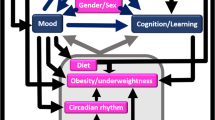
Abstract
Peripartum depression is a common, serious complication in mothers. To assess the influence of infant care, sleep and growth on the risk of peripartum depression, 1,271 mothers of healthy one-month-old infants completed comprehensive questionnaires including the Edinburgh Postnatal Depression Scale. Relationships between high depression scores and variables related to infants’ care, sleep and growth were assessed adjusting for other variables. High depression scores were found in 233 mothers, which were associated with variables related to infants’ care (poor satisfaction with infant care support, p < 0.001; mothers’ passive response to night-time fussing, p = 0.001), sleep (dim bedroom lighting, p < 0.05; short sleep duration, p < 0.05) and growth (poor weight gain, p < 0.05), as well as maternal variables (primiparas, p < 0.001; poor income satisfaction, p < 0.001; poor sleep satisfaction, p < 0.001; daytime sleepiness, p = 0.001). Considering the observed association between high depression scores and infants’ care, sleep and growth, a multidisciplinary approach accounting for infant care would be required to prevent peripartum depression.
Similar content being viewed by others
Introduction
According to a systematic review conducted in the United States, approximately 20% of women have a major depressive episode during the first 3 months postpartum1, which is considerably higher than <10% of women in the general population2. Peripartum depression (PD) is associated with long-term, compromising effects on the mother-infant relationship and development of infants3,4. Researchers investigated the potential dependence of PD on various factors, such as ethnicity, family history of depression, socioeconomic status, type of feeding and maternal oxytocin levels5,6. In addition to maternal variables, several studies investigated the role of factors related to infant care. For example, serious sleeping and crying problems in young infants are associated with a two-fold increase in maternal depression and anxiety7, and inappropriate social infant care support is associated with an elevated incidence of PD8. Vigod et al. described that delivery of low-birth-weight infants and their admission to the neonatal intensive care unit are associated with an increased risk of maternal PD, suggesting that growth and health status of infants might also be associated with the risk of PD9. These findings suggest that strategies to predict and prevent PD may need to account for variables rooted in various aspects of infant care, as well as maternal and socioeconomic variables. Thus far, however, few comprehensive studies have addressed the role of factors related to the infant care, sleep and growth in the development of PD in mothers with healthy infants.
The aim of this study was to assess whether infant care, sleep and growth are associated with the risk of PD even when adjusted for other known and unknown independent variables in a large cohort of mothers with healthy young infants.
Results
Data are presented as mean ± standard deviation, number (%) or median (interquartile range). Clinical backgrounds of the study population have been published previously10. Of the questionnaires collected from 1,302 participating mothers, data from 31 mothers were excluded due to excessive missing data. The 1,271 mothers in the final study cohort were 31.9 ± 4.6 years old (Table 1). The highest education was high school or college in 1,228 (96.6%) mothers; 1,164 (91.6%) mothers were fully or generally satisfied with their family income; the typical night-time sleep duration of the mothers was 5.4 ± 1.5 h, with 619 (48.7%) mothers dissatisfied with their sleep (Table 1). High Edinburgh Postnatal Depression Scale (EPDS) scores (≥9) were noted in 233 (18.3%) mothers, whereas very high EPDS scores (≥12) were found in 115 (9.0%) mothers. The infants were 39.5 ± 1.2 weeks gestation and 3091 ± 374 g at birth, whose body weight gain was 44.1 ± 10.4 g/day. The night-time and daily sleep durations of the infants were 8.3 ± 1.8 h and 15.8 ± 2.8 h, respectively.
Findings from univariate analysis: variables associated with infant care, sleep and growth (Table 2).
Elevated EPDS scores were associated with smaller body weight gain of infants (p < 0.001), dim lighting of infant’s bedroom (p = 0.031 for ‘dim’ compared with ‘bright’), irregular light-off time (p = 0.002 for ‘irregular’ compared with ‘regularly early’), shorter daytime sleep duration (p = 0.009 for ‘short’ and 0.048 for ‘medium’; compared with ‘long’), shorter night-time sleep duration (p < 0.001 for ‘short’ and p = 0.041 for ‘medium’; compared with ‘long’), and shorter daily sleep duration (p < 0.001 for both ‘short’ and ‘medium’ compared with ‘long’). For variables associated with infant care support and child-rearing style, the presence of an additional caregivers (p = 0.016 for ‘one’ compared with ‘no’ extra caregiver), poor satisfaction with social infant care support (p = 0.022 for ‘generally satisfied’ and p < 0.001 for ‘not satisfied’; compared with ‘no support’), formula-milk-based feeding (p = 0.004 for ‘mainly formula milk’ compared with ‘exclusive breastfeeding’), passive response of mothers to night-time fussing (p < 0.001 for ‘observe’ without dandling compared with ‘rock briefly’), and poor confidence with child rearing (p < 0.001 for mothers of both ‘anxious’ and ‘generally confident’; compared with ‘confident’) were associated with high EPDS scores.
Findings from univariate analysis: maternal variables
Higher maternal age (p = 0.032 for ≥35 years old compared with 29≤ and <35 years old) and dissatisfaction with family income (p < 0.001 for ‘not satisfied’ and p = 0.003 for ‘generally satisfied’; compared with ‘fully satisfied’), primipara status (p < 0.001 for primiparas compared with both parity 2 and ≥3) and vaginal delivery (p = 0.030 for vaginal delivery compared with elective caesarean delivery) were associated with high EPDS scores (Table 3). Of variables associated with maternal sleep status during the last 7 days of the baby check, irregular bedtime (p < 0.001 for ‘irregular’ and p = 0.027 for ‘regularly late’; compared with ‘regularly early’), shorter night-time sleep duration (p < 0.001 for ‘short’ and p = 0.043 for ‘medium’; compared with ‘long’), more frequent night-time wakefulness (p < 0.001), poor satisfaction with sleep (p < 0.001 for both ‘dissatisfied’ and ‘mildly satisfied’; compared with ‘satisfied’), and greater Epworth Sleepiness Scale (ESS) scores (p < 0.001) were associated with high EPDS scores (See Supplementary Table 1 for the variables in relation to maternal sleep during the last month of pregnancy).
Multivariate analysis
In the multivariate analysis, high EPDS scores were associated with poor satisfaction with family income (p < 0.001 for ‘unsatisfied’ and p = 0.017 for ‘generally satisfied’; compared with ‘fully satisfied’), primipara status (p = 0.001 and p < 0.001 for primiparas; compared with parity 2 and ≥ 3, respectively), poor satisfaction with social infant care support (p = 0.041 for ‘generally satisfied’ and p < 0.001 for ‘not satisfied’; compared with ‘no social infant care support’), dim room lighting (p = 0.024 for ‘dim’ compared with ‘bright’), passive response of mothers to night-time fussing (p = 0.001 for ‘observation’ without dandling compared with ‘dandling briefly’), lower weight gain of infants (p = 0.044), shorter daily sleep duration of infants (p = 0.045 for ‘short’ and p = 0.049 for ‘medium’; compared with ‘long’), poor maternal satisfaction with sleep (p < 0.001 for both ‘mildly dissatisfied’ and ‘dissatisfied’; compared with ‘satisfied’), and greater postpartum ESS scores (p = 0.001) (Table 4).
Discussion
Using a comprehensive dataset from a survey of mothers of 1-month-old infants, infant care, sleep and growth were identified as potential independent variables of PD even after adjustment for the influence of known independent variables. These findings suggest that, for the prediction, prevention, early diagnosis and efficient treatment of PD, multifactorial approach would be required focusing on both maternal and non-maternal variables, especially on those related to the care, sleep and growth of infants.
Peripartum depression and sleep of mothers and infants
Infants’ sleep duration start aggregating in the night-time approximately one month after birth; however, the timing and progress vary between infants11. A delay in this process may increase the anxiety of mothers, rendering them susceptible to psychiatric conditions. Our data suggest the dependence of maternal PD on the schedule, quantity, and environment of their infants’ sleep, as well as their own sleep quality and quantity. Using the same dataset, we have previously demonstrated that maternal night-time sleep duration is tightly coupled with that of their infants10. These findings suggest the importance of providing good night-time sleep to infants in order to secure good maternal sleep and ultimately reduce the risk of maternal PD. We have previously shown that punctual light-off time is associated with longer night-time sleep duration in infants10. Univariate analysis in the current study suggested that punctual light-off time for infants and punctual bedtime for mothers are associated with a lower risk of maternal PD. Regular, and possibly early, light-off time for infants might promote a swift acquisition of mature sleep patterns, potentially leading to better maternal sleep and lower incidence of PD.
Peripartum depression and infant care
Pathological clinical conditions of newborn infants, such as preterm delivery and hospitalisation of infants at neonatal intensive care centres, have been recognised as prominent risk variables of PD9. Our study suggested that, even after delivery of a healthy infant, relatively poor infant growth might be the risk of PD. Because infant weight is one of the few visible markers of ‘successful infant care’ for mothers, poor weight gain might easily enhance mothers’ anxiety. Univariate analysis in our study suggested that, in addition to the infant growth, mothers’ confidence and anxiety regarding infant care was associated with high EPDS scores. These findings highlight the importance of attending to mothers’ psychological characteristics to predict and prevent PD. To ameliorate the excessive pressure on mothers, it is important to provide appropriate social infant care support8. In the current study, the risk of PD was paradoxically dependent on the type and quality of social infant care support. While dissatisfaction with social infant care support was associated with high EPDS scores, mothers who were caring for their infants on their own showed the lowest risk of PD, suggesting that satisfaction with social infant care support is not necessarily dose-dependent, but is affected by mothers’ psychological characteristics.
With respect to the type of feeding, our findings from the univariate analysis were consistent with previous studies showing the benefits of breastfeeding in reducing the risk of PD12,13. Breastfeeding mothers are exempted from additional night-time tasks required to prepare the formula milk, allowing them longer night-time sleep duration. Increased serum oxytocin levels of breastfeeding mothers may also influence the threshold for developing PD14. Although our data suggest the benefit of exclusive breastfeeding, the supplemental use of formula milk did not increase the risk of PD. Given that failure to establish breastfeeding is associated with a significant increase in the risk of PD15, encouragement of breastfeeding should be accompanied by sufficient support to minimise maternal psychological pressure.
Other independent variables of peripartum depression
Consistent to previous studies, which demonstrated the association between maternal socio-economic status and the development of PD5,6, we confirmed that poor satisfaction of mothers with their family income was associated with high EPDS scores. Dependence of EPDS scores on delivery mode and parity was also suggested. However, unlike a previous study, which observed an elevated risk of PD following caesarean delivery16, our data in the univariate analysis suggested the association between elective caesarean delivery and lower risk of PD. Background characteristics associated with the indication of caesarean delivery might be associated with the paradoxical impact of caesarean delivery to the risk of PD. In our study, the majority of women who underwent caesarean delivery were multipara mothers, however, no further data were available regarding the indication of caesarean delivery. There currently is no large-scale survey, which assessed the indication of caesarean delivery in Japanese women. In Japan, the rate of elective caesarean delivery is 11.0%, which is lower than in other developed countries17. Considering that this delivery mode is covered by the national insurance, it is unlikely that socio-economic status affected the decision of the delivery mode in our study cohort. Future studies need to address the impact of caesarean delivery on the risk of PD.
Strength and limitation of the study
The development of PD is dependent on factors rooted in various aspects of maternal life, such as socio-economic status, pregnancy plan, problems with family and spouse, history of mental illness in the family, and social support5,6, suggesting that maternal background is insufficient to fully explain the mechanism of PD. By incorporating various background variables associated with maternal life during and after delivery, we have demonstrated that variables associated with infant care, sleep and growth are key factors to understand the mechanism of maternal PD. However, considering that clinical variables of mothers, infants, and other family members are often inter-correlated with each other (such as lifestyle and sleep schedule), it remains uncertain whether variables other than those related to maternal background are directly associated with PD, or merely via the inter-correlation between the variables. As previously described, our study was conducted by recruiting typical Japanese mothers in terms of age, academic background, and other socio-economic variables18. Second, we used a relatively low cut-off value of EPDS ≥ 9 to represent increased risks of PD19,20,21. Based on the high-enough sensitivity and specificity of this threshold to predict PD in Japanese women (82% and 95%, respectively), it was speculated that Japanese women are too reluctant to express their feelings and emotions when they are depressed21. Therefore, when generalising our findings to mothers in other parts of the world, careful consideration is required for cultural and traditional factors.
Conclusions
In addition to established maternal factors associated with PD, our survey with mothers of healthy 1-month-old infants highlighted that independent variables rooted in infant care, growth and sleep, were associated with the risk of maternal PD even when adjusted for known independent variables. A comprehensive, multidisciplinary approach is required to predict, prevent, identify, and treat maternal PD. Future studies need to address whether a strategy, which incorporate infant care, growth and sleep, improve prediction and prevention of PD, and thus help minimise the adverse influence of PD on the mother-child relationship.
Methods
Ethics statement
This study was conducted in compliance with the Declaration of Helsinki, and with the approval of the ethics committees of Kurume University School of Medicine and Kyushu University School of Medicine. Informed consent was obtained from all participating mothers, who answered the survey.
Study population
Data collection was performed with the participation of two tertiary perinatal centres (Kurume University Hospital and Kyushu University Hospital) and three private obstetrics hospitals (Izumi Ladies’ Clinic, Fukuoka Birth Clinic, and Sato Women’s Hospital) in Fukuoka, Japan. Preliminary data on the photoperiod and sleep patterns of infants and their mothers were reported previously, and data collection methods were described in detail10. Briefly, mothers of newborn infants were asked to complete a questionnaire at the time of the 1-month baby check between December 2014 and November 2015.
Questionnaires
Information was collected using the originally developed Sleep Questionnaire for Mothers of Newborn Infants10, EPDS20,21,22 and ESS23,24. Our original sleep questionnaire comprised 45 questions in relation to mothers and their family, birth of infants, social infant care support, infants’ sleep pattern and environment, and mothers’ sleep condition10. For the current study, 30 questions were carefully chosen to represent the variables of interest; relatively a wide range of variables (birth information, child-rearing style, growth and sleep of infants, and maternal sleep status before and after delivery, and ESS scores after delivery) were considered to assess the potential dependence of high EPDS scores on infant care, sleep and growth with adjustment for known independent factors of PD and other factors associated with various aspects of maternal life. The ESS assesses sleepiness while engaged in eight different activities (score 0–24), with higher scores indicating higher daytime sleepiness23. The EPDS is a 10-item self-report scale specifically designed to screen for postpartum depressive symptoms (score 0–30), with higher scores indicated higher levels of depressive symptomatology. For the Japanese version of EPDS, a relatively low cut-off score of 8/9 is recommended for the screening of PD19.
Data analysis
Questionnaires with more than 20% missing data without relevant reasons were excluded from the analysis25. To minimise biases derived from missing data, multiple imputation of the variable was performed (n = 5 imputations) based on the correlation between variables with missing values and other participant characteristics using SPSS ver. 21.0 (IBM, Armonk, NY, U.S.A.). A univariate logistic regression model was used to evaluate the association of potential independent variables with EPDS scores ≥9. Continuous variables with normal distribution were categorised into 2–3 levels using quartiles or clinically relevant thresholds when appropriate. P-values for the univariate analysis were not corrected for multiple comparisons because of the exploratory nature of the analysis. However, to avoid type-1 errors, p-values between 0.01 and 0.05 were regarded as chance level. A multivariate logistic regression analysis was performed to identify the final model to explain EPDS scores ≥9. For the multivariate model, mandatory variables (e.g. satisfaction with infant care support, body weight gain, daily sleep duration of infants, maternal satisfaction with sleep, and maternal satisfaction with family income) were chosen based on our hypothesis driven by previous studies, whereas additional variables were chosen based on clinical relevance, collinearity and the univariate analysis results.
References
Gavin, N. I. et al. Perinatal depression: a systematic review of prevalence and incidence. Obstetrics and gynecology 106, 1071–1083, https://doi.org/10.1097/01.AOG.0000183597.31630.db (2005).
Kendler, K. S., Gardner, C. O. & Prescott, C. A. Toward a comprehensive developmental model for major depression in women. The American journal of psychiatry 159, 1133–1145, https://doi.org/10.1176/appi.ajp.159.7.1133 (2002).
Mirhosseini, H. et al. Cognitive Behavioral Development in Children Following Maternal Postpartum Depression: A Review Article. Electronic physician 7, 1673–1679, https://doi.org/10.19082/1673 (2015).
Tronick, E. & Reck, C. Infants of depressed mothers. Harvard review of psychiatry 17, 147–156, https://doi.org/10.1080/10673220902899714 (2009).
Meltzer-Brody, S. et al. Postpartum psychiatric disorders. Nature reviews. Disease primers 4, 18022, https://doi.org/10.1038/nrdp.2018.22 (2018).
Ozcan, N. K., Boyacioglu, N. E. & Dinc, H. Postpartum Depression Prevalence and Risk Factors in Turkey: A Systematic Review and Meta-Analysis. Archives of psychiatric nursing 31, 420–428, https://doi.org/10.1016/j.apnu.2017.04.006 (2017).
Smart, J. & Hiscock, H. Early infant crying and sleeping problems: a pilot study of impact on parental well-being and parent-endorsed strategies for management. Journal of paediatrics and child health 43, 284–290, https://doi.org/10.1111/j.1440-1754.2007.01060.x (2007).
Morikawa, M. et al. Relationship between social support during pregnancy and postpartum depressive state: a prospective cohort study. Scientific reports 5, 10520, https://doi.org/10.1038/srep10520 (2015).
Vigod, S. N., Villegas, L., Dennis, C. L. & Ross, L. E. Prevalence and risk factors for postpartum depression among women with preterm and low-birth-weight infants: a systematic review. BJOG: an international journal of obstetrics and gynaecology 117, 540–550, https://doi.org/10.1111/j.1471-0528.2009.02493.x (2010).
Iwata, S. et al. Dependence of nighttime sleep duration in one-month-old infants on alterations in natural and artificial photoperiod. Scientific reports 7, 44749, https://doi.org/10.1038/srep44749 (2017).
Mirmiran, M., Maas, Y. G. & Ariagno, R. L. Development of fetal and neonatal sleep and circadian rhythms. Sleep Med Rev 7, 321–334 (2003).
Kendall-Tackett, K. A new paradigm for depression in new mothers: the central role of inflammation and how breastfeeding and anti-inflammatory treatments protect maternal mental health. International breastfeeding journal 2, 6, https://doi.org/10.1186/1746-4358-2-6 (2007).
Ystrom, E. Breastfeeding cessation and symptoms of anxiety and depression: a longitudinal cohort study. BMC pregnancy and childbirth 12, 36, https://doi.org/10.1186/1471-2393-12-36 (2012).
Skrundz, M., Bolten, M., Nast, I., Hellhammer, D. H. & Meinlschmidt, G. Plasma oxytocin concentration during pregnancy is associated with development of postpartum depression. Neuropsychopharmacology: official publication of the American College of Neuropsychopharmacology 36, 1886–1893, https://doi.org/10.1038/npp.2011.74 (2011).
Borra, C., Iacovou, M. & Sevilla, A. New evidence on breastfeeding and postpartum depression: the importance of understanding women’s intentions. Maternal and child health journal 19, 897–907, https://doi.org/10.1007/s10995-014-1591-z (2015).
Houston, K. A. et al. Mode of delivery and postpartum depression: the role of patient preferences. American journal of obstetrics and gynecology 212(229), e221–227, https://doi.org/10.1016/j.ajog.2014.09.002 (2015).
Maeda, E. et al. Cesarean section rates and local resources for perinatal care in Japan: A nationwide ecological study using the national database of health insurance claims. The journal of obstetrics and gynaecology research 44, 208–216, https://doi.org/10.1111/jog.13518 (2018).
Cabinet Office, G. o. J. Declinign birth rate: Current state and countermeasures, http://www8.cao.go.jp/shoushi/shoushika/whitepaper/measures/w-2015/27webhonpen/html/b1_s1-1-3.html (2015).
Department of Health, G. o. W. A. Using the Edinburgh Postnatal Depression Scale; Translated into Languages other than English, https://www.mcpapformoms.org/Docs/Edinburgh%20Depression%20Scale%20Translated%20Government%20of%20Western%20Australia%20Department%20of%20Health.pdf (2006).
Okano, T., Murata, M. & Masuchi, S. Reliablilty and feasibility of Japanese version of the Edinburg Postnatal Depression Scale (article in Japanese). Arch Psychiatr Diagn Clin Eval. 7, 525–533 (1996).
Yoshida, K., Yamashita, H., Ueda, M. & Tashiro, N. Postnatal depression in Japanese mothers and the reconsideration of ‘Satogaeri bunben’. Pediatr Int 43, 189–193 (2001).
Cox, J. L., Holden, J. M. & Sagovsky, R. Detection of postnatal depression. Development of the 10-item Edinburgh Postnatal Depression Scale. The British journal of psychiatry: the journal of mental science 150, 782–786 (1987).
Johns, M. W. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep 14, 540–545 (1991).
Fukuhara, S. et al. The Japanese version of the Epworth Sleepiness Scale (article in Japanese). Ann Jpn Resp Soc. 44, 896–898 (2006).
Hawthorne, G. & Elliott, P. Imputing cross-sectional missing data: comparison of common techniques. The Australian and New Zealand journal of psychiatry 39, 583–590, https://doi.org/10.1080/j.1440-1614.2005.01630.x (2005).
Acknowledgements
The authors thank Drs Shigeki Izumi, Norio Sato and Satoshi Hojo for their cooperation in the data collection. This work was supported by the Japan Society for the Promotion of Science, The Ministry of Education, Culture, Sports, Science, and Technology (Grant-in-Aid for Scientific Research 18K07795, 17K16286 and 16K09005).
Author information
Authors and Affiliations
Contributions
S.I., F.F. and O.I. designed the study protocol. S.I., F.F. and M.U. developed the questionnaire. S.I., M.K., F.F., K.T., M.U., T.H. and S.M. recruited study subjects. S.I. and O.I. performed the statistical analyses and contributed to interpretation of findings. S.I. drafted the initial manuscript. M.K., K.T., F.F., M.U., T.H., S.M., S.S., and O.I. critically reviewed and revised the manuscript. All authors have seen and approved the final version of this manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Iwata, S., Kinoshita, M., Fujita, F. et al. Peripartum depression and infant care, sleep and growth. Sci Rep 9, 10186 (2019). https://doi.org/10.1038/s41598-019-46563-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-46563-4
This article is cited by
-
Influence of mothers’ nighttime responses on the sleep–wake rhythm of 1-month-old infants
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.