Abstract
Sulfur oxides (SOx), particularly SO2 emitted by coal-fired power plants, produce long-term risks for cardiovascular disease (CVD). We estimated the relative risks of CVD and ischemic heart disease (IHD) attributable to SOx emission globally. National SOx reduction achieved by emissions control systems was defined as the average SOx reduction percentage weighted by generating capacities of individual plants in a country. We analyzed the relative risk of CVD incidence associated with national SOx reduction for 13,581 coal-fired power-generating units in 79 countries. A 10% decrease in SOx emission was associated with 0.28% (males; 95%CI = −0.39%~0.95%) and 1.69% (females; 95%CI = 0.99%~2.38%) lower CVD risk. The effects on IHD were > 2 times stronger among males than females (2.78%, 95%CI = 1.99%~3.57% vs. 1.18%, 95%CI = 0.19%~2.17%). Further, 1.43% (males) and 8.00% (females) of CVD cases were attributable to suboptimal SOx reduction. Thus, enhancing regulations on SOx emission control represents a target for national and international intervention to prevent CVD.
Similar content being viewed by others
Introduction
CVD has been a leading cause of death globally for decades1. Treating CVD is costly, especially in the United States (US). For the US, the burden of medical cost for CVD was 656 billion United States Dollars (USD) in 2015 and is projected to reach 1,208 billion USD in 20302.
Controlling emissions from power-generating plants is important for human health as well as climate. Among the health problems linked to sulfur oxides (SOx) exposures in air is cardiovascular disease (CVD)3,4. Various air pollutants initiate and promote atherosclerotic progression5,6 and are associated with transient increases in plasma viscosity and thrombus formation7. Clear links have been drawn between SO2 and CVD8,9. Indeed, total suspended particles (TSP) and SO2 are associated with changes in vasomotor tone10 and thus alter heart rate11,12 and cardiac function13. Such mechanisms may underlie the association between SO2 and CVD.
Coal-fired power-generating facilities have long been known to emit pollutants that fuel climate change and adversely impact human health. Among these emissions are SOx, including sulfur dioxide (SO2). Global SO2 emissions, measured by the bottom-up mass balance method, peaked in the early 1970s and decreased for decades14. After the 2000s, these emissions increased again, mostly from developing countries15. The majority of SOx in the air is anthropogenic emission from coal-fired power plants16. For example, in the US, 65% of SO2 emission were from electric utilities, and more than 90% of those were coal-fired power plants17. Similarly, in the European Union, more than 70% of the emission was from electricity sectors18.
To control these emissions, dozens of methods with relatively high efficiencies have been developed for fitting of coal-fired power plants. SOx emissions are determined by (1) the sulfur content in coals burned and (2) the emission control system used19. The principle technology of emission control systems is the use of sorbents to scrub SOx from the flue gas, called a flue gas desulfurization (FGD) system. Another method to reduce SOx emission is the use of low sulfur coal, such as sub-bituminous coal mined in the Powder River Basin of Montana and Wyoming19. However, this method is not efficient and has a limited application globally. Indeed, FGD products represent an efficient and economically feasible approach to control emissions on a large scale. The cost to retrofit US plants with FGD equipment was estimated at about $407 (2008 USD) per kilowatt (kW) for a 500-megawatt (MW) plant in 2009; this cost escalates yearly by $1620. For most nations, coal-fired power plants are either state-owned or government-funded, giving governments direct authority on implementing emission controls; privately-owned power-generating units can be required to follow emissions regulations.
Here, we estimated the relative risks and incident cases of CVD, particularly ischemic heart disease (IHD), attributable to SOx emission from coal-fired power plants from a global perspective. This study sought to determine the potential reduction in preventable CVD that could be attributed to reduced global SOx emissions.
Results
Data on the coal capacities of power plants across the globe were derived from the Utility Data Institute World Electric Power Plants Data Base (UDI WEPP)21. We identified a total of 13,581 generating units in 79 countries that used coal as the primary energy source (Table 1). Most were in Europe (N = 36), the Americas (N = 12), and the Western Pacific (N = 11).
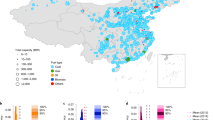
To calculate SOx emission controls, the efficiencies of different SOx reduction control systems in coal-fired power plants were extracted from the literature. Most SOx control systems in the studied countries had relatively high SOx reduction efficiency, by 80% or more (Supplementary Table 1). Data on SOx control technology were only available for larger power plants. As a consequence, 19 countries had no data on control technologies. However, the total capacity of plants with missing control technology data is only 14.15 GW, representing 0.78% of the total coal capacity in the study. We assigned those missing as 0 reduction in the following analysis. We defined national SOx reduction as the average SOx reduction percentage weighted by generating capacities of individual plants in a given country. Total coal capacities and national SOx reduction in included countries in 2012 are summarized in Fig. 1. The lack of installing control systems in small units in many countries produced a bimodal distribution of national SOx reduction, with a median of 58.49% (Supplementary Figure 1).
Total coal capacity (upper panel) and national SOx reduction by country (lower panel) in 2012. The map is created by using R version 3.2.5, Package ‘rworldmap’30.
To determine effects of SOx emission controls on CVD, we extracted age- and sex-adjusted CVD incidence rates from the Global Burden of Disease (GBD) Study22, including two subcategories: ischemic heart diseases (IHD) and rheumatic heart diseases (RHD). The former reflects coronary artery disease, which may have a stronger association with air pollution; the latter is a contagious disease that we used as a falsifying outcome. Table 2 summarizes these and other covariates included in the study. IHD was more common among males, while RHD, accounting for less than 1% of CVD, was more common among females. One behavior risk factor for CVD—smoking prevalence—was almost three times higher among males than females.
We applied a Poisson regression to analyze the relative risk of age-standardized CVD incidence associated with national SOx reduction, adjusted for behavior, economic, and regional factors. A 10% decrease in SOx emission from coal-fired power plants is associated with a 0.75% lower standardized CVD incidence rate [relative risk (RR) = 0.9925, 95% confidence interval (CI) = 0.9892–0.9959], after adjustments (primary model, sex combined, Table 3). The association of SOx reduction was stronger for lower CVD in females (RR = 0.9831, 95% CI = 0.9762–0.9901) than in males (RR = 0.9972, 95% CI = 0.9905–1.0039).
Results of analysis of IHD and RHD as different outcomes are presented in Table 4. Unlike the effects on CVD, the effects of SOx reduction on IHD were stronger in males than in females. A 10% decrease in SOx emission from coal-fired power plants was associated with 0.9722-fold (95% CI = 0.9643–0.9801) lower IHD incidence among males, while females had an analogous association of 0.9882 (95% CI = 0.9783–0.9981). No statistically significant relationships between SOx reduction and RHD incidence rate among either males or females were found.
CVD incident cases attributable to suboptimal emission controls were estimated in all studied countries, assuming every country can reach 95% emission reduction. The fractions of CVD attributable to suboptimal SOx reduction (PAF) were up to 1.43% and 8.00% for males and females, respectively (Supplementary Table 2). Similarly, the PAFs of IHD from suboptimal SOx reduction were up to 13.24% and 5.70% for males and females, respectively. The number of attributable cases varied widely between countries. Take IHD for example, India and China had the highest preventable cases from optimizing SOx reductions in coal-fired power plants, with estimations of 381,843 and 177,756 preventable cases, respectively (Fig. 2).
Discussion
To our knowledge, this is the first study showing the preventable CVD incidence attributable to SOx reductions from coal-fired power plants from a global perspective. We found that 10% reductions in SOx emissions were associated with CVD incidence rates that were 0.28% lower for males and 1.69% lower for females. Up to 13.24% and 5.70% of incident IHD cases are attributable to suboptimal SOx emissions control in coal-fired power plants among males and females, respectively, given a country can reach 95% SOx reduction in the electricity sector. Our falsifying test (see Methods) revealed no relationship between RHD and air pollution, supporting SOx as a risk factor on air pollution related CVD.
Taking SOx reduction in coal-fired power plants as a determinant of CVD incidence was reasonable and adequate from several perspectives: (1) The majority of SO2 emission was from fossil fuel combustion, mostly coal-fired power units. Therefore, using the reduction percentage in coal-fired plants could capture the largest amount of SOx reduction. (2) The implication of national SOx reduction provides an alternative for policy application at the national level. By summarizing a national SOx reduction, policy makers could use the results presented here to help estimate the counterfactual outcome given a country has improved its SOx control system in coal-fired power plants. (3) Our approach provides a direct method to estimate the externality costs from coal-fired power plants, specifically from SOx control systems, by comparing the costs of treatment for CVD attributable to SOx emissions from coal-fired power plants.
Considering the magnitude of estimated costs of CVD, retrofitting FGD equipment in coal-fired power plants could be economically justifiable. Take the US as an example: the national SOx reduction is 82.60% in the US. The US needs to install FGD in a total capacity of 42,093.37 MW (=339462.7 MW*(95%–82.6%)) to reach optimal reduction. Given the the cost of installing FGD at $455 per kW and 30 years lifetime of coal-fired power plants19, the annual cost of SOx emissions control would be $638.42 million, nominal price. In contrast, the estimated cost of CVD is $1,067.96 million (=564.32 billion dollars*(2,756/1,456,342))2,23 under the estimated PAF = 0.0003 for males and 0.002 for females, respectively, in the US (Supplementary Table 2). Yet, for many countries, the situation is more nuanced. For example, China has much higher CVD incidence and PAF than the US, so the health benefits per unit of SOx reduction could be much higher, making FGD installation a cost-effective strategy to improve public health. Moreover, developing countries usually have relatively low SOx reduction rates, such as in the cases of China (59.44%) and India (44.45%). Marginal costs of FGD might rise, while marginal benefits might decrease, when these countries increase their SOx reduction rates. It is possible to find an efficient level of SOx reduction rates (below 95%) when the marginal costs equal marginal benefits. The above examples illustrate the applications of SOx reduction rate and PAF as helpful analytical tools to illuminate policy-making in public health and SOx emissions control.
The log-linear model also provides an interpretation of elasticity. For example, the elasticity of IHD on demanding SOx emission control systems is 0.07 (=ln(0.9722) × 2.5) and 0.03 (=ln(0.9882) × 2.5) among males and females, respectively, given the national SOx reduction is 25% in a given country ceteris paribus (Table 4). This implies the change of IHD is more sensitive to SOx reduction among males than females. Similarly, the elasticity is 0.21 (=ln(0.9722) × 7.5) and 0.09 (=ln(0.9882) × 7.5) among males and females, respectively, given the national SOx reduction is 75% in a given country ceteris paribus. The elasticity becomes larger when SOx reduction improves, which means the incidence of IHD would be even more sensitive to additional improvement of the emission controls for countries having already had better SOx control systems in coal-fired power plants.
Several limitations or concerns should also be addressed. (1) The cross-sectional study did not provide a temporal interpretation of the causal effect of SOx reduction on CVD prevention. However, since the national SOx reduction in 2012 remained relatively constant compared to our 2016 data, it could be deemed as a marker for what has happened over many previous years. (2) Despite using an ecological study design, the potential for “ecological fallacy”24 is unlikely because our analysis on aggregated data is meant to inform policy decisions at the national level and for international comparison, but not at the individual level25. (3) This approach can be regarded as conservative in the sense that some of these plants may have actually reduced emissions more than our approach recognizes, implying that our approach actually underestimates the association between SOx and CVD/IHD. Countries with national SOx controls equal to 0 had lower CVD incidence rates (631 vs. 960 cases per hundred thousand males, on average). However, note that plants with missing control data amount to less than 1% of the total global coal capacity, so different assumptions about these missing data are not expected to have a meaningful impact on the analysis. (4) The study did not adjust for meteorological, geographical and/or other covariates26. If we assume the lack of considering meteorological effect misclassified our exposure, we might underestimate the true effect as well. Other covariates, such as socioeconomic status has an impact on cardiovascular disease at individual27 and national levels28. Also, we’ve adjusted per capita GDP(PPP) and the geographic region as proxy indicators of healthcare expenditure and living standard. However, similar to previous report29, we didn’t find any statistically significant relationship between coal capacity and socioeconomic status at the national level. (5) It is noteworthy that even though the study does not explicitly calculate the effects of detailed secondary formation and/or byproduct of SO2, retrofitting SOx control system reduces both SO2 and its secondary products. It is the cumulative effect that is of interest in this study. We did not consider the influence of seasonal differences on SOx emission, either. Instead of the ambient SOx levels, we focused on the reduction (control) effect of SOx emission (%) from coal-fired power plants, which is assumably constant across seasons. (6) The outcome data were obtained and interpolated from the GBD estimation22. Although we acknowledged the possible inconsistency of over- or under-reports from the global incidence data, GBD estimation provided the most thorough CVD incidences that we could access for a better international comparison.
Conclusion
In conclusion, CVD is a common, costly, and often fatal condition. Improvement in SOx controls in coal-fired power plants has a marked association with lower incidence of CVD and IHD. Although the causality and biological mechanisms need further exploration, SOx emission is a pervasive public health issue with major cardiovascular and healthcare economic consequences. Since SOx emission is primarily from coal combustion, regulations on SOx emission do present a key target for national and international intervention.
Methods
Data source
A total of 79 countries with data available for analyses were included in 2012. We obtained the age- and sex-adjusted CVD incidence rates from the Global Burden of Disease (GBD) Study22. CVD data included two subcategories, ischemic heart diseases (IHD) and rheumatic heart diseases (RHD). The former was coded as 410–410.9, 411–411.1, 411.8–411.9 in the International Classification of Diseases version 9 (ICD-9) and I21.0–I21.4, I21.9, I22.0–I22.2, and I22.8–I22.9 in ICD-10. The latter was coded as 391.0–391.2, 391.8–391.9, 392.0, 394.0–394.2, 394.9, 395.0–395.2, 395.9, 396.0–396.3, 396.8–396.9, 397.0–397.1, 397.9, 398.8–398.9 in ICD-9 and I01.0–I01.2, I01.8–I01.9, I02.0, I05.0–I05.2, I05.8–I05.9, I06.0–I06.2, I06.8–I06.9, I07.0–I07.2, I07.8–I07.9, I08.0–I08.3, I08.8–I08.9, I09.0–I09.2, I09.8–I09.9 in ICD-1022. The GBD study has thorough estimation of incidence rates of CVD in 2010 and 2015, respectively. For CVD incidence rates data between 2011 and 2014, we estimated using linear interpolation.
Coal capacity is defined as the generating capacity of a coal-fired power plant [unit: megawatt (MW)]. The estimation for reduction of each unit is based on the representative SOx reduction percentage of the corresponding control technology from literature review, summarized in Supplementary Table 1. National SOx reduction is the coal-capacity-weighted average SOx reduction in a given country. The formula is written as follows:
where i is a coal-fired power unit and n is the total units in a country. Coal capacities are the weight for the reductions in different power units. Missing data on SOx control are assigned 0. Data on the coal capacity of every plant were derived from the Utility Data Institute World Electric Power Plants Data Base (UDI WEPP)21.
We also collected data exclusively on both behavior and economic covariates at the national level, including smoking prevalence, economy, traffic index, and macroeconomic indicators, and industrialization. Annual smoking prevalence within each country was estimated and sex- and age-adjusted31. The macro level indicator was the annual per capita gross domestic product adjusted for purchasing power parity [GDP (PPP)] and inflation to the base year 2011 to capture a country’s standard of living level32. Traffic index, measured as the proportion of a country’s population living in urban areas, was applied to capture air pollutants emitted from all mechanical vehicles and public transports associated with human activities33. The industrialization level was measured using the shares of CO2 emissions from manufacturing industries and construction in total CO2 emissions (% of total fuel combustion)34. We further grouped studied countries into 6 WHO regions (combination of geographical distribution and mortality): Africa, the Americas, Southeast Asia, Europe, Eastern Mediterranean, and the Western Pacific35.
Data analysis
We took a natural logarithm of coal capacity to approximate normal distribution in the model. A Poisson regression was performed for count data of incidence cases of diseases. Our primary model is as following:
where i denotes each country; ln(E[λ i ]) denotes the natural log of expected standardized incident rates for CVD conditioned on covariates X i ; \({\beta }_{0}\) is the intercept; \({\beta }_{1}to\,{\beta }_{6}\) are coefficients of individual covariates; and I_Region is indicator variable for the six WHO regions to consider the underlying difference in hygiene and healthcare status.
In addition to the above primary model, four other models were specified to assess sensitivity to the inclusion of different adjustment covariates. They were: (1) Univariate model with SOx reduction only; (2) Behavior-adjusted model with smoking prevalence and healthcare index of per capita GDP (PPP); (3) Economic-adjusted model with per capita GDP (PPP), traffic index, and industrialization; and (4) non-regional model with combination of behavior and economics. All models were analyzed for both sexes combined, males, and females, respectively, and weighted by nationwide sex-specific population33 for all multivariate models.
Under the assumption that every country could hypothetically reach an optimal national SOx reduction by a factor of 95%, we estimated the proportional attributable factor (PAF) for IHD for every country. The formula for PAF is written as follows36:
where P i is the proportion of people exposed to suboptimal SOx reduction. In the estimation, we applied WHO mortality strata and assumed the P i is 0.1 in strata A countries and 0.5 in stratum B to E countries37, respectively, as often used in other studies38,39,40. \(R{R}_{i}\) is the relative risks from the primary model, comparing existing national SOx reduction in 2012 vs. the counterfactual optimal reduction (95%). Supplementary Table 2 shows the step-by-step calculation for PAFs. The incident cases of IHD attributable to SOx controls in coal-fired power plants were estimated by multiplying the standardized incidence rates by sex-specific population and PAF.
We performed the PROC GENMOD procedure with a log link function, using SAS version 9·4 (SAS Institute, Cary, NC, US) to estimate the effect of selected factors on standardized incidences of CVD, IHD, and RHD, respectively.
Additional analysis and falsification test
To investigate the possibility that general health improvements correlated with SOx reduction in coal-fired power plants might be obscuring our CVD results, we further analyzed two subcategories of CVD: IHD and RHD. Since the latter is related to previously unsatisfactorily-treated streptococcus infection, we identified RHD as a falsification outcome that might be a marker that are not expected to bear any relationship to air pollution. We applied the primary model for IHDs and rheumatic heart diseases, respectively, as the additional analysis and examined whether the relationship between RHD and SOx reduction existed as a falsification test.
References
World Health Organization. The top 10 causes of death, http://www.who.int/mediacentre/factsheets/fs310/en/ (2017).
Mozaffarian, D. et al. Heart disease and stroke statistics–2015 update: a report from the American Heart Association. Circulation 131, e29–322, https://doi.org/10.1161/CIR.0000000000000152 (2015).
Peters, A., Dockery, D. W., Muller, J. E. & Mittleman, M. A. Increased particulate air pollution and the triggering of myocardial infarction. Circulation 103, 2810–2815 (2001).
Samet, J. M., Dominici, F., Curriero, F. C., Coursac, I. & Zeger, S. L. Fine particulate air pollution and mortality in 20 U.S. cities, 1987-1994. N Engl J Med 343, 1742–1749, https://doi.org/10.1056/NEJM200012143432401 (2000).
Suwa, T. et al. Particulate air pollution induces progression of atherosclerosis. J Am Coll Cardiol 39, 935–942 (2002).
Penn, A. & Snyder, C. A. 1,3 Butadiene, a vapor phase component of environmental tobacco smoke, accelerates arteriosclerotic plaque development. Circulation 93, 552–557 (1996).
Peters, A., Doring, A., Wichmann, H. E. & Koenig, W. Increased plasma viscosity during an air pollution episode: a link to mortality? Lancet 349, 1582–1587, https://doi.org/10.1016/S0140-6736(97)01211-7 (1997).
Shah, A. S. et al. Global association of air pollution and heart failure: a systematic review and meta-analysis. Lancet 382, 1039–1048, https://doi.org/10.1016/S0140-6736(13)60898-3 (2013).
Hoek, G., Brunekreef, B., Fischer, P. & van Wijnen, J. The association between air pollution and heart failure, arrhythmia, embolism, thrombosis, and other cardiovascular causes of death in a time series study. Epidemiology 12, 355–357 (2001).
Harrabi, I., Rondeau, V., Dartigues, J. F., Tessier, J. F. & Filleul, L. Effects of particulate air pollution on systolic blood pressure: A population-based approach. Environ Res 101, 89–93, https://doi.org/10.1016/j.envres.2006.01.012 (2006).
Gold, D. R. et al. Ambient pollution and heart rate variability. Circulation 101, 1267–1273 (2000).
Pope, C. A. III et al. Heart rate variability associated with particulate air pollution. Am Heart J 138, 890–899 (1999).
Van Hee, V. C. et al. Exposure to traffic and left ventricular mass and function: the Multi-Ethnic Study of Atherosclerosis. Am J Respir Crit Care Med 179, 827–834, https://doi.org/10.1164/rccm.200808-1344OC (2009).
Smith, S. J. J. v. A., Z. Klimont, R. J. Andres, A. Volke, S. Delgado Arias. Anthropogenic sulfur dioxide emissions: 1850–2005. Atmos. Chem. Phys. 11, 6 (2011).
Jiang, J., Zha, Y., Gao, J. & Jiang, J. Monitoring of SO2 column concentration change over China from Aura OMI data. International Journal of Remote Sensing 33, 1934–1942, https://doi.org/10.1080/01431161.2011.603380 (2012).
Australian Government Department of the Environment and Energy. Sulfur dioxide (SO2), https://www.environment.gov.au/protection/publications/factsheet-sulfur-dioxide-so2 (2005).
U.S. Environmental Protection Agency. 2011 National Emissions Inventory, version 2 Technical Support Document, https://www.epa.gov/sites/production/files/2015-10/documents/nei2011v2_tsd_14aug2015.pdf (2015).
European Environment Agency. Sulphur dioxide (SO2) emissions, https://www.eea.europa.eu/data-and-maps/indicators/eea-32-sulphur-dioxide-so2-emissions-1/assessment-3 (2014).
U.S. Environmental Protection Agency. Available and Emerging Technologies for Reducing Greenhouse Gas Emissions from the Portland Cement Industry https://www.epa.gov/sites/production/files/2015-12/documents/cement.pdf (2010).
Cichanowicz, J. E. Current Capital Cost And Cost-Effectiveness of Power Plant Emissions Control Technologies (2010).
UDI World Electric Power Plants Database (WEPP). World Electric Power Plants Database, http://www.platts.com/products/world-electric-power-plants-database (2016).
Global Burden of Disease Study. Incidence, Prevalence, and Years Lived with Disability 1990-2015, http://ghdx.healthdata.org/gbd-results-tool (2015).
Lloyd-Jones, D. et al. Executive summary: heart disease and stroke statistics–2010 update: a report from the American Heart Association. Circulation 121, 948–954, https://doi.org/10.1161/CIRCULATIONAHA.109.192666 (2010).
Robinson, W. S. Ecological correlations and the behavior of individuals. Int J Epidemiol 38, 337–341, https://doi.org/10.1093/ije/dyn357 (2009).
Idrovo, A. J. Three criteria for ecological fallacy. Environ Health Perspect 119, A332, https://doi.org/10.1289/ehp.1103768 (2011).
Lin, C. K., Hung, H. Y., Christiani, D. C., Forastiere, F. & Lin, R. T. Lung cancer mortality of residents living near petrochemical industrial complexes: a meta-analysis. Environ Health 16, 101, https://doi.org/10.1186/s12940-017-0309-2 (2017).
Clark, A. M., DesMeules, M., Luo, W., Duncan, A. S. & Wielgosz, A. Socioeconomic status and cardiovascular disease: risks and implications for care. Nat Rev Cardiol 6, 712–722, https://doi.org/10.1038/nrcardio.2009.163 (2009).
Mackenbach, J. P., Cavelaars, A. E., Kunst, A. E. & Groenhof, F. Socioeconomic inequalities in cardiovascular disease mortality; an international study. Eur Heart J 21, 1141–1151, https://doi.org/10.1053/euhj.1999.1990 (2000).
US Energy Informative Administration. International Energy Outlook 2017. (US Energy Informative Administration, 2017).
R Core Team. R: A Language and Environment for Statistical Computing, https://www.r-project.org/ (2017).
Ng, M. et al. Smoking prevalence and cigarette consumption in 187 countries, 1980-2012. JAMA 311, 183–192, https://doi.org/10.1001/jama.2013.284692 (2014).
Feenstra, R., Robert I, Marcel PT. Penn World Table version 9.0, http://www.ggdc.net/pwt (2015).
United Nations. World Population Prospects https://esa.un.org/unpd/wpp/Download/Standard/Population/ (2017).
The World Bank. World Development Indicators, http://data.worldbank.org/data-catalog/world-development-indicators (2016).
World Health organization. Health statistics and information systems, http://www.who.int/healthinfo/global_burden_disease/definition_regions/en/ (2017).
Levin, M. L. The occurrence of lung cancer in man. Acta Unio Int Contra Cancrum 9, 531–541 (1953).
World Health organization. List of Member States by WHO region and mortality stratum, http://www.who.int/whr/2003/en/member_states_182-184_en.pdf (2013).
Lee, L. J., Lin, C. K., Hung, M. C. & Wang, J. D. Impact of work-related cancers in Taiwan-Estimation with QALY (quality-adjusted life year) and healthcare costs. Prev Med Rep 4, 87–93, https://doi.org/10.1016/j.pmedr.2016.05.015 (2016).
Driscoll, T. et al. The global burden of disease due to occupational carcinogens. Am J Ind Med 48, 419–431, https://doi.org/10.1002/ajim.20209 (2005).
Hewett, P. Interpretation and use of occupational exposure limits for chronic disease agents. Occup Med 11, 561–590 (1996).
Acknowledgements
We acknowledge the support of Graduate Consortium, Harvard University Center for the Environmental Health. We are also sincerely grateful to Prof. Henry Lee, Harvard Kennedy School for his important input at the initial stage of study design.
Author information
Authors and Affiliations
Contributions
C.K.L. contributed to study design, data analysis, reporting results, data interpretation, and writing of the manuscript. C.Z. contributed to study design, data interpretation, and preparing the manuscript. R.T.L., P.C.C. and P.W. contributed to data interpretation, and writing of the manuscript. D.C.C. and N.M. contributed to data interpretation and preparing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lin, CK., Lin, RT., Chen, PC. et al. A Global Perspective on Sulfur Oxide Controls in Coal-Fired Power Plants and Cardiovascular Disease. Sci Rep 8, 2611 (2018). https://doi.org/10.1038/s41598-018-20404-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-20404-2
This article is cited by
-
Air pollution modeling to support strategic environmental assessment: case study—National Emission Reduction Plan for coal-fired thermal power plants in Serbia
Environment, Development and Sustainability (2023)
-
Annual and Periodic Variations of Particulates and Selected Gaseous Pollutants in Astana, Kazakhstan: Source Identification via Conditional Bivariate Probability Function
Aerosol Science and Engineering (2023)
-
Association of ambient air pollution with cardiovascular disease risks in people with type 2 diabetes: a Bayesian spatial survival analysis
Environmental Health (2020)
-
A Selective Review of Negative Control Methods in Epidemiology
Current Epidemiology Reports (2020)
-
A global perspective on coal-fired power plants and burden of lung cancer
Environmental Health (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.