Abstract
A topological index of a molecular structure is a numerical quantity that differentiates between a base molecular structure and its branching pattern and helps in understanding the physical, chemical and biological properties of molecular structures. In this article, we quantify four counting polynomials and their related topological indices for the series of a concealed non-Kekulean benzenoid graph. Moreover, we device a new method to calculate the PI and Sd indices with the help of Theta and Omega polynomials.
Similar content being viewed by others
Introduction
Graph theory has numerous applications in modern chemistry. In chemical graph theory, the vertices and edges respectively represent the atoms and bonds of a molecular structure. To predict the chemical structure using numerical quantity (i.e., topological indices) graph theory plays a vital role. Topological indices have many applications in theoretical chemistry, especially in QSPR/QSAR research. Numerous researchers have conducted studies on topological indices for different graph families; these indices have important chemical significance in the fields of chemical graph theory, molecular topology, and mathematical chemistry. Diudea was the first chemist to consider the subject of computing the topological indices of nanostructures1,2,3. A small particle of an object of intermediate size (between the microscopic and molecular structures of the object) is called a nanoparticle of that object. Nanoparticles are products derived through engineering at the molecular scale.
Let G (V, E) be a connected graph with a vertex set V and an edge set E. For any two vertices v1 and v2 in G, the distance between v1 and v2 is denoted by d (v1, v2)—the shortest path between v1, and v2. If e is the edge formed by joining v1 and v2, and f is an edge formed by joining v3 and v4, then e = v1v2 and f = v3v4 are called codistance edges if d (v1, v2) = d (v3, v4) and d (v1, v2) = d (v3, v4) = d (v1, v4) + 1 = d (v3, v2) + 1 and is denoted by ‘e co f’. Here, the corelation is symmetric and reflexive but not transitive. Let C (e) = {f ∈ E (G); f co e}: if the ‘co’ relation is transitive, then the set C (e) is called the orthogonal cut and denoted by co of G. The set of opposite edges that lie along the same face or the same ring, eventually forming a strip of adjacent faces or rings, is called an opposite edge strip and denoted by ‘ops’. This concept is also termed a quasi-orthogonal cut, denoted by ‘qoc’. Here, the co distance edges are defined within the entire graph G, while ‘ops’ are defined in the same face or ring. By m (G, c), we mean the number of strips of length c. In this paper, we constructed four polynomials: Omega, Sadhana, Theta and PI. Counting polynomials are those polynomials whose exponent is the extent of a property partition and whose coefficients are the multiplicity of the corresponding partition. We also calculated the topological indices related to these polynomials and formulae. Each counting polynomial represents interesting topological properties of the molecular graph. These polynomials are constructed on the basis of quasi-orthogonal cut edge strips for the series of concealed non-Kekulean benzenoid graphs. The counting polynomials and matching polynomials are useful for topologically describing bipartite structures as well as for counting some single-number descriptors (i.e., the topological indices). The Omega and Theta polynomials count equidistant edges of the graph, while the Sadhana and PI polynomials count nonequidistant edges. Various results related to counting polynomials and topological indices can be found in1,2,3,4,5,6,7,8,9,10,11,12,13,14.
The Omega polynomial of a graph G (V, E) is denoted byω (G, x); more information can be found in15,16. The Omega polynomial is defined as \(\omega (G,x)=\sum _{c}\,m(G,c){x}^{c}s\), where c is the length of the strip, and ‘m (G, c)’ is the number of strips of length c. The Sadhana polynomial was defined as, \(Sd(G,x)=\sum _{c}\,m(G,c){x}^{|E(G)|-c}\) in17, where |E (G)| is the size of the graph G. The Theta polynomial of graph Gis denoted by ‘θ (G, x)’ and defined as, \(\theta (G,x)=\sum _{c}\,m(G,c)c{x}^{c}\) in18. The PI polynomial is defined in4 as PI (G, x) =\(\sum _{c}\,m(G,c)c{x}^{|E(G)|-c}\).
Series of Concealed Non-Kekulean Benzenoid Graph
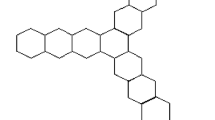
The Kekulean and non-Kekulean structures in benzenoids have important properties from a chemical point of view. It is known that most benzenoids with different numbers of starred and unstarred vertices have no Kekulean structure; these possess color excess and are referred to non-Kekulean benzenoids. In contrast, benzenoids with equal numbers of starred and unstarred vertices necessarily possess Kekulean structures. According to Gutman19, equal numbers of starred and unstarred vertices is a necessary and sufficient condition for a benzenoid structure to be Kekulean. However, it is not true that non-Kekulean with equal numbers of starred and unstarred vertices were detected and later identified as concealed non-Kekulean benzenoids. The total number of edges in the series of concealed non-Kekulean benzenoid graphs shown in Figure 1.1 is 17n + 14, where n is the number of connected edges in the middle of the graph. It has been demonstrated that exactly eight systems of this category exist. If we eliminate the edge cut, which consists of the connected edges, then the graph is decomposed into two parts. Such a structure is called the Kekulean structure of the benzenoid graph. In the present work, we use the concealed non-Kekulean benzenoid graph shown in Fig. 1 19,20,21,22,23.
The series of concealed non-Kekulean benzenoid graphs in Fig. 1 has six quasi-orthogonal cuts (i.e., Si i = 1, 2 … 6) of different lengths. The lengths of the cuts (qoc) and the number of cuts in the series of concealed non-Kekulean benzenoid graphs are n, n + 1, n + 2, 2, 3, and 6 and 1, 2, 2, 4, 4, and 2(n − 1), respectively.
Theorem 1.1
The Omega and Theta polynomials for the series of concealed non-Kekulean benzenoid graph are as follows:
and
Proof
: To calculate the Omega and Theta polynomials of the concealed non-Kekulean benzenoid graphs shown in Figure 1.1, we need to find both the quasi-orthogonal cuts (qoc) and the number of quasi orthogonal cuts of each type. Let Si, where i = 1, 2 … 6 be six ‘qoc’ in a concealed non-Kekulean benzenoid graph. The lengths and cardinalities of these quasi-orthogonal cuts (i.e., Si, where i = 1, 2 … 6) are n, n + 1, n + 2, 2, 3, and 6 and 1, 2, 2, 4, 4, and 2(n − 1), respectively. Because the Omega polynomial is defined as, ω (G, x) =\(\sum _{c}m(G,\,c){x}^{c}\), where c is the length of the cut and m (G, c) represents the number of quasi-orthogonal cuts of length c. Hence, the Omega polynomial calculated from the qocs of a concealed non-Kekulean benzenoid graph is:
Also, by the definition of the Theta polynomial, Θ (G, x) =\(\sum _{c}\,m(G,c)c{x}^{c}\). Hence, the Theta polynomial calculated from the cuts of a concealed non-Kekulean benzenoid graph becomes
Theorem 1.2
The Sadhana and PI polynomials for the series of concealed non-Kekulean benzenoid graphs shown in Figure 1.1, are as follows:
and
Proof
: In the series of concealed non-Kekulean benzenoid graphs, the total number of edges is |E (G)| = 17n + 14, and there are six strips (of qocs) of different lengths, namely, Si,i = 1, 2 … 6. The lengths of these strips are n, n + 1, n + 2, 2, 3, and 6, respectively. The cardinality of the length of Si,i = 1, 2 … 6 is1, 2, 2, 4, 4, and 2(n − 1). From the definition of the Sadhana polynomial, sd (G, x) =\(\sum _{c}m(G,\,c){x}^{|E(G)|-c}\). Therefore, the Sadhana polynomial constructed from the cuts of a concealed non-Kekulean benzenoid graph is:
Additionally, the π Polynomial is defined as π (G, x) =\(\sum _{c}m(G,\,c)c.{x}^{|E(G)|-c}\). By using the lengths of the strips and the number of strips in a concealed non-Kekulean benzenoid graph, the π polynomial becomes;
Table 1. lists the first five polynomials of all four counting polynomials for concealed non-Kekulean benzenoid graphs.
Topological Indices for the Series of Concealed Non-Kekulean Benzenoid Graph
The numerical value of the first derivatives of these counting polynomials at x = 1 yields the interesting properties of the molecular graph. These values are called the topological indices of the graphs. At x = 1, the value of the first derivative of the Omega polynomial gives the total number of edges of the graph, and at x = 1, the Theta polynomials give the same result. The relations for the topological indices related to these polynomials are as follows:
The following Table 2. shows the Omega, Theta, PI, and Sadhana indices calculated from their related polynomials.
In24, John et al. proposed the following formulae to calculate the PI index in terms of the Omega and Theta indices by considering relations (1) and (2).
The omega index and the theta polynomial give the same result at x = 1; therefore, in terms of the theta polynomial relation (5) can be calculated as follows:
The Sadhana index Sd (G) was defined by Khadikar et al. (for more details, we refer readers to6,23,25,26).
The Sadhana index is
where m (G, c) is the number of strips of length c. The Sadhana polynomial Sd (G, x) was defined by Ashrafi et al. in 2008 (see27).
We proposed a new method to calculate the Sadhana index Sd (G) in terms of the Omega index and the Omega polynomial, as given below:
where \({\rm{\Omega }}^{\prime} {({\rm{G}},{\rm{x}})}_{x=1}\) is for the Omega index and Ω(G, x)x=1 is for the Omega polynomial at x = 1.
In Table 3, the PI and Sadhana indices are calculated with the help of the Omega and Theta polynomials.
Results
The Omega and Theta polynomials count the equidistant edges of the graph, while the Sadhana and PI polynomials count the nonequidistant edges of the graph. These polynomials help researchers discuss and predict the molecular structure without necessarily having to refer to quantum mechanics. Hence, we sum up this paper with the following results:
References
Diudea, M. V. Omega polynomial. Carpath. J. Math. 22, 43–47 (2006).
Diudea, M. V. Omega polynomial in all R [8] lattices. Iranian. J. Math. Chem. 1, 69–77 (2010).
Diudea, M. V., Cigher, S. & John, P. E. Omega and related counting polynomials. MATCH Common. Math. Comput. Chem. 60, 237–250 (2008).
Ashraf, A. R., Manoochehrian, M. & Azari, H. Y. On the PI polynomial of a graph. Util. Math. 71, 97–108 (2006).
John, P. E., Vizitin, A. E., Cigher, S. & Diudea, M. V. CI index in tubular nanostructures. MATCH Common. Math. Compute. Chem. 57, 479–484 (2007).
Khadikar., P. V., Joshi, S., Bajaj, A. V. & Mandloi, D. Correlation the benzene character acenes or helicenes and simple molecular discriptors. Bioorg. Med.Chem. Lett. 14, 1187–1191 (2004).
Cyvin, S. J. & Gutman, I. Kekulé structures in benzenoid hydrocarbons. Sepringer-verlag: Berlin, https://doi.org/10.1007/978-3-662-00892-8 (1988).
Baig, A. Q., Imran, M. & Ali, H. Computing omega, sadhana and PI polynomials of benzenoid carbon nanotubes. Optoelectronics and advanced materials-rapid communications. 9, 248–255 (2015).
Prabhu, S., Arulperumjothi, M., Murugan, G., Dhinesh, V. M. & Praveen, J. K. On certain counting polynomial of titanium dioxide nanotubes. Nanoscience & Nanotechnology-Asia. 8, https://doi.org/10.2174/2210681208666180322120144 (2018).
Maqsood, A., Saeed, M., Arshad, M., Cheema, I. Z. & Hussain, M. Algebraic polynomials and invariants of certain chemical networks. Journal of Computational and Theoretical Nanoscience. 15, 1340–1347 (2018).
Yan, L., Yingfang, L., Farahani, M. R. & Imran, M. Sadhana and PI polynomials and their indices of an infinite class of the titania anotubes TiO2 (m, n). Journal of Computational and Theoretical Nanoscience. 13, 8772–8775 (2006).
Imran, M., Baig, A Q., Haidar, S. U., Ali, H. & Hasni, R. Computing topological polynomials of mesh-derived networks. Discrete mathematics, Algorithms and Applications. 10, https://doi.org/10.1142/S1793830918500775 (2018).
Imran, M. & Hayat, S. On counting polynomials of certain polyomino chains. Bulgarian Chemical Communications. 48, 332–337 (2016).
Kwun, Y. C., Munir, M., Nazeer, W., Rafique, S. & Kang, S. M. M-Polynomials and topological indices of V-phenylenic nanotubes and nanotori. Scientific Reports. 7, 8756, https://doi.org/10.1038/s41598-017-08309-y (2017).
Ghorbani, M. & Ghazi, M. Computing omega and PI polynomial of graph. Digest Journal of Nanomaterial and Biostructures. 5, 843–849 (2010).
Ghorbani, M. & Jaddi, M. Computing counting polynomials of leapfrog fullerenes. Optoelectronics and advanced materials-(RC). 4, 540–543 (2010).
Ashrafi, A. R., Ghorbani, M. & Jalali, M. Computing omega and sadhana polynomials of C12n+4 Fullerene. Digest Journal of Nano materials and Biostructures. 5, 403–406 (2009).
Jalali, M. & Ghorbani, M. On Omega polynomial of C40n+6 fullerenes. Studia Universitis Babe-Bolyai. 4, 25–32 (2009).
Gutman, I. The number of Kekul´e structures in conjugated systems containing a linear polyacene fragment. Croatica Chemica Acta. 55, 371–374 (1982).
Gutman, I. & Cyvin, S. J. Introduction to the theory of benzenoid hydrocarbons. Sepringer-verlag: Berlin, https://doi.org/10.1007/978-3-642-87143-6 (1989).
Clar, E. THE AROMATIC SEXTET. Wiley: London, https://doi.org/10.1002/zfch.19730130531 (1972).
Dias, J. R. Resonance structures of benzenoid conjugated radicals. Phys. Chem. Chem. Phys. 1, 5081–5086 (1999).
Khadkar, P. V., Agrawal, V. K. & Karmarkar, S. Prediction of lipophilicity of polyacenes using quantitative structure-activity relationships. Bioorg, Med. Chem. 10, 3499–3507 (2002).
John, P. E., Khadikar, P. V. & Singh, J. A method of computing the PI index of benzenoid hydrocarbons using orthogonal cuts. J. Math. Chem. 42, 27–45 (2007).
Ashrafi, A. R., Jalali, M., Ghorbani, M. & Duidea, M. V. Computing PI and omega polynomials of an infinite family of fullerenes. Communication in Mathematical and in Computer Chemistry. 60, 905–916 (2008).
Farahani, M. R. Sadhana polynomial and its index of hexagonal system B a, b. International journal of computational and theoretical chemistry. 1, 7–10 (2013).
Ashrafi, A. R., Ghorbani, M. & Jalali, M. The vertex PI and szeged indices of an infinite family of fullerenes. Journal of Theoretical and Computational chemistry. 7, 221–231 (2008).
Acknowledgements
This research project was supported by a grant from the Research Center of the Center for Female Scientific and Medical Colleges, Deanship of Scientific Research, King Saud University, Saudi Arabia.
Author information
Authors and Affiliations
Contributions
Mr. Muhammad Nadeem and Dr. Awais Yousaf made substantial contributions to this paper. Mr. Muhammad Nadeem conceived the study and derived the results through the logic of inductive hypotheses. Dr. Awais Yousaf, Mr. Nadeem’s PhD adviser, suggested this problem, validated all the results and helped with manuscript preparation. Hanan Alolaiyan and Abdul Razaq validated the results and prepared the final version of this manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Nadeem, M., Yousaf, A., Alolaiyan, H. et al. Certain polynomials and related topological indices for the series of benzenoid graphs. Sci Rep 9, 9129 (2019). https://doi.org/10.1038/s41598-019-45721-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-45721-y
This article is cited by
-
Valency based novel quantitative structure property relationship (QSPR) approach for predicting physical properties of polycyclic chemical compounds
Scientific Reports (2024)
-
An effective technique for developing the graphical polynomials of certain molecular graphs
Scientific Reports (2023)
-
RETRACTED ARTICLE: Topological analysis of carbon and boron nitride nanotubes
Scientific Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.