Abstract
Physical activity (PA) is known to influence cognitive function. However, the impact of PA on patients with cerebrovascular disease (CVD) has not yet been elucidated. PA and cognitive function have not been measured simultaneously over time, which makes it difficult to evaluate their relationship. The purpose of this study was to investigate the change in the amount of PA recorded by ActiGraph GT3X-BT and six test scores of cognitive function, and the relationship between them in 15 patients with CVD (six women and nine men; 78.0 ± 11.6 years old). Results showed an increase in the amount of PA and scores of cognitive tests, and a significant decrease in the duration of sedentary behavior during the four months (p < 0.05). There were significant correlations between PA Energy Expenditure (PAEE) and Raven’s Colored Progressive Matrices test (RCPM) (r = 0.536, p < 0.0001). There were significant correlations between PAEE and Symbol Digit Modalities Test (SDMT) (r = 0.271, p = 0.036). There were significant correlations between sedentary behavior and RCPM (r = −0.606, p < 0.0001). There were significant correlations between sedentary behavior and SDMT (r = −0.355, p = 0.005). There were significant correlations between Light PA (LPA) and RCPM (r = 0.603, p < 0.0001). There were significant correlations between LPA and SDMT (r = 0.362, p = 0.005).
Similar content being viewed by others
Introduction
Approximately, 5.5 million people worldwide die each year from either primary insult or secondary complications of stroke1. Cerebrovascular Disease (CVD), including stroke (cerebral hemorrhage or cerebral infarction), damages the cerebral tissue. As a result, various forms of neurological symptoms occur in motor and cognitive functions. Concerning the value of physical activity (PA) in patients with CVD, previous studies showed that accelerometers yield valid and reliable data about the PA of patients with stroke2, and that the PA of community dwelling stroke survivors is low3. However, these studies do not show the quantitative change in PA over time.
Regarding cognitive function, cognitive decline resulting from higher brain dysfunction has multiple domains: aphasia, apraxia, agnosia, and unilateral spatial neglect4,5. Moreover, impairments in executive function, attention, and memory (short- and long-term) have been found in 43–78% of individuals with acute stroke, depending on the type, duration, and location of injury6,7. In order to improve these symptoms, cognitive rehabilitation is started on the onset of CVD8. Inpatient rehabilitation is typically initiated when the patient is medically stable enough to be transferred out of acute care. Typically, they are admitted to a dedicated interdisciplinary rehabilitation unit9. There is a wide variability in the length, type, and intensity of services provided in such programs. Kinsella and Ford10 found that the greatest recovery in cognitive function is expected within the first three months. In addition, Desmond et al.11 found that long-term improvement in general cognitive function might be evident after stroke.
Cognition is important for recovery in other neurological domains: patients with higher cognitive status during admission to rehabilitation had better functional outcomes, even when relevant confounding was controlled12. Cognitive impairments can reduce a person’s ability to understand task instructions, plan and initiate self-directed activities, and solve problems4,13. Therefore, further research is necessary in order to improve cognitive function with evidence-based strategies, although there are many potential rehabilitation strategies.
Currently, a growing body of evidence suggests that PA and exercise influence cognitive function. Kirk-Sanchez and McGough 14 reported in the review that evidence from both animal and human studies supports the role of physical exercise in modifying metabolic, structural, and functional dimensions of the brain; and in preserving cognitive performance in older adults. Barnes et al.15 reported that exercise might be one strategy to prevent or delay cognitive decline. In the meta-analysis by Chang et al.16, it was reported that the effects of acute exercise on cognitive performance were generally small. However, larger effects were possible for particular cognitive outcomes and when specific exercise parameters were used. In targeting CVD patients with cognitive decline, the relationship between PA and cognitive function has still not been elucidated. Previous clinical studies of CVD patients reported that acute treadmill exercise improves the function of hemiplegic upper extremity but not cognitive performance17. It has been reported that aerobic exercise improved mobility and selected cognitive domains related to motor learning, which enhances sensorimotor control after stroke18. In summary, regardless of the fact that there is some evidence showing that increased PA after stroke enhances cognitive performance, the pool of studies identified is small, and methodological shortcomings have been identified19. If PA and exercise was related to the cognitive function of patients with CVD, it would be possible to design effective interventions with PA for everyday life and cognitive rehabilitation. Therefore, it is necessary to measure PA and cognitive function simultaneously over time; and to investigate the relationship between them in patients with CVD.
Regarding the measurement of PA level, there are subjective evaluations that use questionnaires and activity record surveys20, and objective evaluations that use accelerometers, pedometers, global positioning systems, and other motion capture devices21. Subjective evaluations have found that memories of activity time and action can be inaccurate and that subjective assessment of activity intensity can vary individually. In objective evaluation, it can be difficult to decide the kind of PA that was specifically performed in the daytime while wearing the monitor, but not while sleeping. On the other hand, cognitive function is, in general, measured by cognitive testing11, although it is sometimes difficult to conduct among people with a damaged ability of comprehension as a result of CVD, among other issues.
The purpose of this study was to investigate the influence of PA on cognitive function in patients with CVD. We investigated the amount of PA and physical inactivity of patients with convalescent CVD over time, and investigated the potential relationship between PA and cognitive function. In our previous study, we evaluated the total energy expenditure as the amount of PA in CVD patients by using the factorial method. We found a significant correlation between total energy expenditure and everyday cognitive function22. In the current study, we identified PA using a wearable accelerometer, ActiGraph. ActiGraph is widely used in clinical settings and home situations. We identified the type of PA that was specifically performed in this study, since the daily activities of patients were observed and determined during 24 hours of hospitalization. This device allows long-term assessment of the patient’s body movements by means of a small solid-state recorder23. In clinical settings, it has been used for sleep medicine24, in treatment studies conducted on patients with Parkinson’s disease25, among patients with Chronic Obstructive Pulmonary Disease (COPD)26, and for stroke patients27. Although hospitalized stroke patients exhibit almost sedentary behaviors, ActiGraph can measure fine movements without burdening the patients. As measurement of cognitive function, we adopted six cognitive tests, which assessed attention, memory, executive function, and general intelligence as comprehensive functions, which can be conducted without difficulties in language comprehension. We hypothesized that there is a relationship between PA and cognitive function in convalescent CVD patients.
Results
Characteristics of subjects
The characteristics of the subjects are listed in Table 1. There were six women and nine men. Their mean age was 78.0 ± 11.6 years (range 48–94 years). Of the 15 subjects, 12 were ischemic, two were hemorrhagic, and one subject had encephalopathy. We recruited one subject with encephalopathy, since we set the inclusion criteria as wide range of brain injury. The mean value of BMI was 20.7 ± 2.7 kg/m2 (range 16.8–25.1), the mean value of years of education was 11.9 ± 3.0 years (range 8–16), the mean value of days since CVD was 44.3 ± 12.6 years (range 19–66), and the mean value of National Institutes of Health Stroke Scale (NIHSS) was 3.3 ± 3.2 points (range 1–12). All the subjects had experienced CVD for the first time.
PA of all the subjects was measured. All the subjects completed the six cognitive tests. The mean raw scores of each physical activity measure over four months are presented in Table 2. The mean raw scores of each of the cognitive assessments over four months are presented in Table 3.
Change in physical activity over four months
We investigated the change in 9 parameters of PA over four months. Significant changes were seen as shown in Fig. 1 over the four months. The value of PAEE and LPA significantly increased, and the value of sedentary behavior significantly decreased over four months. There was a significant main effect of time points (the first month through the fourth month) in Friedman Tests, regarding PAEE (χ2(3) = 10.760, p = 0.013, Fig. 1A), sedentary behavior (χ2(3) = 13.640, p = 0.003, Fig. 1B), and LPA (χ2(3) = 11.000, p = 0.012, Fig. 1C). We conducted Wilcoxon ‘s post hoc test in order to investigate the differences among the 1st, 2nd, 3rd and 4th months. There were significant differences in PAEE, sedentary behavior, and LPA between the 1st month and the 4th month, according to Wilcoxon post-hoc test (Bonferroni-adjusted comparisons), that is, PAEE (z = −3.181, p = 0.006, Fig. 1A), sedentary behavior (z = −2.897, p = 0.024, Fig. 1B), and LPA (z = −2.783, p = 0.03, Fig. 1C). There were no differences between any of the other time points for any of the PA variables.
Box plots showing change in the value of physical activity over four months. (A) PAEE, (B) Sedentary behavior, and (C) LPA. The boxes extend from the 25th to the 75th percentile, with a line at the median and error bars defining the highest and lowest values. Friedman test was performed followed by Wilcoxon post hoc test, comparing the value of each month (*p < 0.05 and **p < 0.01; Bonferroni-adjusted comparisons).
Change in cognitive function over four months
We investigated the change in 6 tests of cognitive function over four months. Significant changes were seen as shown in Fig. 2 in the 15 subjects over the four months. The value of RCPM, SDMT significantly increased over four months. There was a significant main effect of time points (first month through fourth month) in Friedman Tests, regarding RCPM (χ2(3) = 8.2, p = 0.042, Fig. 2A), SDMT (χ2(3) = 9.875, p = 0.020, Fig. 2B). We conducted Wilcoxon ‘s post hoc test in order to investigate the differences among 1st, 2nd, 3rd and 4th months. There were significant differences in RCPM and SDMT, according to Wilcoxon post-hoc test (Bonferroni-adjusted comparisons), between the 1st and 4th month (z = −2.649, p = 0.048, Fig. 2B). There were no differences between any of the other time points for any of the cognitive variables.
Box plots showing change in the value of cognitive function over four months. (A) RCPM, (B) SDMT. The boxes extend from the 25th to the 75th percentile, with a line at the median and error bars defining the highest and lowest values. Friedman test was performed followed by Wilcoxon post hoc test, comparing the value of each month (*p < 0.05; Bonferroni-adjusted comparisons).
Relationship between physical activity and cognitive function
Figure 3(a–f) shows the relationship between the score of PA for each month and the score of cognitive function for each month for 15 subjects.
Relationship between the value of PA and cognitive function. The relationship between the score of PA for each month and the score of cognitive function for each month. Data for 15 individual subjects are presented. Each marker corresponds to an individual subject, and is commonly used in plots (a) through (f).
There were significant correlations between the mean value of PAEE and the mean value of RCPM (r = 0.536, p < 0.0001), that of sedentary behavior and that of RCPM (r = −0.606, p < 0.0001), that of LPA and that of RCPM (r = 0.603, p < 0.0001), that of PAEE and that of SDMT (r = 0.271, p = 0.036), that of sedentary behavior and that of SDMT (r = −0.355, p = 0.005), and that of LPA and that of SDMT (r = 0.362, p = 0.005).
There were significant correlations between the mean value of PAEE and the 4th month’s score of RCPM (r = 0.699, p = 0.004), the mean value of PAEE and the 4th month’s score of SDMT (r = 0.481, p = 0.070), the mean value of sedentary behavior and the 4th month’s score of RCPM (r = −0.744, p = 0.001), the mean value of sedentary behavior and the 4th month’s score of SDMT (r = −0.544, p = 0.036), the mean value of LPA and the 4th month’s score of RCPM (r = 0.735, p = 0.002), and the mean value of LPA and the 4th month’s score of SDMT (r = 0.588, p = 0.021).
Discussion
In the current study, we examined PA and cognitive function for four months, and the relationship between them among 15 CVD patients in convalescent hospital wards. We first evaluated the change in PA by collecting data from the ActiGraph which was worn by subjects, and from cognitive function item scores of six assessments over four months from the time of admission. Next, we examined the correlation between the value of PA measures and cognitive function item scores.
As shown in Figs 1 and 2, significant changes in the PA and cognitive function were found. In previous studies, Manns and Evan 28 reported that minutes of activity and length of activity bouts significantly increased from predischarge to 6 weeks postsdischarge. Although the term of measurement was different from that study, we obtained a similar result. LPA increased significantly, but higher intensity activity measures, such as MVPA did not, and patients gradually engaged less in sedentary behaviors. Regarding cognitive function, RCPM and SDMT scores increased significantly, but other assessments did not. It was indicated that general intelligence, and divided attention in many domains of cognitive function improve in CVD patients in this study. Hochstenbach et al.29 reported that there was room for improvement in all cognitive domains after stroke. The study showed that the largest improvement was found in the attentional domain and the least improvement was found in the memory domain. In the current study, intelligence and divided attention improved, which is consistent with this preceding study. Nonetheless, as we did not have control subjects, the cause-and-effect relation between PA and cognitive function could not be revealed in the current study. On the other hand, selective and alternated attention, executive function, and memory did not improve in four months in the current study. Particularly, the value of the symbol cancellation task was stable. A previous study29 showed that attention domain improved but memory domain did not improve through 2-year follow up study. We obtained controversial result compared to the previous study, and it is necessary to confirm this point recruiting greater numbers of subjects. Regarding memory, there is a possibility that memory is difficult to improve. Or, it could be considered to be a ceiling effect since the subject of the current study was able to almost completely carry out the memory task.
It was suggested that the higher the PAEE and LPA, the higher the cognitive function value; and the lower the sedentary behavior is, the higher the cognitive function value. The present findings indicate that PA among people with CVD is potentially related to their cognitive function.
The current study has several limitations. First, the current study had a small sample size comprising individuals in an inpatient rehabilitation unit. Thus, our findings are not likely to be conclusive. To intensively investigate changes in PA and the cognitive function of CVD patients over time, and to establish evidence of the positive relationship between PA and cognitive function in CVD patients, it is necessary to recruit a greater number of participants. Second, regarding cognitive assessments, the score on Symbol Cancellation, Design Memory, and Mazes did not change over the four months. It is necessary to confirm whether these domains could not actually improve or whether other tests would have been more appropriate. We will conduct a future follow up study that will consider these points.
In the current study, we found a significant change in the score of PAEE, LPA, sedentary behavior, and general intelligence and attention as cognitive functions. We also found that PA was potentially related to cognitive function among people with brain damage. Aarsland et al.30, Larson et al.31, and Podewils et al.32 reported that cognition in Alzheimer’s disease patients was also highly related to energy expenditure and exercise, and that PA helped to prevent disease progression. Likewise, the present study found a potential correlation between PA and cognitive function in patients with CVD. However, the current study did not explore a causal relationship between PA and cognitive function. Thus, in the future, it will be necessary to further investigate through intervention experiments, whether PA is effective in improving the cognitive function of patients with CVD using an intervention study. We will do intervention studies to clarify that PA is valid for cognitive function.
Materials and Methods
Participants
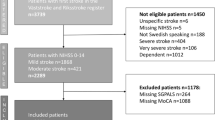
Adults who experienced cerebrovascular disease, and who were suspected to have cognitive deficit based on the Mini-Mental State Examination33, were recruited from Nishiyamato rehabilitation hospital (Nara, Japan). The inclusion criteria were patients who (1) had diseases and disorders of the nervous system, that is, nonspecific cerebral vascular disorders including post stroke (ischemic or hemorrhagic), traumatic brain injury, and encephalitis and encephalopathy, (2) completed their acute treatment and were in stable condition, and (3) were ready for rehabilitation. The exclusion criteria were the (1) appearance of delirium and fever symptoms during hospitalization, (2) large fluctuation in mental state, (3) refusing rehabilitation, (4) inability to stay in the hospital with other patients, and (5) inability to wear the monitoring device on the waist. Subjects in the inpatient rehabilitation unit typically underwent three basic therapies: physical therapy (PT), occupational therapy (OT), and speech language therapy (ST). PT provides fundamental training to perform basic activities, gait training, and balance training. OT provides training to perform activities of daily living (ADL), instrumental activities of daily living (IADL), and upper extremity training. ST provides training on recovering speech, language, and cognitive communication; and on swallowing. OT and ST perform the paper-and-pencil cognitive tasks in a quiet therapy room.
This study was conducted according to the Declaration of Helsinki and approval for the project was obtained from the local ethics committee (Department of Rehabilitation Medicine, Nishiyamato Rehabilitation Hospital, Nara, Japan, protocol number 16). All participants gave their written informed consent to participate in this study, which was approved by Nishiyamato rehabilitation hospital. Procedures in accordance with institutional guidelines were followed in this study.
Procedure
The value of PA, sedentary behavior, and six cognitive test scores were collected per month from the first month of admission to the fourth month. PA and sedentary behavior were recorded for two to seven days (at least two days) in a month. Participants wore a PA monitor, ActiGraph GT3X-BT (ActiGraph Corp., Pensacola, Florida) on the waist. A speech language pathologist conducted six cognitive tests on any day of wearing an ActiGraph. This study was carried out for total for 15 months in total. The subjects screened in this study are shown in the flowchart (see Fig. 4).
First, the average values of PA per day for all subjects were calculated. As PA value, we adopted nine outcomes which were Physical Activity Energy Expenditure (PAEE), sedentary behavior, time spent on Light-intensity Physical Activity (LPA), Moderate-intensity Physical Activity (Moderate Physical Activity), Vigorous-intensity Physical Activity (Vigorous Physical Activity), Very Vigorous-intensity Physical Activity (Very Vigorous Physical Activity), time spent on Moderate-to-vigorous intensity physical Activity (MVPA), vector magnitude overall physical activity counts per minute (cpm; Vector Magnitude Count), and steps. Count per minute (cpm) represents colloquial expression for number of times something (e.g., heartbeat) happens within a 60-second duration34. At the same time, the score of six cognitive tests was measured, and then, these changes were noted. We identified significant changes in the values of nine outcomes of PA value and six tests scores of cognitive measurements. Next, we examined the correlation between PA value and the cognitive test scores.
Measurement of physical activity
PA was measured with an accelerometer, ActiGraph GT3X-BT (ActiGraph Corp., Pensacola, Florida), worn on the waist. Concerning the reliability of Actigraph in people with post-stroke, the Intraclass correlation coefficients (ICC) was high when comparing activity counts for the paretic and non-paretic hips (0.96)35. The location of wearing an accelerometer, e.g., waist, wrist, and ankle, etc. may influence PA estimates. As a validation study, Shiroma et al.36 reported that similar patterns of waist and wrist activity accrual indicated that each location was capable of estimating total PA volume. LaMunion et al.37 also reported that Actigraph y-axis and vector magnitude counts did not level off when the monitoring device was worn at the hip. Therefore, we observed that reliable measurement of PA can be obtained by wearing Actigraph at the waist. We included the hemi-paretic patients in this study. It was considered that the device would be an obstacle, if it was worn on the non-paretic wrist and ankle, which are used actively throughout the day. Therefore, this device was worn on the waist in this study. Each participant wore the device continuously: day and night. The inclusion criteria regarding the use of accelerometers were the ability to wear the device continuously (without removing) for at least two days to a maximum of seven days. This protocol was used because some participants could not continuously wear the device for a long period of time. We used the average value per day for two to seven days in statistical analysis. The activities of the participant were determined per hour. ActiLife software (version 6.13.3; ActiGraph Corp., Pensacola, Florida) was used to initialize the accelerometers and download the data. Customized software was used for data reduction and analysis. We used vector magnitude units (VMU). VMU represents total movements in three planes for each minute, and is used to categorize PA between high and low levels. The outcome variables of PA and the thresholds we used in this study were PAEE (kcal/day), sedentary behavior (min/day; < = 99 cpm), LPA (min/day; 100–1951 cpm), Moderate Physical Activity (min/day; 1952–5274 cpm), Vigorous Physical Activity (min/day; 5725–9498), Very Vigorous Physical Activity (min/day > = 9499), MVPA (min/day; > = 1952 cpm), Vector Magnitude Count (counts/minute), and steps (counts/minute) which is obtained from the algorithms of the Freedson Combination38. We used the Low-Frequency Extension filter. The Low-Frequency Extension option should only be used in very specific cases where PA is at such a low level that it might otherwise be eliminated with the normal filter. We used this filtering because subjects of this study exhibited very slow shuffling movements. The epoch length was 60 seconds, which represent the amount of time the raw acceleration data is summed after the filter is applied.
Measurement of cognitive function
We conducted six cognitive tests in this study: Raven’s Progressive Matrices Test (RCPM); Symbol Digit Modalities Test (SDMT); and four tests from the nonlinguistic tasks of Cognitive Linguistic Quick Test39, including Symbol Trails, Symbol Cancellation, Mazes, and Design Memory. These tests were conducted because they can assess different main aspects of cognitive function, and can be administered easily; even participants with language comprehension difficulties could understand and conduct these tests. Participants of this study could use pencils for writing using their dominant or non-dominant hand.
Raven’s colored progressive matrices test
This test assesses general intelligence. Raven’s Progressive Matrices (Raven, 1976) provide a trusted, nonverbal assessment of intelligence. Since these scales minimize the impact of language skills and cultural bias, they are particularly well suited for measuring the intelligence of individuals with reading problems or hearing impairment, as well as those whose native language is not English. Appropriate for both children and adults, Raven’s Progressive Matrices measure two complementary components of general intelligence: the capacity to think clearly and make sense of complex data (educative ability), and to store and reproduce information (reproductive ability)40,41.
Symbol digit modalities test
This test assesses divided attention. The SDMT (Smith, 1982)42 was developed to identify individuals with neurological impairment. The SDMT assesses key neurocognitive functions that underlie many substitution tasks, including attention, visual scanning, and motor speed. The SDMT requires individuals to identify nine different symbols corresponding to the numbers one through nine, and practice writing the correct number under the corresponding symbol. Afterwards, they manually fill the blank space under each symbol with the corresponding number. The second oral administration is then completed. The participant is given a blank copy of the test and asked to state the correct number for each corresponding symbol. They have 90 seconds to complete each of these administrations. The written and oral score is calculated by totaling the number of correct answers for each section. Oral and written administrations provide two different indices of functioning, which assess attention, scanning abilities, and motor skills43.
Symbol trails
This test assesses the executive functions of planning, working memory, and mental flexibility without placing demands on the language system. However, this task also calls upon visual attention and perception. Two learning trails involving the same concepts of size and shape are used in preparation of the test item, which involves drawing a single line to connect a total of 11 circles and triangles in an alternating fashion according to size and shape and beginning with the smallest circle. The maximum possible score is 10 points39.
Symbol cancellation
This test assesses visual attention, scanning, discrimination, inhibition, and response shifting within four quadrants of space. Thirty-six abstract symbols are arranged in a pseudo-random fashion with the target stimulus appearing thrice in each quadrant of space to allow for assessment of visual field deficits and visual neglect. Discriminating the target stimulus from resembling symbols is necessary. The task is to cross out target symbols. The highest possible score is 12 points39.
Mazes
This test assesses executive functions, specifically those involved in planning a course of action, rejecting/inhibiting incorrect choices, and correcting mistakes. Two mazes of different levels of difficulty are used. The highest possible score for each maze is four (correct solution) with a total of eight points39.
Design memory
This test assesses immediate/working visual memory and attention without language demands. Two target abstract designs are presented one at a time for memorization. They must be identified immediately from arrays of six designs that include four foils that are similar to the targets. The highest possible score is six points39.
Statistical analysis
Due to the small sample size, non-parametric statistics were used. Friedman Tests were performed to compare the values per month from the first month of admission to the fourth month: average kcal values per month for PAEE, sedentary behavior time, time spent on LPA, Moderate-intensity Physical Activity, Vigorous-intensity Physical Activity, Very Vigorous-intensity Physical Activity, and MVPA, vector magnitude overall physical activity counts per minute, step counts per minute, and six cognitive test scores including RCPM, SDMT, Symbol Trails, Symbol Cancellation, Mazes, and Design Memory. When significant differences were found in Friedman Tests, Wilcoxon Tests as post-hoc test were conducted to examine the differences between each value by using Bonferroni-adjusted correction. For the parameters in which significant differences were found in Friedman Tests, the relationship between the mean value of PA for all four months and the mean value of cognitive tests score for all four months, and the relationship between the mean value of PA and the cognitive tests score of the 4th month were analyzed using Spearman’s correlation coefficient. The level of statistical significance was determined as p = 0.05. For the statistical analysis, we used IBM SPSS statistics for Mac, version 25.
References
WHO: World Health Report. Geneva: World Health Organization (2003).
Gebruers, N. et al. Monitoring of physical activity after stroke: a systematic review of accelerometry-based measures. Arch Phys Med Rehabil. 91, 288–97 (2010).
Field, M. J. et al. Physical activity after stroke: a systematic review and meta-analysis. Hindawi Publishing Corporation ISRN Stroke. 1–13 (2013).
Skidmore, E. R. et al. Cognitive and affective predictors of rehabilitation participation after stroke. Arch Phys Med Rehabili. 91, 203–7 (2010).
Gottesman, R. F. & Hillis, A. E. Predictors and assessment of cognitive dysfunction resulting from ischemic stroke. Lancet Neurol. 9, 895–905 (2010).
Lesniak, M. et al. Frequency and prognostic value of cognitive disorders in stroke patients. Dement Geriatr Cogn Disord. 26, 356–63 (2008).
Zinn, S., Bosworth, H. B., Hoeing, H. M. & Swartzwelder, H. S. Executive function deficits in acute stroke. Arch Phys Med Rehabil. 88, 173–80 (2007).
Carney, N. et al. Effect of cognitive rehabilitation on outcomes for persons with traumatic brain injury: A systematic review. Journal of Head Trauma Rehabilitation. 14, 277–307 (1999).
Cullen, N., Chundamala, J., Bayley, M. & Jutai, J. Erabi, Group. The efficacy of acquired brain injury rehabilitation. Brain Inj. 21, 113–32 (2007).
Kinsella, G. & Ford, B. Acute recovery from patterns in stroke patients: neuropsychological factors. Med J Aust. 13, 663–6 (1980).
Desmond, D. W., Moroney, J. T., Sano, M. & Stern, Y. Recovery of cognitive function after stroke. Stroke. 27, 1798–1803 (1996).
Helti, R. J. et al. Rehabilitation outcome of elderly patients after a first stroke: effect of cognitive status at admission on the functional outcome. Arch Phys Med Rehabil. 83, 742–9 (2002).
Cumming, T. B., Marshall, R. S. & Lazar, R. M. Stroke, cognitive deficits, and rehabilitation: still an incomplete picture. International Journal of stroke. 8, 38–45 (2013).
Kirk-Sanchez, N. J. & McGough, E. L. Physical exercise and cognitive performance in the elderly: current perspectives. Clin Interv Aging. 9, 51–62 (2014).
Barnes, J. N. Exercise, cognitive function, and aging. Adv Physiol Educ. 39, 55–62 (2015).
Chang, Y. K., Labban, J. D., Gapin, J. I. & Etnier, J. L. The effects of acute exercise on cognitive performance: a meta-analysis. Brain Res. 9, 87–101 (2012).
Ploughman, M. et al. Does treadmill exercise improve performance of cognitive or upper-extremity tasks in people with chronic stroke? A randomized cross-over trial. Arch Phys Med Rehabil. 89, 2041–7 (2008).
Quaney, B. M. et al. Aerobic exercise improves cognition and motor function poststroke. Neurorehabil Neural Repair. 23, 879–85 (2009).
Cumming, T. B. et al. The effect of physical activity on cognitive function after stroke: a systematic review. Int Psychogeriatr. 24, 557–67 (2012).
Dollman, J. et al. A hitchhiker’s guide to assessing young people’s physical activity: Deciding what method to use. J Sci Med Sport. 12, 518–525 (2009).
Kolt, G. The complexity of measuring physical activity and sedentary behavior. Journal of Science and Medicine in Sport. 16, 1 (2013).
Kojima, M. & Nagano, A. The Association between Total Energy Expenditure and Cognitive Function in Convalescent Patients with Cerebrovascular Disease. Int Journal of Sport and Health Science. 16 (2018).
Klösch, G., Gruber, G., Anderer, P. & Saletu, B. Activity monitoring in sleep research, medicine and psychopharmacology. Wien Klin Wochenschr. 17(113), 288–95 (2001).
Kazenwadel, J. et al. New actigraphic assessment method for periodic leg movements (PLM). Sleep. 18, 689–97 (1995).
van, Hilten, J. J., Middelkoop, H. A., Kerkhof, G. A. & Roos, R. A. A new approach in the assessment of motor activity in Parkinson’s disease. J Neurol Neurosurg Psychiatry. 54, 976–9 (1991).
Ehsan, M. et al. A longitudinal study evaluating the effect of exacerbations on physical activity in patients with chronic obstructive pulmonary disease. Ann Am Thorac Soc. 10, 559–64 (2013).
Reiterer, V. et al. Actigraphy-a useful tool for motor activity monitoring in stroke patients. Eur Neurol. 60, 285–91 (2008).
Manns, P. J. & Baldwin, E. Ambulatory Activity of Stroke Survivors: Measurement Options for Dose, Intensity, and Variability of Activity. Stroke. 40, 864–867 (2009).
Hochstenbach, J. B., den, Otter., R. & Mulder, T. W. Cognitive recovery after stroke: a 2-year follow-up. Arch Phys Med Rehabil. 84, 1499–504 (2003).
Aarsland, D., Sardahaee, F. S., Anderssen, S. & Ballard, C. Is physical activity a potential preventive factor for vascular dementia? A systematic review. Aging Ment Health. 14, 386–95 (2010).
Larson, E. B. et al. Exercise is associated with reduced risk for incident dementia among persons 65 years of age and older. Ann Intern Med. 17, 73–81 (2006).
Podewils, L. J. et al. Physical activity, APOE genotype, and dementia risk: findings from the cardiovascular health cognition study. Am J Epidemiol. 161, 639–651 (2005).
Folstein, S. E., Fostein, P. R. & McHugh, P. R. “Mini-mental state”: A practiceal method for grading the cognitive state of patients for clinician. J. Psychiatri. Res. 12, 189–198 (1975).
Medical Dictionary for the Health Professions and Nursing. Retrieved December 23 2018 from https://medical-dictionary.thefreedictionary.com/counts+per+minute (2012).
Rand, D. et al. How active are people with stroke?: use of accelerometers to assess physical activity. Stroke. 40, 163–8 (2009).
Shiroma, E. J. et al. Daily physical activity patterns from hip- and wrist-worn accelerometers. Physiol Meas. 37, 1852–1861 (2016).
LaMunion, S. R., Bassett, D. R. Jr., Toth, L. P. & Crouter, S. E. The effect of body placement site on Actigraph wGT3X-BT activity counts. Biomed. Phys. Eng. Express. 3, 035026 (2017).
Freedson, P. S., Melanson, E. & Sirard, J. Calibration of the Computer Science and Applications, Inc. accelerometer. Medicine and Science in Sports and Exercise. 30, 777–781 (1998).
Helm-Estabrooks, N. Cognition and aphasia: a discussion and a study. J Commun Disord. 35, 171–86 (2002).
Raven, J. C. & Court, J. H. Manual for Raven’s coloured progressive matrices. HK Lewis London (1976).
Basso, A., Capitani, E. & Laiacona, M. Raven’s coloured progressive matrices: Normative values on 305 adult normal controls. Functional Neurology. 2, 189–194 (1987).
Smith, A. Symbol Digits Modalities Test. Los Angeles: Western Psychological Services (1982).
Drake, A. S. et al. Psychometrics and normative data for the Multiple Sclerosis Functional Composite: replacing the PASAT with the Symbol Digit Modalities Test. Mult Scler. 16, 228–37 (2010).
Acknowledgements
We thank for all the patients for participating in our study. This research was supported in part by grants from The 44th Daiwa Securities Health Foundation. We would like to thank Editage (www.editage.jp) for English language editing.
Author information
Authors and Affiliations
Contributions
Conceived and/or designed the work that led to the submission (all authors); acquired data (M.K.), or played an important role in interpreting the results (all authors); drafted the article (M.K.), revised the article (A.N.), and approved the final version (all authors).
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kojima, M., Nagano, A. Assessment of physical activity and cognitive function and their potential correlation in convalescent patients of cerebrovascular disease. Sci Rep 9, 3782 (2019). https://doi.org/10.1038/s41598-019-40460-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-019-40460-6
This article is cited by
-
Total Sedentary Time and Cognitive Function in Middle-Aged and Older Adults: A Systematic Review and Meta-analysis
Sports Medicine - Open (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.