Abstract
Abdominal aorta calcification (AAC) has been associated with clinical outcomes in peritoneal dialysis (PD) patients. Heart rhythm complexity analysis has been shown to be a promising tool to predict outcomes in patients with cardiovascular disease. In this study, we aimed to analyze the association between heart rhythm complexity and AAC in PD patients. We prospectively analyzed 133 PD patients. Heart rhythm complexity including detrended fluctuation analysis and multiscale entropy was performed. In linear analysis, the patients in the higher AAC group (AAC ≥15%) had a significantly lower standard deviation of normal RR intervals, very low frequency, low frequency, high frequency and low/high frequency ratio. In non-linear analysis, DFAα1, slope 1–5, scale 5 and area 6–20 were significantly lower in the patients with higher AAC. Receiver operating characteristic curve analysis showed that DFAα1 had the greatest discriminatory power to differentiate these two groups. Multivariate logistic regression analysis showed that DFAα1 and HbA1c were significantly associated with higher AAC ratio. Adding DFAα1 significantly improved the discriminatory power of the linear parameters in both net reclassification improvement and integrated discrimination improvement models. In conclusion, DFAα1 is highly associated with AAC and a potential cardiovascular marker in PD patients.
Similar content being viewed by others
Introduction
Cardiovascular disease (CVD) is the leading cause of morbidity and mortality in end-stage renal disease (ESRD) patients1, accounting for about 40% of deaths among these patients during the first 3 years of dialysis2. Because of the high cardiovascular mortality rate in dialysis patients (almost 5–30 times greater than in the general population)3, the burden of CVD on patients starting dialysis and its impact on the survival of dialysis patients has recently received increasing attention2,3.
Possible explanations for the high rates of CVD mortality and mobility in ESRD patients include atherosclerosis-related vascular complications and autonomic nervous system dysfunction4. Abdominal aorta calcification (AAC) has been reported to predict CVD events and mortality in ESRD patients, including those undergoing peritoneal dialysis (PD)5,6,7. The abdominal aorta calcification can be measured via X ray with grading systems8 or computed tomography (CT) with direct measurement of the percentage of AAC (%AAC)9. The %AAC has been shown to be independently associated with mortality and hospitalization in PD patients7. The cutoff value of 15% AAC in CT has been reported that predicting clinical outcomes in PD patients7. However, the clinical use of %AAC measurement is limited by radiation exposure and medical cost.
Analysis of beat-to beat variation of heart rate, also known as heart rate variability (HRV), is commonly used in cardiovascular researches as a simple and noninvasive approach10. HRV has also been commonly used to predict CVD outcomes11. Newer biological signal analysis methods based on nonlinear signal modeling and complexity evaluation including detrended fractal analysis (DFA) and multiscale entropy (MSE) have been developed in recent years12. Compared to traditional HRV parameters, nonlinear heart rhythm complexity analysis has a better prognostic power in patients with CVD. In addition, both DFA and MSE have been shown to be useful in predicting survival of heart failure patients13,14.
To the best of our knowledge, no previous study has investigated the association between heart rhythm complexity and AAC. Therefore, the aim of this study was to analyze the association between heart rhythm complexity and the severity of AAC in PD patients.
Results
Patients
A total of 133 PD patients (61 men) were enrolled in this study, including 59 (26 men) with AAC ≥15% (higher AAC group) and 74 (35 men) with AAC <15% (lower AAC group). The clinical data are shown in Table 1. The AAC ratio of the whole population, AAC ≥15% group and AAC <15% were 10.38 (0.53–30.70), 33.87 (27.25–46.39) and 1.32 (0.00–8.74), respectively. Patients in higher AAC group were significantly older and had higher incidences of diabetes mellitus (DM), HbA1c, fasting serum glucose, C-reactive protein (CRP), and lower serum creatinine and left ventricular ejection fraction (LVEF). Other clinical parameters including peritoneal dialysis efficiency (PD KT/V), percentage of beta-blocker and calcium channel blocker usage were comparable in both groups (Table 1).
Holter data
In linear analysis, the patients in the higher AAC group had a significantly lower standard deviation of normal RR intervals (SDRR), very low frequency (VLF), low frequency (LF), high frequency (HF) and low frequency to high frequency ratio (LH/HF ratio) than those in the lower AAC group. In non-linear analysis, DFAα1 was significantly lower in the higher AAC group. The value of DFAα2 was comparable between the two groups. In MSE analysis, the patients in the higher AAC group had significantly lower slope 1–5, scale 5, and area 6–20 than the patients in the lower AAC group (Table 2).
Differentiation between the higher and lower AAC groups
Receiver operating characteristic (ROC) curve analysis showed that DFAα1 had the greatest discriminatory power to differentiate the two groups compared to other linear, non-linear and clinical parameters (Fig. 1).
(A, B) Analysis of the discrimination power of linear and non-linear parameters to discriminate patients with higher AAC (AAC ≥15) by receiver operating characteristic curve analysis (A) The areas under the curve of SDRR, VLF, LF, HF, LF/HF ratio were 0.663, 0.752, 0.721, 0.605 and 0.723 respectively. (B) The areas under the curve of DFAα1, slope 5, scale 5 and area 6–20 were 0.781, 0.707, 0.657 and 0.667, respectively.
The areas under the curves (AUC) of HRV parameters including SDRR, the percentage of absolute differences in normal RR intervals greater than 50 ms (pNN50), pNN20, VLF, LF, HF, LF/HF ratio DFAα1, DFAα2, slope 1–5, scale 5, area 1–5, and area 6–20 were 0.663, 0.457, 0.548, 0.752, 0.721, 0.605, 0.723, 0.781, 0.509, 0.707, 0.657, 0.578 and 0.667, respectively.
The AUC of clinical parameters including age, DM, fasting serum glucose, HbA1c, creatinine, CRP and LVEF were 0.641, 0.651, 0.683, 0.729, 0.580, 0.622 and 0.619, respectively.
Logistic regression analysis to predict the higher AAC group
In univariate logistic regression analysis, age, DM, fasting serum glucose, HbA1c, creatinine, LVEF, SDRR, VLF, LF, LF/HF ratio, DFAα1, slope 1–5, scale 5, and area 6–20 were significantly associated with the presence of higher AAC. In multivariate logistic regression analysis, only DFAα1 (OR = 0.032, 95% CI 0.005 to 0.212, p < 0.001) and HbA1c (OR = 3.497, 95% CI 1.727 to 7.084, p = 0.001) remained in the model, and both were associated with higher AAC (Table 3).
Correlations of HRV parameters and percentage of AAC
In univariate linear regression analysis, age, DM, fasting serum glucose, HbA1c, creatinine, LVEF, SDRR, VLF, LF/HF ratio, DFAα1, slope 1–5, scale 5, and area 6–20 were significantly associated with the percentage of ACC. In the multivariate linear regression model, DFAα1 (β:−31.189, 95% CI −44.829 to −17.550, p < 0.001), LF/HF ratio (β:1.111, 95% CI 0.161 to 2.060, p = 0.022), age (β:0.293, 95% CI 0.057 to 0.529, p = 0.015) and HbA1c (β:4.744, 95% CI 1.640 to 7.847, p = 0.003) were significantly associated with the percentage of AAC (Table 4).
The advantage of adding DFA or MSE parameters to the linear parameters to discriminate the higher and lower AAC groups
DFAα1 and slope 1–5 significantly improved the discriminatory power of SDRR, VLF, LF, HF and LF/HF ratio in both net reclassification improvement (NRI) and integrated discrimination improvement (IDI) models. In addition, area 6–20 significantly improved the discriminatory power of SDRR, LF, HF and LF/HF ratio in the IDI model, and SDRR and HF in the NRI model. Scale 5 significantly improved the discriminatory power of SDRR, HF and LF/HF ratio in the IDI model and HF in NRI model (Table 5).
Discussion
There were three major findings in this study. First, the PD patients with higher AAC had worse heart rhythm complexity. Second, in all linear and non-linear parameters, DFAα1 had the greatest single discriminatory power to detect PD patients with higher AAC. Third, non-linear parameters, especially DFAα1, significantly improved the discriminatory power of the linear parameters to differentiate PD patients with higher or lower AAC.
In daily practice, predicting the clinical outcomes of PD patients is a challenge. Atherosclerosis-related vascular calcification has been highly associated with morbidity and mortality in ESRD patients5,6,15. In the advanced stage of atherosclerosis such as atheroma formation, a partial or extensive calcium deposit is frequently observed16. Therefore, blood vessel calcification implies the presence atherosclerosis or subclinical CVD17,18. Several traditional risk factors for atherosclerosis such as dyslipidemia, hypertension, smoking, and age have also been associated with vascular calcification in ESRD patients19,20. In addition, uremia, mineral metabolism, chronic inflammation, fetuin-A and osteoprotegerin (OPG) have also been reported to contribute to vascular calcification21,22. Several studies have reported significant associations between qualitative or semiquantitative evaluations of arterial calcification and all-cause and cardiovascular mortality in hemodialysis patients23,24,25. In our study, HbA1c and age were significantly associated with %AAC in multivariate linear regression model. Age and HbA1c are known risk factors of vascular calcification26,27. In addition, HbA1c levels are associated with mortality in ESRD patients28,29. Even in PD patients without diabetes, higher HbA1c is still associated with higher cardiovascular events30.
The AAC severity measurements include X ray with Kauppila score8 and CT with direct measurement of %AAC9. Previous study supported that CT appeared to be more sensitive than plain X-rays at detecting peripheral and aortic vascular calcifications in hemodialysis patients31. Tsushima et al. developed a method to measure the percentage of calcified volume against whole vascular volume using CT9,32 and CT remains the reference standard in AAC evaluation33. AAC was reported to be an important predictor of vascular morbidity and mortality in the Framingham Heart Study34, and it has also been reported to be associated with clinical outcomes in ESRD patients5,6,7. The percentage of AAC has been shown to be independently associated with mortality and hospitalization in PD patients7. In addition, patients with AAC ≥15% had more cardiovascular events than those with AAC <15%7. However, despite the usefulness of %AAC by CT, the radiation exposure and cost were limited the use of this tool.
In contrast, electrocardiography (ECG) is an easy, low cost and radiation-free examination. In the present study, we found high correlations among the HRV parameters (especially DFAα1) and AAC. This indicates that ECG recording followed by HRV analysis using linear and non-linear parameters has the potential to be an alternative to AAC in clinical practice.
Previous studies have reported an association between the progression of coronary and carotid artery atherosclerosis and autonomic dysfunction35,36. Despite the reported association between autonomic dysfunction and atherosclerosis37, the mechanisms linking autonomic imbalance to atherosclerosis are still unclear. In addition to atherosclerosis-related autonomic dysfunction, uremic autonomic neuropathy in ESRD patients has frequently been associated with parasympathetic damage and sympathetic nerve overactivity38, both of which have been associated with worse clinical outcomes in ESRD patients39. In our previous study, PD patients had significantly lower values of several linear and nonlinear parameters than those with normal renal function, and this also supports the hypothesis of prominent autonomic dysfunction in ESRD patients40.
In the present study, DFAα1 had a better correlation with AAC than linear parameters, which implies that non-linear parameters provide more useful information. The non-linear analysis of HRV including MSE and DFA has been reported to be a better predictor of clinical outcomes than traditional linear analysis. MSE has been associated with the prognosis of heart failure13, outcome of acute stroke41, primary aldosteronism42, critical illnesses requiring extracorporeal life support43, and post-myocardial infarction heart function44. Furthermore, long-time scale parameters (area 6–20) in heart failure patients have been shown to have the best prognostic predictive power13, which is similar to our MSE results. DFA as a scaling analysis method to determine the statistical self-affinity of a signal can be used for the evaluation of the fractal behavior in the heart beat dynamics. The short-term (α1; 4–11 beats,) and long-term (α2; 11–64 beats) fractal correlation exponents have been shown to provide a clearer understanding of the fractal correlation property in a physiological system45. DFA has also been associated with the interaction between sympathetic and vagal systems46. In the DIAMOND-CHF trial, after adjusting for clinical parameters, DFAα1 but not linear parameters remained to be an independent predictor of mortality14. Taken together with our findings, non-linear HRV analysis may be a useful tool to evaluate the risk of cardiovascular events.
The traditional linear HRV parameters have also been positively associated with CVD risk factors and multiple cardiovascular outcomes including coronary artery disease and cardiovascular mortality47,48. We also found similar results in that linear HRV parameters including SDRR, VLF, LF, HF and LF/HF ratio were also significantly associated with AAC. In addition, combining linear and non-linear analysis further significantly improved the discriminatory power of the severity of AAC. Combining these linear and non-linear HRV parameters can provide more accurate information to build a ROC curve model to predict the severity of AAC.
There are several limitations to this study. First, this is a small pilot study and the findings should be confirmed by a larger clinical study with long-term follow-up data. Second, our study group is limited to PD patients, and further studies are needed to elucidate whether the same association between AAC and heart rhythm complexity exists in hemodialysis patients.
In conclusion, heart rhythm complexity analysis can predict the severity of AAC in PD patients. DFAα1 had the greatest discriminatory power to differentiate PD patients with higher or lower AAC. In addition, DFAα1 and MSE slope 1–5 significantly improved the discriminatory power of the linear parameters, which suggests the advantage of combining linear and non-linear parameters.
Methods
Patients
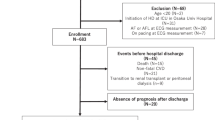
We prospectively enrolled 133 patients who received PD with conventional glucose-based lactate-buffered solution (UltraBag; Baxter Healthcare SA, Singapore) for more than 6 months. Patients with chronic atrial fibrillation, clinical signs of acute infection, and those with a prior renal transplant were excluded. The baseline characteristics, medical history and medication usage were carefully recorded, and biochemical parameters were measured during initial evaluation. All patients received 24-h ambulatory ECG Holter recording (ZymedDigiTrak Plus 24-Hour Holter Monitor Recorder and Digitrak XT Holter Recorder 24 Hour, Philips, Amsterdam, Netherlands). This study was approved by the Institutional Review Board of National Taiwan University Hospital, and all research was performed in accordance with relevant guidelines and regulations. All subjects provided written informed consent including for storage of their information in the hospital database and usage for research.
Data pre-processing
A stable 4 hours segment of daytime RR intervals (between 9AM and 5PM) was selected for analysis. The selected electrocardiograms were automatically annotated via an algorithm and carefully examined by two experienced technicians.
Time and frequency domain analysis
All parameters were calculated according to the recommendations of the North American Society of Pacing Electrophysiology and the European Society of Cardiology10. SDRR and the percentage of absolute differences in normal RR intervals greater than 50 ms (pNN50) were calculated to represent the total variance and vagal modulation of heart rate. The frequency domain parameters including high frequency (HF; 0.15–0.4 Hz), low frequency (LF; 0.04–0.15 Hz), and very low frequency (VLF; 0.003–0.04 Hz) power, were computed by averaging the absolute powers (ms2) after Fourier transformation.
Detrended fluctuation analysis (DFA)
DFA is used to evaluate the fractal behavior beneath seemingly nonstationary RR dynamics by eliminating extrinsic trends to remove spurious long-term correlations. The external trends were assumed to be the linear or polynomial fitted trends over different scales, and by removing these trends from the integrated time series, the intrinsic fractal behavior could be better quantified. Detrended fluctuations were calculated by adding up the detrended integrated time series in individual scales. Then, the logarithmic plot of fluctuations against time scales were further constructed. The slope (α exponent) of the log-log plot was used to indicate the fractal correlation characters of time series.
While the respiratory sinus arrhythmia is responsible for most of the short-term RR dynamics in normal subjects, the crossover phenomenon of α exponents of RR dynamics over short (α1; 4–11 beats) and long (α2; 11–64 beats) time scales are of importance. We calculated both short- and long-term α exponents for better probing the fractal characters of the biological system.
Multiscale entropy (MSE) analysis
MSE takes the predictability of multiple time scales into account and extends the entropy of a single timescale to the information richness structure embedded over different time scales. The profile of the sequential changes of the entropies over different time scales can be further quantifies. In brief, the time series of different time scales were derived by using a coarse-graining process (i.e. averaging consecutive beats to form a new time series), and the sample entropy was adopted to estimate the predictability over different time scales49. The estimated entropy over different time scales can then be used to represent the complexity (meaningful information richness) of the physiological signals. The linear-fitted slope of scale 1 to scale 5 (slope 1–5), the sum of entropy values of scales 1 to scale 5 (area 1–5) or scale 6 to scale 20 (area 6–20) were calculated to quantify the complexity of the beat-to-beat dynamics exhibited in short and long time scales.
Echocardiography
Transthoracic echocardiography (iE33 xMATRIX Echocardiography System, Philips, Amsterdam, Netherlands) was performed in all patients. The LVEF was quantitated by M-mode measurements or area-length methods9.
Computed tomography
A standard 64-MDCT scan (LightSpeed VCT, GE Healthcare, Milwaukee, WI) was performed in all patients. The calcified area was quantified based on an attenuation range of >150 Hounsfield units using image analysis software (ImageJ, version 1.45, National Institutes of Health, Bethesda, MD). The percentages of the area of the whole aorta affected by aortic calcification were calculated from the images of four consecutive slices just above the iliac bifurcation level9,32.
Statistical analysis
Data were expressed as median (25th and 75th percentiles). Comparisons of data between the higher and lower AAC groups were performed by the Mann-Whitney U test. Differences between proportions were calculated by the chi-square test or Fisher’s exact test. Logistic regression analysis was used to validate associations between parameters and the presence of high AAC. Significant determinants in univariate logistic regression analysis (P < 0.05) were then tested in multivariate logistic regression analysis with stepwise subset selection to identify independent factors to predict the presence of high AAC. Linear regression analysis was used to validate associations between parameters and percentage of AAC. Significant determinants in univariate linear regression analysis (P < 0.05) were then tested in multivariate linear regression analysis with stepwise subset selection to identify independent factors to predict the percentage of AAC. The goodness-of-fit of a logistic model was indicated by R2, while the discriminatory power of the model was assessed by the area under the ROC curve (AUC).
Two statistics, net reclassification improvement (NRI) and integrated discrimination improvement (IDI), were used to evaluate improvements in the accuracy of the prediction after adding a single nonlinear parameter into a logistic regression model using only linear parameters50. The significance of NRI and IDI statistics was based on approximate normal distributions. All statistical analyses were performed by R software (http://www.r-project.org/) and SPSS version 25 for Windows (SPSS Inc., IL, USA). The significance level of the statistical analysis was set at 0.05.
References
Szeto, C. C., Wong, T. Y., Chow, K. M., Leung, C. B. & Li, P. K. Are peritoneal dialysis patients with and without residual renal function equivalent for survival study? Insight from a retrospective review of the cause of death. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 18, 977–982 (2003).
de Jager, D. J. et al. Cardiovascular and noncardiovascular mortality among patients starting dialysis. JAMA 302, 1782–1789, https://doi.org/10.1001/jama.2009.1488 (2009).
Roger, V. L. et al. Heart disease and stroke statistics–2012 update: a report from the American Heart Association. Circulation 125, e2–e220, https://doi.org/10.1161/CIR.0b013e31823ac046 (2012).
Koomans, H. A., Blankestijn, P. J. & Joles, J. A. Sympathetic hyperactivity in chronic renal failure: a wake-up call. Journal of the American Society of Nephrology: JASN 15, 524–537 (2004).
Okuno, S. et al. Presence of abdominal aortic calcification is significantly associated with all-cause and cardiovascular mortality in maintenance hemodialysis patients. American journal of kidney diseases: the official journal of the National Kidney Foundation 49, 417–425, https://doi.org/10.1053/j.ajkd.2006.12.017 (2007).
Verbeke, F. et al. Prognostic value of aortic stiffness and calcification for cardiovascular events and mortality in dialysis patients: outcome of the calcification outcome in renal disease (CORD) study. Clinical journal of the American Society of Nephrology: CJASN 6, 153–159, https://doi.org/10.2215/CJN.05120610 (2011).
Huang, J. W. et al. Osteoprotegerin, inflammation and dyslipidemia are associated with abdominal aortic calcification in non-diabetic patients on peritoneal dialysis. Nutr Metab Cardiovasc Dis 24, 236–242, https://doi.org/10.1016/j.numecd.2013.08.006 (2014).
Kauppila, L. I. et al. New indices to classify location, severity and progression of calcific lesions in the abdominal aorta: a 25-year follow-up study. Atherosclerosis 132, 245–250 (1997).
Miwa, Y., Tsushima, M., Arima, H., Kawano, Y. & Sasaguri, T. Pulse pressure is an independent predictor for the progression of aortic wall calcification in patients with controlled hyperlipidemia. Hypertension 43, 536–540, https://doi.org/10.1161/01.HYP.0000117153.48029.d1 (2004).
Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 93, 1043–1065 (1996).
Tsuji, H. et al. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 94, 2850–2855 (1996).
Seely, A. J. & Macklem, P. T. Complex systems and the technology of variability analysis. Critical care 8, R367–384, https://doi.org/10.1186/cc2948 (2004).
Ho, Y. L., Lin, C., Lin, Y. H. & Lo, M. T. The prognostic value of non-linear analysis of heart rate variability in patients with congestive heart failure–a pilot study of multiscale entropy. PloS one 6, e18699, https://doi.org/10.1371/journal.pone.0018699 (2011).
Makikallio, T. H. et al. Fractal analysis and time- and frequency-domain measures of heart rate variability as predictors of mortality in patients with heart failure. The American journal of cardiology 87, 178–182 (2001).
Bhan, I. & Thadhani, R. Vascular calcification and ESRD: a hard target. Clinical journal of the American Society of Nephrology: CJASN 4(Suppl 1), S102–105, https://doi.org/10.2215/CJN.04800709 (2009).
Berliner, J. A. et al. Atherosclerosis: basic mechanisms. Oxidation, inflammation, and genetics. Circulation 91, 2488–2496 (1995).
Rumberger, J. A., Simons, D. B., Fitzpatrick, L. A., Sheedy, P. F. & Schwartz, R. S. Coronary artery calcium area by electron-beam computed tomography and coronary atherosclerotic plaque area. A histopathologic correlative study. Circulation 92, 2157–2162 (1995).
Simon, A., Giral, P. & Levenson, J. Extracoronary atherosclerotic plaque at multiple sites and total coronary calcification deposit in asymptomatic men. Association with coronary risk profile. Circulation 92, 1414–1421 (1995).
Paloian, N. J. & Giachelli, C. M. A current understanding of vascular calcification in CKD. Am J Physiol Renal Physiol 307, F891–900, https://doi.org/10.1152/ajprenal.00163.2014 (2014).
Speer, M. Y. & Giachelli, C. M. Regulation of cardiovascular calcification. Cardiovasc Pathol 13, 63–70, https://doi.org/10.1016/S1054-8807(03)00130-3 (2004).
Dellegrottaglie, S., Sanz, J. & Rajagopalan, S. Vascular calcification in patients with chronic kidney disease. Blood Purif 24, 56–62, https://doi.org/10.1159/000089438 (2006).
Wang, A. Y. et al. Associations of serum fetuin-A with malnutrition, inflammation, atherosclerosis and valvular calcification syndrome and outcome in peritoneal dialysis patients. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 20, 1676–1685, https://doi.org/10.1093/ndt/gfh891 (2005).
Blacher, J., Guerin, A. P., Pannier, B., Marchais, S. J. & London, G. M. Arterial calcifications, arterial stiffness, and cardiovascular risk in end-stage renal disease. Hypertension 38, 938–942 (2001).
London, G. M. et al. Arterial media calcification in end-stage renal disease: impact on all-cause and cardiovascular mortality. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 18, 1731–1740 (2003).
Adragao, T. et al. A simple vascular calcification score predicts cardiovascular risk in haemodialysis patients. Nephrology, dialysis, transplantation: official publication of the European Dialysis and Transplant Association - European Renal Association 19, 1480–1488, https://doi.org/10.1093/ndt/gfh217 (2004).
Leopold, J. A. Vascular calcification: an age-old problem of old age. Circulation 127, 2380–2382, https://doi.org/10.1161/CIRCULATIONAHA.113.003341 (2013).
Scicali, R. et al. HbA1c increase is associated with higher coronary and peripheral atherosclerotic burden in non diabetic patients. Atherosclerosis 255, 102–108, https://doi.org/10.1016/j.atherosclerosis.2016.11.003 (2016).
Morioka, T. et al. Glycemic control is a predictor of survival for diabetic patients on hemodialysis. Diabetes Care 24, 909–913 (2001).
Ricks, J. et al. Glycemic control and cardiovascular mortality in hemodialysis patients with diabetes: a 6-year cohort study. Diabetes 61, 708–715, https://doi.org/10.2337/db11-1015 (2012).
Duong, U. et al. Glycemic control and survival in peritoneal dialysis patients with diabetes mellitus. Clinical journal of the American Society of Nephrology: CJASN 6, 1041–1048, https://doi.org/10.2215/CJN.08921010 (2011).
NasrAllah, M. M. et al. Comparing different calcification scores to detect outcomes in chronic kidney disease patients with vascular calcification. Int J Cardiol 220, 884–889, https://doi.org/10.1016/j.ijcard.2016.06.064 (2016).
Tsushima, M. et al. Noninvasive quantitative evaluation of early atherosclerosis and the effect of monatepil, a new antihypertensive agent. An interim report. Am J Hypertens 7, 154S–160S (1994).
Karohl, C., D’Marco Gascon, L. & Raggi, P. Noninvasive imaging for assessment of calcification in chronic kidney disease. Nat Rev Nephrol 7, 567–577, https://doi.org/10.1038/nrneph.2011.110 (2011).
Wilson, P. W. et al. Abdominal aortic calcific deposits are an important predictor of vascular morbidity and mortality. Circulation 103, 1529–1534 (2001).
Huikuri, H. V. et al. Heart rate variability and progression of coronary atherosclerosis. Arterioscler Thromb Vasc Biol 19, 1979–1985 (1999).
Gottsater, A., Ahlgren, A. R., Taimour, S. & Sundkvist, G. Decreased heart rate variability may predict the progression of carotid atherosclerosis in type 2 diabetes. Clin Auton Res 16, 228–234, https://doi.org/10.1007/s10286-006-0345-4 (2006).
Ulleryd, M. A. et al. The association between autonomic dysfunction, inflammation and atherosclerosis in men under investigation for carotid plaques. PloS one 12, e0174974, https://doi.org/10.1371/journal.pone.0174974 (2017).
Vita, G. et al. Uremic autonomic neuropathy studied by spectral analysis of heart rate. Kidney international 56, 232–237, https://doi.org/10.1046/j.1523-1755.1999.00511.x (1999).
Zoccali, C. et al. Plasma norepinephrine predicts survival and incident cardiovascular events in patients with end-stage renal disease. Circulation 105, 1354–1359 (2002).
Lin, Y. H. et al. Heart rhythm complexity impairment in patients undergoing peritoneal dialysis. Scientific reports 6, 28202, https://doi.org/10.1038/srep28202 (2016).
Tang, S. C. et al. Complexity of heart rate variability predicts outcome in intensive care unit admitted patients with acute stroke. Journal of neurology, neurosurgery, and psychiatry 86, 95–100, https://doi.org/10.1136/jnnp-2014-308389 (2015).
Lin, Y. H. et al. Reversible heart rhythm complexity impairment in patients with primary aldosteronism. Scientific reports 5, 11249, https://doi.org/10.1038/srep11249 (2015).
Lin, Y. H. et al. Multi-scale symbolic entropy analysis provides prognostic prediction in patients receiving extracorporeal life support. Critical care 18, 548, https://doi.org/10.1186/s13054-014-0548-3 (2014).
Chiu, H. C. et al. Serial heart rhythm complexity changes in patients with anterior wall ST segment elevation myocardial infarction. Scientific reports 7, 43507, https://doi.org/10.1038/srep43507 (2017).
Peng, C. K., Havlin, S., Stanley, H. E. & Goldberger, A. L. Quantification of scaling exponents and crossover phenomena in nonstationary heartbeat time series. Chaos 5, 82–87, https://doi.org/10.1063/1.166141 (1995).
Tulppo, M. P. et al. Physiological background of the loss of fractal heart rate dynamics. Circulation 112, 314–319, https://doi.org/10.1161/CIRCULATIONAHA.104.523712 (2005).
Bigger, J. T., Fleiss, J. L., Rolnitzky, L. M. & Steinman, R. C. The ability of several short-term measures of RR variability to predict mortality after myocardial infarction. Circulation 88, 927–934 (1993).
Dekker, J. M. et al. Low heart rate variability in a 2-minute rhythm strip predicts risk of coronary heart disease and mortality from several causes: the ARIC Study. Atherosclerosis Risk In Communities. Circulation 102, 1239–1244 (2000).
Richman, J. S. & Moorman, J. R. Physiological time-series analysis using approximate entropy and sample entropy. American Journal of Physiology-Heart and Circulatory Physiology 278, H2039–H2049 (2000).
Steyerberg, E. W. et al. Assessing the performance of prediction models: a framework for traditional and novel measures. Epidemiology 21, 128–138, https://doi.org/10.1097/EDE.0b013e3181c30fb2 (2010).
Acknowledgements
We thank the staff of the Second Core Lab of the Department of Medical Research in National Taiwan University Hospital for technical assistance. This study was supported by grants from National Taiwan University (NTU)-National Taiwan University Hospital (NTUH)-MediaTek Innovative Medical Electronics Research Center (NTUH PC1094, MTKC-2016-1234 2016-1234), and the Ministry of Science and Technology (MOST 103-2221-E-008-006-MY3). M.T.L. and C.L. gratefully acknowledge support from the Ministry of Science and Technology (grant no. 104-3115-E-008 -001 and 104-2745-B-008 -001) and the joint foundations (grant no. 103CGH-NCU-A1, VGHUST103-G1-3-3). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Y.H.L. and J.W.H. conceived and designed the experiments. C.L., Y.H.H., L.Y.D.L., and M.T.L. analyzed the data. C.H.T and Y.H.L. wrote the paper. C.T.L. and C.K.P. made scientific comments on the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tsai, CH., Lin, C., Ho, YH. et al. The association between heart rhythm complexity and the severity of abdominal aorta calcification in peritoneal dialysis patients. Sci Rep 8, 15627 (2018). https://doi.org/10.1038/s41598-018-33789-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-33789-x
Keywords
This article is cited by
-
Usefulness of heart rhythm complexity in heart failure detection and diagnosis
Scientific Reports (2020)
-
Heart Rhythm Complexity Impairment in Patients with Pulmonary Hypertension
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.