Abstract
Human KLK8/neuropsin, a kallikrein-related serine peptidase, is mostly expressed in skin and the hippocampus regions of the brain, where it regulates memory formation by synaptic remodeling. Substrate profiles of recombinant KLK8 were analyzed with positional scanning using fluorogenic tetrapeptides and the proteomic PICS approach, which revealed the prime side specificity. Enzyme kinetics with optimized substrates showed stimulation by Ca2+ and inhibition by Zn2+, which are physiological regulators. Crystal structures of KLK8 with a ligand-free active site and with the inhibitor leupeptin explain the subsite specificity and display Ca2+ bound to the 75-loop. The variants D70K and H99A confirmed the antagonistic role of the cation binding sites. Molecular docking and dynamics calculations provided insights in substrate binding and the dual regulation of activity by Ca2+ and Zn2+, which are important in neuron and skin physiology. Both cations participate in the allosteric surface loop network present in related serine proteases. A comparison of the positional scanning data with substrates from brain suggests an adaptive recognition by KLK8, based on the tertiary structures of its targets. These combined findings provide a comprehensive picture of the molecular mechanisms underlying the enzyme activity of KLK8.
Similar content being viewed by others
Introduction
Initially, kallikrein-related peptidase 8 was named neuropsin, since the mouse ortholog Klk8 or mK8 had been cloned from mouse brain, in particular as hippocampus cDNA1. Similarly, the human KLK8 gene was cloned from human cDNA2, while expression of the KLK8 protein was detected in skin, esophagus, tonsils, salivary gland, breast milk, cervico-vaginal fluid, and in ovarian cancer extracts3,4. KLK8, formerly termed hK8, belongs to the 15 human kallikrein-related peptidases of the trypsin-like family S1A, with the MEROPS code S01.2445. It is even present in primitive mammals, such as platypus and wallabies6. KLK8 is secreted as an inactive pro-enzyme, whereby a signal peptide is cut off, followed by propeptide cleavage at the Lys15-Val16 bond, releasing active enzyme7. First hints of a specific function came from mouse Klk8 expression in the limbic system, with mRNA level changes in pyramidal neurons upon epileptic seizures8,9. Similarly, KLK8 mRNA transcripts were found in the human hippocampus and neocortex10.
Further studies suggested that mouse Klk8 (mKlk8) participates in important brain processes, such as neural plasticity and memory formation, whereby long term potentiation (LTP) and the formation of synaptic boutons in mice require the active mKlk8 protease, which remodels the extracellular matrix11. LTP processes at synapses of higher mammals involve a complicated interplay of KLK8/neuropsin with the proteases tPA, plasmin, MMP-9, ADAM-10, and neurotrypsin12. Also, during LTP the activity of voltage-gated calcium channels correlates with elevated extracellular Ca2+ concentration, which has a stimulatory effect on the enzymatic activity of KLK87,13. At synapses it cleaves the cell adhesion molecule L1CAM, neuregulin-1, a signal ligand of the ErbB4 receptor, and the tyrosine kinase EphB2, which modulates the expression of the NMDA receptor12. Since single nuclear polymorphisms of KLK8 have been discovered in humans with bipolar disorder and learning disability, a similar role in synaptic processes as in mice can be assumed11. Real time PCR results demonstrated expression of KLK8 in all parts of the developing embryo brain, while the expression in adult brain is much more restricted14. Remarkably, the splice variant KLK8-II from human brain, with a 45 residue longer signal peptide14, was discovered in archaic hominins, such as Neanderthals and Denisovans, but is absent chimpanzees15. Thus, it has been hypothesized that the splice variant KLK8-II is a distinguishing feature of humans compared with the great apes and other primates16.
Apart from its functions in brain, KLK8 contributes to wound healing, as demonstrated with knockout mice17,18. Also, elevated KLK8 levels were found in the inflamed psoriatic plaques of humans and in a corresponding mouse psoriasis model19,20. KLK8 and its mouse ortholog degrade extracellular matrix proteins, such as fibronectin and type-IV collagen21,22. Several natural inhibitors of KLK8 have been described, such as SerpinB6, SerpinB9 and the protein C inhibitor, as well as the skin-specific Kazal-type serine protease inhibitor SPINK9 blocks human KLK8 activity23.
In order to explore human KLK8, the recombinant enzyme from an E. coli expression system was used for X-ray and enzymatic studies, which are the basis for a comparison with the structure of mKlk824 and for a mechanistic analysis of the structure-functional modules of this intriguing protease.
Results
Extended substrate specificity profiles of KLK8
Active, non-glycosylated KLK8 was prepared from a chimeric construct expressed in E. coli, refolded and activated by enterokinase25. According to SDS-PAGE analysis, this recombinant KLK8 was more than 95% pure, and its integrity was shown by N-terminal sequencing and mass spectrometry. Since only scattered information on natural or synthetic KLK8 substrates was available, the extended substrate specificity of KLK8 was determined by a positional-scanning screening of a synthetic diverse tetrapeptide library (PSSCL) and the proteomic identification of cleavage sites (PICS), which employs a peptide library from digested E. coli proteins26,27. Regarding the non-prime side, KLK8 possesses a typical tryptic S1 subsite and cleaves mainly at P1-Arg residues and with less than 25% the efficiency at Lys in both approaches (Fig. 1A,B). Both methods indicate that the S2 pocket prefers the aliphatic residues Leu, Val, and Ile, while PSSCL ranks polar residues, such as Asn, Ser, and Thr relatively high. Unusual for trypsin-like serine proteases the S3 subsite favors the basic side chains Lys and Arg, besides Ser and Ala in case of PSSCL and only Arg according to PICS. PSSCL shows that the S3 subsite disfavors Asp and Glu as P3 residues, while the overall less specific S4 subsite accommodates best Thr and Trp significantly above average. The PICS method alone provided information on the prime side specificity, where the S1′ subsite prefers the small or polar residues Ser and Ala, the S2′ pocket exhibits a preference for Ala, along with the aliphatic residues Ile and Val, which also appears to be better accepted in the S4′ subsite than other residues. A moderate preference for P5′-His and P6′-Tyr shows up after correction for the natural occurrence of amino acids in E. coli proteins (Fig. 1B). Based on the PSSCL results for the range of P1 to P4 residues, optimized fluorogenic 7-amino-4-methyl coumarin (AMC) and 7-amino-4-carbomoylmethyl coumarin (ACC) substrates were synthesized.
Specificity profiling with a positional scanning and proteomic identification of cleavage sites. (A) Peptide substrate specificity of KLK8 measured by positional scanning for peptide positions P4 to P1. The height of the columns represents substrate cleavage rates by released ACC, while the x-axis indicates the amino acids in one-letter code, whereby n represents norleucine, which substitutes Cys. (B) PICS heat maps with amino acids listed in alphabetical order along the y-axis and peptide positions run from P6 to P6′ on the x-axis. The left panel displays cleavages in 73 substrates and the right panel contains corrected values for the natural abundance of each residue. Both PICS and PSSCL agree in a moderate preference for Thr and Trp in P4 and for basic residues in P3, such as Lys or Arg. Aliphatic residues Leu, Val, and Ile are found by both methods in P2, whereby Val dominates in the corrected PICS, while PSSCL ranks the polar Asn, Ser, and Thr relatively high. The major primary specificity for Arg over Lys in P1 is found by both methods, albeit stronger in PICS. The corrected PICS data show a preference for Ser and Met in P1′, for Ile and Trp in P2′, and no distinct specificity for P3′ and P4′. By contrast, S5′ and S6′ subsites are moderately specific for His and Tyr.
Ca2+ stimulation and Zn2+ inhibition of recombinant KLK8 activity
Active-site titration with the burst reagent p-nitrophenyl-p’-guanidino-benzoate (NPGB) revealed that 62% of the recombinant KLK8 was active. This value was used as molar fraction of active protease for the calculation of the turnover number (kcat). The ideal substrates Ac-Thr-Lys-Leu-Arg-AMC and Ac-Thr-Lys-Leu-Arg-ACC were checked for their turnover by KLK8 and compared with other tetrapeptidyl substrates, regarding their enzyme kinetic parameters kcat, KM, and the resulting catalytic efficiency (Table 1). For example, Ac-Thr-Lys-Leu-Arg-ACC exhibited a Michaelis constant (KM) around 15 µM, a kcat around 45 s−1 and a catalytic efficiency kcat/KM = 3 137 000 M−1 s−1, whereas the previously described best KLK8 substrate Bz-Val-Pro-Arg-AMC had a kcat/KM = 20 280 M−1 s−1 7. Both optimized substrates turned out to be at least 10-times more active than commercial substrates such as Ac-Thr-Lys-Pro-Arg-AMC, whereas Tos-Arg-AMC was not cleaved at all. No turnover was seen for Boc-Glu-Lys-Lys-AMC, which is cleaved by mKlk8 to some extent28, or for Ac-Trp-Glu-Asp-Arg, in line with the PSSCL findings that basically exclude negatively charged P2 and P3 residues.
Zinc ions have a strong inhibitory effect on the hydrolytic activity of KLK8. At pH 8.0 and 20 °C, we determined an IC50 value of 3.6 ± 0.3 µM (see section on the zinc binding 99-loop), close to the IC50 value of 3.3 µM measured for different KLK8 preparation at pH 8.5 and 37 °C7. Eadie-Hofstee plots of the activity and substrate concentration at different Zn2+ concentrations indicate that the mechanism of inhibition is non-competitive, which suggests that Zn2+ binds the free and substrate saturated enzyme with similar affinity.
In contrast to Zn2+, Ca2+causes an increase of the hydrolytic activity of KLK8 with the para-nitroanilide Bz-Pro-Phe-Arg-pNA exhibiting a maximum around 1 mM which agrees with a previous publication7. However, KLK8 cleaved the optimal substrates Ac-Thr-Lys-Leu-Arg-ACC and Ac-Thr-Lys-Leu-Arg-AMC with a 2-fold increase of activity at 200 to 300 µM Ca2+. For the substrate Bz-Pro-Phe-Arg about 60% higher KLK8 activity was observed in the presence of 1 mM Ca2+ at 37 °C, while KLK8 that had been preincubated with 5 µM Zn2+ showed nearly 70% increase of activity upon addition of Ca2+.
Overall structure of KLK8
Originally, one form of KLK8 could be crystallized in the presence of the aldehyde inhibitor leupeptin. Several data sets were collected of these crystals in space group P212121, with one molecule per asymmetric unit and a maximum resolution of 2.3 Å. The corresponding KLK8 structure will be referred to as KLK8-Ca. Upon soaking the crystals with ZnCl2, one cell constant approximately doubled in length, accompanied by a transition to space group P21, with four molecules per asymmetric unit. The corresponding 2.1 Å structure of KLK8, in which Zn2+ was not clearly identified, is referred to as KLK8-leup and resembles largely the KLK8-Ca molecule.
As in most trypsin-like serine proteinases the KLK8 chain consists of two six-stranded β-barrels that can be considered as half-domains (Fig. 2A). At their interface the catalytic triad of Ser195, His57 and Asp102 is situated above the specificity subsites S4 to S4′, aligned from left to right in the standard orientation. Compared to the coordinates of mKlk8 (PDB code: 1NPM) human KLK8 exhibits a relatively low root mean square deviation (RMSD) of 0.63 Å for 222 equivalent residues atoms, agreeing well with respect to 73% identical residues7. Similar to other mature KLKs, the N-terminal α-ammonium group of Val16 forms the crucial salt bridge to the side chain carboxylate of Asp194, which stabilizes the oxyanion hole and a properly shaped S1 pocket (Fig. 2A). Also, KLK8 contains the KLK-specific cis-Pro219 and six disulfide bridges.
Overall structure of KLK8-Ca. (A) Ribbon stereo plot of KLK8-Ca shown in standard orientation. The side chains of the catalytic triad, the activating salt bridge Val16-Asp194, the major specificity-determining Asp189 and Thr190, as well as the six disulfide bridges and Cys93 are shown as sticks (carbon teal, nitrogen blue, oxygen red, and sulfur yellow). Also, the three major ligands of Ca2+ (green sphere) are depicted, namely Asp70, Asp77 and Glu80. Functionally important loops are displayed in different colors with labels. (B) Surface model of KLK8 with inhibitor and Ca2+. The left panel shows KLK8 in standard orientation with a stick model of leupeptin, occupying the subsites S4 to S1. The electrostatic surface potential is depicted red for negative and blue for positive potential in the range −10 e/kBT to +10 e/kBT. The right panel shows the KLK8 backside, rotated by 180°. The 60-loop and the area around Arg109 correspond to the thrombin anion binding exosite I, while the positive patch at the back around Lys166 and Lys186A has no counterpart.
The active site cleft from the non-prime specificity pockets S4 to S1 and S1′ to S4′ is predominantly negatively charged (Fig. 2B). Several basic residues cluster around the 60-loop in a positively charged surface patch, extending above the prime side region of the active-site cleft in standard orientation. Together with the positively charged patch around Arg109 this area resembles the more extended basic regions of KLK5 and KLK7 and the functionally important anion binding exosite I of thrombin29.
Active-site cleft and specificity pockets
Both KLK8 structures exhibit a similar active site architecture, whereby in the KLK8-Ca structure the substrate binding cleft is occupied by several water molecules, located mostly at polar or charged positions of the specificity pockets. By contrast, the P21 form of KLK8-leup contains four copies of the aldehyde inhibitor leupeptin, with the sequence acetyl-Leu1i-Leu2i-Argininal3i. For this reversible inhibitor an IC50 of 66 µM was reported for KLK8 from insect cell expression, while it seems less than 10 µM for mKlk87,28. Leupeptin binds covalently to Ser195 as analogue of an acyl intermediate in the canonical conformation (Fig. 3A). The P1-Arg3i aldehyde group in KLK8-leup is covalently linked to Ser195 Oγ as hemiacetal, with the oxygen occupying the oxyanion hole in the four KLK8 copies. This orientation of the hemiacetal oxygen corresponds to one of the two alternative conformations observed in a trypsin leupeptin complex (PDB code 1JRT)30. It forms hydrogen bonds to the backbone amide N-H atoms of Gly193 and Ser195 or a nearby water at average distances of 3.3 Å, while the other conformation with a hydrogen bond to the Nε2 atom of His57 is not observed.
Leupeptin inhibitor and ideal substrate binding to the active site. (A) Stereo model of leupeptin in KLK8-leup, with the most relevant interacting residues displayed as sticks, e.g. the covalently linked Ser195. The electron density is a 2Fo-Fc map shown as green grid, contoured at 1.0 σ, hydrogen bonds are represented by grey dots. A water molecule (red sphere) occupies the oxyanion hole at the NH groups of Gly193 and Ser195 in this chain. (B) Molecular docking of an ideal substrate to KLK8 according to PICS profiling for residues P6 to P6′. Binding of residues P4 to P4′ agrees well with the specificity profiles. Moreover, the calculated interactions of P4-Thr with Glu97 and P3-Arg with Glu149 and Asp218 (red patches) can explain the preference of the S4 subsite for Thr over Trp, and the unique S3 preference for basic side chains. Binding of the hydrophobic residues P3′-Leu and P4′-Met may depend on the subsite shaping residues Leu41 and Phe151 (green patches).
Similar to a substrate, leupeptin forms a short antiparallel β-sheet with two backbone hydrogen bonds between P3-Leu1i and Gly216, and the amide of P1-Arg3i and the carbonyl oxygen of Ser214 (Fig. 3A). The Arg3i side chain is buried in the S1 specificity pocket, which is rather hydrophobic in the upper region, whereas at the bottom the side-chains of Thr190, Ser217, Tyr228, and a water molecule bound to the Ser217 carbonyl O contribute to a polar environment. The negatively charged carboxylate of Asp189 forms a tight salt bridge to the positively charged Arg3i guanidyl group. Both positional scanning and PICS analysis corroborated that Arg is strongly favored over Lys as P1 residue (Fig. 1). This finding can be explained by the presence of Thr190, which enhances the specificity for P1-Arg similar to Ser190 of many trypsin-like proteinases, whereas an Ala190 would shift the P1 preference towards Lys31.
The funnel-shaped S2 subsite of KLK8 is bordered by the hydrophobic side-chains of His57, His99, and partially Trp215, which explains very well the accommodation of the Leu2i side-chain of the inhibitor and the preference for the aliphatic Val and Leu residues in the substrate profiling (Fig. 1). Also, the hydroxyl group of Tyr94 may interact with polar residues, such as Asn or Ser, which are accepted according to the PSSCL. Overall, this subsite appears to be quite flexible and adopts in all five KLK8 molecules different side chain conformations, in particular for His99, while water molecules sit at varying positions.
Although most P3 side chains of serine protease substrates extend to the bulk solvent, two major alternative conformations are possible for Leu1i and, consequently, for the acetyl group. In three KLK8-leup copies, the Leu side chain prefers the canonical conformation in the bulk solvent, whereby its backbone NH can form a hydrogen bond to the carbonyl O of Gly216 as in KLK532, whereas in one copy it occupies the S4 pocket as in trypsin30. As Leu is not a preferred P3 residue according to PSSCL and PICS, it is difficult to relate structural and functional information for the unusual specificity of the S3 subsite for basic residues (Fig. 1). The variable arrangement of the predominantly hydrophobic His99, Trp215, Tyr172 and the more polar Gln175 and Glu97 may suffice to explain the relatively low specificity for P4 residues, including the uncommon preference for Thr and Trp, and the rejection of negatively charged P4 residues.
A molecular docking calculation for an ideal peptide substrate of KLK8, derived from the PICS data, resulted in proper binding of P4 to P4′ (Fig. 3B). Besides the expected accommodation in the S2 to S1 pockets, the interaction of the Arg guanidyl group by the carboxylate groups of Asp218 and Glu149 explains the specificity profiles for the preferred basic P3 residues. Also, a polar interaction of the P4-Thr with Glu97 may be the basis for the unusual S4 specificity. The S1′ subsite is a shallow pocket that is mostly shaped by two main chain stretches with polar character, while the hydrophobic bottom consists of the disulfide Cys42-Cys58, which agrees with a moderate preference for P1′-Ser and Met. Eventually, the accommodation of the aliphatic P2′-Ile, P3′-Leu and P4′-Met depends on the hydrophobic environment, such as Leu40, Leu41, Lys60 and Phe151 bordering the S2′ to S4′ subsites, respectively. The adjacent S5′ and S6′ subsites possess a mixed character, which tolerates aromatic, but also charged or polar residues, such as His and Tyr, while still putative prime side subsites may extend even beyond the 75-loop.
The Ca2+ binding 75-loop
Among the loops shaping the active-site cleft of KLK8, the 75- or “calcium binding”-loop is the most distant from the catalytic triad. This loop is named after the central residue, but it is also known as the 70–80-loop. The 75-loop of KLK8-Ca exhibits an ideal coordination geometry compared to the slightly deviating KLK8-leup loops and holds the Ca2+ ion with six ligands at an average distance of 2.3 Å, which is slightly below standard values (Fig. 4A,B)33. Among them are the side chain carboxylate groups of Asp70, Asp77, and Glu80, which provide a negative charge excess with respect to the divalent cation. Since the Oδ1 and Oδ2 of Asp77 bind the backbone N-H group of Ser72 at a distance of roughly 3.1 Å, some portion of the third negative charge is compensated. Similarly, the carboxylate of Glu80 binds the N-H groups of Asp77 and Gly78 with its Oε2 and Oε1 atoms, respectively. Also, the positively charged side chain of Arg66, appears to compensate some portion of the negative excess charge by a contact of the Nη1 to the Oδ2 of Asp70 at around 3.3 Å, albeit not with an ideal geometry. Additionally, the Ca2+ ion is bound with an average distance of 2.3 Å by the carbonyl O atoms of Ser72 and Asn75, as well as by one water molecule. The Ca2+ coordination sphere is octahedral, as it is known for coagulation factors or trypsin where the 75-loop binds Ca2+ via three glutamates, including a mediating water34. By introducing the substitution Asp70Lys in the 75-loop, the stimulatory effect of Ca2+ was essentially abolished in enzymatic assays with Boc-Val-Pro-Arg-AMC or Bz-Pro-Phe-Arg-pNA compared with normal activity (Fig. 4C). The function of Ca2+ bound to the 75-loop can be dual as in trypsin, where it serves both to stabilize the enzyme and enhance the activity35.
The Ca2+ and Zn2+ binding loops of KLK8 as regulatory modules. (A) The 75-loop of KLK8-Ca with Ca2+ ligands surrounded by the 2Fo-Fc electron density map, contoured at 1.0 σ. Ca2+ is depicted in green with electron density contoured at 3.0 σ, while the water ligand is shown as red sphere. (B) Ligand bonds to Ca2+ are shown as red dots and relevant hydrogen bonds as black dots, including distances in Å. The excess charge of three carboxylates is compensated by Arg66 near Asp70 and hydrogen bonds of Asp77 and Glu80 with backbone N-H groups. (C) The Ca2+ stimulation of the KLK8 activity is maximal in the range 100 µM to about 1 mM. In the D70K variant no significant stimulatory effect of Ca2+ was observed. (D) The mutant His99Ala confirmed the inhibitory Zn2+ site with a 13-fold higher IC50, i.e. 47 µM versus 3.6 µM of wild-type KLK8. (E) In the 99-loop, Cys93 can be excluded as potential Zn2+ ligand, since Zn2+ inhibition of the Cys93Ser variant was unchanged compared to the wild-type. A water molecule (grey) between His99 and Tyr94 in KLK8-leup is in a suitable position to bind Zn2+. The variant Tyr94Phe has a 2-fold higher IC50, suggesting a minor role as Zn2+ ligand for Tyr94. Hydrogen bonds are shown as grey dots. (F) Two complex structures of KLK5 and KLK7 with Zn2+ and Cu2+ feature ligands for cations, such as the carbonyl O of residue 96 and Tyr94. The KLK7-His99Ala mutant nearly abolished the non-competitive Zn2+ inhibition, as for KLK8. This inhibition type can be explained by Zn2+ binding to the His57 side chain, which has to switch from the catalytic triad. A rotation of His99 would bring both ligands in ideal distances to Zn2+ 36.
Zn2+ binding in the 99-loop
The 99-loop of KLK8 exhibits an intermediate length between those of KLKs 4, 5, 6 and 7 and the long, exposed “kallikrein-loops” of KLKs 1 to 3 with an insertion of 11 residues23. Guided by the crystal structures of murine neuropsin, KLK5 and other KLKs with confirmed Zn2+ inhibition we generated mutants in order to investigate the functional role of critical residues located in the 75- and 99-loops23,24. The substitution of His99Ala resulted in an active KLK8 variant that exhibited a 13-fold higher IC50 (46.7 ± 3.2 µM) for Zn2+ inhibition with respect to the wild type (Fig. 4D). Lack of the hydroxyl group in the variant Tyr94Phe resulted in a doubled IC50 (6.9 µM), which hints to a role as secondary or water bridged ligand of Zn2+ (Fig. 4E). Since cysteines are favored Zn2+ ligands, the residual inhibition of the His99Ala mutant could depend on Cys93 (Fig. 4E), which was mutated to a serine. However, the Cys93Ser variant was as active as the wild type and displayed similar inhibition by Zn2+, which excludes Cys93 as ligand, since Ser is as Tyr not an ideal ligand for Zn2+ 36. This finding suggests that substrates still can bind to the active site in the inhibited state, while the inactivation is based on another process, such as a the disruption of the catalytic triad, which was found in the crystal structure of rat tonin (klk2) in complex with Zn2+ (PDB code 1TON)37. Similarly, His57 in rat trypsin relocated from the triad to bind Cu2+ together with an engineered His96 (1AND)38. Since both KLK5 and KLK7 structures exhibit either Zn2+ or Cu2+ bound to the equivalent His99 ligands and His96 or the Thr96 carbonyl O, other possible ligands are His57 or perhaps Asp102 (Fig. 4F). At least, we can narrow down the most likely Zn2+ binding residues to the region around His99, Tyr94 and His57 in the S2 subsite (Fig. 4F). There, the Tyr94 OH and the carbonyl O atoms of Val96 and Asp98 could serve as additional ligands or secondary ones by bridging water molecules in the coordination sphere of the metal ion.
Discussion
KLK8/neuropsin is among the major suspects that make a substantial difference in human neuronal processes with respect to the corresponding activity in other primate brains. A structure based sequence alignment demonstrates that only three insignificant amino acid exchanges are present in the active KLK8 protease of our second closest relative in evolution, the gorilla, whereas chimpanzees have exactly the same sequence as humans (Fig. 5). Apparently, the major difference in all non-human primates is the lack of KLK8 isoform 2, with a 45 residue longer signal/prepeptide, which could be responsible for altered protein trafficking, as observed for long signal peptides of other protein isoforms14,39. In case the insertion is part of an elongated propeptide of pro-KLK8, it might not influence the distorted loop network at all, whereas KLK8 activation might be altered, favoring a more efficient auto-activation as observed for KLKs 2 and 540. Since isoform 2 is mostly expressed in the hippocampus, the special role of KLK8 in learning and memory formation, may be highly important for human consciousness.
Structure-based sequence alignment of KLK8. Other species are gorilla (Gorilla gorilla), mouse (Mus musculus) and rat (Rattus norvegicus) (mKlk8 and rKLK8), including bovine trypsin (bTRY) and chymotrypsinogen A (bCTRA) as numbering standard. Signal peptides of isoforms precede the propeptide, whereby isoform 2 with a 45 residue insertion is supposed to be unique for humans. Human and chimpanzee KLK8 are identical, whereas the gorilla has three exchanges. Mice share 73% identical residues in KLK8 with humans, similar to rats with about 71%. β-sheets, α-helices, and the short 310-helix (residues 56–59) of KLK8 are indicated by arrows and screws, respectively. Relevant surface loops are labeled according to the central residue. Darker shades of grey represent higher conservation, while the catalytic triad residues are depicted with black background. Ca2+-ligands are displayed with green background and Zn2+ ligands are shown with magenta background, triangles indicate interacting side chains. The sequons of the glycosylation site at Asn95 are highlighted in blue.
Regarding natural KLK8 substrates, pro-KLKs 1, 2, 3, 5, 6, 9, 11, and 12 appear to be physiologically relevant in KLK activation cascades40. The growth hormone somatotropin is another interesting natural substrate, as well as single-chain t-PA, which after activation by KLK8 activates plasmin, facilitating cancer cell invasion22,41. Among neuronal mouse substrates is the signal protein neuregulin 1 (NRG1), which is cleaved by Klk8 at three sites with P1-Arg42. Although KLK8 expression correlates with cleavage of the neural cell adhesion molecule L1-CAM, the specific sites have not been determined yet43. However, murine Klk8 cleaves the receptor tyrosine kinase ephrin type-B receptor 2 (EphB2) after Arg518, resulting in NMDA receptor dependent synaptic plasticity events in stress response44. Recently, KLK8 inhibition was proposed as therapeutic target in Alzheimer’s disease, which is accompanied by elevated KLK8 protein levels, while in a murine Alzheimer’s model Klk8 inhibition by antibodies significantly attenuated the pathology45.
Both the synthetic library and the proteomic specificity profiling agree very well regarding the non-prime side, while there is basically no discrepancy with the MEROPS specificity matrix (Fig. 1). Also, the specific interactions of the P4 to P1 residues can be well explained on a structural level by the complex of KLK8 with the leupeptin inhibitor. In the prime side region, the MEROPS matrix is biased by the IVGG motif that is often present at the N-terminus of serine proteases, which are substrates in KLK activation cascades40. Otherwise, this part of the matrix, summarized as Ia/vi/gn/G with small letters for lower preference, resembles more the corresponding raw profile of PICS (as/avi/-/v) than the corrected one (ms/iw/-m) (Fig. 1B). Comprising 25 cleavages, the MEROPS matrix is just below the theoretical limit of 30 substrates for a reliable specificity determination within a 95% confidence interval, using the information entropy as measure of specificity46. By contrast, the PICS measurement comprised 73 substrate cleavages, which increases the reliability substantially, as evidenced by the concordance with the virtually unbiased positional scanning library results (Fig. 1A).
A structure based comparison of the specificity matrix and the profiling results with physiological substrates from murine and human neuronal tissue, i.e. NRG1, EphB2, and neuroserpin (SERPINI1), reveals that several cleavage sites are located in disordered regions as it is usually expected, but some comprise secondary structural elements as it was established for other proteases47. Some cleavage sites resemble the extended conformation found in surface loops and strands, which also coincide mainly with P1 to P3 sequence that is determined by mainly electrostatic and to some extent by hydrophobic and polar interactions. The cleavage sites KKE18R-GSGK of NRG1 and NRL38R-ATGE of neuroserpin belong to this category, probably requiring the α-helix of the non-prime side (PDB code 3FGQ) to unwind to some extent (Fig. 6A)42,48. Also, the reactive center loop of neuroserpin, which has been suggested as physiological inhibitor of KLK8 in the brain, with the sequence ISM362R-MAVL and an α-helical turn could fit in here49. However, the cleavage sites ELN79R-KNKP of NRG1 and GYG518R-YSGK of EphB2 hardly agree with the established substrate specificity of KLK8, except for the presence of P1-Arg42,44. Intriguingly, the three-dimensional models of both sites exhibit accessible surface stretches with basic residues in neighbor strands, such as Arg67 and Lys69 in NRG1 or Arg509 and Arg511 in EphB2 (Figs 6B,C). They are in reach of Asp218 and Glu149, which appear to be the determinants of the specificity for basic P3 residues. Thus, only by considering the three-dimensional structure of substrates with large deviations from the ideal recognition sequence, we can explain the preference of KLK8 for its known physiological targets. Since the phenomenon of significantly differing synthetic and natural substrate preferences is well known for other proteases, systematic analyses of similar cases may indeed reveal this type of alternative presentation of preferred cleavage sites as a more general pattern.
Models of natural protein substrates bound to the active site of KLK8. The molecular surface of KLK8 is shown with red patches of acidic residues and green for hydrophobic ones. (A) Human neuroserpin, a serine protease inhibitor from the hippocampus, is cleaved at Arg38 as P1 residue, which is located in the last turn of an α-helix, as observed in the crystal structure (PDB 3FGQ). Apparently, Arg36, Leu37 and Ala39 are suitable P3, P2 and P1′ residues in accordance with our profiling results. Proper binding and turnover by KLK8 requires a transition of the helical turn into a more linear, extended conformation. (B) The synaptic regulator neuregulin 1 (NRG1) possesses only P1-Arg79 and P2-Asn78 as KLK8 specific substrate residues at its cleavage site. According to a structure-based homology model, created by SWISS-MODEL79 they belong to a β-strand, while the Leu77 side-chain might occupy the S4 instead of the S3 subsite. However, an adjacent β-strand contains Lys69, which is located close to Glu149 and Asp218, substitute the preferred basic P3 side-chain. (C) Similarly, a model from SWISS-MODEL of the neuronal KLK8 substrate ephrin type-B receptor 2 (EphB2), exhibits an arrangement of two strands, with P1-Arg518 as only specific residue for cleavage by KLK8. Interestingly, Arg509 and Arg511 might be ideally positioned for electrostatic interaction with Glu149 and Asp218.
Concerning the kinetic parameters, the glycan-free E. coli expressed KLK8 can be compared with an identical, refolded KLK8 construct, which cleaved Boc-VPR-AMC with a catalytic efficiency of about 23 300 M−1 s−1, which is nearly identical to our result (Table 1)22. Similarly, KLK8 from Sf9 insect cells and yeast expression (Pichia pastoris) had a catalytic efficiency of 21 000 M−1 s−1 for Boc-VPR-AMC and 20 000 M−1 s−1 for the substrate H-D-IPR-pNA, respectively7,50. Since the KLK8 variants from eukaryotic expression are most likely glycosylated at Asn95 in the 99-loop, it seems that this modification does not influence the basic enzymatic activity, in contrast to the corresponding glycan of the KLK2 99-loop51. Nevertheless, the glycan carrying 99-loop, as it was observed in the mouse Klk8 structure (PDB code 1NPM) may contribute to resistance against proteolysis, protection of Cys93 against oxidation, or conformational stability. In this latter context, it may favor a distinct conformation as proposed by the conformational selection model for KLKs. In particular, the glycan at Asn95 might regulate the concerted conformational rearrangements of loops surrounding the active site and concomitantly enhance the turnover of larger polypeptides51,52.
In the synaptic cleft, the Ca2+ concentration is about 0.7 to 2 mM, which overlaps with the reported stimulatory range for KLK8 of 100 µM up to 10 mM53. Similar to the prototypic serine protease trypsin, the activity increases up to 4-times for small synthetic substrates by Ca2+ stimulation7. However, some coagulation factors, such as FXa exhibit more than 30-fold Ca2+ stimulation of their enzymatic activity against chromogenic substrates54. Regarding the mechanism of the stimulation molecular dynamics (MD) simulations showed that some parts of the KLK8 molecule tend to rearrange in the presence and absence of Ca2+ at the 75-loop (Fig. 7A). Ca2+ binding in the 75-loop results in significant conformational changes in the 37-, 61- and mainly the 99-loop (represented as root mean square deviations, RMSD), whereas smaller rearrangements occur in the Ca2+-free state. Intriguingly, MD calculations with the core glycan GlcNAc2Man3 linked to Asn95 resulted in a wide open S2 pocket, a conformation that seems to be stabilized by the glycan (Fig. 7B). This observation might explain the stronger stimulatory effect of Ca2+ on the activity of recombinant KLK8 from insect cells, which produce glycosylated proteins7. Apparently, the allosteric effect is transmitted upon Ca2+ binding from the 75-loop via the neighboring 37- and 61-loops to the 99-loop. A similar allostery was seen in the comparison of a Ca2+-bound and –free factor IXa triple mutant, with a long-ranging conformational rearrangement from the 75-loop to the center of the active site, which was termed “communication line”, involving the 148-loop, the activating N-terminal salt bridge and the S1 pocket55. Without Ca2+ the 75-loop reorients itself and covers parts of the prime side region from S6′ on, which would interfere with the binding of polypeptide substrates (Fig. 7C). Concomitantly, the 99-loop closes the upper part of the S2 subsite with Val96 and His99 in a way that would hamper substrate binding in the non-prime side, which further corroborates the importance of this alternative communication line in KLK8. Such a blocked S2 pocket, involving Lys98 and Tyr99, has been observed for factor IXa in complex with the inhibitor benzamidine in the S1 pocket, albeit with Ca2+ bound in the 75-loop56. The closed or “locked” conformation of the fIX 99-loop is based on the interaction of Tyr177 with asparagines 97 and 100, which is physiologically released by the major stimulatory factor VIIla57. In contrast to factor IXa, KLK8 lacks the specific residues that lock the closed conformation, as it seems to shift easier between the open and closed conformations. By contrast, mKlk8 adopts a relatively open conformation even in the absence of inhibitors or Ca2+. Furthermore, it has been demonstrated that Ca2+-binding to the 75-loop of factor IXa is allosterically linked to the Na+-binding/225-loop58. The corresponding 220-loop of most KLKs cannot bind Na+, due to the presence of cis-Pro219. A molecular dynamics study based on free and inhibitor bound KLK4 crystal structures described an allosteric interplay of loops surrounding the active site, in particular the 37-, 75-, and 220-loops, although it involves an inhibitory cation site at Glu77 and His2559. However, a comparative analysis of many trypsin-like serine proteases, including all known KLKs, suggested that the 99-, 148-, and 220-loop, surrounding the catalytic triad, open and close in a concerted manner, according to the conformational selection mechanism52. Thus, it would not be surprising, if all loops around the substrate binding cleft were connected in an allosteric network, with different characteristics in individual serine proteases.
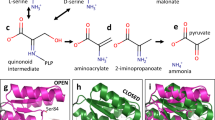
Molecular dynamics and the allosteric loop network of KLK8. (A) Plots of root mean square deviations (RMSD) in Å of KLK8 residue coordinates for five sequential MD production cycles at 298 K after the warm-up cycles, with respect to the final warm-up model. Data are depicted Ca2+ bound to the 75-loop and the last cycle without Ca2+ (chymotrypsinogen numbering). The significance level 2 Å (red line) is MOE default. For better visualization of loop motions, the RMSD difference (∆) of the models from cycle 5 [(+Ca2+) − (−Ca2+)] was calculated. Whereas the Ca2+ bound 75-loop seems more stationary, the 37-, 61-, and mainly the 99-loop exhibit large motions. This finding indicates an allosteric communication line between these loops. (B) Model from MD calculations of KLK8 shown as surface with colored loops with a core glycan linked to Asn95, consisting of two GlcNAc (blue spheres) and three mannoses (light green). Ca2+ (green) is bound to the 75-loop and leupeptin from the crystal structure is overlayed as stick model at subsites S4 to S1, in order to emphasize the wide open 99-loop (yellow) conformation, which seems to be facilitated by the presence of the glycan. The glycan may prevent cleavage in the 99-loop and disulfide formation of Cys93. (C) In MD calculations of glycan-free KLK8 were similar as for glycosylated KLK8. Upon removal of Ca2+, the 75-loop closes the substrate binding cleft in the prime side near the 37-loop. The concomitant closure of the S2 pocket by Val96 and His99 (orange) in the 99-loop might hamper binding of small substrates. The labeled loops around the active site represent the allosteric network of various trypsin-like serine proteases, which may regulate substrate access52. (D) Zn2+-binding would similarly close the S2 pocket and inactivate KLK8 by rotating the His57 side chain away from the catalytic triad. Such a conformation was observed in the structure of rat tonin (1TON).
In line with this general model, Zn2+ was identified as second cationic modulator of KLK8 activity, which may fix or “lock” the closed 99-loop conformation (Fig. 7D). While its physiological role at synapses is less clear than the one of Ca2+, its extracellular synaptic concentration is around 1 µM, but may reach much higher concentrations during zincergic signaling in the hippocampus60. Moreover, Zn2+ is a regulator of KLK5, 7, and 14 activity in skin, besides the LEKTI or SPINK inhibitors, which do not target KLK823,61. Thus, Ca2+ and Zn2+ appear to be antagonistic regulators of KLK8 activity in brain and skin, especially during wound healing that is usually accompanied by an increase of the Ca2+ concentration62. By contrast, in a mouse model of spinal cord injury, mKlk8 exhibited increased expression, which was confirmed in corresponding human cases63. In a multiple sclerosis mouse model upregulation of mKlk8 and mKlk6 was observed in brain encephalitis and in the spinal cord during disease development to axon degeneration. Both conditions are serious medical problems, which await proper treatment, perhaps by employing KLK8 as target for pharmaceuticals64.
Materials and Methods
Expression and purification of KLK8
KLK8 cDNA was obtained from ovarian tumor tissue mRNA and cloned into the pQE30 vector at BamH/HindIII restriction sites (Qiagen). The KLK8 constructs are coding for the mature protease domain with an N-terminal Met-Arg-Gly-Ser-His6-tag-Gly-Ser sequence followed by an enterokinase (EK) cleavage site (Asp4-Lys↓Ile-Ile). Site-directed PCR mutagenesis was performed with Pfu Turbo (Stratagene) and template digestion with DpnI (New England Biolabs).
Wild-type KLK8 and the variants D70K, Y94F, H99A, and C93S were obtained from E. coli M15[pREP4] cells (Qiagen) as inclusion bodies. After treatment with denaturing lysis buffer (6 M guanidinium-HCl, 100 mM NaH2PO4, 10 mM Tris-HCl, pH 8.0) and removal of cell debris, KLK8 was purified with nickel-nitrilotriacetic acid Sepharose chromatography (Qiagen). Column washing was done by stepwise reduction of the pH from 8.0 to 5.0, followed by KLK8 elution with 8 M urea, 100 mM NaH2PO4, 10 mM Tris-HCl, pH 4.0. The solution was titrated with NaOH to pH 8.0 and incubated with 10 mM DTT overnight at 25 °C and dialyzed against the 100-fold volume of 4 M urea, 50 mM Tris-HCl, pH 8.0, 100 mM NaCl, and 0.005% Tween-20 for 12 h at 4 °C. Refolding of KLK8 was done by dropwise dilution in 2 M urea, 50 mM Tris-HCl, pH 8.0, 100 mM NaCl, 5 mM reduced glutathione, 0.5 mM oxidized glutathione, 0.002% NaN3, 0.005% Tween-20 in the 100-fold sample volume at 4 °C for 30 h. Afterwards, the KLK8 solution was concentrated with 10 kDa cutoff double cassette membranes. The latter procedure was repeated with 1 M urea. Final concentration in 150 mM NaCl, 50 mM Tris-HCl, pH 8.0, 0,005% Tween-20 (storage buffer) was done with microconcentrators (Vivaspin, 10 kDa cutoff, Sartorius). The N-terminal His6-tag was cleaved from KLK8 with EK (Sigma), of which 1 U yielded 25 μg/ml of KLK8 to 98% in 12 h at 4 °C in storage buffer, followed by treatment with the EK antibody capture kit (Sigma) for 60 min. Active KLK8 was purified with benzamidine-Sepharose affinity chromatography (Amersham Pharmacia), washed with storage buffer, containing 300 mM NaCl, and eluted with this buffer containing 20 mM p-amino-benzamidine. Eventually, size exclusion chromatography on Superdex 2000 (16/26) was performed with storage buffer. The identity of mature KLK8 was confirmed by SDS-PAGE, mass spectrometry and N-terminal sequencing.
Positional scanning with a synthetic combinatorial peptide library
The positional scanning procedure has been previously described in detail52. Briefly, the diverse library with the general composition acetyl-P4-P3-P2-P1-7-ACC consists of 20 P1-, 20 P2-, 20 P3-, and 20 P4-libraries with one of the 19 canonical amino acids and norleucine (Nle) as substitute for Cys in the P1, P2, P3, or P4 positions. The three other positions contain an equimolar mixture of all these amino acids, resulting in 8000 compounds. The final concentration of activated KLK8 was 60 nM, whereby the single and total concentrations of substrates in 100 µl were 31.25 nM and 250 µM, respectively. Assays were performed in triplicate samples for 10 min at 25 °C in 150 mM NaCl, 50 mM Tris-HCl, pH 8.0, 0.005% Tween 20, and 1% dimethyl sulfoxide (DMSO), using 96-well Microfluor-1 Black U bottom plates (Dynex Technologies, Chantilly, VA) and a SpectraMax Gemini fluorescence spectrometer (Molecular Devices) with excitation at 380 nm and emission at 460 nm.
Proteomic identification of cleavage sites
The general procedure has been described previously for KLK2, while a more detailed description on enhanced PICS was recently published26,51. The peptide library was generated by proteolysis of E. coli proteins by GluC. KLK8 samples were incubated with the library with a 1:300 ratio in 50 mM Tris (pH 7.5), 100 mM NaCl at 37 °C for 3 h. After the reaction, isotope labeling of protease-treated and control samples was performed, followed by liquid chromatography-tandem mass spectrometry (Q-Exactive plus MS with an Easy nanoLC 1000, Thermo Scientific). The spectrum to sequence assignment was done with X! Tandem (Version 2013.09.01) with E. coli strain K12 as reference proteome. Semi-specific peptides with an increase >8-fold in the KLK8 samples were accepted as cleavage products. Prime side and non-prime side sequences were assigned according to the reference database and the corresponding protease specificity presented as heat-maps employing Web-PICS65.
Enzyme kinetic measurements
Prior to measurements with fluorogenic substrates, active site titration of mature KLK8 was done with pNPGB (BACHEM). The final enzyme concentration was 120 nM and the one of p-NPGB was 100 µM 150 mM NacCl, 50 mM Tris-HCl, pH, pH 8.0, 0.005% Tween-20, 1% DMSO at 25 °C. The release of p-nitrophenol was monitored at 410 nm, which allowed to calculate the molarity of active KLK866. Enzymatic activity of KLK8 using AMC substrates (BACHEM) was measured in 150 mM NaCl, 50 mM Tris-HCl, pH, pH 7.5, 0.005% Tween-20, 1% DMSO at 25 °C on a Perkin-Elmer LS50B spectrofluorimeter at excitation and emission wavelength of 380 nm and 460 nm. According to the results of the specificity profiling an ideal substrate was synthesized with the formula Ac-Thr-Lys-Leu-Arg-ACC27. Substrate final concentrations were 40 µM and the KLK8 concentration employed was 60 nM, Ca2+ and Zn2+ were added in the range of 1 to 1000 µM and 0.5 to 500 µM, respectively.
In measurements with pNA substrates, the following KLK8 final concentrations were employed: 175 nM wt, 250 nM D70K, and 320 nM H99A in the assay buffer with 50 mM Tris-HCl pH 7.5, 100 mM NaCl, 0.005% (v/v) Tween-20, 0.02% NaN3, and 5% (v/v) DMSO. For determination of kcat and KM the concentration of Bz-Pro-Phe-Arg-pNA (BACHEM) ranged from 50 to 1000 µM, while 250 µM were used in the Zn2+ inhibition and Ca2+ activation experiments at 37 °C. The release of pNA was monitored by the absorption at 405 nm every 20 seconds for 5 minutes. The fraction of active KLK8 was determined by active site titration with BPTI (82% active) that had been titrated with trypsin, which was 48% active according to the p-NPGB burst titration67.
Crystallization, data collection and processing
Using the sitting-drop vapor diffusion method, KLK8 crystals were grown at 18 °C from drops containing 1 μl protein solution (8 mg/ml), 10 mM leupeptin and 1 μl precipitant (100 mM tri-Na citrate, pH 5.6, 35% (v/v) tert-Bu-OH), equilibrated against 500 μl of precipitant solution. KLK8-Ca crystals grew in the orthorhombic space group P212121 with one molecule per asymmetric unit (asu), whereas KLK8-leup crystals were obtained by 2 min soaking with 100 µM ZnCl2 and belonged to the monoclinic space group P21 with four molecules per asu, respectively (Table 2). Data for KLK8-Ca were collected with a wavelength of 0.97469 Å at the EMBL beamline X12 (DESY, Hamburg) and for KLK8-leup with a wavelength of 1.00000 Å at the beamline PX II (SLS, Switzerland). Data were indexed and integrated in XDS and scaled with SCALA and AIMLESS68,69. Two KLK8-Ca data sets were merged in order compensate the strong anisotropy of the 2.0 Å resolution data set. A data cutoff at 2.3 Å was suggested by the CC(1/2) values from AIMLESS. Analysis of KLK8-leup data in CTRUNCATE and XTRIAGE showed a pseudomerohedral twin fraction of 0.24, which was taken into account in following steps of refinement70,71.
Molecular replacement, model building, and refinement
For the KLK8-Ca data in space group P212121 a molecular replacement search was performed with PHASER in the automated search mode using the mouse Klk8 model with 73% identical residues (PDB code 1NPM)72. The best solution with one mol/asu exhibited Z values for the rotation function (RFZ) of 17.0 and of 29.3 for the translation function (TFZ) with a log-likelihood gain (LLG) of +970 and an R-factor after rigid body refinement of 52.2%. After inspection of proper molecular packing, the solution was evaluated with a total omit map from SFCHECK, which had a figure-of-merit (FOM) = 80.3%73. In case of the KLK8 data in space group P21, the refined polypeptide KLK8-Ca model was employed in PHASER auto mode, resulting in a LLG = +6792 with Z-values of RFZ = 18.5 and TFZ = 21.8, and an R-factor of 44.0%.
Model building for KLK8-Ca was done iteratively with COOT and refinement with PHENIX, resulting in final Rcryst and Rfree values of 22.4% and 26.6%, respectively, for a maximum resolution of 2.30 Å (Table 2)71. The Ca2+ site was fully occupied, while no significant electron density was observed for leupeptin. Due to the high anisotropy, real space refinement was required to reduce RSRZ outliers to acceptable 8.8%. Asn and Gln side chain orientations were corrected according to NQ-Flipper V2.774. Similarly, the model of KLK8-leup was refined, requiring the pseudomerohedral twin operator (h, -k, -l), resulting in Rcryst and Rfree values of 21.2% and 25.9%, respectively, at a resolution of 2.1 Å (Table 2). An anomalous Fourier map showed no distinct peaks corresponding to potential Zn2+ sites, as well as density correlation and real space R-factors from OVERLAPMAP in CCP4i did not confirm transition metal ions. Overall, the main chain of all polypeptides is well defined, except for the loop around Arg148 and two C-terminal residues. Also, the 75-loops of KLK8 copies C and D are not well defined, while leupeptin shows some variations in the individual Leu side chain positions and the acetyl group. The Ramachandran plots show 97.3% (KLK8-Ca) and 94.9% (KLK8-leup) of residues in the most favored regions and 2.7% and 5.1% in additionally allowed regions, while no outliers were observed. All figures were created with PyMOL v1.7rc1 and electrostatic potential phi maps were calculated with the PDB2PQR Server and APBS tool75 (CIT). Root mean square deviations (RMSD) were calculated with the program SUPERPOSE76.
Molecular dynamics in solution and docking calculations
To perform the molecular dynamics calculations in an aqueous solution environment model coordinates were first titrated at pH 7.0 using the Protonated3D function of MOE 2015.10 (www.chemcomp.com)77. The resulting structure was solvated in a cubic box of water molecules with specified edges (83.6 Å × 83.6 Å × 83.6 Å) centered on the KLK8 using periodic boundary conditions. Cl− counter ions were added to maintain overall neutrality. Afterwards, a series of equilibration steps were performed by molecular dynamics annealing runs, without using a barostat, for 100 ps at temperatures 50 K, 150 K, 200 K, 250 K and 298,15 K. The production was performed for 500 ps at 298.15 K. The molecular dynamics calculations were accomplished using the AMBER99 force field as implemented in NWChem 6.678. During warm up 10000 replicas were collected at each temperature and 5 × 100000 replicas during each production cycle. In each case the time step was 0.001 ps. After each warm-up cycle and each production cycle a PDB-structure was generated and used for further analysis. The single replicas were not compared with each other. Unbound molecular docking to KLK8 was similarly performed, using a model based on the PAR-1 fragment in complex with thrombin (PDB code 1LU9) and the PICS results. The coordinates of the best hit with acceptable binding of P1-Arg to the S1 pocket were further optimized with MOE.
Accession codes
Mass spectrometry proteomics data for PICS were deposited to the ProteomeXchange Consortium via the PRIDE archive with the dataset identifier PXD006884. Coordinates have been deposited at the Protein Data Bank (www.rcsb.org) under the accession numbers 5MS3 (KLK8-Ca) and 5MS4 (KLK8-leup).
References
Suzuki, J. et al. Ontogeny of neuropsin mRNA expression in the mouse brain. Neuroscience Research 23, 345–351 (1995).
Yoshida, S., Taniguchi, M., Hirata, A. & Shiosaka, S. Sequence analysis and expression of human neuropsin cDNA and gene. Gene. 213, 9–16 (1998).
Kishi, T., Grass, L., Soosaipillai, A., Shimizu-Okabe, C. & Diamandis, E. P. Human Kallikrein 8: Immunoassay Development and Identification in Tissue Extracts and Biological Fluids. Clin Chem 49, 87–96, https://doi.org/10.1373/49.1.87 (2003).
Shaw, J. L. V. & Diamandis, E. P. Distribution of 15 Human Kallikreins in Tissues and Biological Fluids. Clin Chem. 53, 1423–1432, https://doi.org/10.1373/clinchem.2007.088104 (2007).
Rawlings, N. D., Barrett, A. J. & Bateman, A. MEROPS: the database of proteolytic enzymes, their substrates and inhibitors. Nucleic Acids Res. 40, D343–350, https://doi.org/10.1093/nar/gkr987 (2012).
Koumandou, V. L. & Scorilas, A. Evolution of the plasma and tissue kallikreins, and their alternative splicing isoforms. PLoS One 8, 1–12 (2013).
Kishi, T., Cloutier, S. M., Kündig, C., Deperthes, D. & Diamandis, E. P. Activation and enzymatic characterization of recombinant human kallikrein 8. Biol. Chem. 387, 723–731, https://doi.org/10.1515/BC.2006.091 (2006).
Chen, Z. L. et al. Expression and activity-dependent changes of a novel limbic-serine protease gene in the hippocampus. J. Neurosci. 15, 5088–5097 (1995).
Okabe, A. et al. Kindling induces neuropsin mRNA in the mouse brain. Brain Research 728, 116–120 (1996).
Mitsui, S., Tsuruoka, N., Yamashiro, K., Nakazato, H. & Yamaguchi, N. A novel form of human neuropsin, a brain-related serine protease, is generated by alternative splicing and is expressed preferentially in human adult brain. Eur J Biochem. 260, 627–634 (1999).
Shiosaka, S. & Ishikawa, Y. Neuropsin—A possible modulator of synaptic plasticity. Journal of Chemical Neuroanatomy 42, 24–29, https://doi.org/10.1016/j.jchemneu.2011.05.014 (2011).
Sonderegger, P. & Matsumoto-Miyai, K. Activity-controlled proteolytic cleavage at the synapse. Trends in Neurosciences 37, 413–423, https://doi.org/10.1016/j.tins.2014.05.007 (2014).
Evans, R. C. & Blackwell, K. T. Calcium: Amplitude, Duration, or Location? The Biological Bulletin 228, 75–83 (2015).
Lu, Z.-x, Huang, Q. & Su, B. Functional characterization of the human-specific (type II) form of kallikrein 8, a gene involved in learning and memory. Cell Res 19, 259–267 (2009).
Paixão-Côrtes, V. R., Viscardi, L. H., Salzano, F. M., Hünemeier, T. & Bortolini, M. C. Homo sapiens, Homo neanderthalensis and the Denisova specimen: New insights on their evolutionary histories using whole-genome comparisons. Genet Mol Biol 35, 904–911 (2012).
Hrvoj-Mihic, B., Bienvenu, T., Stefanacci, L., Muotri, A. R. & Semendeferi, K. Evolution, development, and plasticity of the human brain: from molecules to bones. Frontiers in Human Neuroscience 7, https://doi.org/10.3389/fnhum.2013.00707 (2013).
Kirihara, T. et al. Prolonged recovery of ultraviolet B-irradiated skin in neuropsin (KLK8)-deficient mice. Brit J Dermatol 149, 700–706 (2003).
Kishibe, M. et al. Kallikrein-related peptidase 8-dependent skin wound healing is associated with upregulation of kallikrein-related peptidase 6 and PAR2. J Invest Dermatol. 132, 1717–1724 (2012).
Eissa, A. et al. Serum kallikrein-8 correlates with skin activity, but not psoriatic arthritis, in patients with psoriatic disease. Clin Chem Lab Med. 51, 317–325 (2013).
Iinuma, S. et al. Klk8 is required for microabscess formation in a mouse imiquimod model of psoriasis. Experimental Dermatology 24, 887–889, https://doi.org/10.1111/exd.12794 (2015).
Kato, K. et al. Serine Proteinase Inhibitor 3 and Murinoglobulin I Are Potent Inhibitors of Neuropsin in Adult Mouse Brain. J Biol Chem. 276, 14562–14571, https://doi.org/10.1074/jbc.M010725200 (2001).
Rajapakse, S., Ogiwara, K., Takano, N., Moriyama, A. & Takahashi, T. Biochemical characterization of human kallikrein 8 and its possible involvement in the degradation of extracellular matrix proteins. FEBS letters 579, 6879–6884 (2005).
Goettig, P., Magdolen, V. & Brandstetter, H. Natural and synthetic inhibitors of kallikrein-related peptidases (KLKs). Biochimie 92, 1546–1567, https://doi.org/10.1016/j.biochi.2010.06.022 (2010).
Kishi, T. et al. Crystal structure of neuropsin, a hippocampal protease involved in kindling epileptogenesis. The Journal of biological chemistry 274, 4220–4224 (1999).
Debela, M. et al. Specificity Profiling of Seven Human Tissue Kallikreins Reveals Individual Subsite Preferences. J. Biol. Chem. 281, 25678–25688, https://doi.org/10.1074/jbc.M602372200 (2006).
Biniossek, M. L. et al. Identification of Protease Specificity by Combining Proteome-Derived Peptide Libraries and Quantitative Proteomics. Mol. Cell. Proteomics 15, 2515–2524, https://doi.org/10.1074/mcp.O115.056671 (2016).
Harris, J. L. et al. Rapid and general profiling of protease specificity by using combinatorial fluorogenic substrate libraries. Proc Natl Acad Sci USA 97, 7754–7759 (2000).
Shimizu, C. et al. Characterization of Recombinant and Brain Neuropsin, a Plasticity-related Serine Protease. J Biol Chem. 273, 11189–11196, https://doi.org/10.1074/jbc.273.18.11189 (1998).
Debela, M. et al. Structures and specificity of the human kallikrein-related peptidases KLK 4, 5, 6, and 7. Biol. Chem. 389, 623–632, https://doi.org/10.1515/BC.2008.075 (2008).
Kurinov, I. V. & Harrison, R. W. Two crystal structures of the leupeptin-trypsin complex. Protein Science 5, 752–758 (1996).
Sichler, K. et al. The influence of residue 190 in the S1 site of trypsin-like serine proteases on substrate selectivity is universally conserved. FEBS Letters 530, 220–224 (2002).
Debela, M. et al. Structural Basis of the Zinc Inhibition of Human Tissue Kallikrein 5. J. Mol. Biol. 373, 1017–1031 (2007).
Harding, M., Nowicki, M. W. & Walkinshaw, M. D. Metals in protein structures: a review of their principal features. Crystallogr. Rev. 16, 247–302, https://doi.org/10.1080/0889311X.2010.485616 (2010).
Bode, W. & Schwager, P. The single calcium-binding site of crystallin bovin beta-trypsin. Febs Lett 56, 139–143 (1975).
Sipos, T. & Merkel, J. R. Effect of calcium ions on the activity, heat stability, and structure of trypsin. Biochemistry 9, 2766–2775, https://doi.org/10.1021/bi00816a003 (1970).
Laitaoja, M., Valjakka, J. & Jänis, J. Zinc Coordination Spheres in Protein Structures. Inorg. Chem. 52, 10983–10991, https://doi.org/10.1021/ic401072d (2013).
Fujinaga, M. & James, M. N. Rat submaxillary gland serine protease, tonin. Structure solution and refinement at 1. 8 A resolution. J. Mol. Biol 195, 373–396 (1987).
McGrath, M. E., Haymore, B. L., Summers, N. L., Craik, C. S. & Fletterick, R. J. Structure of an engineered, metal-actuated switch in trypsin. Biochemistry 32, 1914–1919 (1993).
Hiss, J. A. & Schneider, G. Architecture, function and prediction of long signal peptides. Brief. Bioinform. 10, 569–578, https://doi.org/10.1093/bib/bbp030 (2009).
Yoon, H. et al. Activation Profiles and Regulatory Cascades of the Human Kallikrein-related Peptidases. J Biol Chem. 282, 31852–31864, https://doi.org/10.1074/jbc.M705190200 (2007).
Komatsu, N. et al. Proteolytic processing of human growth hormone by multiple tissue kallikreins and regulation by the serine protease inhibitor Kazal-Type5 (SPINK5) protein. Clinica Chimica Acta 377, 228–236 (2007).
Tamura, H., Kawata, M., Hamaguchi, S., Ishikawa, Y. & Shiosaka, S. Processing of neuregulin-1 by neuropsin regulates GABAergic neuron to control neural plasticity of the mouse hippocampus. J Neurosci. 32, 12657–12672, https://doi.org/10.1523/JNEUROSCI.2542-12.2012. (2012).
Konar, A. & Thakur, M. K. Neuropsin Expression Correlates with Dendritic Marker MAP2c Level in Different Brain Regions of Aging Mice. Molecular Neurobiology 51, 1130–1138, https://doi.org/10.1007/s12035-014-8780-z (2014).
Attwood, B. K. et al. Neuropsin cleaves EphB2 in the amygdala to control anxiety. Nature 473, 372-375, http://www.nature.com/nature/journal/v473/n7347/abs/10.1038-nature09938-unlocked.html#supplementary-information (2011).
Herring, A. et al. Kallikrein-8 inhibition attenuates Alzheimer’s disease pathology in mice. Alzheimer’s & Dementia 12, 1273–1287, https://doi.org/10.1016/j.jalz.2016.05.006 (2016).
Schauperl, M. et al. Characterizing Protease Specificity: How Many Substrates Do We Need? PLoS One 10, e0142658, https://doi.org/10.1371/journal.pone.0142658 (2015).
Timmer, J. C. et al. Structural and kinetic determinants of protease substrates. Nat. Struct. Mol. Biol. 16, 1101–U1128, https://doi.org/10.1038/Nsmb.1668 (2009).
Takehara, S. et al. The 2.1-Å Crystal Structure of Native Neuroserpin Reveals Unique Structural Elements That Contribute to Conformational Instability. Journal of Molecular Biology 388, 11–20, https://doi.org/10.1016/j.jmb.2009.03.007 (2009).
Krueger, S. R. et al. Expression of Neuroserpin, an Inhibitor of Tissue Plasminogen Activator, in the Developing and Adult Nervous System of the Mouse. The Journal of Neuroscience 17, 8984 (1997).
Stefansson, K. et al. Activation of proteinase-activated receptor-2 by human kallikrein-related peptidases. J Invest Dermatol 128, 18–25 (2008).
Guo, S. et al. A Single Glycan at the 99-Loop of Human Kallikrein-related Peptidase 2 Regulates Activation and Enzymatic Activity. J. Biol. Chem. 291, 593–604, https://doi.org/10.1074/jbc.M115.691097 (2016).
Skala, W. et al. Structure-Function Analyses of Human Kallikrein-Related Peptidase 2 Establish the 99-Loop as Master Regulator of Activity. J. Biol. Chem. 289, 34267–34283, https://doi.org/10.1074/jbc.M114.598201 (2014).
Cohen, J. E. & Fields, R. D. Extracellular Calcium Depletion in Synaptic Transmission. The Neuroscientist 10, 12–17, https://doi.org/10.1177/1073858403259440 (2004).
Rezaie, A. R. & Esmon, C. T. Asp-70–> Lys mutant of factor X lacks high affinity Ca2+ binding site yet retains function. Journal of Biological Chemistry 269, 21495–21499 (1994).
Zögg, T. & Brandstetter, H. Structural Basis of the Cofactor- and Substrate-Assisted Activation of Human Coagulation Factor IXa. Structure 17, 1669–1678, https://doi.org/10.1016/j.str.2009.10.011 (2009).
Hopfner, K.-P. et al. Coagulation factor IXa: the relaxed conformation of Tyr99 blocks substrate binding. Structure 7, 989–996, https://doi.org/10.1016/S0969-2126(99)80125-7 (1999).
Sichler, K. et al. Physiological fIXa Activation Involves a Cooperative Conformational Rearrangement of the 99-Loop. Journal of Biological Chemistry 278, 4121–4126, https://doi.org/10.1074/jbc.M210722200 (2003).
Rezaie, A. R. & He, X. Sodium Binding Site of Factor Xa: Role of Sodium in the Prothrombinase Complex. Biochemistry 39, 1817–1825, https://doi.org/10.1021/bi992006a (2000).
Riley, B. T. et al. Direct and indirect mechanisms of KLK4 inhibition revealed by structure and dynamics. Sci Rep. 6, 35385, https://doi.org/10.1038/srep35385, http://www.nature.com/articles/srep35385#supplementary-information (2016).
Takeda, A. & Tamano, H. Cognitive decline due to excess synaptic Zn2+ signaling in the hippocampus. Front Aging Neurosci. 6, https://doi.org/10.3389/fnagi.2014.00026 (2014).
Kantyka, T. et al. Inhibition of kallikrein-related peptidases by the serine protease inhibitor of Kazal-type 6. Peptides 32, 1187–1192, https://doi.org/10.1016/j.peptides.2011.03.009 (2011).
Lansdown, A. B. G. Calcium: a potential central regulator in wound healing in the skin. Wound Repair and Regeneration 10, 271–285 (2002).
Radulovic, M. et al. Kallikrein Cascades in Traumatic Spinal Cord Injury: In Vitro Evidence for Roles in Axonopathy and Neuron Degeneration. J. Neuropathol. Exp. Neurol. 72, 1072-1089, 1010.1097/NEN.0000000000000007 (2013).
Panos, M., Christophi George, P., Rodriguez, M. & Scarisbrick Isobel, A. Differential expression of multiple kallikreins in a viral model of multiple sclerosis points to unique roles in the innate and adaptive immune response. In Biol. Chem. 395, 1063 (2014).
Schilling, O. auf dem Keller, U. & Overall, C. M. Factor Xa subsite mapping by proteome-derived peptide libraries improved using WebPICS, a resource for proteomic identification of cleavage sites. Biol. Chem. 392, 1031–1037, https://doi.org/10.1515/BC.2011.158 (2011).
Chase, T. Jr. & Shaw, E. Comparison of the esterase activities of trypsin, plasmin, and thrombin on guanidinobenzoate esters. Titration of the enzymes. Biochemistry 8, 2212–2224 (1969).
Debela, M. et al. Crystal structures of human tissue kallikrein 4: activity modulation by a specific zinc binding site. J. Mol. Biol. 362, 1094–1107 (2006).
Kabsch, W. X. D. S. Acta Crystallographica Section D 66, 125–132, https://doi.org/10.1107/S0907444909047337 (2010).
Evans, P. An introduction to data reduction: space-group determination, scaling and intensity statistics. Acta Crystallographica Section D 67, 282–292, https://doi.org/10.1107/S090744491003982X (2011).
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallographica Section D 67, 235–242, https://doi.org/10.1107/S0907444910045749 (2011).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallographica Section D 66, 213–221, https://doi.org/10.1107/S0907444909052925 (2010).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Cryst. 40, 658–674 (2007).
Vaguine, A. A., Richelle & Wodak, S. J. SFCHECK: a unified set of procedures for evaluating the quality of macromolecular structure-factor data and their agreement with the atomic model. Acta Cryst. D55, 191–205 (1999).
Weichenberger, C. X. & Sippl, M. J. NQ-Flipper: validation and correction of asparagine/glutamine amide rotamers in protein crystal structures. Bioinformatics 22, 1397–1398, https://doi.org/10.1093/bioinformatics/btl128 (2006).
DeLano, W. L. The PyMOL Molecular Graphics System (2002).
Krissinel, E. & Henrick, K. Secondary-structure matching (SSM), a new tool for fast protein structure alignment in three dimensions. Acta Crystallographica Section D 60, 2256–2268, https://doi.org/10.1107/S0907444904026460 (2004).
Molecular Operating Environment (MOE) v. 2015.10 (Chemical Computing Group, Inc., 1010 Sherbooke St. West, Suite #910, Montreal, QC, Canada, H3A 2R7, 2016).
Valiev, M. et al. NWChem: A comprehensive and scalable open-source solution for large scale molecular simulations. Computer Physics Communications 181, 1477–1489, https://doi.org/10.1016/j.cpc.2010.04.018 (2010).
Biasini, M. et al. SWISS-MODEL: modelling protein tertiary and quaternary structure using evolutionary information. Nucleic Acids Research 42, W252–W258, https://doi.org/10.1093/nar/gku340 (2014).
Acknowledgements
We are thankful to Martina Wiesbauer and Sabine Creutzburg for technical support. Support for this study came from the European Commission (project CAMP; LSHG-2006-018830), the Fonds der Chemischen Industrie (W.B.), and by the “Graduiertenkolleg 333 der Deutschen Forschungsgemeinschaft” (M.D.), the German Federal Ministry of Education and Research, Leading Edge Cluster m4 (V.M.), NIH GM104659 and CA196276 (C.S.C.), the Austrian science fund FWF, projects I631-B11 (W.S.) and P25003-B21 (P.G.).
Author information
Authors and Affiliations
Contributions
M.D., V.M., W.S., B.E., E.L.S., M.L.B., P.G. performed experiments and analyzed the data; V.M., C.S.C., O.S., W.B., H.B. and P.G. designed the study; M.D., V.M., B.E., C.S.C., O.S., W.B., H.B. and P.G. interpreted the results and wrote the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Debela, M., Magdolen, V., Skala, W. et al. Structural determinants of specificity and regulation of activity in the allosteric loop network of human KLK8/neuropsin. Sci Rep 8, 10705 (2018). https://doi.org/10.1038/s41598-018-29058-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-29058-6
This article is cited by
-
Coordination chemistry suggests that independently observed benefits of metformin and Zn2+ against COVID-19 are not independent
BioMetals (2024)
-
Visualizing protein–ligand binding with chemical energy-wise decomposition (CHEWD): application to ligand binding in the kallikrein-8 S1 Site
Journal of Computer-Aided Molecular Design (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.