Abstract
Stratospheric ozone has begun to recover in Antarctica since the implementation of the Montreal Protocol. However, the effects of ultraviolet (UV) radiation on tundra greenhouse gas fluxes are rarely reported for Polar Regions. In the present study, tundra N2O and CH4 fluxes were measured under the simulated reduction of UV radiation in maritime Antarctica over the last three-year summers. Significantly enhanced N2O and CH4 emissions occurred at tundra sites under the simulated reduction of UV radiation. Compared with the ambient normal UV level, a 20% reduction in UV radiation increased tundra emissions by an average of 8 μg N2O m−2 h−1 and 93 μg CH4 m−2 h−1, whereas a 50% reduction in UV radiation increased their emissions by an average of 17 μg N2O m−2 h−1 and 128 μg CH4 m−2 h−1. No statistically significant correlation (P > 0.05) was found between N2O and CH4 fluxes and soil temperature, soil moisture, total carbon, total nitrogen, NO3−-N and NH4+-N contents. Our results confirmed that UV radiation intensity is an important factor affecting tundra N2O and CH4 fluxes in maritime Antarctica. Exclusion of the effects of reduced UV radiation might underestimate their budgets in Polar Regions with the recovery of stratospheric ozone.
Similar content being viewed by others
Introduction
Atmospheric nitrous oxide (N2O) and methane (CH4) are two main greenhouse gases (GHGs). In addition, N2O partly contributes to stratospheric ozone depletion1. Increases in N2O and CH4 emissions and their roles in aggravating global warming, have caused great concern in the past three decades1. Currently, the fluxes of N2O and CH4 and their influencing factors have been extensively investigated from subtropical, tropical, temperate terrestrial ecosystems and boreal tundra in the Northern Hemisphere2,3,4,5,6,7. However, the related studies were conducted very late in the Antarctic terrestrial ecosystem.
Recent studies of N2O and CH4 fluxes from the Antarctic terrestrial ecosystem mainly concentrated on the McMurdo Dry Valleys of continental Antarctica8,9,10,11, the Antarctic Peninsula and other islands of maritime Antarctica12,13,14,15. Microtopography, mineralizing substrate availability, soil temperature, soil moisture and O2 availability could affect tundra N2O or CH4 fluxes9,10,11,12,14,15,16,17. Climate change might affect N2O and CH4 emissions from the tundra, because some soil parameters, e.g., soil moisture and temperature, are associated with microbial activity and the mineralization of organic carbon and nitrogen in soils17,18,19. In addition, significantly enhanced N2O and CH4 emissions were found from penguin and seal colonies, which have been identified as “hot spots” for N2O and CH4 emissions in maritime Antarctica because of the high load of readily available organic carbon and nitrogen through penguin or seal excreta12,14,16. Nevertheless, the effects of solar ultraviolet (UV) radiation on N2O and CH4 fluxes have received little attention in the Antarctic tundra.
Strong UV (UV-A and UV-B) radiation has occurred in Antarctica because of the serious destruction of stratospheric ozone20. Enhanced UV radiation resulted in a 75% decrease in the ATP content of the microorganisms in the upper water of the Weddell Sea, Antarctica21. Pakulski et al.22 reported a 57% reduction in marine bacteria around Palmer Station during low total ozone column episodes. A significant correlation has been identified between DNA damage in Antarctic pelagic icefish eggs and UV irradiance23. The survival rates of Antarctic krill decrease in response to increased UV radiation24. Both UV-A and UV-B are major drivers of the decomposition of vegetation litter in the Antarctic terrestrial ecosystem through the process of photodegradation25,26,27. In addition, they have the potential to affect the structure and function of Antarctic mosses, Ceratodon purpureus and Bryum subrotundifolium28 and to influence indirectly the soil microbial populations and activities26. UV radiation is also a key regulator of vegetation morphology and genetic processes and is important in vegetation growth27,29. Vegetation growth and soil microbial activities are the main factors influencing plant respiration and N2O and CH4 emissions from the tundra28. Sunlight could greatly affect N2O and CH4 emissions from tundra ecosystem because of O2 release via vegetation photosynthesis30. The UV-induced release of carbon from plant litter and soils might contribute to global warming27. Therefore, it is important to investigate the effects of UV intensity on tundra N2O and CH4 fluxes and carbon and nitrogen cycles, in maritime Antarctica.
Currently, stratospheric ozone has recovered somewhat in Antarctica since the implementation of the Montreal Protocol in 198931. The Antarctic ozone hole has shrunk by nearly 400,000 square miles since it was discovered around 30 years ago. The ozone layer in the Polar Regions is projected to recover to pre-1980 levels by 2048, thus less solar UV radiation will reach the earth’s surface32. However, the effects of a reduction in UV radiation on N2O and CH4 emissions to date have not been investigated in the Antarctic tundra. During the austral summers of 2011/2012, 2013/2014 and 2014/2015, we selected a tundra ecosystem in the maritime Antarctica as study area (Fig. 1) and for the first time, investigated tundra N2O and CH4 fluxes under the conditions of simulated reduction in UV (UV-A and UV-B) radiation, to explore whether natural UV radiation reduction could stimulate tundra N2O and CH4 emissions. This is an important attempt to increase the Antarctic GHGs data sets to reasonably evaluate the potential effects of UV radiation reduction on tundra N2O and CH4 fluxes.
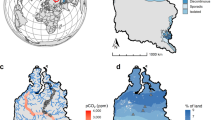
Study area and the N2O and CH4 fluxes at the observation sites. Panel (a), the dot indicates the location of the investigation area in maritime Antarctica. Panel (b), location of the investigation sites on Fildes Peninsula and Ardley Island, King George Island. Three upland tundra sites (GW1–GW3) are shown. Panel (c), the sites of the flux chambers in the eastern tundra and the western tundra on Ardley Island, including six regular sites AE1–AE3 and AW1–AW3. Note: The map was drawn using CorelDRAW 2017 software (http://www.corel.com/cn/).
Results
UV radiation and environmental variables between experimental treatments
In the summer of 2011/2012, UV radiation intensity showed similar temporal variation patterns between the control site and the sites covered by 0.03 mm and 0.06 mm filter membranes (Fig. 2a). The use of filter membrane between experimental treatments significantly decreased (analysis of variance (ANOVA) and least significant difference (LSD) test, P < 0.05) the amount of UV radiation penetrating into the chamber (Table 1). Compared with the control tundra site, the UV-A and UV-B through the sites with 0.03 mm and 0.06 mm filter membrane decreased by 20% and 50%, respectively (Fig. 2b). The highest mean UV-A and UV-B intensity occurred at the control site (14.4 ± 2.1 mW cm−2 and 4.7 ± 0.3 mW cm−2, respectively), followed by the site covered by 0.03 mm membrane (11.4 ± 1.6 mW cm−2 and 3.8 ± 0.3 mW cm−2, respectively) and the lowest at the site covered by 0.06 mm membrane (7.1 ± 1.0 mW cm−2 and 2.4 ± 0.2 mW cm−2, respectively). However, no significant differences (ANOVA and LSD test, P > 0.05) were found in terms of chamber temperatures (CTs) between the different treatment groups (Table 1) and the CTs showed similar temporal variation patterns at different tundra sites (Fig. 2c). Thus, the use of filter membranes between experimental treatments did not significantly alter chamber micrometeorological conditions, except for the UV intensity. Therefore, the filter membranes could be used to stimulate various UV intensities and explore the effects of UV radiation on tundra N2O and CH4 fluxes in maritime Antarctica. In addition, soil environmental properties, including pH, soil moisture, soil total organic carbon (TOC) and total nitrogen (TN) were similar to each other among the sites: AW1, AW2 and AW3 in the western tundra; AE1, AE2 and AE3 in the eastern tundra on Ardley Island; and GW1, GW2 and GW3 in the upland tundra on Fildes Peninsula. Detailed information about the climatic conditions and soil physiochemical properties is given in Supplementary Figures S1 and Tables S1 and S2.
Tundra N2O fluxes under reduced UV radiation
During the three observation periods, tundra N2O fluxes showed similar fluctuations between experimental treatments under reduced UV radiation (Fig. 3). In the western tundra marsh on Ardley Island, the highest mean N2O flux (24.2 ± 7.1 μg N2O m−2 h−1 in summer 2011/2012, 8.0 ± 3.6 μg N2O m−2 h−1 in summer 2013/2014 and 13.8 ± 4.7 μg N2O m−2 h−1 in summer 2014/2015) occurred at the site AW3 under 50% reduction in UV radiation, followed by AW2 (12.2 ± 3.4 μg N2O m−2 h−1 in summer 2011/2012, 4.8 ± 3.4 μg N2O m−2 h−1 in summer 2013/2014 and 2.3 ± 3.9 μg N2O m−2 h−1 in summer 2014/2015) under 20% reduction in UV radiation and the lowest was at the control site AW1 (mean fluxes were close to the detection limit) (Fig. 3a,b,c). Similarly, in the eastern tundra on Ardley Island substantial N2O emissions (mean 29.5 ± 2.6 μg N2O m−2 h−1) were observed at site AE3 under 50% reduction in UV radiation in summer 2011/2012, which was almost twice as high as that at site AE2 under 20% reduction in UV radiation (mean 13.8 ± 5.5 μg N2O m−2 h−1), whereas the control site AE1 was a weak N2O sink (mean −3.2 ± 5.2 μg N2O m−2 h−1) (Fig. 3d). For the upland tundra, site GW3 under 50% reduction in UV radiation showed the highest N2O emissions (mean 8.8 ± 3.6 μg N2O m−2 h−1) at all the sites in summer 2014/2015, whereas the control site GW1 was a weak N2O sink with a mean flux of −3.0 ± 1.2 μg N2O m−2 h−1 (Fig. 3e). Overall, the reduction in UV radiation significantly increased tundra N2O emissions in maritime Antarctica, although the N2O fluxes fluctuated markedly between the summers of 2011/2012, 2013/2014 and and 2014/2015.
The N2O flux from the eastern, western and upland tundra sites with different experimental treatments under the reduction of UV radiation during the summers of 2011/2012, 2013/2014 and 2014/2015. Panels a–c show the western tundra N2O flux under the different UV radiation intensities in the summers of 2011/2012, 2013/2014 and 2014/2015, respectively; panel d shows the eastern tundra N2O flux under the different UV radiation intensities in 2011/2012 summer; and panel e shows the upland tundra N2O flux under the different UV radiation intensities in summer 2014/2015. The squares represent the mean fluxes and solid lines represent median values. Boxes enclose the interquartile range; whiskers show the full range. Analysis of variance (ANOVA) and the least significant difference (LSD) tests on the N2O emission rates from all three sites showed a significant difference (P < 0.05) between the sites with different UV-radiation treatments.
ANOVA and LSD tests on the N2O emission rates from all three sites showed a significant difference (P < 0.05) among the sites with different UV-radiation treatments (Fig. 4). Relative to the controls, the 20% reduction in UV radiation increased the tundra N2O emissions by more than 5 μg N2O m−2 h−1, reaching as high as 14 μg N2O m−2 h−1. The 50% reduction in UV radiation increased tundra N2O emissions by more than 9 μg N2O m−2 h−1, reaching as high as 27 μg N2O m−2 h−1 during the observation periods (Table 2). Therefore, UV radiation intensity had an important effect on the N2O fluxes in maritime Antarctic tundra. Tundra N2O fluxes showed no significant correlations (Pearson correlation test, P > 0.05) with total organic carbon, soil moisture, total nitrogen, 0 cm soil temperature, 5 cm soil temperature, 10 cm soil temperature and NO3−-N and NH4+-N contents when the data at all the tundra sites were combined (Table S3), thus these environmental variables might not be the key factors affecting tundra N2O fluxes.
Comparisons of tundra N2O and CH4 fluxes under different UV radiation intensities in maritime Antarctica. Note: All the data from the sites AW1–AW3, AE1–AE3 and GW1–GW3 were analyzed for N2O and CH4 fluxes. For all the tundra sites, there were significant differences (ANOVA and LSD tests, P < 0.05) between the mean N2O, CH4 fluxes under the different UV radiation intensities.
Tundra CH4 fluxes under reduced UV radiation
During the summers of 2013/2014 and 2014/2015, the western tundra sites showed a large fluctuation, ranging from −324.9 to 594.4 μg CH4 m−2 h−1, with a mean of 89.5 ± 24.4 μg CH4 m−2 h−1 (Fig. 5a,b). Relatively strong CH4 uptake occurred at the control site AW1, with a mean flux of −11.4 ± 41.2 μg CH4 m−2 h−1. The flux at site AW2 under 20% reduction in UV radiation ranged between a weak sink and a weak source, with the mean of 122.4 ± 33.9 μg CH4 m−2 h−1. The CH4 flux at site AW3 under 50% reduction in UV radiation ranged between a weak sink (as low as −66.9 μg CH4 m−2 h−1) and a strong source (up to 594.4 μg CH4 m−2 h−1), with the greatest mean CH4 emission rate (157.7 ± 40.9 μg CH4 m−2 h−1) among all the sites. Similarly, the upland tundra acted as stronger CH4 sink at the control site GW1 (mean −102.4 ± 88.3 μg CH4 m−2 h−1 with the maximum uptake of 520.1 μg CH4 m−2 h−1) compared with site GW2 (mean −14.3 ± 58.9 μg CH4 m−2 h−1) under 20% reduction in UV radiation, whereas tundra site GW3 under 50% reduction in UV radiation showed weak CH4 emission (mean 42.5 ± 94.5 μg CH4 m−2 h−1) in summer 2014/2015 (Fig. 5c). Therefore, the reduction of UV radiation decreased tundra CH4 uptake rates over all three sites and could even convert the tundra from CH4 sinks into net sources in maritime Antarctica.
The CH4 flux from the western and upland tundra sites during the summers of 2013/2014 and 2014/2015. Panels a and b. shows the western tundra CH4 flux under the different UV radiation intensities in the summers of 2013/2014 and 2014/2015; panel c shows the upland tundra CH4 flux under the different UV radiation intensities in 2014/2015 summer. The squares represent the mean fluxes and solid lines represent median values. Boxes enclose the interquartile range; whiskers show the full range. ANOVA and the LSD test on the CH4 emission rates from all three sites showed a significant difference (P < 0.05) between the sites with different UV-radiation treatments.
There were significant differences (ANOVA and LSD test, P < 0.05) between the mean CH4 fluxes under the different UV radiation intensities for all tundra sites (Fig. 4). Relative to the controls, the 20% reduction in UV intensity increased tundra CH4 emissions by more than 77 μg CH4 m−2 h−1, reaching as high as 109 μg CH4 m−2 h−1. The 50% reduction in UV intensity increased tundra CH4 emissions by more than 106 μg CH4 m−2 h−1, reaching as high as 150 μg CH4 m−2 h−1 during the observation periods (Table 3). Therefore, UV radiation intensity had an impact on tundra CH4 fluxes in maritime Antarctica. Except for 0 cm soil temperature, CH4 fluxes showed no significant correlations (Pearson correlation analysis, P > 0.05) with total organic carbon, soil moisture, total nitrogen, 5 cm soil temperature, 10 cm soil temperature and NO3−-N and NH4+-N contents (Table S3), indicating that these environmental variables might not be the key factors affecting tundra CH4 fluxes.
Discussion
In this study, no significant correlation (Pearson correlation analysis, P > 0.05) was found between tundra N2O fluxes and soil biogeochemical properties (Table S3). However, reduced UV radiation significantly (ANOVA and LSD test, P < 0.05) increased tundra N2O emissions in maritime Antarctica, confirming that the variability in UV radiation has an important effect on tundra N2O fluxes and a reduction in UV radiation might increase tundra vegetation N2O production. Some wetland plants can produce and release some N2O via the physiological reaction of plant tissues33,34. Generally nitrate reductase (NR), which is responsible for reducing nitrate into nitrite in some plants, plays a key role in the nitrogen metabolism pathway26. Furthermore, the reduction in UV radiation significantly stimulated the activities of NR and glutamine synthetase in plants35,36. In maritime Antarctica, tundra vegetation might also produce some N2O, which is probably related to the content of nitrate and the activity of NR. Indeed, exposure to enhanced UV radiation caused a decrease in the growth rate of Deschampsia antarctica and the activities of NR in maritime Antarctica26. Therefore, the reduction in UV radiation might increase NR activity, thereby stimulating nitrate reduction and N2O formation in tundra vegetation, which would lead to an increase in N2O emissions from tundra vegetation.
The increase in N2O emissions might also be caused by stimulation of tundra vegetation growth under reduced UV radiation. The response of tundra vegetation photosynthetic rates and vegetation-soil respiration rates to the change in light intensity was almost immediate in the static chambers15,37. Reduced UV radiation significantly increased photosynthesis, the leaf cross-section and the proportion of aerenchyma in most of wetland plants34,36,38. The growth of the two phanerogamic Antarctic plants, Deschampsia antarctica and Colobanthus quitensis, appeared to be affected by manipulated surface solar UV levels during the severe ozone depletion in field experiments39 and leaf growth of Deschampsia antarctica decreased with elevated UV-B40. Plant growth affected the available nitrogen, soluble organic carbon and O2 in the soil; and accelerated N2O production and release from the plant-soil system14,38. In addition, plants also serve as a conduit to transport the N2O produced in the soil to the atmosphere14,36. Therefore, the stimulation of tundra vegetation growth under reduced UV radiation might influence soil properties and further promote N2O emissions from the soil-vegetation system.
In addition, N2O is produced naturally through nitrification and denitrification by soil microorganisms41. Although UV radiation cannot penetrate into the soil below 5 mm, enhanced UV radiation may impose indirect effects on the dynamics of microbial communities, mainly via its direct influence on vegetation growth and physiological metabolism, which in turn reduces the absorption of available N and affects root secretion42. Many studies have shown that reduced UV radiation significantly increased total abundance and activities of bacteria, such as nitrifiers and denitrifiers, in the rhizosphere soil of wetland vegetation26,34,43. Therefore, reduced UV radiation might increase the activities of tundra soil microorganisms associated with the nitrogen cycle in maritime Antarctica.
Similarly, the lack of a significant correlation (Pearson correlation analysis, P > 0.05) between tundra CH4 fluxes and soil properties (Table S3) indicated that soil temperature, soil moisture and other soil properties had an insignificant effect on tundra CH4 fluxes. In this study, the reduction of UV intensity could significantly (ANOVA and LSD test, P < 0.05) increase tundra CH4 emission in maritime Antarctica, which was very similar to that observed at peatland sites in Finland44. Direct effects of UV radiation on CH4 producing or oxidizing bacteria were not likely because solar radiation penetrates only a few centimeters into the ground45,46. However, there are some indirect effects between UV radiation and CH4 emission, because the reduction of UV radiation induced changes in root exudates, which indirectly affect CH4 production in the soil42,47. Unlike higher plants, lichens and mosses in Antarctica lack a well-developed root system; therefore, most C/N organic material entering the extracellular pools in polar soils probably comes from root and microbial turnover48,49. Vegetation root exudates provide carbon and energy sources for the growth of methanogens, thus promoting CH4 production in the tundra26,47. Intense UV radiation might decrease the distribution of carbohydrates into the roots of vegetation in the Antarctic summer, which was thought to be the major reason why enhanced UV radiation inhibited CH4 emissions in wetlands29,50. UV radiation induced changes in the contents of soil root exudates and decreased UV radiation led to an increase of 15.8% in the rate of CH4 emissions from the wetlands36. Therefore, decreased UV radiation stimulated the secretion of root exudates, which might be an important mechanism underlying the effect of UV radiation on CH4 emissions from tundra wetland.
By contrast, in general, ground vegetation might exhibit morphological changes under different ultraviolet intensities34,51. Outdoor species may be sensitive to an increase in UV and decreased UV radiation significantly increased the leaf cross section and proportion of aerenchyma in most wetland plants44,51. In our study area, tundra vegetation, including short mosses and lichens, grow very close to the ground and some of them were even buried in the tundra soils14, therefore aerenchymatous tissue of tundra vegetation might have an important role in transporting CH4 from the soil to the atmosphere. In this experiment, the increased cross-sectional area of the plant aerenchyma caused by the reduction of UV radiation is one possible explanation for the stimulated transport of CH4 from the soil to the atmosphere. However, it remains unclear whether the stomatal functioning controls CH4 transport through the mosses or lichens. If the UV induces changes in the stomatal conductance of tundra plants, as shown in several studies with higher plants44,52,53, it could alter CH4 emission rates. Therefore, the reduction in UV radiation might stimulate CH4 emission by affecting tundra vegetation development.
In this study, atmospheric photochemical reactions in the chamber should also be considered. The UV-induced photolysis of N2O comprises approximately 90% of the global N2O sink54 and it is very likely that the enhanced N2O emissions under lower UV intensity were caused by reduced photolysis of N2O. In addition, an important atmospheric sink for CH4 is the reaction between OH and CH455 and less OH might be generated when UV radiation is reduced in the chambers, thus the “apparent” CH4 flux from the tundra sites might also be enhanced when the chambers are covered by the thicker filter membranes. More research is needed to test these hypotheses in the future. In general, our results indicated that a reduction of natural UV radiation significantly (ANOVA and LSD test, P < 0.05) increased tundra N2O and CH4 emissions compared with the control under ambient UV levels (Tables 2 and 3). Solar UV radiation might have an important effect on N2O and CH4 budgets in the maritime Antarctic tundra. Although strong solar UV radiation still exists in maritime Antarctica, recovery of stratospheric ozone has occurred since the implementation of the Montreal Protocol in 1989 and the amount of solar UV radiation reaching the earth’s surface would be decreased31,32. The effects of UV radiation on tundra N2O and CH4 fluxes and their budgets, should be evaluated in the Arctic and Antarctic regions. The exclusion of its effects might underestimate N2O and CH4 budgets in the tundra ecosystem of Polar Regions. To assess the regional N2O and CH4 budget precisely, long-term measurements of GHG fluxes should be designed in the Antarctic or Arctic tundra ecosystems to show effects of UV radiation intensities on N2O and CH4 fluxes.
Methods
Study area and investigation sites
One research area was located on Ardley Island (62° 13′ S, 58° 56′W; an area of 2.0 × 1.5 km) (Fig. 1). This island is recognized by the Scientific Committee of Antarctic Research as an area of special scientific interest. The western region of this island is a costal lowland tundra marsh and the vegetation cover was around 95%14. The middle on this island is a non-level, hilly and relatively dry upland tundra, with vegetation coverage of 90–95%14. The middle upland and western lowland tundra are free of active penguin populations. The active penguin populations only concentrate in the east of this island12 and tundra patches have formed in the marginal zones of penguin nesting sites and are almost totally (90–95%) covered by mosses, algae and lichens in the east15.
Another research area was situated on Fildes Peninsula (61° 51′−62° 15′S, 57° 30′−59° 00′W; an area of 30 km2) in the southwestern area of King George Island (Fig. 1a,b). Communities of mosses and lichens represent the vegetation on this peninsula. An upland tundra was well-developed in the northwest of the Chinese Great Wall Station, at a distance of about 500 m from the station. The upland tundra was nearly dry, with an elevation of around 40 m a.s.l. The sampling ground was totally covered by mosses (Bryum Pseudotriquetrum and Bryum muelenbeckii) and lichens (Usnea sp.), with a depth of around 5–10 cm for the vegetation layer. Under the vegetation cover is an organic clay layer, with the depth of around 10–15 cm. A more detailed description about the study area was given by Zhu et al.15.
During the summers of 2011/2012, 2013/2014 and 2014/2015, three observation sites were set up in the western tundra marsh on Ardley Island, equipped with three chamber collars each. The chambers were covered by special polyester filter membranes (Mylar-D, 0.03-mm/0.06-mm thick; DuPont Co., Wilmington, DE, USA), which removed part of the UV-A and UV-B wavelengths and had no effect on other wavelengths of light56, to simulate the effect of natural UV-radiation reduction on tundra GHG fluxes: (1) the control site AW1 had transparent chambers; (2) site AW2 had transparent chambers covered by a 0.03-mm filter membrane; and (3) site AW3 had transparent chambers covered by a 0.06-mm filter membrane (Fig. 1c). In addition, during the summer of 2012/2013, three other observation sites were established in the eastern tundra of Ardley Island: (1) the control site AE1 had transparent chambers; (2) site AE2 had transparent chambers covered by a 0.03-mm filter membrane; and (3) site AE3 had transparent chambers covered by a 0.06-mm filter membrane (Fig. 1c). During summer 2014/2015, N2O and CH4 fluxes were also measured at three observation sites in the upland tundra on the Fildes Peninsula: (1) the control site GW1 had transparent chambers; (2) site GW2 had transparent chambers covered by a 0.03-mm filter membrane; (3) site GW3 had transparent chambers covered by a 0.06-mm filter membrane (Fig. 1b). There were no differences in the dominant vegetation species and phytomass among the three sites in each study area15. These observation sites were characteristic of the typical surface and vegetation within the tundra ecosystems in maritime Antarctica.
UV radiation measurement
To test whether the UV radiation polyester filter membrane with different thicknesses could decrease solar ultraviolet radiation, we used an UV radiation instrument (Photoelectric Instrument Factory, Beijing Normal University, Beijing, China) with UV radiation sensors and data loggers (model UV-II) to measure the UV intensity. The sensors, which were manually mounted under the chambers with different thickness polyester filter membrane, collected UV data at 5-min intervals and the measured data displayed by the instrument was the radiant exposure (mW cm−2). The instrument was calibrated by the manufacturer and was used within the one-year interval of the validity for this calibration. The order of measurements was randomized to ensure that the measuring sequence did not bias the results and each site had three replicate measurements. During the period from Dec 24, 2011 to Feb 5, 2012, the UV radiation intensity was measured eight times at sites AW1, AW2 and AW3. These data indicated that the filter membrane significantly (ANOVA and LSD test, P < 0.05) decreased the UV radiation transmitted to the chamber (Fig. 2a). The UV radiation through site AW1 plots was not affected, the UV-A and UV-B decreased by 20% through the site AW2 plots and by 50% through the AW3 plots (Fig. 2b).
In situ N2O and CH4 flux measurement
A static chamber technique was used to measure N2O and CH4 fluxes from the tundra sites12,15. Gas samples were taken from the clear plexiglass chambers (area: 0.25 m2, volume: 0.06 m3) placed on the PVC collars installed at the measurement sites. The collars were pushed 5 cm into the soil and air samples were taken within the headspace after 0, 10 and 20 min using a both ends needle. Gas samples were immediately transferred to 17.8 ml glass vials, which had been evacuated in advance14,15. More information on the in situ N2O and CH4 flux measurement is given in Supplementary Materials S1. During the summer of 2011/2012, N2O fluxes were measured at the sites (AW1, AW2, AW3 and AE1, AE2, AE3) from Dec 1, 2011 to Feb 21, 2012. During the summer of 2013/2014, N2O and CH4 fluxes were simultaneously measured at the western sites (AW1, AW2 and AW3) from Feb 14 to Mar 14, 2014. During the summer of 2014/2015, their fluxes were measured at the sites (AW1, AW2, AW3 and GW1, GW2, GW3) from Dec 1, 2014 to Feb 21, 2015.
Analysis of N2O and CH4 concentrations and calculation of flux
The methods of analyzing N2O and CH4 concentrations and flux calculation were described in detail in our previous papers12,15. In brief, gas samples were analyzed using gas chromatography (GC-HP5890 II, USA; Shimadzu GC-14B, Japan; Shimadzu GC-12A, Japan) to measure N2O and CH4 concentrations. Their emission fluxes were calculated by fitting the experimental data to a linear least squares plot (N2O and CH4 concentrations vs. time). More information is given in Supplementary Materials S2.
Measurements of environmental variables and soil properties
Soil temperatures (ST0, ST5 and ST10) were measured in situ using a ground thermometer inserted into the corresponding depth at the sampling sites. Meteorological data, e.g. air temperature (AT), daily sunlight time (ST), precipitation and total daily radiation (TDR) were acquired at the weather station of Great Wall Station. Soil samples were collected in the chamber plots after the fieldwork was completed in the summers of 2011/2012 and 2014/2015. The soils were sampled using a PVC tube (height: 15 cm; diameter: 6 cm), which was sealed and stored at 4 °C until analysis. Soil moisture was determined by oven drying at 105 °C to a constant weight. Each soil sample was homogenized manually and a subsample (fresh weight: 10 g) was extracted with 100 mL of 1 M KCl for 1 h and then filtered and analyzed for NH4+-N and NO3−-N, which were determined using a colorimetric method based on Berthelot’s reaction and ion chromatography14,15. The TOC content in the soils was determined by the chemical volumetric method12 and and TN was analyzed using automatic elemental analysis (Elementar Vario EL, Hanau, Germany). The pH was determined after a 1:3 (soil:solution) dilution of soil with distilled water15.
Statistical analysis
The standard error (SE) was used to estimate the uncertainty of the mean of individual fluxes. All the data for N2O and CH4 fluxes were expressed as the mean ± SE. Differences in N2O fluxes or CH4 fluxes under different UV radiation intensities were examined using one-way repeated ANOVA and LSD tests at the P = 0.05 level. The relationships between soil parameters and N2O and CH4 fluxes were addressed using Pearson correlation analysis (P = 0.05 level). The contribution of the reduction in UV radiation to tundra N2O or CH4 fluxes was calculated as: CT0.03 = MF0.03-MFcon and CT0.06 = MF0.06-MFcon. CT0.03 and CT0.06 indicate the contribution of the 20% and 50% reduction in UV radiation to tundra N2O or CH4 fluxes, respectively. MF0.03, MF0.06 and MFcon indicate the mean N2O or CH4 fluxes under the 20% and 50% reduction in UV radiation and under the control at the ambient UV level, respectively. All statistical analyses were performed using SPSS 20.0 (http://www.spss.com.cn/) and Microsoft Excel 2016 (https://products.office.com/zh-cn/excel) for Windows 10.
References
IPCC. IPCC Climate Change 2013: The Physical Science Basis. Cambridge University Press, Cambridge, United Kingdom and New York, NY, USA (2013).
Oechel, W. C. et al. Acclimation of ecosystem CO2 exchange in the Alaskan Arctic in response to decadal climate warming. Nature 406, 978–981 (2000).
Corradi, C., Kolle, O., Walter, K., Zimov, S. A. & Schulze, E.-D. Carbon dioxide and methane exchange of a north-east Siberian tussock tundra. Glob. Change Biol. 11, 1910–1925 (2005).
Repo, M. E. et al. Large N2O emissions from cryoturbated peat soil in tundra. Nat. Geosci. 2, 189–192 (2009).
Marushchak, M. E. et al. Hot spots for nitrous oxide emissions found in different types of permafrost peatlands. Glob. Change Biol. 17, 2601–2614 (2011).
Ullah, S. & Moore, T. R. Biogeochemical controls on methane, nitrous oxide and carbon dioxide fluxes from deciduous forest soils in eastern Canada. J. Geophys. Res. 116, G03010 (2011).
Kirschke, S. et al. Three decades of global methane sources and sinks. Nat. Geosci. 6, 813–823 (2013).
Burkins, M. B., Virginia, R. A. & Wall, D. H. Organic carbon cycling in Taylor Valley, Antarctica: quantifying soil reservoir and soil respiration. Glob. Change Biol. 7, 113–125 (2001).
Gregorich, E. G. et al. Emissions of CO2, CH4 and N2O from lakeshore soils in an Antarctic dry valleys. Soil Biol. Biochem. 38, 3120–3129 (2006).
Barret, J. E., Virginia, R. A., Parson, A. N. & Wall, D. H. Soil carbon turnover in the McMurdo Dry Valleys, Antarctica. Soil Biol. Biochem. 38, 3019–3034 (2006).
Ball, B. A. & Virginia, R. A. Controls on diel soil CO2 flux across moisture gradients in a polar desert. Antarct. Sci. 27, 527–534 (2015).
Sun, L. G., Zhu, R. B., Xie, Z. Q. & Xing, G. X. Emissions of nitrous oxide and methane from Antarctic tundra: role of penguin dropping deposition. Atmos. Environ. 36, 4977–4982 (2002).
Convey, P. & Smith, R. I. L. Response of terrestrial Antarctic ecosystems to climate change. Plant Ecol. 182, 1–10 (2006).
Zhu, R. B., Liu, Y. S., Xu, H., Ma, D. W. & Jiang, S. Marine animals significantly increase tundra N2O and CH4 emissions in maritime Antarctica. J. Geophys. Res. 118, 1773–1792 (2013).
Zhu, R. B., Ma, D. W. & Xu, H. Summertime N2O, CH4 and CO2 exchanges from a tundra marsh and an upland tundra in maritime Antarctica. Atmos. Environ. 83, 269–281 (2014).
Zhu, R. B., Sun, L. G. & Ding, W. X. Nitrous oxide emissions from tundra soil and snowpack in the maritime Antarctic. Chemosphere 59, 1667–1675 (2005).
Bokhorst, S., Huiskes, A., Convey, P. & Aerts, R. Climate change effects on organic matter decomposition rates in ecosystems from the Maritime Antarctic and Falkland Islands. Glob. Change Biol. 13, 2642–2653 (2007).
Hopkins, D. W. et al. Carbon, nitrogen and temperature controls on microbial activity in soils from an Antarctic dry valley. Soil Biol. Biochem. 38, 3130–3140 (2006).
Carvalho, J. V. S. et al. Impact of expected global warming on C mineralization in maritime Antarctic soils: results of laboratory experiments. Antarct. Sci. 22, 485–493 (2010).
Thompson, D. W. et al. Signatures of the Antarctic ozone hole in Southern Hemisphere surface climate change. Nat. Geosci. 4, 741–749 (2011).
Vosjan, J. H., Döhler, G. & Nieuwland, G. Effect of UV-B irradiance on the ATP content of microorganisms of the Weddell Sea (Antarctica). NL. J. Sea Res. 25, 391–393 (1990).
Pakulski, J. D., Kase, J. P., Meador, J. A. & Jeffrey, W. H. Effect of stratospheric ozone depletion and enhanced ultraviolet radiation on marine bacteria at Palmer Station, Antarctica in the early austral spring. Photochem. Photobiol. 84, 215–221 (2008).
Malloy, K. D., Holman, M. A., Mitchell, D. & Detrich, H. W. Solar UVB-induced DNA damage and photoenzymatic DNA repair in Antarctic zooplankton. PNAS. 94, 1258–1263 (1997).
Ban, S. et al. Effect of solar ultraviolet radiation on survival of krill larvae and copepods in Antarctic Ocean. Polar Biol. 30, 1295–1302 (2007).
Stewart, K. J., Brummell, M. E., Farrell, R. E. & Siciliano, S. D. N2O flux from plant-soil systems in polar deserts switch between sources and sinks under different light conditions. Soil Biol. Biochem. 48, 69–77 (2012).
Krywult, M., Smykla, J. & Wincenciak, A. The presence of nitrates and the impact of ultraviolet radiation as factors that determine nitrate reductase activity and nitrogen concentrations in Deschampsia antarctica Desv. around penguin rookeries on King George Island, Maritime Antarctica. Water Air Soil Poll. 224, 1563 (2013).
Bornman, J. F. et al. Solar ultraviolet radiation and ozone depletion-driven climate change: effects on terrestrial ecosystems. Photoch. Photobio. Sci. 14, 88–107 (2015).
Green, T. G. A., Kulle, D., Pannewitz, S., Sancho, L. G. & Schroeter, B. UV-A protection in mosses growing in continental Antarctica. Polar Biol. 28, 822–827 (2005).
He, Y. et al. Effect of enhanced UV-B radiation on methane emission in a paddy field and rice root exudation of low-molecular-weight organic acids. Photoch. Photobio. Sci. 15, 735–743 (2016).
Li, F., Zhu, R., Bao, T., Wang, Q. & Xu, H. Sunlight stimulates methane uptake and nitrous oxide emission from the High Arctic tundra. Sci. Total Environ. 572, 1150–1160 (2016).
Steinbrecht, W. et al. Ozone and temperature trends in the upper stratosphere at five stations of the Network for the Detection of Atmospheric Composition Change. Int. J. Remote Sens. 30, 3875–3886 (2009).
Bais, A. F., Tourpali, K. & Kazantzidis, A. Projections of UV radiation changes in the 21st century: impact of ozone recovery and cloud effects. Atmos. Chem. Phys. 11, 7533–7545 (2011).
Hakata, M., Takahashi, M., Zumft, W., Sakamoto, A. & Morikawa, H. Conversion of the nitrate nitrogen and nitrogen dioxide to nitrous oxides in plants. Acta Biotechnol. 23, 249–257 (2003).
Robson, T. M. et al. Reduction of solar UV-B mediates changes in the Sphagnum capitulum microenvironment and the peatland microfungal community. Oecologia 140, 480–490 (2004).
Quaggiotti, S., Trentin, A. R., Vecchia, F. D. & Ghisi, R. Response of maize (Zea mays L.) nitrate reductase to UVB radiation. Plant Sci. 167, 107–116 (2004).
Hu, Z. H. et al. Impacts of enhanced UV-B radiation on respiration rate, CH4 and N2O emission fluxes from rice paddy. Environ. Sci. 32, 3018–3022 (2011).
Ding, W., Zhu, R. B., Ma, D. W. & Xu, H. Summertime fluxes of N2O, CH4 and CO2 from the littoral zone of Lake Daming, East Antarctica: effects of environmental conditions. Antarct. Sci. 25, 752–762 (2013).
Del Grosso, S. J. et al. General model for N2O and N2 gas emissions from soils due to denitrification. Global Biogeochem Cycles 14, 1045–1060 (2000).
Xiong, F. S. & Day, T. A. Effect of solar ultraviolet-B radiation during springtime ozone depletion on photosynthesis and biomass production of Antarctic vascular plants. Plant Physiol. 125, 738–751 (2011).
Rozema, J. et al. Consequences of depletion on stratospheric ozone for terrestrial Antarctic ecosystems: the response of Deschampsia antarctica to enhanced UV-B radiation in a controlled environment. Plant Ecol. 154, 101–115 (2001).
Hink, L., Nicol, G. W. & Prosser, J. I. Archaea produce lower yields of N2O than bacteria during aerobic ammonia oxidation in soil. Environ. Microbiol. 19, 4829–4837 (2017).
Bang-Andreasen, T., Schostag, M., Priemé, A., Elberling, B. & Jacobsen, C. S. Potential microbial contamination during sampling of permafrost soil assessed by tracers. Sci. Rep. 7, 43338 (2017).
Johnson, D., Campbell, C. D., Lee, J. A. & Callaghan, T. V. Arctic microorganisms respond more to elevated UV-B radiation than CO2. Nature 416, 82–83 (2002).
Niemi, R. et al. Elevated UV‐B radiation alters fluxes of methane and carbon dioxide in peatland microcosms. Glob. Change Biol. 8, 361–371 (2002).
Erickson, D. J. III, Sulzberger, B., Zepp, R. G. & Austin, A. T. Effects of stratospheric ozone depletion, solar UV radiation and climate change on biogeochemical cycling: Interactions and feedbacks. Photochem. Photobiol. Sci. 14, 127–148 (2015).
Johnson, M. G. et al. Evolution of niche preference in Sphagnum peat mosses. Evolution 69, 90–103 (2015).
Liu, D., Ding, W., Yuan, J., Xiang, J. & Lin, Y. Substrate and/or substrate-driven changes in the abundance of methanogenic archaea cause seasonal variation of methane production potential in species-specific freshwater wetlands. Appl. Microbiol. Biotechnol. 98, 4711–4721 (2014).
Osyczka, P., Dutkiewicz, E. M. & Olech, M. Trace elements concentrations in selected moss and lichen species collected within Antarctic research stations. Pol. J. Ecol. 55, 39 (2007).
Roberts, P., Newsham, K. K., Bardgett, R. D., Farrar, J. F. & Jones, D. L. Vegetation cover regulates the quantity, quality and temporal dynamics of dissolved organic carbon and nitrogen in Antarctic soils. Polar Biol. 32, 999–1008 (2009).
Zhang, Y., Chu, D., Li, Y., Wang, L. & Wu, Y. Effect of elevated UV-B radiation on CH4 emissions from the stands of Spartina alterniflora and Phragmites australis in a coastal salt marsh. Aquat. Bot. 111, 150–156 (2013).
Robson, T., Klem, K., Urban, O. & Jansen, M. A. Re-interpreting plant morphological responses to UV-B radiation. Plant Cell Environ. 38, 856–866 (2015).
Jansen, M. A. & Van Den Noort, R. E. Ultraviolet-B radiation induces complex alterations in stomatal behaviour. Physiol. Plantarum 110, 189–194 (2000).
Von Fischer, J. C. & Hedin, L. O. Controls on soil methane fluxes: tests of biophysical mechanisms using stable isotope tracers. Glob. Biogeochem. Cycles 21, GB2007, https://doi.org/10.1029/2006GB002687 (2007).
Griffith, D. W. T., Toon, G. C., Sen, B., Blavier, J.-F. & Toth, R. A. Vertical profiles of nitrous oxide isotopomer fractionation measured in the stratosphere. Geophys. Res. Lett. 27, 2485–2488 (2000).
Tyler, S. C., Ajie, H. O., Rice, A. L., Cicerone, R. J. & Tuazon, E. C. Experimentally determined kinetic isotope effects in the reaction of CH4 with Cl: Implications for atmospheric CH4. Geophys. Res. Lett. 27, 1715–1718 (2000).
Robson, T. M. et al. Six years of solar UV-B manipulations affect growth of Sphagnum and vascular plants in a Tierra del Fuego peatland. New Phytol. 160, 379–389 (2003).
Acknowledgements
This work was funded in part by the National Natural Sciences Foundation of China (Grant No. 41776190; 41576181). We thank Dr. Chuangneng Lou and other members of Chinese Antarctic Research Expedition for field support in gas sampling and also thank Prof. Shaocai Yu and six anonymous reviewers for their comments.
Author information
Authors and Affiliations
Contributions
R.B.Z., T.B. and H.X. developed the idea and designed the experiments, D.W.M. and T.B. collected the samples from the Antarctica, T.B., P.W., W.J.Y. and D.W.M. analyzed the samples, R.B.Z. and T.B. analyzed the data and wrote the manuscript. All authors have reviewed this manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Bao, T., Zhu, R., Wang, P. et al. Potential effects of ultraviolet radiation reduction on tundra nitrous oxide and methane fluxes in maritime Antarctica. Sci Rep 8, 3716 (2018). https://doi.org/10.1038/s41598-018-21881-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-21881-1
This article is cited by
-
Forest canopy mitigates soil N2O emission during hot moments
npj Climate and Atmospheric Science (2021)
-
Effects of multiple environmental variables on tundra ecosystem respiration in maritime Antarctica
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.