Abstract
Hand, foot and mouth disease (HFMD) is a substantial burden throughout Asia, but the effects of temperature pattern on HFMD risk are inconsistent. To quantify the effect of temperature on HFMD incidence, Wuhan was chosen as the study site because of its high temperature variability and high HFMD incidence. Daily series of HFMD counts and meteorological variables during 2010–2015 were obtained. Distributed lag non-linear models were applied to characterize the temperature-HFMD relationship and to assess its variability across different ages, genders, and types of child care. Totally, 80,219 patients of 0–5 years experienced HFMD in 2010–2015 in Wuhan. The cumulative relative risk of HFMD increased linearly with temperature over 7 days (lag0–7), while it presented as an approximately inverted V-shape over 14 days (lag0–14). The cumulative relative risk at lag0–14 peaked at 26.4 °C with value of 2.78 (95%CI: 2.08–3.72) compared with the 5th percentile temperature (1.7 °C). Subgroup analyses revealed that children attended daycare were more vulnerable to temperature variation than those cared for at home. This study suggests that public health actions should take into consideration local weather conditions and demographic characteristics.
Similar content being viewed by others
Introduction
Hand, foot and mouth disease (HFMD) is an emerging infectious disease that is caused by several non-polio enteroviruses1. In China, HFMD is the most frequent infectious disease affected children under 5 years old2. Most HFMD cases have self-limiting symptoms, including fever, oral ulcers and maculopapular rashes or vesicular sores on the hands, feet, or buttocks3. However, a small portion of HFMD patients can develop serious health consequences, even death, due to complications from aseptic meningitis, encephalitis, acute flaccid paralysis, and neurogenic pulmonary edema4. Many studies on the epidemiology of HFMD have observed seasonality of HFMD incidence5,6. Furthermore, meteorological factors, particularly daily average temperature, have been reported to affect on the incidence of HFMD in recent studies.
Temperature as a key meteorological factor has been reported to be associated with HFMD occurrence in many countries. However, findings are inconsistent among studies. Several studies have demonstrated that the incidence of HFMD increased with rising temperature7,8, but a negative correlation has also been documented9. Several studies have reported non-linear effect of temperature on HFMD incidence and have found a cut-off point at 25.0–27.5 °C for the association between temperature and the risk of HFMD incidence10,11. In fact, different patterns of HFMD incidence have been found in studied areas with different climate conditions. For example, there is only one annual peak in summer in North China, while there are two peaks, in early summer and autumn, in South China. Since temperature has a similar effect on enterovirus-associated HFMD, one question then arises: why are there different relationships between temperature and incidence of HFMD even within similar populations. The possible effect of temperature on HFMD incidence may be determined by a study in an area with high temperature variability and high incidence of HFMD. Wuhan is an ideal area for such study.
Wuhan is the capital of Hubei Province and the largest city in Central China with a population of over ten million. It is also the site of serious HFMD outbreaks and has over ten thousand HFMD cases reported each year. In addition, the weather in Wuhan is characterized by a hot and wet summer and a cold and humid winter. The summer in Wuhan is similar to that in South China, while the winter is similar to that in North China. The uniqueness of Wuhan’s climate is particularly suitable for studying the association between temperature and HFMD incidence. The aim of the present study was to quantify the effect of temperature on HFMD occurrence in Wuhan, and to assess whether this association varies by age, gender, and type of child care (children cared for at home, children attended daycare). A time-series analysis using surveillance data in Wuhan, China, was performed to identify the association of daily HFMD counts with temperature, which will assist in public health prevention and control measures.
Results
Data description
A total of 80,219 HFMD cases among patients aged 0–5 years were reported in Wuhan from 1 January 2010 to 31 December 2015. Only 7,280 cases (9.07%) were lab-confirmed. In this period, HFMD occurred more commonly in males with a male-to-female ratio of 1.67:1. The proportions of each age group were 14.4% (11,586 cases), 47.5% (38,088 cases) and 38.1% (30,545 cases) in children under 1 year old, aged 1–2 years and aged 3–5 years, respectively. There were 46,117 children (57.5%) cared for at home and 34,102 children (42.5%) who attended daycare. A descriptive summary of daily HFMD cases and meteorological variables is provided in Table 1. The mean values of average temperature and humidity were 16.7 °C and 78.7%, respectively. The time-series distribution of daily HFMD cases and meteorological variables in Wuhan from 2010 to 2015 revealed seasonality in HFMD occurrence (Supplementary Fig. S2). Summer peaks in the number of HFMD cases were observed in April-July, with secondary peaks occurring in November-December.
Relationship between daily average temperature and HFMD occurrence at different lags
Figure 1 shows the effect of average temperature at different percentiles (25th, 50th, 75th and 95th) on HFMD counts, compared with its 5th percentile, at different lag days. The average temperature of the four above mentioned percentiles demonstrated a similar pattern of association with HFMD incidence. An immediate increase in relative risk (RR) at lag0 (the current day) was followed by a decrease at lag1 (the previous day) and lag2 (the previous two day). Subsequently, the RRs exhibited an inverted V-shape at lag2-lag14, with a peak at lag5. The RR values at lag5 of different percentiles (25th, 50th, 75th and 95th) of temperature on HFMD incidence were 1.07 (95%CI: 1.03–1.12), 1.19 (95%CI: 1.13–1.26), 1.31 (95%CI: 1.23–1.39), and 1.34 (95%CI: 1.24–1.45), respectively.
The relative risks of different average temperature for HFMD cases at different lags. The 5th percentile values of average temperature (1.7 °C) were defined as the reference for calculating relative risk. The temperature 8.6 °C, 17.8 °C, 24.7 °C and 30.8 °C represented the 25th percentile, 50th percentile, 75th percentile and 95th percentile of average temperature, respectively.
Cumulative RRs of average temperature on HFMD occurrence stratified by age, gender and type of child care
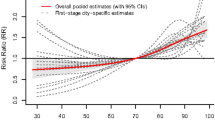
Figure 2 illustrates the cumulative RRs, aggregating all contributions up to 7 days of average temperature on HFMD incidence in different subgroups. The exposure-response relationship was shown to be linear in all subgroups, except for children who attended daycare. A positive association between average temperature and HFMD incidence was observed, with the cumulative RR for 1 °C rise being 4.51% (95CI: 3.12–6.30%) at lag0–7. A 1 °C increase in temperature elevated the risk of HFMD incidence by 5.16% (95%CI: 3.55–7.27%) and 3.49% (95%CI: 2.10–5.38%) in male and female subgroups. A 1 °C increase in temperature led to 8.74% (95%CI: 4.95–14.73%), 3.42% (95%CI: 2.21–5.03%), and 5.56% (95%CI: 3.39–8.67%) increases in HFMD incidence among children aged <1 year, 1-2 years, and 3–5 years, respectively. Each 1 °C increase in temperature was associated an increase of 3.74% (95%CI: 2.44–5.44%) in HFMD incidence among children cared for at home. For children who attended daycare, the cumulative RR increased with average temperature up to 27.5 °C and then began to decrease.
Figure 3 describes the cumulative RRs over 14 days of average temperature on HFMD incidence in different subgroups. The exposure-response relationship presented an approximately inverted V-shape. The cumulative RR reached a peak at 26.4 °C with a value of 2.78 (95%CI: 2.08–3.72). The overall effect of average temperature on HFMD occurrence in both males and females showed similar trends with respect to the entire study population. And it also presented to be an inverted V-shape, with different peak values for children in different age groups, i.e., 4.10 (95%CI: 2.35–7.16) at 27.9 °C for children younger than 12 months; 2.73 (95%CI: 2.00–3.71) at 26.7 °C for children aged 1-2 years old and 3.43 (95%CI: 2.30–5.13) at 25.9 °C for children aged 3-5 years old. The association between cumulative exposure to average temperature with HFMD incidence was approximately linear in children cared for at home, while it presented an inverted V-shape in children who attended daycare, with a peak cumulative RR of 4.22 (95%CI: 2.82–6.31) at 25.6 °C.
The cumulative RRs of different average temperatures on HFMD incidence along the lags in different subgroups are summarized in Table 2. Average temperature had higher cumulative RR on HFMD occurrence in children who attended daycare than that in children cared for at home (both P < 0.001 at lag0-7 and lag0–14). The differences of cumulative RRs of temperature on HFMD incidence between male and female were not significant (P = 0.977 at lag0–7 and P = 0.978 at lag0–14). Insignificant differences of cumulative RRs of temperature on HFMD incidence were observed in different age groups (P = 0.150 at lag0–7 and P = 0.289 at lag0–14).
Sensitivity analyses
In the sensitivity analyses, when changing df (3–6) for temperature and humidity, we found that the patterns of temperature’s effect on HFMD incidence at different lags (Supplementary Fig. S3), cumulative RRs over 7 days and 14 days did not vary substantially (Supplementary Fig. S4 and Supplementary Fig. S5, respectively). When the maximum lag was set to different values (13–21 days), the effect of temperature on HFMD incidence presented similar patterns (Supplementary Figs. S6–S8).
Discussion
This study provides evidence of association between daily average temperature and HFMD occurrence among children aged 0–5 years using population-wide monitoring data in Wuhan, China. After adjusting other climate variables, the relationship was found to be linear between cumulative exposure to average temperature over 7 days and HFMD occurrence, while it appeared to be an inverted V-shape when cumulative exposure to average temperature amounted to 14 days, with a peak in cumulative RR at 26.4 °C observed for the entire study population. Children who attended daycare were more sensitive to temperature variation than those cared for at home. The cumulative relative risks of temperature on HFMD incidence were not found to be significantly different in different age and gender groups. The results of this study may be informative in forecasting HFMD risk for children with different types of child care based on daily average temperature change.
Temperature influences the survival and spread of infectious pathogens in the environment, as well as the behavior and activities of the population, thereby influencing the dynamics of infection transmission12,13. Several laboratory-based studies have shown that the stability of enteric viruses is influenced by temperature and relative humidity14,15. Warm seasons may extend virus survival and improve virulence in the environment; such changes could increase the opportunities for hosts to become infected following contact with contaminated surfaces16. In addition, several studies have indicated that temperature is connected to changes in human contact behaviors, which could affect the incidence of HFMD17,18. For example, during warmer periods, people are more likely to spend their time outside rather than stay at home. This may contribute to the spread of enteroviruses through respiratory droplets, ruptured skin vesicles, or direct contact with contaminated toys and environmental surfaces, all of which increase the frequency of contact, and therefore the occurrence of HFMD.
The pattern of association between temperature and HFMD risk is not consistent among studies. Our study suggested a linear relationship over 7 days and an inverted V-shape over 14 days. The association between temperature and HFMD occurrence has been reported to be linear in several regions, such as Guangzhou19, Rizhao20, Hong Kong21, Japan22 and Shenzhen7. The excessive relative risk of HFMD was 2.75% for 1 °C increase in average temperature at lag6 in Shenzhen7. But the present study found 4.51% increase in HFMD incidence for 1 °C rise in temperature at lag0–7 in Wuhan. The larger estimates of HFMD risk in Wuhan may partly due to cumulative effect for the previous 7 days rather than a single day. Nevertheless, non-linear relationships similar to the relationship indicated in the present study have also been documented in several studies23,24,25. Different average temperature ranges may be the reason for this inconsistency. The mean values of average temperature varied from 13 °C to 23 °C in these study regions. One study reported an increased association between average temperature and HFMD incidence below 26.2 °C, but a decreased association above 26.2 °C in Beijing23. The incidence of HFMD rose at temperatures above 14 °C; at temperatures above approximately 23 °C, it began to decline, producing an inverted V-shaped relationship in Chengdu25. The thresholds of average temperature differed between these studies, potentially due to the different serotypes of enterovirus circulating during the study period. The discrepancy could also be attributed to different climatic and geographic conditions among the cities. Despite these differences, the same pattern of association between average temperature and the incidence of HFMD revealed that the appropriate average temperature for an HFMD outbreak in Wuhan is approximately 26 °C. Higher temperatures could lead to the activation of enteroviruses and affect children’s activities, reducing the time spent outdoors and thus the possibility of contact with other children26.
We observed that types of child care may be an important modifier of temperature effect on HFMD incidence. In spite of insignificant difference, stratified analyses indicated that children younger than 1 year old and those aged 3–5 years (children who attended daycare) were more vulnerable to temperature change on HFMD incidence than those aged 1–2 years. A recent study also indicated that children younger than 1 year old were more sensitive to temperature25. It has been reported that the positive rate of maternal EV-A71-neutralizing antibody was 48–85% at birth27,28,29 and decreased to 1–7% at 6 months27,29,30. After that, the positive rate of natural infection increased gradually with age, and stayed stable at 50 to 80% for children aged 6 years or older31,32,33. Therefore, the susceptibility of children aged 0–1 years to temperature may increase because of the lack of immunity. A study in Taiwan suggested that attending a daycare center or kindergarten significantly increased the likelihood of EV-A71 infection34. It is important to note that children’s activities and behaviors differ across different age groups. Personal hygiene practices among children aged 3–5 years are sub-optimal, and proper hygienic supervision of children in kindergarten is difficult for caregivers. Children of this age subgroup are generally cared for at preschool or kindergarten, where they may share beds during noontime breaks and toys, which can be easily contaminated with enteroviruses.
The present study provides a better understanding of the relationship between temperature variation and HFMD risk in Wuhan. Even though it could not fully capture HFMD dynamics from a causal perspective, the finding could serve as an effective way for identifying the risk of HFMD incidence based on short-term weather change. Thus, prevention measures such as good hygiene practices and temporary closure of educational institutions could be adopted before the actual upsurge of HFMD occurrence and thus prevent epidemics in a local scale. Moreover, this study found that children attended daycare (aged 3–5 years old) were more susceptible to temperature variation on HFMD incidence. This helps us enhance measures focused on this specific population and inform parents or caregivers to be aware of HFMD occurrence when temperature changed.
However, some limitations of this study should be acknowledged. First, our study was an ecological one, which did not permit us to explore the association based on individuals and inhibit assessment of causal inference. Second, the present study was focused in one city, and the results may not be applicable to other locations, particularly those with different climates. Third, air pollutants, such as PM10, may play an important role in HFMD risk35. Since we have not available air pollutants data for the present study, we have not discussed its effect on the HFMD risk. Further study is needed to address air pollutants’ roles on HFMD risk. Furthermore, this study did not analyze the pathogenic origin of HFMD as EV-A71, CV-A16 or other enteroviruses. It has been reported that in some regions, EV-A71 and CV-A16 are the predominant viruses causing HFMD in spring and summer, while other enteroviruses cause HFMD during autumn and winter36. Future research should therefore concentrate on the relationship between enterovirus serotypes and meteorological variables. Despite these limitations, our findings contribute to the understanding of temperature effects on HFMD incidence, which may inform health authorities in the development of efficient prevention strategies.
In conclusion, this study suggests that daily average temperature may be an important predictor of HFMD occurrence in different subgroups of children in Wuhan, China. It could be helpful in guiding health resource allocation and development of public health preparedness and intervention strategies.
Materials and Methods
Study area
Wuhan, the capital city of Hubei Province, is located in Central China and features a land size of 8,569.15 km2 and population density of 1,206 persons per km2 as of 2014. It experiences a subtropical monsoon climate, with an annual average temperature of 16.7 °C and relative humidity of 81%37.
Data sources
HFMD has been listed among the notifiable infectious diseases in China since May 2008, and cases must be reported via the national enhanced surveillance system in mainland China. The system data are collected nationwide and are therefore more representative of an actual HFMD epidemic. A 2011 data-quality inspection report indicated that the collected data are of high quality, particularly in the eastern regions of China, with reporting completeness of 99.84% and accuracy of information reported of 92.76%38. In our study, daily reported cases of HFMD in Wuhan from January 2010 to December 2015 were obtained from the Hubei Provincial Center for Disease Control and Prevention. Patients were diagnosed with HFMD if they presented with the following symptoms: scattered vesicular lesions on oral mucosa, papules and vesicular lesions on palms, soles and, occasionally, the buttocks with or without fever in accordance with the National Guidelines on clinical management of Hand, Foot and Mouth Disease disseminated by the Chinese Ministry of Health39. If patient presented with laboratory evidence of enterovirus infection (including EV-A71, CV-A16, or other non-EV71 and non-CV-A16 enterovirus), he or she was defined as a lab-confirmed case. All HFMD cases were requested to be reported to the system online within 24 hours of diagnosis. The gender, age, and child care types of HFMD patients were also obtained. Because 94.9% of HFMD cases occurred in children aged 0–5 years in Wuhan, only children of this age group were chosen for the present study. Daily meteorological data, including average temperature, average air pressure, average vapor pressure, average relative humidity and average wind speed for the same period, were obtained from the China Meteorological Data Sharing Service System (http://www.cma.gov.cn/).
Statistical analyses
Meteorological data and HFMD cases were linked by date and analyzed using a time-series design. A distributed lag non-linear model (DLNM) was constructed to represent a modeling framework to flexibly describe associations showing potentially non-linear and delayed effects in time-series data40,41. In the present study, DLNM combined with a quasi-Poisson distribution was performed to quantify the relationship between daily temperature and HFMD counts adjusted with other meteorological variables. The model was specified as follows:
where E(Yt) was the expected number of HFMD cases on day t; α was the intercept; cb denoted the cross-basis function; df was the degree of freedom; ns() indicated a smooth function based on natural cubic splines; DOY referred to the day of the year, while time represented the day of the entire study period; DOW referred to the day of the week; PH presented a binary variable for public holidays. An autoregressive term for daily HFMD cases was also included in the model to account for autoregression. The relative risks (RRs) of a chosen percentile value of a meteorological variable with 95% confidence intervals (CI), compared with its 5th percentile, were estimated. We also assessed the cumulative exposure-response relationship accounting for the entire lag period.
To investigate the effect of the meteorological variables on HFMD occurrence in different subgroups, stratified analyses were performed for subgroups of different ages (<1, 1–2, 3–5 years), types of child care (children cared for at home, children attended daycare) and genders (male and female). Significance test for subgroup analysis was based on a multivariate Wald test of the coefficients42. These sets of coefficients represented the change in the overall cumulative exposure–response curves in each potential effect modifier (such as male and female). The null hypothesis was that no difference for subgroups.
A preliminary analysis was performed to explore the correlation between meteorological variables by a Spearman’s rank correlation analysis. Air pressure and vapor pressure were not included in the model because of high correlation with temperature, while wind speed was excluded because of insignificant association with HFMD counts (Supplementary Table S1). Therefore, only temperature and humidity were incorporated into the models. The maximum lag was set to 14 days, given the incubation period of HFMD and our data’s suggestion that the relationships between temperature and childhood HFMD were negligible after a 14-day lag. The cumulative effect of temperature on HFMD over a lag of 0–7 days has also been estimated, as HFMD has an incubation period of 3–7 days. In our preliminary model, the degree of freedom in the smoothing function of covariables, i.e., temperature, humidity, DOY and time, and lag, was selected based on previous studies. Specifically, the cross-basis function was applied to temperature and humidity to model the non-linear exposure association with 5 df and the lagged association with 4 df23,43. DOY with 6 df was applied to control for seasonality, while time with 4 df was adjusted for long-term trends10,44. DOW and PH were incorporated into the models to control for the fluctuation in one week and public holidays. An autoregressive term for lag1–7 days of daily HFMD cases was included in the models based on partial autocorrelation function plot (Supplementary Fig. S1).
Because the effect estimation may vary with parameters specification in the model, the following sensitivity analyses were performed: (1) varying the df (3–6) for temperature and relative humidity; (2) varying the maximum lag (13–21 days). Quasi Akaike Information Criterion (QAIC) was used to assess the model fits and ascertain the final model parameters. All data manipulation and analyses were performed using the “dlnm”, “mgcv” and “spline” packages in the R statistical software (Version 3.2.5, http://cran.r-project.org).
Ethical approval
HFMD has been listed among the notifiable infectious diseases in China since May 2008. According to the national surveillance protocol in China, the collection of individual data for all notifiable diseases, including HFMD, was part of an ongoing public health response and was thus exempt from institutional review board assessment. The information contained in the patients’ records was anonymized and deidentified prior to analysis. Only aggregated data were analyzed and reported.
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Chan, K. P. et al. Epidemic hand, foot and mouth disease caused by human enterovirus 71, Singapore. Emerging infectious diseases 9, 78–85, https://doi.org/10.3201/eid0901.020112 (2003).
Yang, S. et al. Epidemiological features of and changes in incidence of infectious diseases in China in the first decade after the SARS outbreak: an observational trend study. The Lancet. Infectious diseases, https://doi.org/10.1016/s1473-3099(17)30227-x (2017).
Chong, P., Liu, C. C., Chow, Y. H., Chou, A. H. & Klein, M. Review of enterovirus 71 vaccines. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America 60, 797–803, https://doi.org/10.1093/cid/ciu852 (2015).
Ooi, M. H., Wong, S. C., Lewthwaite, P., Cardosa, M. J. & Solomon, T. Clinical features, diagnosis, and management of enterovirus 71. The Lancet. Neurology 9, 1097–1105, https://doi.org/10.1016/S1474-4422(10)70209-X (2010).
Xing, W. et al. Hand, foot, and mouth disease in China, 2008–12: an epidemiological study. The Lancet. Infectious diseases 14, 308–318, https://doi.org/10.1016/s1473-3099(13)70342-6 (2014).
Solomon, T. et al. Virology, epidemiology, pathogenesis, and control of enterovirus 71. The Lancet. Infectious diseases 10, 778–790, https://doi.org/10.1016/S1473-3099(10)70194-8 (2010).
Zhang, Z. et al. Short-term effects of meteorological factors on hand, foot and mouth disease among children in Shenzhen, China: Non-linearity, threshold and interaction. The Science of the total environment 539, 576–582, https://doi.org/10.1016/j.scitotenv.2015.09.027 (2016).
Wei, J. et al. The effect of meteorological variables on the transmission of hand, foot and mouth disease in four major cities of shanxi province, China: a time series data analysis (2009-2013). PLoS neglected tropical diseases 9, e0003572, https://doi.org/10.1371/journal.pntd.0003572 (2015).
Zou, X. N., Zhang, X. Z., Wang, B. & Qiu, Y. T. Etiologic and epidemiologic analysis of hand, foot, and mouth disease in Guangzhou city: a review of 4,753 cases. The Brazilian journal of infectious diseases: an official publication of the Brazilian Society of Infectious Diseases 16, 457–465, https://doi.org/10.1016/j.bjid.2012.08.001 (2012).
Wang, P., Zhao, H., You, F., Zhou, H. & Goggins, W. B. Seasonal modeling of hand, foot, and mouth disease as a function of meteorological variations in Chongqing, China. International journal of biometeorology, https://doi.org/10.1007/s00484-017-1318-0 (2017).
Huang, Y. et al. Effect of meteorological variables on the incidence of hand, foot, and mouth disease in children: a time-series analysis in Guangzhou, China. BMC infectious diseases 13, 134, https://doi.org/10.1186/1471-2334-13-134 (2013).
Lin, H. et al. Short-term effect of El Nino-Southern Oscillation on pediatric hand, foot and mouth disease in Shenzhen, China. PloS one 8, e65585, https://doi.org/10.1371/journal.pone.0065585 (2013).
Imai, C., Armstrong, B., Chalabi, Z., Mangtani, P. & Hashizume, M. Time series regression model for infectious disease and weather. Environmental research 142, 319–327, https://doi.org/10.1016/j.envres.2015.06.040 (2015).
Abad, F. X., Pinto, R. M. & Bosch, A. Survival of enteric viruses on environmental fomites. Applied and environmental microbiology 60, 3704–3710 (1994).
Mbithi, J. N., Springthorpe, V. S. & Sattar, S. A. Effect of relative humidity and air temperature on survival of hepatitis A virus on environmental surfaces. Applied and environmental microbiology 57, 1394–1399 (1991).
Altizer, S. et al. Seasonality and the dynamics of infectious diseases. Ecology letters 9, 467–484, https://doi.org/10.1111/j.1461-0248.2005.00879.x (2006).
Robinson, M., Drossinos, Y. & Stilianakis, N. I. Indirect transmission and the effect of seasonal pathogen inactivation on infectious disease periodicity. Epidemics 5, 111–121, https://doi.org/10.1016/j.epidem.2013.01.001 (2013).
Wang, Y. et al. Hand, foot, and mouth disease in China: patterns of spread and transmissibility. Epidemiology 22, 781–792, https://doi.org/10.1097/EDE.0b013e318231d67a (2011).
Chen, C. et al. Short-term effects of meteorological factors on children hand, foot and mouth disease in Guangzhou, China. International journal of biometeorology 58, 1605–1614, https://doi.org/10.1007/s00484-013-0764-6 (2014).
Wu, H., Wang, H., Wang, Q., Xin, Q. & Lin, H. The effect of meteorological factors on adolescent hand, foot, and mouth disease and associated effect modifiers. Global health action 7, 24664, https://doi.org/10.3402/gha.v7.24664 (2014).
Ma, E., Lam, T., Wong, C. & Chuang, S. K. Is hand, foot and mouth disease associated with meteorological parameters? Epidemiology and infection 138, 1779–1788, https://doi.org/10.1017/S0950268810002256 (2010).
Onozuka, D. & Hashizume, M. The influence of temperature and humidity on the incidence of hand, foot, and mouth disease in Japan. The Science of the total environment 410–411, 119–125, https://doi.org/10.1016/j.scitotenv.2011.09.055 (2011).
Xu, M. et al. Non-Linear Association between Exposure to Ambient Temperature and Children’s Hand-Foot-and-Mouth Disease in Beijing, China. PloS one 10, e0126171, https://doi.org/10.1371/journal.pone.0126171 (2015).
Chang, H. L. et al. The association between enterovirus 71 infections and meteorological parameters in Taiwan. PloS one 7, e46845, https://doi.org/10.1371/journal.pone.0046845 (2012).
Yin, F., Zhang, T., Liu, L., Lv, Q. & Li, X. The Association between Ambient Temperature and Childhood Hand, Foot, and Mouth Disease in Chengdu, China: A Distributed Lag Non-linearAnalysis. Scientific reports 6, 27305, https://doi.org/10.1038/srep27305 (2016).
Suminski, R. R., Poston, W. C., Market, P., Hyder, M. & Sara, P. A. Meteorological conditions are associated with physical activities performed in open-air settings. International journal of biometeorology 52, 189–197, https://doi.org/10.1007/s00484-007-0110-y (2008).
Luo, S. T. et al. Enterovirus 71 maternal antibodies in infants, Taiwan. Emerging infectious diseases 15, 581–584, https://doi.org/10.3201/eid1504.081550 (2009).
Mao, Q. Y. et al. Dynamic change of mother-source neutralizing antibodies against enterovirus 71 and coxsackievirus A16 in infants. Chinese medical journal 123, 1679–1684 (2010).
Tran, C. B. et al. The seroprevalence and seroincidence of enterovirus71 infection in infants and children in Ho Chi Minh City, Viet Nam. PloS one 6, e21116, https://doi.org/10.1371/journal.pone.0021116 (2011).
Ji, H. et al. Seroepidemiology of human enterovirus71 and coxsackievirusA16 in Jiangsu province, China. Virology journal 9, 248, https://doi.org/10.1186/1743-422X-9-248 (2012).
Li, W. et al. Seroepidemiology of human enterovirus71 and coxsackievirusA16 among children in Guangdong province, China. BMC infectious diseases 13, 322, https://doi.org/10.1186/1471-2334-13-322 (2013).
Zhu, Z. et al. Retrospective seroepidemiology indicated that human enterovirus 71 and coxsackievirus A16 circulated wildly in central and southern China before large-scale outbreaks from 2008. Virology journal 7, 300, https://doi.org/10.1186/1743-422X-7-300 (2010).
Ooi, E. E., Phoon, M. C., Ishak, B. & Chan, S. H. Seroepidemiology of human enterovirus 71, Singapore. Emerging infectious diseases 8, 995–997, https://doi.org/10.3201/eid0809.010397 (2002).
Chang, L. Y. et al. Risk factors of enterovirus 71 infection and associated hand, foot, and mouth disease/herpangina in children during an epidemic in Taiwan. Pediatrics 109, e88 (2002).
Huang, R., Bian, G., He, T., Chen, L. & Xu, G. Effects of Meteorological Parameters and PM10 on the Incidence of Hand, Foot, and Mouth Disease in Children in China. International journal of environmental research and public health 13, https://doi.org/10.3390/ijerph13050481 (2016).
Zhang, J. et al. Characterization of hand, foot, and mouth disease in China between 2008 and 2009. Biomedical and environmental sciences: BES 24, 214–221, https://doi.org/10.3967/0895-3988.2011.03.002 (2011).
Statistics, W. Wuhan statistical yearbook, 2014).
Ji, B. Y., Guo, G. Y. & Analysis, Q. of notifiable infectious disease surveillance and reporting of medical units in 6 provinces. Mod Preventive Med 38, 4226–4271 (2011).
Health, C. M. o. Clinical management guideline for hand, foot and mouth disease, 2010, http://www.moh.gov.cn/mohyzs/s3586/201004/46884.shtml (2010).
Gasparrini, A. Distributed Lag Linear and Non-Linear Models in R: The Package dlnm. Journal of statistical software 43, 1–20 (2011).
Ding, Z. et al. Association of cold temperature and mortality and effect modification in the subtropical plateau monsoon climate of Yuxi, China. Environmental research 150, 431–437, https://doi.org/10.1016/j.envres.2016.06.029 (2016).
Gasparrini, A. et al. Temporal Variation in Heat-Mortality Associations: A Multicountry Study. Environmental health perspectives 123, 1200–1207, https://doi.org/10.1289/ehp.1409070 (2015).
Xiao, X. et al. The exposure-response relationship between temperature and childhood hand, foot and mouth disease: A multicity study from mainland China. Environment international 100, 102–109, https://doi.org/10.1016/j.envint.2016.11.021 (2017).
Wang, P., Goggins, W. B. & Chan, E. Y. Hand, Foot and Mouth Disease in Hong Kong: A Time-Series Analysis on Its Relationship with Weather. PloS one 11, e0161006, https://doi.org/10.1371/journal.pone.0161006 (2016).
Acknowledgements
We thank staff members at the county and province level CDCs as well as at hospitals for the data collection. We would like to thank Dr. Antonio Gasparrini from Department of Medical Statistics, London School of Hygiene and Tropical Medicine and William B Goggins from Division of Biostatistics, School of Public Health and Primary Care, The Chinese University of Hong Kong for their significant contributions on statistical advice. This study was financially supported by the 2016–2017 CDC Special Program of Health and Family Planning Commission of Hubei Province: Integration Research on Public Health Emergency Prepared Technology [grant number WJ2016JT-007] and the 2017–2018 Key Special Program of Health and Family Planning Commission of Hubei Province: Community Intervention Research on EV-A71 Vaccine Prevention Effect [grant number WJ2017Z018].
Author information
Authors and Affiliations
Contributions
S.W. and X.G. conceptualized the design of the study. J.H. and S.C. participated in the analysis and prepared for the tables and figures. J.H. drafted the initial manuscript. S.W. critically reviewed the manuscript. Y.W., Y.T., L.W. and X.G. participated in data collection and data interpretation. M.Z. and S.H. did literature research and contributed to data interpretation. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Huang, J., Chen, S., Wu, Y. et al. Quantifying the influence of temperature on hand, foot and mouth disease incidence in Wuhan, Central China. Sci Rep 8, 1934 (2018). https://doi.org/10.1038/s41598-018-20318-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-20318-z
This article is cited by
-
Influence of social and meteorological factors on hand, foot, and mouth disease in Sichuan Province
BMC Public Health (2023)
-
Non-linear association between daily mean temperature and children’s hand foot and mouth disease in Chongqing, China
Scientific Reports (2023)
-
Long-term air pollution levels modify the relationships between short-term exposure to meteorological factors, air pollution and the incidence of hand, foot and mouth disease in children: a DLNM-based multicity time series study in Sichuan Province, China
BMC Public Health (2022)
-
Influence and prediction of meteorological factors on brucellosis in a northwest region of China
Environmental Science and Pollution Research (2022)
-
The lagged effect and attributable risk of apparent temperature on hand, foot, and mouth disease in Changsha, China: a distributed lag non-linear model
Environmental Science and Pollution Research (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.