Abstract
The objective of the study was to demonstrate the non-inferiority of low-frequency spa therapy combined with rehabilitation (Spa-rehab) versus standard spa therapy at 6 months for symptomatic knee osteoarthritis (KOA). A prospective, randomized, monocenter, non-inferiority trial with recruitment of community-based symptomatic KOA patients was performed. Standard spa therapy comprised standardized spa treatment, 6 days a week for 3 weeks, and Spa-rehab therapy comprised spa sessions, 3 days a week for 3 weeks, followed by a dedicated rehabilitation program, 3 days a week for 3 weeks. The primary endpoint was achieving at 6 months a minimal clinically important improvement (MCII) for pain on a visual analog scale and/or an MCII for function on the WOMAC index and no knee surgery (composite MCII). Secondary endpoints were composite MCII at 3 months and achieving a Patient Acceptable Symptom State (PASS) for pain and function at 3 and 6 months. Among 283 patients included, 145 were allocated to standard spa therapy and 138 to Spa-rehab therapy. We could not demonstrate the non-inferiority of Spa-rehab therapy for the primary endpoint: difference for responders − 0.08 [90% CI (− 0.18 to 0.02), p = 0.14]. However, the difference test between the groups was not significant (p = 0.18). Spa-rehab therapy was not inferior to standard spa therapy for the composite MCII at 3 months or the PASS at 3 and 6 months. Spa-rehab therapy can reasonably be proposed to patients with symptomatic KOA. This protocol may be more cost-effective than standard spa therapy and avoid absenteeism from work and accommodation costs for patients who live close to a centre.
Similar content being viewed by others
Introduction
Estimates from the 2017 Global Burden of Disease study highlight that hip and knee osteoarthritis (KOA) is one of the leading causes of global disability1. Its population-based prevalence in western countries is increasing2 and estimates of symptomatic KOA prevalence range from 5.4 to 29.8%3,4.
Several professional recommendations for KOA care (European League Against Rheumatism5, American College of Rheumatology6 and the Osteoarthritis Research Society International7,8) highlight the role of non-pharmacological approaches9,10, including weight reduction, education, aerobics, muscle strengthening and water-based exercises and spa therapy5. In a systematic review, aerobic, resistance, and performance exercises had similar effects on reducing pain11. However, the benefit seemed to be short (< 6 months)11,12 and most clinical trials assessing the impact of physical activity (and especially land-based exercise) have a short-term follow-up ranging from 6 to 12 weeks13. High-quality evidence indicates that land-based therapeutic exercise provides short-term benefit in terms of reduced knee pain, and moderate-quality evidence shows improvement in physical function among people with knee OA6. The magnitude of the treatment effect would be considered moderate (immediate) to small (2–6 months) but comparable with estimates reported for non-steroidal anti-inflammatory drugs6.
An approach combining exercises to increase strength, flexibility and aerobic capacity seems to be most effective in managing KOA14.
Several randomized controlled trials (RCTs) have assessed the efficacy of spa therapy for KOA9,15,16,17. The estimated effect size (ES) for spa therapy on pain to support recommendations was 0.46 (0.17–0.75)18, which is moderate but comparable than that for pharmacologic treatments. In a recent systematic review, different types of spa therapy were considered as effective to reduce pain, nonsteroidal anti-inflammatory drug consumption, and functional limitation and to improve quality of life19. However, because spa therapy (defined as thermal baths containing mineral waters and exercising in a health near a mineral or hot spring) is a complex intervention, analyzing its efficacy is difficult and the quality of evidence is only fair10.
Efficacy has been demonstrated with some spa protocols; this is the case for the usual and only reimbursed protocol in France; however, patients often complain of fatigue and the inability to follow such intensive and long spa stays. Moreover, spa protocols for OA have seldom been compared. An outpatient protocol, less intensive, but for a longer time, combining usual spa therapy and rehabilitation care, is an innovative format and could be more appropriate than standard spa therapy for some patients.
In this RCT, we hypothesized that the effect of this modified spa therapy protocol is not inferior to the usual standard protocol but is not clinically superior. The aim was to develop an intervention better suited to patients who do not want to spend 3 weeks off work. Indeed, non-pharmacological treatment and exercise recommendations to patients should focus on the patient’s preferences and access, both of which may be important barriers to participation. If a patient does not find a certain form of care acceptable or cannot afford it, he/she is not likely to pursue this treatment.
The primary objective of the study was to demonstrate the non-inferiority at 6 months of a combined spa therapy and rehabilitation protocol consisting of spa sessions every other day for 3 weeks, followed by rehabilitation sessions 3 days a week for 3 weeks (Spa-rehab therapy) versus the current standard spa protocol that is reimbursed in France: daily spa sessions for 3 consecutive weeks (standard spa therapy). Secondary objectives were to demonstrate the non-inferiority of Spa-rehab therapy versus standard spa therapy at 3 months, to describe the evolution of pain, function, quality of life (QoL), fatigue, drug consumption and satisfaction with the 2 therapies, and to determine the predictive factors of response to therapy at 6 months.
Patients and methods
Study design
We conducted a prospective, randomized, monocenter non-inferiority study comparing Spa-rehab and standard spa therapy for symptomatic KOA (Fig. 1).
Intervention
Standard spa therapy
The standardized spa therapy protocol has been published and has been proven effective for KOA12. Patients received 18 days of spa therapy over 3 weeks, 6 days a week. Each day treatment included 4 techniques: mineral hydrojet sessions at 37 °C for 15 min, manual massages under mineral water at 38 °C by a physiotherapist for 10 min, applications of mineral-matured mud at 45 °C to the knee for 15 min and supervised general mobilization in a collective mineral water pool at 32 °C with groups of 6 patients for 25 min. The different sessions were interspersed with rest periods in a comfortable resting room.
Spa-rehab therapy (spa therapy + rehabilitation program)
Patients received 9 days of spa therapy over 3 weeks, for 3 days a week, with the same 4 techniques as for standard spa therapy. Then, patients followed a 9-day rehabilitation program over the next 3 weeks, for 3 sessions a week (supplemental material). This rehabilitation program aimed to maintain the effect of spa therapy; improve joint mobility, strength and proprioception; and develop appropriate behavior and attitudes. This program was designed by experienced rehabilitative physicians (J Paysant, R Ceconnello, P Boisseau) according to the Société Française de Chirurgie Orthopédique and Société Française de Médecine Physique recommendations20,21 and was personalized and adapted (type and intensity) for each patient. Rehabilitation sessions included 4 sequences: a preliminary sequence based on physiotherapy, personalized work with stretching exercises followed by muscular strengthening of the lower limb, global work with functional and proprioceptive exercises and a collective education session giving information on OA and treatment, and a program of exercises to perform at home. A booklet with all the information was delivered.
The Spa-rehab therapy was designed to improve the availability to follow spa therapy for patients with busy daily lives, to decrease fatigue perception and to extend the spa therapy effects.
All patients continued their usual non-steroidal anti-inflammatory drug (NSAID) or analgesic treatment.
Patients
Inclusion criteria were meeting the American College of Rheumatology criteria for KOA22, unilateral or bilateral OA, pain on a visual analog scale (VAS, 0–100) > 30, and tibiofemoral Kellgren and Lawrence (K&L)23 grade ≥ 2. Exclusion criteria were total knee replacement surgery (TKR) scheduled in the next year, contraindication to spa therapy (evolving cardiovascular conditions, immune deficiency, cancer, current infection), severe comorbidity leading to significant deterioration of QoL, other symptomatic musculoskeletal disease, spa treatment within the previous 12 months, physiotherapy, rehabilitation or knee steroid injection within the past month and hyaluronic acid injection or change in symptomatic slow-acting drug use for OA in the past 3 months. Before the baseline visit, patients were asked not to change their NSAID regimen in the previous 5 days and their painkiller regimen in the previous 12 h.
Patients were recruited in the Lorraine region, east of France, by advertisements in the regional press and by posting flyers in waiting rooms of offices of general practitioners, rheumatologists, and physiotherapists. A free-access telephone number was available to take appointments. Patients were examined for inclusion criteria by rheumatologists working in the regional tertiary care center with no vested interest in the spa.
All patients gave their written informed consent before inclusion. The ethics committee “Comité de Protection des Personnes” (CPP) Est III gave approval for the study (2011-A01319-32; No. CPP: 11.12.02) and the study was registered at ClinicalTrials.gov (NCT01544647; March 6, 2012). The methods were carried out in accordance with the relevant guidelines and regulations. Spa mineral water and treatments were authorized by the French authority.
Randomization
A computer program was used to randomly assign eligible patients to the 2 spa therapies. Because complete blinding of patients is not possible in non-pharmacologic treatments, the Zelen double-consent randomisation technique was used to avoid measurement bias and “resentful demoralisation” linked to not receiving the preferred treatment or another negative effect of preference bias: patients refusing to be included because they are not assigned to their preferred group24. Indeed, some patients could find the active spa therapy too long or not providing enough spa sessions, and some patients could find standard spa therapy too hectic and tiring, which could prevent them from participating because they have to stop their usual activities. Accordingly, patients were randomized and gave their signed inform consent after randomization24,25. Each treatment group had its own consent form and information letter explaining the practical details of the intervention. Also, to avoid measurement bias, treatment in the spa setting was not performed at the same time.
Data collection
After the pre-inclusion visit and randomization, data were collected at baseline (beginning of spa therapy), at 3 weeks (end of spa therapy), 6 weeks (end of rehabilitation therapy for Spa-rehab group) and 3 and 6 months.
Outcome criteria
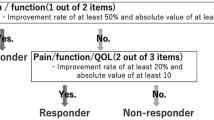
The primary endpoint was achieving at 6 months a minimal clinically important improvement (MCII), defined for KOA as ≥ 19.9 mm on a VAS for pain (0–100), and/or ≥ 9.1 points26 on the Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC) function subscale (0–100)27 and no TKR – the “composite MCII”. Secondary endpoints were achieving at 3 and 6 months an MCII for pain and function and a patient acceptable symptom state (PASS) for pain (≤ 32.3 mm on a VAS for pain (0–100)), or function (≤ 31.0 on the WOMAC function subscale)26; evolution of pain and fatigue scores on a VAS (0–100); Medical Outcomes Survey 36-item Short Form (SF-36)28,29 and OsteoArthritis Knee and Hip Quality Of Life questionnaire (OAKHQOL) scores30; drug consumption; and satisfaction. We thus assessed the core set of patient-reported outcome measures (PROMs) (pain, physical function and patient global assessment) recommended31.
Other data collected
Sociodemographic data (age, gender, education level, marital status, employment status), and clinical data (comorbidity, body mass index, treatments, knee effusion, knee malalignment, previous injury, symptoms duration) were recorded at baseline. Knee X-rays, including a Schuss view of < 6 months, were graded according to the K&L classification23.
At each visit, the spa physician performed a clinical examination and inquired about drug consumption and adverse events (AEs).
Patients were asked to assess their level of pain and fatigue on a VAS (0–100) and to complete a functional ability survey [WOMAC (0–100): higher scores indicating greater severity]27 and QoL questionnaires [SF-36 and OAKHQOL (0–100): lower scores indicating greater severity]29,30. Patients were also questioned about their satisfaction with treatment on a VAS (0–100). The number of sessions each patient attended was recorded. Adherence to the program of exercises to perform at home for the Spa-rehab therapy group was not monitored.
Postural control was also measured by static posturography but reported elsewhere32,33.
Safety analysis
All patients who attended at least one spa therapy session were included in the safety analysis. All AEs were reported whatever their severity. A serious AE was defined as any untoward medical occurrence that results in death, is life-threatening, requires inpatient hospitalization or prolongation of existing hospitalization or results in persistent significant disability or incapacity.
Statistical analysis
Assuming a response rate of 50% with standard spa at 6 months15, a power of 80% and an alpha risk of 5%, we needed 135 patients per group to demonstrate the non-inferiority of Spa-rehab therapy as compared with standard spa therapy, with a lower confidence limit of the difference in proportions of 15%. To allow for an expected 5% attrition rate, we aimed to recruit 284 patients overall. Categorical data are expressed as number (%) and continuous data as mean ± SD. We checked differences between groups at baseline by Student t test and chi-square test as appropriate. Per-protocol analyses were performed to maximize the contrast between the 2 groups34 and intent-to-treat analysis to preserve randomization35. Patients who failed to attend half of the sessions were not included in the per-protocol analysis. As recommended in non-inferiority trials34,36, the 90% CI for the difference was used and the Spa-rehab therapy was considered not inferior to the standard spa therapy if the lower boundary of the 90%CI for the difference between the 2 groups (Spa-rehab minus standard spa therapy), was not < 15% for the primary endpoint (proportion of patients achieving the composite MCII). The same analyses were repeated at 3 months for the proportions of patients achieving the MCII for pain, MCII for function and composite MCII. The proportion of patients achieving the PASS for pain and function was computed before spa therapy and at 3 and 6 months after therapy and compared by the non-inferiority tests as above. The evolution of function and QoL are reported graphically. We computed the standardized ES, equal to the mean change in score from baseline to 6 months, divided by the SD of the baseline score in each group. The relationship between baseline variables (sociodemographic and clinical data, X-rays, season) and proportion of patients achieving the composite MCII was analyzed by logistic regression analysis. Candidate variables with p < 0.1 on bivariate analysis were included in the multivariate models. Variables with p < 0.05 were retained in the models. Between-group comparisons of AEs were analyzed by chi-square test. All analyses involved use of SAS 9.3 (SAS Inst., Cary, NC). p < 0.05 was considered statistically significant.
Results
We screened 566 patients between February and September 2013; 305 were eligible, pre-included and randomized. Overall, 22 patients declined participation before the inclusion visit for logistic reasons (too-long delay between signing the consent form and the intervention or unable to find available time or transportation). Overall, 283 remaining participants were allocated to standard spa (n = 145) or Spa-rehab therapy (n = 138). None of the patients refused the treatment allocated when they signed the consent form.
Twelve patients failed to attend half of the sessions and were not included in the per-protocol analysis, but they were also lost to follow-up, so the population for per-protocol and intent-to-treat analyses was the same. In all, 131 (92.3%) patients with standard spa therapy and 118 (91.5%) with Spa-rehab therapy completed the 6-month follow-up (Fig. 2).
Among the patients included in the analysis, the mean (SD) number of days of spa therapy completed was 17.7 (0.7) in the standard spa therapy group (18 were expected) and 8.7 (0.9) in the Spa-rehab therapy group (9 were expected). Over the 9 days of rehabilitation expected, the mean (SD) number of days completed was 8.7 (0.5).
Patient characteristics
The baseline characteristics of patients are in Table 1. Baseline sociodemographic or disease characteristics of patients did not differ between the groups, except that pain and functional disability was greater with standard than Spa-rehab therapy (Table 1), but the differences were low: pain and functional disability difference of 4.1 and 5.0, respectively, on a 0–100 scale.
As well, patients lost to follow-up or not respecting the protocol (n = 34) and patients completing the study (n = 249) did not differ except for employment status: 17 (50%) were retired among those lost to follow-up and 182 (72.7%) in the completer group (p = 0.007).
One patient in each group underwent TKR between the 3- and 6-month visits.
Primary outcome measure
At 6 months, the 90% CI for the difference in responders between the two groups according to the composite MCII ranged from − 0.18 to 0.02 (p = 0.14), which excluded the non-inferiority of the Spa-rehab therapy (Table 2) (− 0.18 was below the non-inferiority limit of 15%). Analysis was completed with chi-square test assessing the difference between the 2 groups, which was not significant at 6 months (p = 0.18). Adjustment on baseline VAS for pain did not change the result.
Secondary outcome measure
At 3 months, the 90% CI for the difference between the two groups of responders according to the composite MCII ranged from 0.04 to 0.24 (p < 0.0001) (Table 2), which demonstrated the non-inferiority of the Spa-rehab versus standard spa therapy for the composite MCII. The 90% CI for the difference excluded 0, which indicates a significantly greater response with Spa-rehab than standard spa therapy.
The number of patients achieving the PASS increased from baseline to 3 months and then decreased at 6 months, especially with Spa-rehab therapy (Table 3). Non-inferiority was shown for all analyses (p = 0.007 to < 0.0001).
Pain and physical function dimensions of QoL or functional instruments improved at the end of spa treatment (3 weeks). They still improved from 3 to 6 weeks with Spa-rehab therapy and then reached a plateau up to 6 months. Emotional and vitality dimension scores also improved up to 6 weeks (supplemental material). Fatigue decreased with both therapies (Fig. 3).
The ES for the outcome measures between baseline and 6 months for the 2 groups are in Table 4.
The number of patients who took at least one pain killer since the last visit decreased from baseline to 6 months: 72 (50.7%) to 36 (29.0%) with standard spa therapy and 81 (63.3%) to 43 (37.7%) with Spa-rehab therapy. Satisfaction on the VAS was greater with standard than Spa-rehab therapy (p = 0.01) at 3 weeks and was lower at 6 weeks (p = 0.01) (figures a, b, c, supplemental material).
Factors associated with the spa therapy response
Factors independently associated with the number of responders according to the composite MCII were baseline WOMAC function score [OR for each 10-point increase, 1.8, 95% CI (1.5–2.2)] and the season of the 6-month visit [OR 2.7 (1.2–6.2); and 1.4 (0.7–3.1)], for completing the 6-month visit in autumn (n = 85) and winter (n = 88) versus spring (n = 53).
Adverse events (AEs)
No serious AEs linked to the intervention were reported. Four serious AEs not linked to the intervention (ovarian cyst, uncontrolled diabetes, pulmonary embolism, hydrocele) were reported with standard spa therapy and 3 (prolapse, carotid surgery, TKR) with Spa-rehab therapy. These all occurred well after the spa treatment. The AEs are described in supplemental material (table d). Patients mainly reported knee or other musculoskeletal symptoms, fatigue, infections or cutaneous symptoms. The number of AEs did not differ with the 2 therapies (p < 0.2).
Discussion
Non-inferiority of Spa-rehab versus standard spa therapy could not be demonstrated for the main outcome criteria (composite MCII) at 6 months. However, the difference test was not significant. Spa-rehab therapy was not inferior to standard spa therapy for the MCII criteria at 3 months or the PASS at 3 and 6 months.
Spa care or balneotherapy protocols are diverse, but few have demonstrated a clinically significant efficacy and they are rarely compared among themselves37,38,39. In a systematic review, only 9 RCTs analysing thermal mineral water care in KOA were included, with small sample sizes and short follow-up9. A Cochrane review of balneotherapy highlighted the poor quality of studies40. When present, the effect is usually small; however, the ES is comparable to that with medical treatment for OA18.
One large RCT of spa therapy showed a favorable effect on pain and function after 6 months as compared with usual treatment alone. However, the treatment was long and intensive (daily sessions during a 3-week period)15.
With our Spa-rehab therapy, designed to improve availability to follow spa therapy, to decrease fatigue perception and to prolong the spa therapy effects, pain and physical functioning dimensions improved at the end of the spa treatment and still improved from 3 to 6 weeks and then reached a plateau up to 6 months. Patients with Spa-rehab therapy showed better results at 6 weeks and 3 months than those with standard spa therapy, but more patients seemed to lose the clinically significant effect at 6 months. This finding confirms the short effect of rehabilitation11. Fatigue followed the same evolution and was similar in the 2 groups.
We found only 2 factors predicting the achievement of the MCII at 6 months: high WOMAC function score at baseline and season of assessment. The first factor can be explained in part by regression to the mean. The association with season of assessment could be explained by climatic conditions, changes in implementation of the spa protocol, longer delay between inclusion and treatment, and time of the year.
Some weaknesses of the study should be highlighted. First, despite randomization, pain and functional limitations in patients at baseline were slightly higher with standard spa than Spa-rehab therapy. This finding may have been responsible for a greater number of patients achieving the MCII with standard spa than Spa-rehab therapy and the lack of non-inferiority at 6 months41. Second, the results of the trial for the main criteria are difficult to interpret because we could not demonstrate non-inferiority or a difference between interventions. We calculated the sample size with an a priori hypothesis of an expected difference of 0, as is usual in a non-inferiority trial. If the hypothesis of the expected difference was 8%, as we observed, a post-hoc statistical power analysis showed that 600 subjects per group would have been required to demonstrate the non-inferiority, with 577 subjects per group to demonstrate a statistically significant difference from 0. However, a true difference of 8% between the 2 groups is not clinically significant, and we set the meaningful lower limit of non-inferiority at 0.15. The study was monocentric, which may limit the generalizability of the results. Adherence to the program of exercises to perform at home for the Spa-rehab group was not monitored. Finally, the objective of the study was to develop an intervention better suited to patients and easier to follow than the current standard spa protocol of 3 weeks and to demonstrate its non-inferiority but not its mechanism of action. Therefore, we did not investigate how and why the intervention works.
Our study has several strengths. First, PROMs are now widely used, because taking into account patients’ perception of their health state has become a priority. Analyzing the number of responders based on meaningful thresholds is an appropriate way to compare treatments but is rare in spa studies. As now recommended, data were analyzed considering a combination of change (e.g., MCII) and final state (e.g., PASS) to determine the response to the intervention42. Indeed, minimally important difference (MID)-only approaches tend to lack specificity. Few studies have included a number of patients sufficient to analyze the MID, and our study size was among the largest of studies of spa therapy in OA. Finally, the program of rehabilitation combining exercises to increase strength, flexibility and aerobic capacity with a large number of supervised sessions met actual recommendations11.
Future research is needed to determine the best protocol for maintaining work productivity in workers with symptomatic KOA and the intervention with the best cost-effectiveness ratio as compared with usual care. As well, we need to better understand how and why spa therapy is efficient43,44 because numerous elements play a role besides balneotherapy and physical exercises: remoteness from usual life environment, group dynamics, frequent contacts with health care professionals, absence of work duties and personal investment.
In conclusion, we could not demonstrate the non-inferiority of Spa-rehab versus standard spa therapy for the main outcome criteria at 6 months. However, the difference test was not significant. As well, Spa-rehab therapy was not inferior to standard spa therapy for the MCII criteria at 3 months or the PASS at 3 and 6 months. Spa-rehab therapy may be an acceptable alternative to standard spa therapy for patients with symptomatic KOA. This protocol could be cost-effective and allow patients who are still employed to benefit from spa therapy without absenteeism from work or for patients living close to the spa centre to avoid accommodation costs.
Ethics approval and consent to participate
All patients gave their written informed consent before inclusion. The ethics committee CPP Est III gave approval for the study (2011-A01319-32; No. CPP: 11.12.02) and the study was registered at ClinicalTrials.gov (NCT015446476. March, 2012). Spa mineral water and treatments were authorized by the French authority.
References
GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet Lond. Engl. 392, 1789–1858 (2018).
WHO Scientific Group on the Burden of Musculoskeletal Conditions at the Start of the New Millennium. World Health Organ Tech. Rep. Ser. 919, 1–218 (2003).
Guillemin, F. et al. Prevalence of symptomatic hip and knee osteoarthritis: a two-phase population-based survey. Osteoarthr. Cartil. 19, 1314–1322 (2011).
Eumusc. Musculoskeletal Health in Europe. Eumusc (accessed 10 June 2018); https://www.eumusc.net/myUploadData/files/30%2520August%2520v5%2520Final%2520draft%2520report.pdf (2011).
Fernandes, L. et al. EULAR recommendations for the non-pharmacological core management of hip and knee osteoarthritis. Ann. Rheum. Dis. 72, 1125–1135 (2013).
Kolasinski, S. L. et al. 2019 American College of Rheumatology/Arthritis Foundation Guideline for the Management of Osteoarthritis of the Hand, Hip, and Knee. Arthr. Rheumatol. 72, 220–233 (2020).
Zhang, W. et al. OARSI recommendations for the management of hip and knee osteoarthritis, Part II: OARSI evidence-based, expert consensus guidelines. Osteoarthr. Cartil. 16, 137–162 (2008).
Bannuru, R. R. et al. OARSI guidelines for the non-surgical management of knee, hip, and polyarticular osteoarthritis. Osteoarthr. Cartil. 27, 1578–1589 (2019).
Harzy, T., Ghani, N., Akasbi, N., Bono, W. & Nejjari, C. Short- and long-term therapeutic effects of thermal mineral waters in knee osteoarthritis: a systematic review of randomized controlled trials. Clin. Rheumatol. 28, 501–507 (2009).
McAlindon, T. E. et al. OARSI guidelines for the non-surgical management of knee osteoarthritis. Osteoarthr. Cartil. 22, 363–388 (2014).
Juhl, C., Christensen, R., Roos, E. M., Zhang, W. & Lund, H. Impact of exercise type and dose on pain and disability in knee osteoarthritis: a systematic review and meta-regression analysis of randomized controlled trials. Arthr. Rheumatol. 66, 622–636 (2014).
Fransen, M. et al. Exercise for osteoarthritis of the knee. Cochrane Database Syst. Rev. 1, CD004376 (2015).
Zampogna, B. et al. The role of physical activity as conservative treatment for hip and knee osteoarthritis in older people: a systematic review and meta-analysis. J .Clin. Med. 9, 1167 (2020).
Uthman, O. A. et al. Exercise for lower limb osteoarthritis: systematic review incorporating trial sequential analysis and network meta-analysis. BMJ 347, f5555 (2013).
Forestier, R. et al. Spa therapy in the treatment of knee osteoarthritis: a large randomised multicentre trial. Ann. Rheum. Dis. 69, 660–665 (2010).
Kovács, I. & Bender, T. The therapeutic effects of Cserkeszölö thermal water in osteoarthritis of the knee: a double blind, controlled, follow-up study. Rheumatol. Int. 21, 218–221 (2002).
Guillemin, F., Virion, J. M., Escudier, P., De Talance, N. & Weryha, G. Effect on osteoarthritis of spa therapy at Bourbonne-les-Bains. Joint Bone Spine 68, 499–503 (2001).
Zhang, W. et al. OARSI recommendations for the management of hip and knee osteoarthritis: part III: changes in evidence following systematic cumulative update of research published through January 2009. Osteoarthr. Cartil. 18, 476–499 (2010).
Fraioli, A. et al. Efficacy of spa therapy, mud-pack therapy, balneotherapy, and mud-bath therapy in the management of knee osteoarthritis a systematic review. BioMed. Res. Int. 2018, 1042576 (2018).
Delarue, Y., de Branche, B., Anract, P., Revel, M. & Rannou, F. Supervised or unsupervised exercise for the treatment of hip and knee osteoarthritis. Clinical practice recommendations. Ann. Readaptation Med. Phys. Rev. Sci. Soc. Francaise Reeducation Fonct Readaptation Med. Phys. 50, 759–768, 747–758 (2007).
Mazieres, B. et al. Adherence to, and results of, physical therapy programs in patients with hip or knee osteoarthritis. Development of French clinical practice guidelines. Joint Bone Spine 75, 589–596 (2008).
Altman, R. et al. Development of criteria for the classification and reporting of osteoarthritis. Classification of osteoarthritis of the knee. Diagnostic and Therapeutic Criteria Committee of the American Rheumatism Association. Arthritis Rheum. 29, 1039–49 (1986).
Kellgren, J. H. & Lawrence, J. S. Radiological assessment of osteo-arthrosis. Ann. Rheum. Dis. 16, 494–502 (1957).
Adamson, J., Cockayne, S., Puffer, S. & Torgerson, D. J. Review of randomised trials using the post-randomised consent (Zelen’s) design. Contemp. Clin. Trials 27, 305–319 (2006).
Schellings, R. et al. Indications and requirements for the use of prerandomization. J. Clin. Epidemiol. 62, 393–399 (2009).
Tubach, F. et al. Evaluation of clinically relevant states in patient reported outcomes in knee and hip osteoarthritis: the patient acceptable symptom state. Ann. Rheum. Dis. 64, 34–37 (2005).
Bellamy, N., Buchanan, W. W., Goldsmith, C. H., Campbell, J. & Stitt, L. W. Validation study of WOMAC: a health status instrument for measuring clinically important patient relevant outcomes to antirheumatic drug therapy in patients with osteoarthritis of the hip or knee. J. Rheumatol. 15, 1833–1840 (1988).
Leplege, A., Ecosse, E., Verdier, A. & Perneger, T. V. The French SF-36 Health Survey: translation, cultural adaptation and preliminary psychometric evaluation. J. Clin. Epidemiol. 51, 1013–1023 (1998).
Kosinski, M., Keller, S. D., Hatoum, H. T., Kong, S. X. & Ware, J. E. Jr. The SF-36 Health Survey as a generic outcome measure in clinical trials of patients with osteoarthritis and rheumatoid arthritis: tests of data quality, scaling assumptions and score reliability. Med. Care 37, 10–22 (1999).
Rat, A.-C. et al. OAKHQOL: a new instrument to measure quality of life in knee and hip osteoarthritis. J. Clin. Epidemiol. 58, 47–55 (2005).
McAlindon, T. E. et al. OARSI Clinical Trials Recommendations: design, conduct, and reporting of clinical trials for knee osteoarthritis. Osteoarthr. Cartil. 23, 747–760 (2015).
Zhang, Z. et al. Diurnal variation on balance control in patients with symptomatic knee osteoarthritis. Arch. Gerontol. Geriatr. 61, 109–114 (2015).
Peultier, L. et al. Influence of meteorological elements on balance control and pain in patients with symptomatic knee osteoarthritis. Int .J. Biometeorol. 61, 903–910 (2017).
Elie, C., De Rycke, Y., Jais, J.-P., Marion-Gallois, R. & Landais, P. Methodological and statistical aspects of equivalence and non inferiority trials. Rev. Epidemiol. Sante Publique 56, 267–277 (2008).
Wiens, B. L. & Zhao, W. The role of intention to treat in analysis of noninferiority studies. Clin. Trials 4, 286–291 (2007).
Piaggio, G. et al. Reporting of noninferiority and equivalence randomized trials: extension of the CONSORT 2010 statement. JAMA 308, 2594–2604 (2012).
Kamioka, H. et al. Effectiveness of aquatic exercise and balneotherapy: a summary of systematic reviews based on randomized controlled trials of water immersion therapies. J. Epidemiol. 20, 2–12 (2010).
Vaht, M., Birkenfeldt, R. & Ubner, M. An evaluation of the effect of differing lengths of spa therapy upon patients with osteoarthritis (OA). Complement. Ther. Clin. Pract. 14, 60–64 (2008).
Verhagen, A. P., Cardoso, J. R. & Bierma-Zeinstra, S. M. A. Aquatic exercise & balneotherapy in musculoskeletal conditions. Best Pract. Res. Clin. Rheumatol. 26, 335–343 (2012).
Verhagen, A. P. et al. Balneotherapy for osteoarthritis. Cochrane Database Syst. Rev. https://doi.org/10.1002/14651858.CD006864 (2007).
Copay, A. G., Subach, B. R., Glassman, S. D., Polly, D. W. Jr. & Schuler, T. C. Understanding the minimum clinically important difference: a review of concepts and methods. Spine J. 7, 541–546 (2007).
Beaton, D. E. et al. Minimal change is sensitive, less specific to recovery: a diagnostic testing approach to interpretability. J .Clin. Epidemiol. 64, 487–496 (2011).
Blaise, P., Marchal, B., Lefèvre, P., Kegels, G. In Au-delà des méthodes expérimentales: l'approche réaliste en évaluation. Réduire les inégalités sociales en santé, pp 285–296 (eds Potvin, L., Moquet, M. J., & Jones, C. M.) (Institut National de Prévention et d'Education pour la Santé (INPES), Saint-Denis, 2010) (Dossiers Santé en Action).
Pawson, R. & Tilley, N. Realistic Evaluation (Sage, Beverly Hills, 1997).
Acknowledgements
R. Aussedat, R. Ceconnello, P. Boisseau, P. Perrin, V. Royaux, S. Ruscade-Cholay. This study was conducted with the support of Inserm CIC-EC 1433 Nancy. This study was funded by the Grand Nancy. Opinions expressed in the present article are those of the authors and do not necessarily reflect those of the sponsors. The study sponsors did not take part in the study design, collection, analysis and interpretation of data, writing of the report or the decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
A.C.R.: conceived and designed the study, collected data, analysed and interpreted the data, drafted the manuscript. D.L.: conceived and designed the study, collected data, analysed and interpreted the data, helped to revise the manuscript. A.V.: coordinated the study, analysed and interpreted the data, helped to revise the manuscript. L.B.: did the statistical analyses, analysed and interpreted the data, helped to revise the manuscript. E.S.: collected data, helped to revise the manuscript. A.D.: collected data, helped to revise the manuscript. M.B.: conceived and designed the study, helped to revise the manuscript. J.P.: conceived and designed the study, helped to revise the manuscript. F.G. conceived and designed the study, analysed and interpreted the data, helped to revise the manuscript. I.C.-V.: conceived and designed the study, collected data, analysed and interpreted the data, helped to revise the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Rat, AC., Loeuille, D., Vallata, A. et al. Spa therapy with physical rehabilitation is an alternative to usual spa therapy protocol in symptomatic knee osteoarthritis. Sci Rep 10, 11004 (2020). https://doi.org/10.1038/s41598-020-67436-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-020-67436-1
This article is cited by
-
Characteristics of patients with knee and/or hip osteoarthritis undergoing spa treatment: the prospective KHOALA cohort study
International Journal of Biometeorology (2022)
-
Importance of the duration of treatment in Balneotherapy
International Journal of Biometeorology (2021)
-
It is not just balneotherapy; it is spa therapy consisting of balneological treatments including balneotherapy as well
International Journal of Biometeorology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.