corresponding author
Additional information
Refereed paper
Rights and permissions
About this article
Cite this article
Brand, H., Gonggrijp, S. & Blanksma, C. Cocaine and oral health. Br Dent J 204, 365–369 (2008). https://doi.org/10.1038/sj.bdj.2008.244
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.bdj.2008.244
This article is cited by
-
Are periodontitis, dental caries and xerostomia more frequently present in recreational ecstasy users?
British Dental Journal (2022)
-
Cocaine and crack cocaine: the prevalence of dental caries among users
BDJ Team (2022)
-
Are periodontitis, dental caries and xerostomia more frequently present in recreational ecstasy users?
BDJ Team (2022)
-
Recreational reluctance
British Dental Journal (2021)
-
Salivary flow rates and buffer capacity and its relationship with oral health status: a cross-sectional study on crack-cocaine-addicted males
Environmental Science and Pollution Research (2020)