Abstract
Diploid strawberry (Fragaria vesca ‘Baiguo’) is a model plant for studying functional genomics in Rosaceae. Adventitious shoot regeneration is essential for functional genomics by Agrobacterium tumefaciens-mediated transformation. An efficient shoot regeneration method using diploid strawberry leaf explants was conducted on 1/2MS + 1/2B5 medium that contained 2.0 mg L−1 TDZ over 14 days of dark culture; this induced the maximum percentage of shoot regeneration (96.44 ± 1.60%) and the highest number of shoots per explant (23.46 ± 2.14) after 11 weeks of culture. The explants considerably enlarged after 12 days; then, turned greenish brown after 30 days, yellowish brown after 36 days, and completely brown and necrotic after 48 days. Large numbers of adventitious shoots were produced from 48 to 66 days, and the shoots elongated from 66 to 78 days; this represents a critical period of reinvigoration, which included 30 days for leaf explant chlorosis, 36 days for adventitious shoot appearance, and 48 days for generation of numerous shoots. During the reinvigoration process, higher expressions of the hormone synthesis-related genes Ciszog1, CKX2, CKX3, CKX7, YUC2, YUC6, YUC10, YUC9, and GA2ox were detected from 30 to 48 days. Our results indicate that these genes may regulate reinvigoration of shoot regeneration.
Similar content being viewed by others
Introduction
Diploid strawberry (Fragaria vesca), also known as the woodland strawberry, has a small genome (2n = 2× = 14, 240 Mbp), easy vegetative propagation, and a small, herbaceous stature1. These advantages make diploid strawberry more suitable as an attractive model than the commercial octoploid strawberry (Fragaria × ananassa) (2n = 8× = 56) and other plants of Rosaceae for functional genomics research2. Adventitious shoot regeneration is essential for subsequent functional genomics research by Agrobacterium tumefaciens-mediated transformation and is affected by internal and external influences.
In strawberry, the effects of basic medium3, plant growth regulators4,5,6, explant types6,7, cultivars5,7, and duration of dark culture8 have been demonstrated on adventitious shoot regeneration of leaf explants. In basic medium, the shoots that developed from the buds were subcultured onto 1/2 Murashige and Skoog (MS) + 1/2B5 + 0.2 mg L−1 6-benzyladenine (BA) + 0.1 mg L−1 indole-3-butyric acid (IBA), MS + Fe-EDDHA (Ethylenediamine di-2-hydroxyphenyl acetate ferric)9 + 0.2 mg L−1 BA + 0.1 mg L−1 IBA, and 1/2MS + 0.2 mg L−1 BA + 0.1 mg L−1 IBA. Diploid strawberry grew best in 1/2MS + 1/2B5 + 0.2 mg L−1 BA + 0.1 mg L−1 IBA (Fig. S1). When exposed to plant growth regulator, the best shoot regeneration frequency in cultivated octoploid strawberry (F. ananassa cv. ‘Honeoye’), 94.7%, was obtained on MS medium supplemented with 2.0 mg L−1 thidiazuron (TDZ)8. TDZ + IBA promoted the highest shoot regeneration efficiencies in leaves of nearly all genotypes, whereas the TDZ/3-Benzo[b] selenienyl acetic acid (BSAA) and TDZ/2,4-Dichlorophenoxy acetic acid (2,4-D) combinations promoted high regeneration efficiencies in only some genotypes5. Additionally, explants kept for 14 days in dark culture showed the highest regeneration percentage of adventitious shoots in explants (100%), and produced an average of 3.6 shoots per explant in ‘Pingyitiancha’ (Malus hupehensis var. pinyiensis)10. Based on these results, we report an efficient protocol obtained using 1/2MS + 1/2B5 medium supplemented with 2.0 mg L−1 TDZ, 20 g L−1 sucrose, and 6 g L−1 agar (pH 5.875) for in vitro shoot regeneration of diploid strawberry F. vesca ‘Baiguo’.
Notably, we found that regenerating competence recovered in the nearly brownish diploid strawberry explants, and shoots were regenerated from 30 to 48 days of culture. This may represent a selective process in which cells with low competence in response to the plant growth regulator treatment are killed, whereas a few competent cells from specific leaf tissue, potentially parenchymatic cells, start to divide and differentiate into a new adventitious shoot over time11. Similarly, “reinvigoration” of plants has been pursued by methods such as grafting and 6-benzyladenine metabolism. The term reinvigoration refers to the reversion of senescence12,13 and was used to describe the reversion of senescence that occurs when leaf explants are cultured in the regeneration medium14. This senescence is a general response that occurs in all strawberry explants, cultivated or wild, and it is reflected by chlorosis of the explant prior to regeneration8,15. Reinvigoration is critical for efficient shoot regeneration in diploid strawberry. However, less is known about molecular aspects of leaf explant reinvigoration events. Molecular mechanisms underlying shoot regeneration have been extensively studied, and considerable evidence has demonstrated important roles of plant growth regulators, such as cytokinins16, auxin17, and gibberellin18, in regulating shoot regeneration. Consequently, the genes involved in hormonal control may affect reinvigoration of leaf explants during shoot regeneration.
In this study, we reported a reinvigoration phenomenon during adventitious shoot regeneration of diploid strawberry, analysed the detailed changes over a long period (at least 11 weeks), and carried out a detailed transcript analysis of related genes during reinvigoration. The relationships between reinvigoration of adventitious shoot regeneration and transcript level variations were also revealed.
Results and Discussion
Reinvigoration during adventitious shoot regeneration of diploid strawberry F. vesca ‘Baiguo’ leaf explants
The morphogenic responses of somatic tissue have been studied in vitro. There are several lines of F. vesca and F. ananassa that have been tested for in vitro regeneration and showing a different behaviour19. Several blackberry and raspberry lines have been tested for in vitro regeneration and showed different morphogenic responses to plant growth regulator combinations20. Our results showed that adventitious shoot regeneration of diploid strawberry F. vesca ‘Baiguo’ differed from that of octoploid strawberry ‘Honeoye’. The octoploid strawberry leaf explants were green during regeneration (Fig. 1A). The explants showed elongation and enlargement after 6 days of culture. The shoot organogenesis from very small callus was formed from the cuts of explants after 15 days culture by histological observations8. A large callus formed after 18 days of culture. Relatively small shoots were observed that were sporadically dispersed within the callus on day 21 of culture, and clustered shoots were observed on day 27. The number of adventitious shoots and their growth increased over time, with most shoots being longer on day 33 of culture8. During whole shoot regeneration of octoploid strawberry, the leaf explants remained green and invigorated, and adventitious shoots typically appeared after 33 days.
Compared with octoploid strawberry ‘Honeoye’, adventitious shoots regenerated more in browning leaf explants of F. vesca ‘Baiguo’ (Fig. 1B). After 12 days of culture, the F. vesca ‘Baiguo’ explants considerably enlarged. After 30 days of culture, the explants turned greenish brown. After 36 days of culture, the explants turned yellowish brown, produced a yellow-green callus, and sporadically formed adventitious shoots around the wound edges. After 48 days of culture, the explants turned completely brown, but many shoots appeared. From 48 to 66 days of culture, adventitious shoots nearly doubled in number and elongated. After 66 days of culture, adventitious shoots were even more elongated. Therefore, the critical periods occurred when there was reinvigoration of completely brown explants that resulted in shoot regeneration, which included 30 days for chlorotic leaf explants, 36 days for adventitious shoot appearance, and 48 days for generation of several shoots.
Dark treatment effects on reinvigoration during adventitious shoot regeneration from the leaf explant of F. vesca
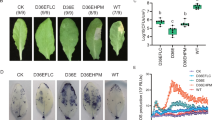
Subsequently, to study the effect of dark treatment on reinvigoration, various periods of initial dark culture (from 0 to 28 days) were applied. Although dark treatments did not influence the reinvigoration outcomes, the duration of dark culture did (Tables 1 and S1). Both the control explants (0 days, no initial dark culture treatment) and explants initially kept under dark culture conditions regenerated adventitious shoots. The adventitious shoot regeneration rate (from 90.67 ± 7.34% to 96.44 ± 1.60%) and number of shoots per explant (from 19.62 ± 1.88 to 23.46 ± 2.14) after the initial 7, 14, and 21 days of dark culture were not different from that in the control, but were significantly higher than that during the initial 28 days of dark culture (63.56 ± 10.45% and 7.86 ± 1.52 for adventitious shoot regeneration rate and number of shoots per explant, respectively) (p < 0.05) (Tables 1 and S1). On the other hand, dark treatments did not influence the reinvigoration process, the duration of dark culture did (Fig. 2A). However, dark treatments only influenced reinvigoration for the first 7 days. The other dark treatments are lower than 0 day in adventitious shoot regeneration rate between 42–60 days after culture (Fig. 2A). Furthermore, chlorosis of leaf explants after the initial 7, 14, and 21 days of dark culture from 30 to 48 days was less obvious than in the control, but more obvious than that in the initial 28 days of dark culture (Figs 2B and S2); this might be attributed to light having negative interactions with hormones that induces oxidative stress which kills cells21, dark incubation enhancing the accumulation of endogenous IAA22, and dark treatments leading to the reduction of photosynthetic capacity and a reduced efficiency of the chloroplasts8,10,23. We also found that, when the regenerated shoots were inoculated on 1/2MS + 1/2B5 medium, the leaves of shoots turned yellow after 66 days; then, there was reinvigoration, which facilitated more continuous shoot growth and vigorous shoot generation after 66 days of culture (Fig. 2C). The results showed that dark treatments did not affect the leaf explant reinvigoration, but the duration of dark culture did. The best dark culture condition for reinvigoration (14 days) was then used to study the molecular aspects of reinvigoration.
Dark treatment effects on reinvigoration during adventitious shoot regeneration of diploid strawberry leaf explants. (A) Adventitious shoot regeneration of diploid strawberry F. vesca ‘Baiguo’ leaf explants in the light (DT0) and in the dark for 7 days (DT7), 14 days (DT14), 21 days (DT21), or 28 days (DT28) after 78 days on 1/2MS + 1/2B5 medium that contained 2.0 mg L−1 TDZ. Arrows indicate the reinvigoration period of adventitious shoot regeneration. (B) Adventitious shoot regeneration on 0, 18, 30, 36, 42, 48, 54, 60, 66, 72, and 78 days of culture under different dark treatments. (C) Culture of the explants including adventitious shoots inoculated on 1/2MS + 1/2B5 medium contained 0.2 mg L-1 BA and 0.1 mg L-1 IBA.
The relation between transcript level of hormone synthesis related genes and morphological characteristics such as explant reinvigoration, shoot regeneration of the leaf explant of ‘F. vesca’
Cytokinins play important roles in plant growth and development. Zeatin O-glucosyltransferase (ZOG) and cis-zeatin O-glucosyltransferase (Ciszog) are very important in cytokinin homeostasis24. The ZOG and Ciszog transcript levels were relatively higher at 6, 30, and 36 days of culture, during which leaf explants began to enlarge or produce shoot primordia (Fig. 3A,B). This is consistent with zeatin concentration variation in in vitro-cultivated strawberry (F.ananassa cv. ‘Honeoye’) leaf explants; the zeatin concentrations peaked at 5.98 ng g−1 Fw during meristemoid formation and reached 7.69 ng g−1 Fw during shoot primordia formation8. Furthermore, the ZOG and Ciszog transcript levels were the highest at 72 days of culture, during which senescence was delayed and adventitious shoots very elongated. This could be explained by the two-step adventitious shoot regeneration process that was first observed in in vitro-derived lingonberry leaves25.
Relationships between hormone transcript levels and morphological characteristics (explant reinvigoration and shoot regeneration). (A) Morphological characteristics of leaf explants in initial 14 days dark treatment during shoot regeneration. (B) Transcript levels of key hormone synthesis-related genes, including Zeatin O-glucosyltransferase (ZOG), cis-zeatin O-glucosyltransferase (Ciszog), cytokinin hydroxylase (CKH), cytokinin oxidase/dehydrogenase (CKX) families, YUCCA (YUC) families, gibberellin 2-beta-dioxygenase (GA2ox), and GA20ox during diploid strawberry ‘Baiguo’ shoot regeneration.
Cytokinin hydroxylase (CKH) is involved in trans-zeatin biosynthesis, and contributes to tissue and organ formation26. The CKH transcript level significantly increased from 0 to 24 days of culture, which was during leaf explant shoot primordia formation. The CKH transcript level was higher from 30 to 48 days of culture, during which reinvigoration occurred and leaf explants began to produce multiple shoots (Fig. 3A,B).
Cytokinin oxidase/dehydrogenase (CKX) is involved in cytokinin degradation and plays important roles in plant meristem activity and morphogenesis, including causing changes in root growth and xylem differentiation27, and has opposite regulatory functions in root and shoot meristems28,29. The transcript levels of some CKX genes (CKX2, CKX3, CKX5, CKX6, and CKX7) increased during the initial days of culture (from 0 to 6 days), which was when there was leaf explant enlargement (Fig. 3A,B). The CKX2, CKX3, and CKX7 transcript levels significantly increased from 30 to 48 days of culture, during which leaf explants were reinvigorated and many shoots appeared. However, the CKX2 and CKX3 levels were lower at 12 and 18 days than at 6, 24, 30, and 36 days. Additionally, the CKX7 level was substantially lower at 30 and 36 days compared with 12, 42, and 48 days. These peaks were consistent with the findings of zeatin concentration variation in in vitro-cultivated strawberry leaf explants8.
Noticeable increases in the transcript levels of cytokinin biosynthesis-related genes were associated with the critical periods, including the first 6 days, during which there was explant enlargement, and 30 to 48 days of culture, during which there was diploid strawberry explant reinvigoration. The results obtained are in consistent with the initial phase of leaf senescence due to cytokinin degradation by CKX.
YUCCA (YUC) catalyses hydroxylation of the tryptamine amino group, which is a rate-limiting step in auxin biosynthesis30,31. The YUC genes play essential roles in auxin biosynthesis and plant development, including floral organ and vascular tissue formation in Arabidopsis32, crown root development control in rice33, and soybean growth and development alteration34. The YUC1, YUC2, YUC3, YUC5, YUC7, YUC8, YUC9, YUC10, and YUC11 transcript levels significantly increased from 0 to 6 days of culture, which was when there was leaf explant enlargement. This can be explained by dark incubation having a positive effect on auxin-related gene stimulation, and thus endogenous auxin production and eventually leaf explant enlargement22,35. The transcript level of YUC6 is higher in 0 day than in 6 days may be ascribed to wounding first triggers short term wound signaling that lasts from seconds to hours, then the wound signal spreads very rapidly from the wound site to mesophyll cells and activates YUC expression in mesophyll cells36. The YUC2, YUC5, YUC6, YUC9, and YUC10 transcript levels significantly increased during reinvigoration (Fig. 3A,B). These noticeable increases in YUC gene transcript levels during the critical periods (including 30, 36, and 48 days of culture) indicate that they have roles in diploid strawberry reinvigoration.
Gibberellin 2-beta-dioxygenase (GA2ox) is involved in GA inactivation, which is associated with Glycine max development37, might play critical roles in cell division38. The GA2ox transcript level was substantially higher from 30 to 48 days of culture, which was when explant reinvigoration was observed (Fig. 3A,B). GA20ox might modify cell growth and development associated with explant reinvigoration. The GA20ox transcript level was much higher before 48 days of culture, which indicates that it is related to explant reinvigoration (Fig. 3A,B). The increased GA2ox levels prior to shoot regeneration, concomitant with a reduction in GA20oxydase (involved in GA synthesis), might indicate that a reduction in GAs preceded adventitious shoot formation.
Briefly, we report an efficient protocol that used 1/2MS + 1/2B5 medium supplemented with 2.0 mg L−1 TDZ, 20 g L−1 sucrose, and 6 g L−1 agar (pH 5.875) for in vitro shoot regeneration of diploid strawberry. During adventitious shoot regeneration, the obvious reinvigoration of nearly brownish diploid strawberry explants aroused our attention. The transcript level changes of shoot regeneration-related genes may provide a possible explanation of such reinvigoration. Plant hormone synthesis-related genes play essential roles in plant development. Higher expressions of the hormone synthesis-related genes Ciszog1, CKX2, CKX3, CKX7, YUC2, YUC6, YUC10, YUC9, and GA2ox were detected from 30 to 48 days of culture during leaf explant reinvigoration. Thus, we conclude that efficient recovery of nearly brownish diploid strawberry explants is closely associated with hormone synthesis-related genes.
Methods
Plant materials
Plant materials of diploid strawberry (F.vesca) and octoploid strawberry (F. ananassa) were obtained from the Beijing Academy of Forestry and Pomology Sciences, Beijing, China. The protocols for sterilisation and sample preparation were optimised according to the protocol described by Jin et al., 2014 and Wang et al.8,10. The disinfected lateral and apical buds were then placed on MS medium that contained 0.5 mg L−1 BA and 0.1 mg L−1 IBA. After 30 days, the shoots that developed from the buds were subcultured onto 1/2MS + 1/2B5 medium that contained 0.2 mg L−1 BA and 0.1 mg L−1 IBA. The basic 1/2MS + 1/2B5 medium contained 20 g L−1 sucrose and 6.0 g L−1 agar (Shishi Co., Fujian, China). The pH was adjusted to 5.875 and media were autoclaved for 20 min at 121 °C. Cultures were maintained in a growth room at 25 ± 2 °C under a 16-hour light photoperiod at approximately 25 µmol m−2s−1 supplied by cool-white fluorescent lamps. The strawberry plantlets were used as experimental materials for adventitious shoot regeneration analysis.
Reinvigoration during adventitious shoot regeneration of diploid strawberry (F. versa)
The protocol for adventitious shoot regeneration of diploid strawberry F. versa was optimised as described by Jin et al., 2014 and Wang et al. 20158,10. 1/2MS + 1/2B5 medium supplemented with 2.0 mg L−1 TDZ, 20 g L−1 sucrose, and 6 g L−1 agar (pH 5.875) was applied for in vitro analysis of F. vesca adventitious shoot regeneration. During this process, the explants were reinvigorated from being completely brown to shoot regeneration.
Subsequently, to study the effects of an initial dark period on reinvigoration, the explants were kept in the dark for 0, 7, 14, 21, and 28 days under the growth conditions described in “Plant materials”. All dark treatments (DT0:78 days at 16-h light photoperiod; DT7:7 days at dark and 71 at 16-h light photoperiod; DT14:14 days at dark and 64 at 16-h light photoperiod; DT21: 21 days at dark and 57 at 16-h light photoperiod; DT28: 28 days at dark and 50 at 16-h light photoperiod) tested for shoot induction were analysed by preparing three subsequent repetitions of three 100 mL glass flasks (90 mm in diameter with 40 mL of medium added), each of which contained 25 segmented leaf explants. The adventitious shoot regeneration rate (%) and number of regenerated shoots per explant were calculated for each glass flask. The morphogenic responses of segmented leaf explants were observed and photographed every 6 days. The adventitious shoot regeneration rates were also recorded. There were three repetitions of each dark duration treatment (225 leaf explants for each dark treatment), and the experiment was repeated three times (repetition1: from 12, Feb. to 30, Apr., 2015; repetition 2: from 12, Jun. to 30, Aug., 2015; repetition 3: from 18, Mar. to 5, Jun., 2016).
Finally, the best regeneration condition was determined (initial 0, 7, 14, 21, or 28 days of dark culture) and applied to prepare leaf explants for transcript-level analysis of related genes at 0, 6, 12, 18, 24, 30, 36, 42, 48, 54, 60, 66, 72, and 78 days of culture. In this study, the best regeneration conditions were observed in the initial 14 days of dark culture, then 0, 6, 12, 18, 24, 30, 36, 42, 48, 54, 60, 66, 72 and 78 days of culture would be as follows: 0:0 days at dark; 6:6 days at dark; 12:12 days at dark; 18:14 days at dark and 4 days at 16-h light photoperiod; 24:14 days at dark and 10 days at 16-h light photoperiod; 30:14 days at dark and 16 days at 16-h light photoperiod; 36:14 days at dark and 22 days at 16-h light photoperiod; 42:14 days at dark and 28 days at 16-h light photoperiod; 48:14 days at dark and 34 days at 16-h light photoperiod; 54:14 days at dark and 40 days at 16-h light photoperiod; 60:14 days at dark and 46 days at 16-h light photoperiod; 66:14 days at dark and 52 days at 16-h light photoperiod; 72:14 days at dark and 58 days at 16-h light photoperiod; 78:14 days at dark and 64 days at 16-h light photoperiod. The leaf explants in the initial 14 days of dark treatment were then sampled every 6 days from 0 to 78 days (0, 6, 12, 18, 24, 30, 36, 42, 48, 54, 60, 66, 72, and 78 days) to conduct the following transcript level analysis of related genes.
The adventitious shoot regeneration rate (%) was calculated as the total number of regenerating explants divided by total number of explants after 78 days of culture and then multiplied by 100%. The number of regenerated shoots per explant was calculated as the total number of adventitious shoots divided by total number of regenerating explants after 78 days of culture.
Transcript analysis of related genes during reinvigoration
Total RNA was extracted, and first-strand cDNA was synthesised from leaf discs every 6 days from 0 to 78 days during F. vesca shoot regeneration, as described by Jin et al.39. To further investigate the transcript levels of hormone synthesis-genes, including the YUC and CKX families, specific primers were designed with Primer 5.0 (Premier Biosoft International, Palo Alto, CA, USA) (Table 2) and acquired from Sangon Inc. (Shanghai, China). qRT-PCR was conducted with on a Bio-Rad CFX96 Real-Time PCR Detection System (Bio-Rad, Hercules, CA, USA). For qRT-PCR, the 10-μL reaction mixture included 5 μL 2 × SYBR Premix (Bio-Rad, Hercules, CA, USA), 1 μL forward primer (10 μM), 1 μL reverse primer (10 μM), 1 μL cDNA template (20 ng), and 2 μL ddH2O; the PCR conditions were as follows: 1 cycle at 95 °C for 30 s, 40 cycles at 95 °C for 5 s, and 1 cycle at 60 °C for 30 s. Finally, the transcription levels of different genes were analysed using the 2−ΔΔCT method40. All analyses were performed with three biological replicates.
Statistical analysis
The results shown are the mean values of three independent experiments. In each independent experiment, all dark treatments (75 leaf explants for each treatment) tested for shoot induction were analysed by three repetitions of three 100 mL glass flasks, each of which contained 25 segmented leaf explants. Statistical analyses were performed in SPSS 16.0 (SPSS Inc., Chicago, IL, USA). One-way ANOVA was applied for parameters (adventitious shoot regeneration rate, number of shoots per explant) to evaluate the effect of dark treatment. The significance of differences was determined according to Duncan’s multiple range tests. Differences at p < 0.05 were considered significant.
References
Shulaev, V. et al. The genome of woodland strawberry (Fragaria vesca). Nat Genet 43, 109–116 (2011).
Oosumi, T. et al. High-efficiency transformation of the diploid strawberry (Fragaria vesca) for functional genomics. Planta 223, 1219–1230 (2006).
Owen, H. & Miller, A. R. Haploid plant regeneration from anther cultures of three north american cultivars of strawberry (Fragaria x ananassa Duch.). Plant Cell Rep 15, 905–909 (1996).
Haddadi, F., Aziz, M. A., Kamaladini, H. & Ravanfar, S. A. Thidiazuron- and zeatin-induced high-frequency shoot regeneration from leaf and shoot-tip explants of strawberry. HortTechnology 23, 276–281 (2013).
Landi, L. & Mezzetti, B. TDZ, auxin and genotype effects on leaf organogenesis in Fragaria. Plant Cell Rep 25, 281–288 (2006).
Mezzetti, B. Genetic transformation in strawberry and raspberry. In: Plant Genetic Engineering, Vol. 6 Improvement of Fruit Crops. (eds Jaiwal, P. K. & Singh, R. P.). SCI Tech Publishing LLC, USA (2003).
Passey, A., Barrett, K. & James, D. Adventitious shoot regeneration from seven commercial strawberry cultivars (Fragaria × ananassa Duch.) using a range of explant types. Plant Cell Rep 21, 397–401 (2003).
Wang, H., Li, M., Yang, Y., Dong, J. & Jin, W. Histological and endogenous plant growth regulators changes associated with adventitious shoot regeneration from in vitro leaf explants of strawberry (Fragaria × ananassa cv. ‘Honeoye’). Plant Cell Tissue Organ Cult 123, 479–488 (2015).
Vergne, P. et al. Somatic embryogenesis and transformation of the diploid Rosa chinensis cv Old Blush. Plant Cell Tissue Organ Cult 100, 73 (2009).
Jin, W., Wang, Y. & Wang, H. Adventitious shoot regeneration from leaves of apple rootstock ‘Pingyitiancha’ (Malus hupehensis var. pinyiensis) and genetic fidelity of regenerated plantlets using SSR markers. Can J Plant Sci 94, 1345–1354 (2014).
Mezzetti, B., Pandolfini, T., Navacchi, O. & Landi, L. Genetic transformation of Vitis viniferavia organogenesis. BMC Biotechnol 2, 18 (2002).
Revilla, M. A., Pacheco, J., Casares, A. & Rodríguez, R. In vitro reinvigoration of mature olive trees (Olea europaea L.) through micrografting. In Vitro Cell Dev Biol-Pl 32, 257–261 (1996).
Zhang, H., Horgan, K. J., Reynolds, P. H. & Jameson, P. E. 6-Benzyladenine metabolism during reinvigoration of mature Pinus radiata buds in vitro. Tree physiol 30, 514–526 (2010).
Sriskandarajah, S., Mibus, H. & Serek, M. Regeneration and transformation in adult plants of Campanula species. Plant Cell Rep 27, 1713–1720 (2008).
Nehra, N. S., Stushnoff, C. & Kartha, K. K. Regeneration of plants from immature leaf-derived callus of strawberry (Fragaria × ananassa). Plant Sci 66, 119–126 (1990).
Magyar-Tábori, K., Dobránszki, J., Teixeira da Silva, J., Bulley, S. & Hudák, I. The role of cytokinins in shoot organogenesis in apple. Plant Cell Tissue Organ Cult 101, 251–267 (2010).
Ljung, K. et al. Biosynthesis, conjugation, catabolism and homeostasis of indole-3-acetic acid in Arabidopsis thaliana. In: Auxin Molecular Biology (eds Perrot-Rechenmann, C. & Hagen, G.). Springer Netherlands (2002).
Rademacher, W. Growth retardants: effects on gibberellin biosynthesis and other metabolic pathways. Annu Rev Plant Physiol Plant Mol Biol 51, 501–531 (2000).
Mezzetti, B., Landi, L., Pandolfini, T. & Spena, A. The defH9-iaaM auxin-synthesizing gene increases plant fecundity and fruit production in strawberry and raspberry. BMC Biotechnology 4, 4 (2004).
Mezzetti, B., Savini, G., Carnevali, F. & Mott, D. Plant genotype and growth regulators interaction affecting in vitro morphogenesis of blackberry and raspberry. Biol Plantarum 39, 139–150 (1997).
O’Donovan, P. et al. Azathioprine and UVA light generate mutagenic oxidative DNA damage. Science 309, 1871–1874 (2005).
Fattorini, L. et al. Jasmonate promotes auxin-induced adventitious rooting in dark-grown Arabidopsis thaliana seedlings and stem thin cell layers by a cross-talk with ethylene signalling and a modulation of xylogenesis. BMC Plant Biol 18, 182 (2018).
Morini, S., Muleo, R., Sciutti, R. & Fortuna, P. Relationship between evolution of CO2 and growth of plum shoot tips cultured in vitro under different light/dark regimes. Physiol Plantarum 87, 286–290 (1993).
Martin, R. C., Mok, M. C., Habben, J. E. & Mok, D. W. S. A maize cytokinin gene encoding an O-glucosyltransferase specific to cis-zeatin. Proc Natl Acad Sci USA 98, 5922–5926 (2001).
Debnath, S. C. A two-step procedure for adventitious shoot regeneration from in vitro-derived lingonberry leaves: shoot induction with TDZ and shoot elongation using zeatin. HortScience 40, 189–192 (2005).
Cheng, Y., Zhao, Y., Liu, J., Yang, B. & Ming, Y. Comparison of phytohormone biosynthesis and signal transduction pathways in developing and abortive hazelnut ovules. Plant Growth Regul 81, 147–157 (2017).
Ireen, K., Ondřej, N., Miroslav, S., Thomas, S. & Tomáš, W. Overexpression of the cytosolic cytokinin oxidase/dehydrogenase (CKX7) from Arabidopsis causes specific changes in root growth and xylem differentiation. Plant J 78, 359–371 (2014).
Werner, T., Motyka, V., Strnad, M. & Schmülling, T. Regulation of plant growth by cytokinin. Proc Natl Acad Sci USA 98, 10487–10492 (2001).
Werner, T. et al. Cytokinin-deficient transgenic Arabidopsis plants show multiple developmental alterations indicating opposite functions of cytokinins in the regulation of shoot and root meristem activity. Plant Cell 15, 2532–2550 (2003).
Takeshi, N. et al. Yucasin is a potent inhibitor of YUCCA, a key enzyme in auxin biosynthesis. Plant J 77, 352–366 (2014).
Zhao, Y. et al. A role for flavin monooxygenase-like enzymes in auxin biosynthesis. Science 291, 306–309 (2001).
Cheng, Y., Dai, X. & Zhao, Y. Auxin biosynthesis by the YUCCA flavin monooxygenases controls the formation of floral organs and vascular tissues in Arabidopsis. Gene Dev 20, 1790–1799 (2006).
Zhang, T. et al. The YUCCA-Auxin-WOX11 module controls crown root development in rice. Front Plant Sci 9, 523 (2018).
Wang, Y., Liu, H., Wang, S. & Li, H. Genome-wide identification and expression analysis of the YUCCA gene family in soybean (Glycine max L.). Plant Growth Regul 81, 265–275 (2017).
Yang, H. et al. Role of auxin homeostasis and response in nitrogen limitation and dark stimulation of adventitious root formation in petunia cuttings. Annals of botany, https://doi.org/10.1093/aob/mcz095 (2019).
Chen, L., Sun, B., Xu, L. & Liu, W. Wound signaling: The missing link in plant regeneration. Plant Signaling & Behavior 11, e1238548 (2016).
Han, Y. et al. Genome-wide transcriptome profiling reveals the mechanism of the effects of uniconazole on root development in. Glycine Max. J Plant Biol 60, 387–403 (2017).
Jiang, S. et al. Comparative analysis of transcriptomes to identify genes associated with fruit size in the early stage of fruit development in Pyrus pyrifolia. Int J Mol Sci 19, 2342 (2018).
Jin, W. et al. The R2R3 MYB transcription factor PavMYB10.1 involves in anthocyanin biosynthesis and determines fruit skin colour in sweet cherry (Prunus avium L.). Plant Biotechnol J 14, 2120–2133 (2016).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT method. Methods 25, 402–408 (2001).
Theologis, A. et al. Sequence and analysis of chromosome 1 of the plant Arabidopsis thaliana. Nature 408, 816 (2000).
Hytönen, T., Elomaa, P., Moritz, T. & Junttila, O. Gibberellin mediates daylength-controlled differentiation of vegetative meristems in strawberry (Fragaria × ananassa Duch). BMC Plant Biol 9, 18 (2009).
Acknowledgements
This work was supported by Beijing Municipal Science & Technology Commission, China (KJCX20170203, KJCX20170104), National Natural Science Foundation for Young Scholars of China (31600418), National Key R&D Program of China (2016YFE0112400), and European Union’s Horizon 2020 Research and Innovation Program (679303).
Author information
Authors and Affiliations
Contributions
W.J. conceived the study. H.W., Y.Y., M.L. and J.L. carried out the experiments. H.W. and Y.Y. analyzed the data. H.W. and W.J. wrote and revised the manuscript. All authors have read and approved of the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, H., Yang, Y., Li, M. et al. Reinvigoration of diploid strawberry (Fragaria vesca) during adventitious shoot regeneration. Sci Rep 9, 13007 (2019). https://doi.org/10.1038/s41598-019-49391-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-49391-8
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.