Abstract
This study aimed to assess characteristics of pregnant women taking antiepileptic drugs with inadequate folic acid intake. This cross-sectional study examined pregnant women taking antiepileptic drugs who were registered in the Japanese Drug Information Institute in Pregnancy (JDIIP) database between October 2005 and December 2016. Participants were classified into two groups according to when they started folic acid supplementation (before pregnancy: ‘adequate’, after pregnancy or never: ‘inadequate’). Logistic regression analysis was performed to investigate factors associated with inadequate folic acid intake. Of 12,794 registrants, 468 pregnant women were taking antiepileptics during the first trimester. Of these, we analysed data from 456 women who had no missing data. As a result, inadequate folic acid intake was noted among 83.3% of them, suggesting that the current level of folic acid intake is insufficient overall. Younger age, smoking, alcohol drinking, multiparity, unplanned pregnancy, and being prescribed AEDs by paediatric or psychiatric departments were independent factors associated with inadequate folic acid intake. As unplanned pregnancy was the strongest factor, healthcare professionals should ensure that childbearing women taking antiepileptics are informed of the importance of planned pregnancy. In addition, healthcare professionals must gain a better understanding of folic acid intake, as the prevalence of adequate intake differed according to which departments prescribed antiepileptic drugs.
Similar content being viewed by others
Introduction
Perinatal use of antiepileptic drugs (AEDs) increases the risk for both congenital malformation and neurodevelopmental impairment in children1,2. The risk varies by type of AED and dosage. A comparison of different AEDs revealed that children exposed to valproate sodium had the greatest risk of malformation1, with an even higher risk for those whose mothers underwent AED polytherapy as opposed to monotherapy3. The presence of physical abnormalities in children of women with epilepsy was more dependent on the use of AEDs during pregnancy than epilepsy itself4. Therefore, perinatal AED use should be approached with great caution, as most women with epilepsy must continue AED treatment during pregnancy to ensure adequate seizure control. Notably, AEDs are used to treat a broader range of diseases during pregnancy aside from epilepsy, including headache, neuropathic pain, bipolar disorder, and other mental disorders5. Valproate sodium is the only AED with specific instructions to avoid use in childbearing women to the extent possible even in monotherapy in various clinical guidelines6,7,8, and the need to select AEDs with caution is stressed when used in polytherapy6,7.
Folic acid supplementation before conception reduces the incidence of neural tube defects in infants9,10,11. Preconceptional folic acid supplementation is recommended for childbearing women, as it is difficult to ensure adequate folate intake solely through diet. In addition, synthetic folic acid has a higher bioavailability than the natural form of folate. In particular, fertile women undergoing AED treatment should take folic acid, because some AEDs promote folate metabolism and lower folic acid blood levels12, which may contribute to the impaired cognitive function in children and foetal malformation including neural tube defects. Folic acid supplementation has also been found to reduce the risk of autism in children exposed to AEDs during pregnancy13.
Accordingly, childbearing women undergoing AED treatment should ensure that their folic acid intake is adequate. Unfortunately, the current status of folic acid intake among pregnant women taking AEDs is unclear. The present study aimed to clarify folic acid intake among pregnant women taking AEDs and determine characteristics of those for whom intake is inadequate.
Results
Setting and participants
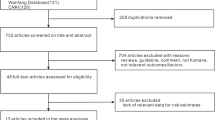
Data were collected from women registered in the Japan Drug Information Institute in Pregnancy (JDIIP) database. JDIIP provides consultations and collects data on “the influence of medication on pregnant women and nursing mothers/foetuses”. Of the 12,794 registrants in the JDIIP database from October 2005 to December 2016, 468 pregnant women were taking AEDs during the first trimester; we analysed data from 456 of these women who had no missing data. Twelve women were excluded as they were missing data for the following items: folic acid intake (n = 6), planned pregnancy (n = 5), fertility treatment (n = 5), body mass index (n = 1), parity (n = 1), and history of spontaneous and artificial abortion (n = 1) (Fig. 1).
Participant characteristics
Maternal characteristics are shown in Table 1. Of our study population, 35.1% had planned pregnancies (65.8% among those with adequate folic acid intake, 34.6% among women who started folic acid supplementation after finding out that they were pregnant, and 24.9% among those with no folic acid intake). Mental and behavioural disorders (59.9%) were the most common diseases under treatment, followed by epilepsy (39.0%). AED treatment during the first trimester (Table 2) was undergone as monotherapy in 85.1% and polytherapy in 14.9%. Valproate sodium was the most frequently taken AED (49.3%). The prevalence of valproate sodium use among women with and without planned pregnancies was 37.5% and 55.7%, respectively. Among 225 participants taking valproate sodium, epilepsy, mood disorders, and other diseases with neither epilepsy nor mood disorders were noted as the diseases under treatment by 28.9%, 44.4%, and 35.6%, respectively (Table 3).
Folic acid intake
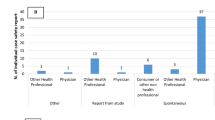
Folic acid intake included that for both supplements available over-the-counter as well as prescription folic acid. Among all participants, 76 (16.7%) demonstrated adequate folic acid intake before pregnancy, 159 (34.9%) began supplementation after pregnancy, and 221 (48.5%) did not take any (Table 1). Those who received folic acid prescriptions from medical institutions comprised 89 participants (19.5%). Daily doses of folic acid prescription ranged from 0.5 to 20 mg, with 4 mg or more in 70 participants and unclear doses in 5 participants. For supplements available over-the-counter, the exact number of tablets taken daily was recorded only for 3 of 152 participants. Figure 2 shows folic acid intake according to department prescribing AEDs. Most participants were prescribed AEDs by the department of psychiatry (n = 154), followed by neurosurgery (n = 53), neurology (n = 52), and psychosomatic medicine (n = 50). 117 women left blank the department that prescribed AEDs (a “blank” answer meant either “unanswered” or “department other than those listed as options”). The prevalence of adequate folic acid intake ranged from 0 to 33.3% depending on the departments that prescribed AEDs.
Folic acid intake according to department prescribing antiepileptic drugs. The following departments are not shown. n = 2; Obstetrics and gynaecology. n = 1; Dentistry, Otorhinolaryngology, Rheumatology (all subjects had no folic acid supplementation), Headache clinic, and Anaesthesiology (all subjects had adequate folic acid supplementation).
Characteristics of participants with inadequate folic acid intake
Multivariate analysis identified younger age, smoking, alcohol drinking, multiparity, unplanned pregnancy, and being prescribed AEDs by paediatric or psychiatric departments as independent factors associated with inadequate folic acid intake (Table 4). Multicollinearity between explanatory variables was not observed as the maximum of variance inflation factor was 2.18.
Discussion
The present study examined folic acid intake and determined characteristics of those with inadequate folic acid intake among pregnant women taking AEDs, using data from the JDIIP database. Of our study population, 83.3% did not take folic acid before pregnancy. These women with inadequate folic acid intake were more likely to be younger, have a history of smoking or alcohol drinking, multiparous, prescribed AEDs by paediatric or psychiatric departments, and less likely to have a planned pregnancy. The prevalence of adequate folic acid intake ranged from 0 to 33.3% in the various departments that had prescribed AEDs. Mental and behavioural disorders were the most common diseases for which AEDs were prescribed as treatment, followed by epilepsy.
Folic acid intake, including both supplemental over-the-counter and prescription folic acid, was insufficient among pregnant women taking AEDs. Although the prevalence of inadequate folic acid intake in the present study (83.3%) was lower than that of general women in a nationwide population-based cohort study in Japan (92.6%)14, the current state of folic acid intake should be improved given that children of pregnant women taking AEDs are at greater risk of developing congenital malformations than those of general childbearing women. Participants who started folic acid intake after pregnancy (comprising one-third of all participants) intended to take folic acid as well as those with adequate folic acid intake. Nonetheless, these participants did not start folic acid intake before finding out that they were pregnant and thus the full effect of folic acid supplementation cannot be expected in such cases. One reason for this may be related to whether or not the pregnancy was planned (planned pregnancies were noted for 65.8% with adequate folic acid intake and 34.6% of those starting folic acid after pregnancy). Further investigation is required to identify factors to explain why some women did not take any folic acid.
Women with planned pregnancies were more likely to have adequate folic acid intake. This was consistent with results from previous population-based studies15,16,17 and a hospital-based study18 of general pregnant women. In the present study, a planned pregnancy was the strongest factor associated with adequate folic acid intake. Another study found a non-significant association between planned pregnancy and prescription of folic acid, but women with epilepsy who had planned pregnancies tended to have lower foetal exposure to AEDs through the selection of monotherapy and by avoiding valproate sodium use19. The present study also found a lower prevalence of valproate sodium use in those with planned pregnancies than in those with unplanned pregnancies (37.5% versus 55.7%, respectively), even though the prevalence of AED polytherapy was comparable in both of these groups, at approximately 15%. Having a planned pregnancy may improve the mother’s behaviour, such as preconceptional folic acid intake and AED adjustment to allow for lower teratogenic risk. On the other hand, since unplanned pregnancies currently represent the majority, a system may need to be developed to guide women toward adequate folic acid intake prior to attempting to get pregnant. One potential solution would be to implement mandatory national folic acid fortification, as is done in many other countries20, but not currently in Japan.
The prevalence of adequate folic acid intake differed by the department that prescribed AEDs. Those who were prescribed AEDs by paediatric or psychiatric departments were less likely to have adequate folic acid intake, potentially due to differences in attitudes among healthcare professionals about the benefits of preconceptional folic acid intake or in preparation for unexpected pregnancies. Although medical treatment policies are not solely dependent on guidelines, actual clinical practice in each department might differ depending on the contents of guidelines and recommendations. In Japan, preconceptional folic acid intake for childbearing women requiring AED treatment is recommended in guidelines for epilepsy6 and chronic headaches7, but is not mentioned in guidelines for neuropathic pain8, bipolar disorder21, or schizophrenia22. An increased awareness about folic acid is needed among healthcare professionals, as well as among women of childbearing age, and more information must be provided about folic acid supplementation in disease guidelines. Targeted healthcare professionals would include physicians prescribing AEDs as well as those in various other occupations, e.g., pharmacists obligated to refer to doctors when there is any doubt about prescriptions, nurses assisting in medical treatment, and public health nurses performing educational activities. Notably, interpretation of our findings about folic acid intake according to department prescribing AEDs should be made with caution, since approximately 25% of participants left blank the answer regarding the department that prescribed AEDs.
Other characteristics of inadequate folic acid intake identified in this study included younger age, multiparity, and a history of smoking and/or alcohol drinking. These characteristics were consistent with those identified by other studies of general pregnant women14,15,16,17. Younger women were less likely to have planned pregnancies17 and knowledge of folic acid and its benefits23, demonstrating the necessity of school health education on this subject as well as continuous efforts to increase the awareness of the importance of folic acid intake.
One questionnaire study revealed that pregnant women obtained information on folic acid through mass media (47%), the Internet (17%), healthcare providers (13%), and the maternal and child health handbook (11%)24. The maternal and child health handbook is a booklet given to pregnant women in Japan, contains information on safe pregnancy, childbirth, and children’s health, and is used to record their own health conditions as well as growth parameters of their children. Given the popularity of the Internet and mass media to obtain this information, it may be most effective to use these resources to increase awareness about folic acid intake among pregnant women and those considering pregnancy. Typically, communication with maternity healthcare providers begins after the pregnancy is confirmed, or if pregnancy issues such as spontaneous abortion occur. In addition, women receive their maternal and child health handbooks only after a healthcare professional confirms their pregnancy. Therefore, the impact of promoting preconceptional folic acid intake by either healthcare professionals or through information in the maternal and child health handbook might be limited.
Our study also found inappropriate use of valproate sodium among pregnant women taking AEDs. Perinatal valproate sodium use has been reported to increase the risk of congenital malformations1 and decrease IQ scores in children25. Indications for valproate sodium use during pregnancy are limited to those with epilepsy or bipolar disorder (a type of mood disorder), and the drug should only be prescribed if all other medications are ineffective in treating the condition or if there are no other alternatives. However, the present study found that 35.6% of pregnant women taking valproate sodium reported diseases other than epilepsy or mood disorders as diseases under treatment. Use of valproate sodium to prevent migraines is contraindicated for pregnant women because the risks outweigh any benefits. On a related note, all non-pregnant women of childbearing age taking valproate sodium to prevent migraines should use effective birth control26. Patients with chronic pain disorders or mental disorders other than mood disorders should also be treated in accordance with this policy.
The present study had some limitations worth noting. First, our participants may not be representative of all pregnant women using AEDs, as they had taken some AEDs during pregnancy and voluntarily consulted the JDIIP. That said, the proportion of AED users among pregnant women is very low (0.2 to 2.0%)5,27,28, and the present study included more than 450 participants, providing highly valuable data. Second, for supplements available over-the-counter, the exact number of tablets taken daily was recorded only by 3 participants because supplements were not considered medication. However, we estimated their daily doses of folic acid to be approximately 0.4 mg based on records of the product names. Third, since the term “before/after pregnancy” as it applies to when folic acid supplementation began was not strictly defined in the self-report questionnaires, potential misclassification might lead to overesting the prevalence of adequate folic acid intake. The accuracy of information could be improved with further efforts to reduce misclassification and missing data. Fourth, we did not consider the effects of marital status, educational history, or income, all of which are important factors related to folic acid intake. Despite these limitations, we gained valuable insight on pregnant women taking AEDs to treat diseases other than epilepsy because we did not collect data from registries limited to patients with epilepsy.
In conclusion, folic acid intake is currently insufficient among pregnant women taking AEDs. Younger age, smoking, alcohol drinking, multiparity, unplanned pregnancy, and being prescribed AEDs by paediatric or psychiatric departments were independent factors associated with inadequate folic acid intake. Having a planned pregnancy was the strongest factor associated with adequate folic acid intake, and thus healthcare professionals should educate women of childbearing age taking AEDs about the importance of planned pregnancy. Awareness among healthcare professionals including clinicians and pharmacists, among others, should also be increased, as the prevalence of adequate folic acid intake differed depending on which department prescribed AEDs.
Methods
Study design and setting
This is a cross-sectional study of data from the JDIIP database. The JDIIP, a project approved by the Ministry of Health, Labour and Welfare, was conducted by the National Center for Child Health and Development beginning in October 2005.
Participants
Pregnant women who used AEDs during the first trimester of pregnancy and who were registered in the JDIIP database from October 2005 to December 2016 were eligible to participate. Those with missing data for items about maternal characteristics and folic acid intake were excluded. AEDs used by participants included carbamazepine, ethosuximide, gabapentin, lacosamide, lamotrigine, levetiracetam, perampanel, phenytoin, phenobarbital, primidone, topiramate, valproate sodium, and zonisamide. Oxcarbazepine was not included since it has not been released in Japan.
Data collection
Women desiring a consultation completed a questionnaire form containing information on their medication use and mailed it to apply for consultation. Consultations could occur by three ways: (1) By telephone: a physician/pharmacist responds to common consultations about items such as cold medicine and analgesics; (2) A woman visits one of the 39 JDIIP outpatient clinics nationwide and a medical specialist responds to the consultation; or (3) A woman visits her attending physician, who then provides an explanation based on the response from the JDIIP.
Items in the database were maternal age, height, body weight before pregnancy, smoking status, alcohol drinking, parity, medical history, fertility treatment, planned pregnancy, medication use (product name, generic name, date of start and discontinuation, daily dose, department that prescribed the medication), and folic acid intake (both prescription and supplementary over-the-counter; for the latter, product name and timing of initiation), as collected from the questionnaire surveys. In addition, some women agreed to return a postcard survey on neonatal outcomes to indicate outcomes including congenital malformation. Women who reportedly never smoked or who quit smoking before pregnancy (i.e., before the day the last menstrual period started) were defined as non-smokers and women who reported that they were still smoking or had smoked until pregnancy confirmation were defined as smokers. The same guidelines were used to define alcohol drinking. The definition of a planned pregnancy was that the woman intended her pregnancy. If there was any ambiguity in the description of the questionnaire, research coordinators either confirmed by phone, or pharmacists or doctors provided confirmation at the consultation.
Outcome and statistical analysis
We assessed maternal characteristics and AED treatment during the first trimester (from 0 weeks 0 days until 13 weeks 6 days of gestation). As maternal characteristics, we extracted information on age, body mass index, smoking status, alcohol drinking, parity, history of spontaneous and artificial abortion, fertility treatment, planned pregnancy, and diseases under treatment. With regard to AED treatment during the first trimester, we extracted information on type of AEDs (e.g., valproate sodium), date of AED use, monotherapy or polytherapy, and the department that prescribed AEDs.
The outcome was prevalence of folic acid intake. We classified participants into two groups, depending on when folic acid intake began: those who began folic acid supplementation before pregnancy were defined as ‘adequate’ intake, while those who started after pregnancy or who never started (no intake) were defined to have ‘inadequate’ intake. Univariable and multivariable logistic regression analyses were performed to calculate the odds ratio and the 95% confidence interval of the association between maternal characteristics as explanatory variables and inadequate intake of folic acid as the outcome variable. Multicollinearity among explanatory variables was confirmed by variance inflation factors. All statistical analyses were conducted with JMP Pro 13.0 (SAS Institute Inc., Cary, NC, USA).
Ethical considerations
Researchers and other personnel could not identify individuals from analytical data, as the information was input by a research coordinator based on questionnaire forms and postcard surveys for neonatal outcomes. Data were anonymised by an information administrator in the JDIIP. Written informed consent regarding the construction of the JDIIP database was obtained from all subjects. For the purpose of the present study, the opt-out was adopted (we disclosed the information on this study and provided potential participants with the opportunity to refuse to participate). All procedures were conducted in accordance with ethical guidelines for medical and health research involving human subjects established by the Ministry of Health, Labour and Welfare in Japan. All study protocols were approved by the ethics committees of the National Center for Child Health and Development (No. 1502) and Kyoto University (No. R1244-1).
Data Availability
Datasets generated during the current study are not publicly available due to a lack of agreement from participants and lack of approval from the ethics committees regarding data provision to third parties. However, the datasets are available from the corresponding author on reasonable request for collaborative research and with permission from the National Center for Child Health and Development.
Change history
14 April 2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
References
Weston, J. et al. Monotherapy treatment of epilepsy in pregnancy: congenital malformation outcomes in the child (Review). Cochrane Database Syst. Rev. Available at: https://doi.org/10.1002/14651858.CD010224.pub2 (2016).
Bromley, R. et al. Treatment for epilepsy in pregnancy:neurodevelopmental outcomes in the child. Cochrane Database Syst. Rev. Available at: https://doi.org/10.1002/14651858.CD010236.pub2 (2014).
Montouris, G. Importance of monotherapy in women across the reproductive cycle. Neurology 69, S10–S16 (2007).
Holmes, L. B. et al. The teratogenicity of anticonvulsant drugs. N. Engl. J. Med. 344, 1132–1138 (2001).
Bobo, W. V. et al. Trends in the use of antiepileptic drugs among pregnant women in the U.S., 2001–2007: a medication exposure in pregnancy risk evaluation program (MEPREP) study. Paediatr. Perinat. Epidemiol. 26, 578–588 (2012).
Clinical Practice Guideline for Epilepsy Development Committee. Clinical Practice Guideline for Epilepsy 2010. Igakushoin (2010).
Chronic Headache Clinical Practice Guideline Development Committee. Clinical Practice Guideline for Chronic Headache 2013. Igakushoin (2013).
The Committee for the Guidelines for the Pharmacologic Management of Neuropathic Pain (Second Edition) of JSPC. The Committee for the Guidelines for the Pharmacologic Management of Neuropathic Pain Second Edition. Shinko Trading Company Ltd., Publication Department of Medical Books (2016).
Berry, R. J. et al. Prevention of neural-tube defects with folic acid in China. N. Engl. J. Med. 341, 1485–1490 (1999).
Czeizel, A. E. & Dudas, I. Prevention of the first occurence of neural-tube defects by periconceptional vitamin supplementation. N. Engl. J. Med. 327, 1832–1835 (1992).
MRC Vitamin Study Resarch Group. Prevention of neural tube defects: results of the medical research council vitamin study. Lancet 338, 131–137 (1991).
Sener, U. et al. Effects of common anti-epileptic drug monotherapy on serum levels of homocysteine, vitamin B12, folic acid and vitamin B6. Seizure 15, 79–85 (2006).
Bjørk, M. et al. Association of folic acid supplementation during pregnancy with the risk of autistic traits in children exposed to antiepileptic drugs in rtero. JAMA Neurol. 75, 160–168 (2018).
Obara, T. et al. Prevalence and determinants of inadequate use of folic acid supplementation in Japanese pregnant women: the Japan environment and children’s study (JECS). J. Matern. Fetal Neonatal Med. 30, 588–593 (2017).
Timmermans, S. et al. Determinants of folic acid use in early pregnancy in a multi-ethnic urban population in the Netherlands: the generation R study. Prev. Med. 47, 427–432 (2008).
Nilsen, R. M. et al. Patterns and predictors of folic acid supplement use among pregnant women: the Norwegian mother and child cohort study. Am. J. Clin. Nutr. 84, 1134–1141 (2006).
Rosenberg, K. D., Gelow, J. M. & Sandoval, A. P. Pregnancy intendedness and the use of periconceptional folic acid. Pediatrics 111, 1142–1145 (2003).
Paulik, E., Császár, J., Kozinszky, Z. & Nagymajtényi, L. Preconceptional and prenatal predictors of folic acid intake in Hungarian pregnant women. Eur. J. Obstet. Gynecol. Reprod. Biol. 145, 49–52 (2009).
Abe, K. et al. Impact of planning of pregnancy in women with epilepsy on seizure control during pregnancy and on maternal and neonatal outcomes. Seizure 23, 112–116 (2014).
Copp, A. J. et al. Spina bifida. Nat. Rev. Dis. Prim. 1, 15007 (2015).
The Committee for Treatment Guidelines of Mood Disorders (the Japanese Society of Mood Disorders). Guideline for Treatment of Bipolar Disorder 2012. Available at: http://minds4.jcqhc.or.jp/minds/utsu/bipolardisorder2012.pdf (2012).
The Japanese Society of Neuropsychopharmacology. Guideline for Pharmacological Therapy of Schizophrenia. Igakushoin (2016).
Kim, M. J. et al. Awareness, knowledge, and use of folic acid among non-pregnant Korean women of childbearing age. Nutr. Res. Pract. 12, 78–84 (2018).
Kondo, A. et al. Folic acid reduces the risk of neural tube defects: awareness and folate intake among pregnant women in 2006. Hinyoukika Kiyo 54, 537–542 (2008).
Meador, K. J. et al. Fetal antiepileptic drug exposure and cognitive outcomes at age 6 years (NEAD study): a prospective observational study. Lancet Neurol. 12, 244–252 (2013).
U.S. Food and Drug Administration. FDA drug safety communication: valproate anti-seizure products contraindicated for migraine prevention in pregnant women due to decreased IQ scores in exposed children. Available at: https://www.fda.gov/Drugs/DrugSafety/ucm350684.htm (2013).
Nishigori, H. et al. Drug Use before and during pregnancy in Japan: the Japan environment and children’s study (JECS). Pharmacy 5, 1–18 (2017).
Charlton, R. et al. Antiepileptic drug prescribing before, during and after pregnancy: a study in seven European regions. Pharmacoepidemiol. Drug Saf. 24, 1144–1154 (2015).
Acknowledgements
This work was supported by a grant from the Japan Agency for Medical Research and Development (Grant number: 17mk0101086h0001). We are grateful to everyone involved with the JDIIP database. In particular, we thank Ken Nakajima, Kunihiko Takahashi, and Takahiro Yamada for their support.
Author information
Authors and Affiliations
Contributions
T.O., M.G. and A.M. acquired the data and prepared the JIIDP database. A.M. obtained research funding. Y.I.S. and T.N. designed the study. Y.T. provided statistical advice. Y.I.S. and Y.S. analysed the data. T.S., T.O. and H.H. contributed to data interpretation. Y.I.S. drafted the manuscript. All authors contributed to the revision of the manuscript and approved the final version.
Corresponding author
Ethics declarations
Competing Interests
The spouse of Y.I.S. is an employee of Daiichi Sankyo Co., Ltd. M.G. received fees from Chugai Pharmaceutical Co., Ltd. for work unrelated to the present manuscript. T.N. received consultant fees from Ohtsuka Pharamaceutical Co., Ltd. and Dainippon Sumitomo Pharmaceutical Co., Ltd., donations from Nakamura Hospital, the Japan Medical Data Center, and HANSHIN Dispensing Holding Co., Ltd., and personal fees from Ono Pharamaceutical Co., Ltd., Chugai Pharamaceutical Co., Ltd., Dentsu Inc., Takeda Pharamaceutical Co., Ltd., Novo Nordisk Pharma. Ltd., Janssen Pharmaceutical K.K., and Boehringer Ingelheim International GmbH for work unrelated to the present manuscript. A.M. received grants from Chugai Pharmaceutical Co., Ltd. and Astellas Pharma Inc., and personal fees from Chugai Pharmaceutical Co., Ltd., Astellas Pharma Inc., and Mitsubishi Tanabe Pharma Corporation during the conduct of the study. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Ikeda-Sakai, Y., Saito, Y., Obara, T. et al. Inadequate Folic Acid Intake Among Women Taking Antiepileptic Drugs During Pregnancy in Japan: A Cross-Sectional Study. Sci Rep 9, 13497 (2019). https://doi.org/10.1038/s41598-019-49782-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-49782-x
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.