Abstract
Study Design:
Retrospective chart review.
Objective:
To compare autonomic dysreflexia (AD) severity during urodynamics and cystoscopy in individuals with spinal cord injury (SCI).
Setting:
Outpatient urological clinic.
Methods:
Demographic and clinical data were collected from charts of individuals with SCI who had blood pressure (BP) monitoring during urological procedures. Cardiovascular parameters were collected at baseline and during the various stages of two examinations.
Results:
A total of 21 SCI individuals (mean age 49.4 years) who underwent both procedures developed episodes of AD. The majority of individuals had cervical SCI (85.7%). The median duration of injury was 183 months (ranging from 3 to 530 months). There was statistically more of an increase (P=0.039) in systolic BP during cystoscopy (67.1±33.8 mm Hg) in comparison with urodynamics (51.8±21.8 mm Hg). The BP response during episodes of AD was more pronounced in individuals with more than 2 years post SCI than with less than 2 years post SCI during both urodynamics and cystoscopy (P=0.047 and P=0.010, respectively).
Conclusion:
Even though cystoscopy filled the bladder to lesser volumes than did urodynamics (150 ml vs 500 ml), during cystoscopy the individuals developed greater changes in systolic BP, indicating that stimulation of the urethra/prostate/internal sphincter region probably is a more potent stimulus of AD than just the filling of the bladder. The severity of AD also increased with time post SCI during both procedures. Considering the high incidence of silent episodes of AD during the urological procedures, it is recommended that monitoring of cardiovascular parameters during these procedures be routinely performed.
Similar content being viewed by others
Introduction
Individuals with spinal cord injury (SCI) suffer from various secondary complications including disordered cardiovascular (CV) control.1 Unpredictable fluctuations of blood pressure (BP) from a low baseline arterial BP to episodes of hypertension known as autonomic dysreflexia (AD) are very common among individuals with SCI. These fluctuations commonly occur among individuals with SCI at and above the sixth thoracic (T6) spinal segment and sometimes can be a result of iatrogenic manipulations.2
One of the most common physician-induced causes of AD in individuals with SCI is the use of procedures that involve urinary bladder and urogenital areas.3 Previously, numerous studies using different groups of individuals with SCI examined CV responses either during urodynamic3, 4, 5, 6 or cystoscopic examination.7, 8 These two procedures both involve urinary bladder distension. However, cystoscopy includes greater stimulation of the urethra and bladder neck afferents during the procedure through urethral/prostate/internal sphincter passage and dilation, which could then provide more intense afferent stimulation to the spinal cord and trigger more pronounced episodes of AD. Considering the physiological difference in urinary bladder and urethral afferents (bladder afferent receptors are influenced predominantly by the viscoelastic properties of the bladder wall, while urethral afferents do not respond to bladder distension, but are excited by low-threshold mechanical stimulation induced by movements of a urethral catheter),9 we could expect a difference in AD responses between these two procedures. However, no previous studies have examined the differences and severity of AD between these two procedures in the same group of individuals.
Clinically, episodes of AD can present with a variety of symptoms including increased systolic and diastolic BP, bradycardia, tachycardia, pounding headache, cutis anserine (goose bumps), anxiety, flushing of the skin, sweating above the neurologic level of injury, malaise and nausea. The intensity of symptoms can either vary from asymptomatic4 to mildly uncomfortable or even life-threatening, as is a case with a myocardial infarction,10 stroke11 or death.12
The aim of this study was to compare the changes in intensity of CV responses due to AD when both urodynamic and cystoscopic examinations are performed on the same individuals. We also examined the effect that the time post SCI had on the severity of AD during these examinations. The possible differences in mechanisms triggering episodes of AD during these procedures are also discussed.
Materials and methods
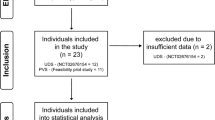
Following approval of the study protocol by the Clinical Research Ethics Board, a retrospective chart analysis was conducted of individuals with SCI followed at the outpatient Spinal Cord Clinic who underwent both urodynamic and cystoscopic examinations during a 1-year period. Only individuals who developed episodes of AD were selected for this study.
Data collection
Data on demography, SCI, urological procedure and CV parameters during the procedures were extracted. This data included the following: age, sex, American Spinal Injury Association Impairment Scale (AIS),13 time post SCI, BP and heart rate (HR) at baseline and during urological evaluations, and any symptoms or signs of AD. Data regarding lower urinary tract (LUT) function were collected using the Lower Urinary Function, Bowel and Sexual Function Subscale of the International Standards to Document Remaining Autonomic Function after Spinal Cord Injury (ISAFSCI).14 The variables within the bladder emptying method originated from the definition in the International Lower Urinary Tract Basic Data Set.15 Only the main methods used by the SCI individuals were collected in our study. Bladder reflex triggering included voluntary triggering such as supra-pubic tapping, thigh scratching or anal/rectal manipulation and involuntary triggering such as reflex detrusor contraction from neurogenic detrusor overactivity. The urodynamic parameters are based on the International Spinal Cord Injury Urodynamic Basic Data Set.16
Urodynamic examination (Aquarius TT, Laborie, Montreal, Quebec, Canada) was done by the same group of urologists according to the principles set forth by the International Continence Society. The examination consisted of cystometry with warm sterile water (37 °C) filled at a fixed 30 ml per minute rate through a pump to a double lumen catheter (6Fr, Laborie, Canada) while patients were supine. Abdominal pressure was measured with an intrarectal balloon catheter (10Fr, Laborie, Canada). Pelvic floor electromyography was recorded during cystometry with a bipolar wire electrode in the urethral sphincter. Filling was stopped when the patient subjectively reported the sensation of fullness, urine leakage occurred or bladder filling reached 500 ml. All individuals were asked to stop anticholinergics 3 days before these two procedures.
Cystoscopy was conducted with the patient in a lithotomy position. Before the procedure, 10 ml of 2% lidocaine jelly was introduced intra-urethrally, followed by the instillation of 150 ml of warm sterile water (37 °C) into the bladder. The filling rate varied from 30 to 50 ml per minute through gravity filling while a flexible cystoscope (CV-180 Evis EXERA II, Olympus, Tokyo, Japan) was introduced into the urethra. When the scope entered the bladder, a systematic inspection of the bladder mucosa was carried out. Once cystoscopy was completed, the scope was withdrawn and bladder was emptied.
CV parameters were based on continuous BP and HR monitoring (1-min interval) with a left arm cuff (Dinamap 100, GE, Fairfield, CT, USA) during urodynamic and cystoscopic examinations. Resting BP and HR were recorded before starting the urodynamic and cystoscopic examinations. Symptoms and signs of AD were also documented throughout the procedure.
To examine the effect of time post SCI, we compared CV responses to urodynamics and cystoscopy in individuals with less than 2 years post SCI (n=7) and more than 2 years post SCI (n=14).
Statistical analyses
Statistical analysis was performed using SPSS 19.0 (IBM-SPSS, Chicago, IL, USA). The data of continuous variables are expressed as mean±s.d. Categorical variables are expressed as percentages and absolute frequencies. Differences between means of continuous variables were calculated with a Student's t-test if it was a normal distribution: a paired test was used between the same individuals who underwent different procedures and an independent test was used between different times post SCI. Otherwise the Mann–Whitney U-test was used for nonparametric comparisons. A Chi-square test (or Fisher’s exact test if any of the sections of the table were less than 5) was used to compare categorical variables. The relationship between maximal detrusor pressure and changes in systolic BP (SBP) was quantified by linear regression analysis. Significance was set at the P<0.05 level.
Results
A total of 76 individuals (66 males and 10 females) with SCI at or above T6 were examined in the urological outpatient clinic with continuous BP monitoring during urodynamics. From this group, 48 individuals (43 males and 5 females) experienced a SBP elevation greater than 20 mm Hg, which met the criteria for AD diagnosis. Among them, 21 individuals (all males) developed AD while undergoing consecutive urodynamic and cystoscopic examinations. The mean age of individuals was 49.4±11.2 years. The majority of individuals had cervical SCI (85.7%). The median duration of injury was 183 months (range from 3 to 530 months). The demographic characteristics, injury level and AIS of the study population are shown in Table 1. In our study, symptoms of AD were present among 8 (38%) individuals during urodynamics and in 10 (48%) individuals during cystoscopic examinations. Most commonly individuals developed headache, flushing of the skin and sweating. There was no difference in severity of AD among symptomatic and asymptomatic individuals during urodynamics (52.9±22.1 mm Hg vs 51.1±22.5 mm Hg, P=0.860) and cystoscopy (68.8±39.6 mm Hg vs 65.6±29.6 mm Hg, P=0.837). In addition, there was no difference between the severity of AD between SCI complete and incomplete individuals during urodynamics (54.4±21.5 mm Hg vs 46.6±23.0 mm Hg, P=0.454) and cystoscopy (66.4±31.6 mm Hg vs 68.6±40.7 mm Hg, P=0.895).
The majority of individuals in our sample required catheters for bladder management (intermittent catheterization 28.6% and indwelling catheter 38.1%). The urodynamic evaluation determined that the majority of these individuals present with neurogenic detrusor overactivity (71.4%) and detrusor sphincter dyssynergia (76.2%). During urodynamics, the maximal detrusor pressure during bladder filling was estimated as 44.1±29.8 cm H2O. However, there was no correlation (r=0.354, P=0.116) between maximal detrusor pressure and changes in SBP due to AD in our sample.
CV parameters at baseline before urodynamics and cystoscopy were similar in these individuals. Table 2 displays the CV parameters, which include: SBP, diastolic BP, HR at baseline and changes during the two examinations. Both urodynamics and cystoscopy triggered episodes of AD. Of the CV parameters, only SBP was significantly different between cystoscopy and urodynamics (67.1±33.8 mm Hg and 51.8±21.8 mm Hg, respectively; P=0.039) (Table 2).
Individuals with less than 2 years post SCI and more than 2 years post SCI were similar with respect of their age, SCI level, SCI severity and baseline CV parameters (Table 3). However, the increase in SBP during AD triggered by urodynamics in individuals more than 2 years post SCI (58.4±21.0 mm Hg) was more pronounced (P=0.047) than in individuals with less than 2 years post SCI (38.6±18.0 mm Hg). Similar changes in SBP were found during cystoscopy in individual less than 2 years and more than 2 years post SCI (41.4±20.4 mm Hg vs 80.0±32.2 mm Hg, P=0.010) (Table 3). Furthermore, when comparing SBP increases between the two procedures, no significant difference was found in the group with less than 2 years post SCI (P=0.483), but in individuals with more than 2 years post SCI, there was a significant increase in SBP during cystoscopy than urodynamics (P=0.049).
Discussion
Numerous studies have documented the presence of AD during urological evaluation among individuals with SCI.3, 4, 5, 6, 7, 8 Our study presents novel information on comparison of the CV responses of the same group of individuals with SCI during two consecutive urological procedures: urodynamics and cystoscopy. The results of our study indicate that as a result of AD, the increase in SBP during cystoscopy was significantly higher (67.1±33.8 mm Hg) than during urodynamics (51.8±21.8 mm Hg, P=0.039). Furthermore, to our knowledge this is the first documentation that the severity of AD in individuals with SCI worsens with time post SCI during both urodynamic and cystoscopic examinations. Our data demonstrate that individuals who have had SCI for more than 2 years develop more pronounced episodes of AD during both urodynamics and cystoscopy. Furthermore, cystoscopy was associated with more severe episodes of AD in those with more than 2 years post SCI.
In comparison with previous studies of AD during urodynamics, Huang et al.6 reported the average increase in SBP in cervical SCI individuals during urodynamics was ∼21±23 mm Hg. However, in our study the change in SBP was noticeably higher (58.4±21.0 mm Hg). One reason could be that the authors did not identify a separate subgroup of individuals with AD (SBP increase of more than 20 mm Hg12) or without AD. The authors indicate that 44.7% of individuals developed episodes of AD. In contrast, we specifically selected for our study only individuals who demonstrated AD during both procedures. Another possible explanation for the low increase of SBP during urodynamics is the differences in BP measurement protocol. In our center, we established a BP measurement protocol during the urological procedures (interval of 1 min using the automated parameters of the Dinamap). These allow us more frequent measurements and more accurate documentation of the BP and HR during the procedures. Interestingly, in the study by Huang et al.,6 the BP recording interval was only set for 3 min.The maximal BP increase during an AD episode is often a short and transient phenomenon, and less frequent BP monitoring adds the possibility of missing the maximum BP increase in the study by Huang et al.6 However, a study by Giannantoni et al.3 reported that individuals with AD during urodynamics had an SBP increase between 112.1±16.3 mm Hg and 167.5±24.0 mm Hg with BP recorded every 2 min. These changes in SBP were very similar to those being reported in the present study.
As we know from previous investigations, both urodynamics and cystoscopy are frequent iatrogenic triggers of episodes of AD.3, 4, 5, 6, 7, 8 Typically, AD is triggered by noxious or non-noxious stimuli below the level of injury. In the majority of situations for individuals with SCI, these triggers originate within the urinary bladder.2 One of the questions in this study asks if either urodynamics or cystoscopy predisposes individuals for more significant CV responses during urological evaluations. During these procedures, one potential trigger of AD is bladder distension due to bladder filling. However, the volume of water instilled during cystoscopy was typically standard and smaller (150 ml) in comparison with urodynamics, where volume varied depending on bladder capacity (in our study, mean bladder volume was 335.3±246.2 ml). Although bladder distension during cystoscopy was less than that in urodynamics, the severity of AD was more pronounced during cystoscopy in our study. Because the increase in SBP during the cystoscopy was 29.5% greater than during urodynamics, other stimuli in addition to bladder distention must be contributing to the development of AD during cystoscopy. Urethral stimulation during cystoscopy is one possible potent stimulus that may add to the severity of AD. Further evidence for possible triggers of more pronounced AD during cystoscopy were also found when comparing the protocol between these two procedures. Cystoscopy typically uses a flexible cystoscope (17 Fr–21 Fr) larger in size than catheters required for urodynamics (6 Fr–8 Fr). Differences in the technique of each procedure, including the filling rate and method of filling, could also explain the difference in severity of AD between cystoscopy and urodynamics. However, we can infer from the cystoscopy that gravity filling of the bladder resulted in only 150 ml of bladder volume filling, much lower than the volume (up to 500 ml) instilled by continuous pump during urodynamics. In our 21 subjects, the bladder filling during urodynamics induced detrusor pressure (average 44.1±29.8 cm H2O), which was consistent with the study by Wallin et al.17 However, in this study investigators reported a positive correlation (r=0.52, P<0.05) between the maximal detrusor pressure and the maximal increase in BP. This correlation was not present in our study. Thus, the relationship between maximal detrusor pressure and BP increase still needs to be further investigated.
The episodes of AD in our study were associated with an increase in BP of up to 256/117 mm Hg. However, only 38% of individuals during urodynamics and 48% during cystoscopy developed symptoms of AD. A significant number of individuals in our study were asymptomatic during the episodes of AD. We previously, reported episodes of asymptomatic (silent) AD during vibrostimulation procedure for sperm retrieval in individuals with SCI.18 Vibrostimulation associated with asymptomatic episodes of AD in 62% of participants (average increase in SBP was 65±44 mm Hg). In this study, we reported an individual with a peak BP of 229/139 mm Hg who still did not experience any subjective symptoms of AD. The results of this previous study indicated that those subjects with complete injuries may not experience the symptoms of AD and, thus, be unaware of the risk of silent AD. In the present study, the majority of individuals also had motor complete SCI and experienced silent episodes of AD during urological examination.
Another aspect that needed to be addressed is the use of local anesthesia. There are still some controversies with respect of using local anesthesia to prevent episodes of AD triggered by medical procedures during urogenital or anorectal procedures.19, 20, 21 As both areas have similar afferent innervations (S4–5) and limited information is available on urogenital procedures, we have included the literature available that addresses issues of local anesthesia and AD. In a review study of flexible cystoscopy in SCI patients, conducted by Rivas et al.,19 lidocaine 2% gel was suggested as a topical local anesthetic and lubricant for routine use as a means to decrease afferent stimulation and prevent episodes of AD. Although there is no strong evidence in the literature supporting the effect of local anesthesia in blocking AD during cystoscopy, the use of a local anesthetic is a common component of cystoscopic procedures. However, Cosman et al.20 found topical lidocaine unsuccessful for preventing AD during anorectal procedures in individuals with SCI, whereas a lidocaine anal block was able to eliminate episodes of AD.21 In our study, 2% lidocaine gel was used with all cystoscopies; however, it was not used during urodynamics, as sensation had to be examined and assessed. Despite this, AD severity was still more pronounced during cystoscopy than urodynamics. Therefore, our results suggest that urethral/bladder neck stimulation during cystoscopy would be a more potent and additional trigger of AD.
We also demonstrated that there is a significant increase in severity of AD in individuals with more than 2 years post SCI. Previously, Giannantoni et al.3 indicated that there was no correlation between AD and disease duration, though no direct CV data on this analysis was presented in the manuscript. However, there are detailed animal data presently available for time course changes in the severity and possible mechanisms of AD.22, 23 It was shown that in the acute period post SCI, AD is typically less developed, and over time, as plastic changes within the spinal cord and aberrant inappropriate connections within the central and peripheral nervous system are established, AD becomes more prominent.24
We also have to acknowledge a few limitations of this study. There are only 21 subjects included in this study and thus we are cognizant of the limitations of our analysis with respect to duration of effect of injury on the severity of AD. As only individuals who underwent both procedures consecutively and developed episodes of AD were selected for this study, this strict inclusion criterion contributed to the small sample size in the study. Furthermore, the bladder filling rate during urodynamics and cystoscopy is inherently different in technique and thus could also account for some differences in the severity of AD between these procedures.
Conclusion
Urodynamic and cystoscopic examinations resulted in the development of AD in individuals injured at or above the T6 spinal segment. Cystoscopy, which involves potent stimulation of the urethra and bladder neck, was associated with more intense episodes of AD compared with urodynamics, although bladder-filling volumes were higher during urodynamics. The severity of AD also increased with time post SCI during both urodynamic and cystoscopic procedures. Considering the high incidence of episodes of silent AD, monitoring of CV parameters during these procedures should be part of clinical practice to minimize the risk of possible life-threatening complications of AD.
Data archiving
There were no data to deposit.
References
Teasell RW, Arnold JM, Krassioukov A, Delaney GA . Cardiovascular consequences of loss of supraspinal control of the sympathetic nervous system after spinal cord injury. Arch Phys Med Rehabil 2000; 81: 506–516.
Krassioukov A, Warburton DE, Teasell R, Eng JJ . A systematic review of the management of autonomic dysreflexia after spinal cord injury. Arch Phys Med Rehabil 2009; 90: 682–695.
Giannantoni A, Di Stasi SM, Scivoletto G, Mollo A, Silecchia A, Fuoco U et al. Autonomic dysreflexia during urodynamics. Spinal Cord 1998; 36: 756–760.
Linsenmeyer TA, Campagnolo DI, Chou IH . Silent autonomic dysreflexia during voiding in men with spinal cord injuries. J Urol 1996; 155: 519–522.
Curt A, Nitsche B, Rodic B, Schurch B, Dietz V . Assessment of autonomic dysreflexia in patients with spinal cord injury. J Neurol Neurosurg Psychiatry 1997; 62: 473–477.
Huang YH, Bih LI, Chen GD, Lin CC, Chen SL, Chen WW . Autonomic dysreflexia during urodynamic examinations in patients with suprasacral spinal cord injury. Arch Phys Med Rehabil 2011; 92: 1450–1454.
Vaidyanathan S, Watt JW, Soni BM, Krishnan KR . Intravenous salbutamol treatment for penile erection arising during cystoscopy of cervical spinal cord injury patients. Spinal Cord 1996; 34: 691–695.
Snow JC, Sideropoulos HP, Kripke BJ, Freed MM, Shah NK, Schlesinger RM . Autonomic hyperreflexia during cystoscopy in patients with high spinal cord injuries. Paraplegia 1978; 15: 327–332.
de Groat WC, Yoshimura N . Afferent nerve regulation of bladder function in health and disease. Handb Exp Pharmacol 2009; 194: 91–138.
Ho CP, Krassioukov AV . Autonomic dysreflexia and myocardial ischemia. Spinal Cord 2010; 48: 714–715.
Pan SL, Wang YH, Lin HL, Chang CW, Wu TY, Hsieh ET . Intracerebral hemorrhage secondary to autonomic dysreflexia in a young person with incomplete C8 tetraplegia: A case report. Arch Phys Med Rehabil 2005; 86: 591–593.
Eltorai I, Kim R, Vulpe M, Kasravi H, Ho W . Fatal cerebral hemorrhage due to autonomic dysreflexia in a tetraplegic patient: case report and review. Paraplegia 1992; 30: 355–360.
Kirshblum SC, Burns SP, Biering-Sorensen F, Donovan W, Graves DE, Jha A et al. International standards for neurological classification of spinal cord injury (revised 2011). J Spinal Cord Med 2011; 34: 535–546.
Krassioukov A, Biering-Sørensen F, Donovan W, Kennelly M, Kirshblum S, Krogh K et al. International standards to document remaining autonomic function after spinal cord injury. J Spinal Cord Med 2012; 35: 201–210.
Biering-Sørensen F, Craggs M, Kennelly M . International lower urinary tract function basic spinal cord injury data set. Spinal Cord 2008; 46: 325–330.
Biering-Sørensen F, Craggs M, Kennelly M . International urodynamic basic spinal cord injury data set. Spinal Cord 2008; 46: 513–516.
Wallin BG, Burton AR, Elam M, Tamaddon K, Millard R, Macefield VG . Viscerosympathetic reflexes in human spinal cord injury: relationships between detrusor pressure, blood pressure and skin blood flow during bladder distension. Exp Physiol 2013; 98: 1081–1091.
Ekland MB, Krassioukov AV, McBride KE, Elliott SL . Incidence of autonomic dysreflexia and silent autonomic dysreflexia in men with spinal cord injury undergoing sperm retrieval: implications for clinical practice. J Spinal Cord Med 2008; 31: 33–39.
Rivas DA . Chancellor MB. Flexible cystoscopy in spinal cord injury. Review article. Paraplegia 1994; 32: 454–462.
Cosman BC, Vu TT . Plowman BK. Topical lidocaine does not limit autonomic dysreflexia during anorectal procedures in spinal cord injury: a prospective, double-blind study. Int J Colorectal Dis 2002; 17: 104–108.
Cosman BC, Vu TT . Lidocaine anal block limits autonomic dysreflexia during anorectal procedures in spinal cord injury: a randomized, double-blind, placebo-controlled trial. Dis Colon Rectum 2005; 48: 1556–1561.
Krassioukov AV, Weaver LC . Episodic hypertension due to autonomic dysreflexia in acute and chronic spinal cord-injured rats. Am J Physiol 1995; 268: H2077–H2083.
Krassioukov AV, Weaver LC . Reflex and morphological changes in spinal preganglionic neurons after cord injury in rats. Clin Exp Hypertens 1995; 17: 361–373.
Ramer LM, van Stolk AP, Inskip JA, Ramer MS, Krassioukov AV . Plasticity of TRPV1-expressing sensory neurons mediating autonomic dysreflexia following spinal cord injury. Front Physiol 2012; 3: 257.
Acknowledgements
Dr Nan Liu is supported by a State scholarship from the China Scholarship Council to the Laboratory of Dr Krassioukov, International Collaboration on Research Discoveries (ICORD), University of British Columbia, Canada; Dr Andrei Krassioukov is supported by grants from the Canadian Heart and Stroke Foundation, Canadian Institute of Health Research, Canadian Foundation for Innovation, Rick Hansen Institute, Craig Neilsen Foundation and Christopher and Dana Reeve Foundation. We also acknowledge support from staff at the Blusson Spinal Cord Clinic during the preparation of this manuscript, Ms Teresa Lim and Ms Colleen McLean.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Liu, N., Fougere, R., Zhou, MW. et al. Autonomic dysreflexia severity during urodynamics and cystoscopy in individuals with spinal cord injury. Spinal Cord 51, 863–867 (2013). https://doi.org/10.1038/sc.2013.113
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sc.2013.113
Keywords
This article is cited by
-
Autonomic dysreflexia in urological practice: pathophysiology, prevention and treatment considerations
World Journal of Urology (2024)
-
Guideline for the management of pre-, intra-, and postpartum care of women with a spinal cord injury
Spinal Cord (2020)
-
Prediction of autonomic dysreflexia during urodynamics: a prospective cohort study
BMC Medicine (2018)
-
International spinal cord injury urodynamic basic data set (version 2.0)
Spinal Cord Series and Cases (2018)
-
Current Management Strategies for Autonomic Dysreflexia
Current Bladder Dysfunction Reports (2018)