Abstract
Stomatal regulation is important for water transpiration from plants. Stomatal opening and closing are controlled by many transporter proteins in guard cells. AtABCG22 is a member of the ATP-binding cassette (ABC) transporters and is a stomatal regulator; however, the function of AtABCG22 has not yet been determined fully, although a mutant phenotype included a significant effect on stomatal status. Here, we further investigated the function of the AtABCG22 gene and its functional relationships with other subfamily genes. Among close family members, we found a functional relationship of stomatal phenotypes with AtABCG21, which is also expressed specifically in guard cells. Based on an analysis of double mutants, adding the atabcg21 mutation to atabcg22 mutant partially suppressed the open-stomata phenotype of atabcg22. Multiple-mutant analyses indicated that this suppression was independent of abscisic acid signaling in guard cells. We also found that atabcg22 mutant showed a unique time course-dependent phenotype, being defective in maintenance of stomatal status after initial stomatal opening elicited by light signaling. The function of AtABCG22 and its relationship with AtABCG21 in stomatal regulation are considered.
Similar content being viewed by others
Introduction
Stomata consist of a pair of guard cells, a unique cell type with an important role in regulating water transpiration from plant surfaces. Many regulators in guard cells have been shown to function in the closing or opening of stomata. For example, open stomata1 (ost1)/snf1-related protein kinases 2e (srk2e) mutants defective in abscisic acid (ABA) signaling have been isolated as major open-stomata (OST) mutant due to the inability to close its stomata1,2. Additionally, cell membrane transporter proteins are important as stomatal regulators3,4, because guard cells are isolated symplastically from neighboring cells5,6,7.
The superfamily of ATP-binding cassette (ABC) transporters is one of the most abundant protein families in nature. These proteins are broadly conserved from prokaryotes to higher eukaryotes in all phyla that use energy to transport substrates in an ATP-dependent manner against concentration gradients. Plant genomes, in particular, have large ABC families of more than 100 genes, indicating that some ABC transporters likely have important roles in plant-specific developmental and environmental responses8,9.
In plant ABC subfamilies, the ABCG subfamily is the largest, composed of both ‘half-size’ and ‘full-size’ transporters. In Arabidopsis, 28 gene members have been classified as AtABCG half-size transporters8, about half of which have been described in the literature. For example, AtABCG1 and AtABCG16 are required for pollen nexine layer formation, and AtABCG2, AtABCG6, and AtABCG20 are required for suberin barriers10,11,12. AtABCG9 is involved in pollen coat maturation, related to steryl glycosides, in concert with a full-size transporter, AtABCG3113. AtABCG11, AtABCG12, and AtABCG13 are involved in transporting precursors of wax and cutin to the epidermis14,15,16,17,18. AtABCG14 controls the root-to-shoot translocation of cytokinins and plant development19,20,21. AtABCG19 has a role in antibiotic resistance22. AtABCG25 is involved in ABA transport and responses23. AtABCG26 is essential for exine formation, by transporting polyketide sporopollenin precursors24,25,26,27,28,29. Most of these transporters are consequently related to roles in maintaining water content or in prohibiting water loss from plant bodies or pollen cells.
In addition to the members above, we previously isolated AtABCG22, which also functions in retaining water in plants30. Mutant AtABCG22 plants show increased water transpiration and drought susceptibility, suggesting a relationship with ABA function. However, we additionally found enhanced phenotypes of atabcg22 mutant which exhibited an additive effect to ABA signaling or ABA biosynthesis, so that the function of AtABCG22 in guard cells remains to be determined.
Here, we show that AtABCG22 has a functional relationship with AtABCG21, which is a closely related but as yet unanalyzed member of the ABCG subfamily in Arabidopsis. We also examined an atabcg22 mutant phenotype involved in stomatal regulation.
Results
AtABCG21 gene expression patterns in plant organs
We previously reported AtABCG22 was involved in stomatal regulation, because atabcg22 mutants that exhibited a typical open-stomata (OST) phenotype30. To study the relationship between AtABCG22 and other ABCG members that were closely related based on a phylogenetic tree17,30, we focused one of the family members, AtABCG21. To investigate the gene expression patterns of AtABCG21 in wild-type (WT) tissues, we used ~2 kb of the AtABCG21 promoter region (pAtABCG21) to drive expression of a β-glucuronidase (GUS) reporter gene. In pAtABCG21::GUS transgenic plants, GUS activity of the transformants was detected in guard cells of seedlings, but not in roots (Fig. 1a,b). The leaf surface was also stained in adult plants, and GUS staining was detected primarily in guard cells in magnified images (Fig. 1c,d). This gene expression pattern was similar to that of AtABCG22 30, suggesting a guard-cell function for AtABCG21.
Subcellular localization of the AtABCG21 protein
We showed previously that AtABCG22 was localized to the cell membrane in plant cells30. To study the subcellular localization of AtABCG21, we made a construct that expressed green fluorescent protein (GFP) fused to AtABCG21 protein under the control of the Cauliflower mosaic virus 35 S promoter. The AtABCG21 open reading frame was placed downstream of 35 S::GFP. The 35 S::GFP-AtABCG21 recombinant gene was expressed transiently in Arabidopsis protoplasts. Subcellular localization of the fusion protein was visualized by confocal imaging of green fluorescence in protoplast cells. The green fluorescence of the GFP-AtABCG21 recombinant protein was present around the cell surface (Fig. 2a). Additionally, we transformed the 35 S::GFP-AtABCG21 recombinant vector into Arabidopsis plants. GFP-AtABCG21 recombinant protein fluorescence was observed clearly around the cell surface in root cells of the transgenic plants (Fig. 2b). These results indicated that AtABCG21 was localized to the cell membrane.
Subcellular localization of AtABCG21. (a) Transient expression of GFP protein and GFP-AtABCG21 fusion protein in Arabidopsis protoplasts. The left panels show images of GFP fluorescence, the middle panels are images of chloroplast autofluorescence, and the right panels are merged images. (b) Subcellular localization of GFP-AtABCG21 fusion proteins in root cells in 35Spro:GFP-AtABCG21 transgenic plants. The left panel shows an image of GFP fluorescence, the middle panel is a bright-field image, and the right panel is a merged image.
Suppression effect of an atabcg21 mutation on an atabcg22 mutant phenotype
We previously reported that the leaf temperature of atabcg22 mutant plants was lower than that of WT plants under normal growth conditions, because of the OST phenotype30. To investigate the functional relationship between AtABCG21 and AtABCG22, we isolated a homozygous T-DNA insertional mutant on AtABCG21 by genotyping (Supplemental Fig. S1a), and confirmed this line was a gene knockout mutant (Supplemental Fig. S1b). We did not find any significant phenotype related to water transpiration for the atabcg21 mutant plants (Supplemental Fig. S1c,d, Fig. S2a,b). However, when we crossed an atabcg21 mutant with an atabcg22 mutant to produce double mutants, we found that the leaf temperature of atabcg21/atabcg22 double-mutant plants was similar to that of WT plants, indicating that transpiration was lower in the leaves of the double mutants than the atabcg22 mutants (Fig. 3a). This was confirmed by the results of a water-loss experiment in which the rate of weight loss from detached leaves of the double-mutant plants was slower than that from detached leaves of atabcg22 mutants, partially recovered to WT plants or atabcg21 single mutants (Fig. 3b). These results indicated that the atabcg21 mutation partially suppressed the OST phenotype of atabcg22 mutants, suggesting a functional relationship between AtABCG21 and AtABCG22 in stomatal regulation.
Functional relationship between atabcg21 and atabcg22 in transpiration phenotypes. (a) Thermal images of atabcg21, atabcg22, and double-mutant plants. Rosette leaves of 5-week-old wild-type plants (Col), atabcg21 mutant plants (abcg21), double-mutant plants (abcg21/abcg22), and atabcg22 mutant plants (abcg22) were imaged with a visible-light camera (left panel) and an infrared thermography device (right panel). Average temperatures are shown with SD under each line of thermal images. (b) Transpiration ratio of atabcg21, atabcg22, and double-mutant plants. Water loss in detached rosette leaves of 5-week-old plants was determined as a percentage of the initial fresh weight. Values are shown as means ± SD of the three independent plants photographed in (a).
The atabcg21 mutation does not suppress srk2e and nced3 mutant phenotypes
Next, we investigated whether the atabcg21 mutation could suppress the phenotype of other OST-type mutants, particularly those related to ABA biosynthesis or ABA signaling. We crossed atabcg21 mutant plants with srk2e or nine-cis-epoxycarotenoid dioxygenase3 (nced3) mutants to produce each double mutant. SRK2E encodes a kinase involved in cellular ABA signaling in guard cells and NCED3 encodes a key enzyme in ABA biosynthesis1,2,31. Single mutants of srk2e or nced3 are typical OST-type mutants, and showed lower leaf temperatures and accelerated transpiration from leaves, compared with WT plants (Supplemental Figs S1,S2). The leaf temperature and transpiration rates of the atabcg21/srk2e and atabcg21/nced3 double mutants were almost the same as those of the srk2e and nced3 single mutants, respectively (Supplemental Figs S1,S2). These results indicated that the atabcg21 mutation did not generally suppress the phenotype of all OST-type mutants, but may repress atabcg22 specifically, suggesting a specific relationship with the function of AtABCG22.
Phenotypes of multiple-mutant plants with ABA signaling defects
When we first isolated atabcg22 mutants, we suspected a relationship between AtABCG22 function and ABA responses. However, double mutants of atabcg22 and ABA signaling mutants showed additive effects, so this issue was not resolved30. In the process of investigating whether the suppression effect of the atabcg21 mutation on the atabcg22 mutant was dependent on ABA in guard cells, we crossed atabcg21/atabcg22 double-mutant plants with srk2e mutants to produce triple mutants. As reported previously for the enhanced phenotypes of atabcg22/srk2e 30, the leaf temperature of the triple-mutant plants, including srk2e, was lower than that of the atabcg21/atabcg22 double mutants (Fig. 4a). Notably, the leaf temperature of triple-mutant plants was slightly, but significantly, higher than that of atabcg22/srk2e double mutants (Fig. 4a). This result was consistent with that of a water-loss experiment, in which the rate of weight loss from detached leaves of the triple-mutant plants was lower than that from detached leaves of atabcg22/srk2e double mutants (Fig. 4b). These results indicated that the atabcg21 mutation still suppressed the OST phenotype of atabcg22/srk2e to some extent, even in a srk2e mutant background, having ABA-signaling defects. This suggested that the suppression effect of the atabcg21 mutation on atabcg22 mutants may be independent of ABA signaling in guard cells.
Suppression phenotype of atabcg21 on atabcg22 and an ABA signaling mutant. (a) Thermal images of double and triple mutant plants of atabcg21, atabcg22, and srk2e. Rosette leaves of 5-week-old atabcg22 mutant plants (abcg22), atabcg22/srk2e double-mutant plants (abcg22/srk2e), atabcg21/atabcg22/srk2e triple-mutant plants (abcg21/abcg22/srk2e), and atabcg21/atabcg22 double-mutant plants (abcg21/abcg22) were imaged with a visible-light camera (left panel) and an infrared thermography device (right panel). Average temperatures are shown with SD under each line of thermal images. (b) Transpiration ratio of double and triple mutant plants of atabcg21, atabcg22, and srk2e. Water loss in detached rosette leaves of 5-week-old plants was determined as a percentage of the initial fresh weight. Values are shown as means ± SD of the three independent plants photographed in (a).
Light signal-dependent phenotype of the atabcg22 mutant
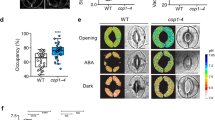
The atabcg22 mutant plants exhibited lower leaf temperatures and increased water loss, indicating elevated transpiration through an effect on stomatal regulation. To address the function of AtABCG22, we measured stomatal conductance over a time course, including light and dark periods. Overall, the stomatal conductance of atabcg22 mutants was higher than that of WT plants, consistent with having an OST-type phenotype (Fig. 5). That of atabcg21 mutants was same as WT plants.
Stomatal conductance in response to changes in light status. Stomatal conductance of wild-type (Col) and atabcg22 (abcg22) was measured using a gas-exchange system (LI-6400). Data are plotted at 10 min intervals for 12 h, including 8 h of dark, in a conditioned green house. The CO2 concentration of the air flow was set at 400 ppm during experiments. Values are shown as means ± SD (n = 3).
In this experiment, we found a unique behavior of atabcg22 mutants in the initial period of lighting. When the light was turned on, stomatal conductance increased quickly, as in WT plants. However after this increase, it decreased rapidly in atabcg22 mutants while it stabilized in WT plants (Fig. 5). When the light was turned off, there appeared to be a normal response in atabcg22 mutants. These results suggest that AtABCG22 may have a function in balancing the stomatal status after light signaling for stomatal opening.
Discussion
When we crossed atabcg21 and atabcg22 mutants to generate double mutants, we found that addition of the atabcg21 mutation partially suppressed atabcg22 mutant phenotypes (Fig. 3). In case of signal transduction, when the phenotype of the first mutation was suppressed by the addition of the second mutation, it could suggest bypassing the first mutation by the second mutation in the signaling pathway. However, the mutated genes in this case encode transporter proteins, rather than signaling factors. Furthermore, both genes are expressed predominantly in guard cells, and both proteins were shown to be localized to the cell membrane (Figs 1,2). Additionally, the result that atabcg21 mutations do not repress other OST-type mutants may indicate a specific relationship between AtABCG21 and AtABCG22 (Supplemental Figs S1,S2).
Accordingly, the simplest interpretation would be that the two transporters have opposite activities in transporting the same substrate. Generally, it has been assumed that eukaryotic ABC proteins uniformly transport a substrate present at the side of the membrane where the nucleotide-binding domain is located to the other side of a membrane; for example, transporters localized to the cell membrane are usually efflux transporters, moving substrates from the cytosol to the outside. However, recent findings show that at least some plant ABC transporters can also act in the opposite direction, as influx transporters, moving substrates from the apoplastic space to the cytosol9,32,33,34.
ABA is a strong effector enhancing stomatal closure. Several AtABCG family members have been reported to be ABA-transporting factors23,34,35. AtABCG22 transcripts are enriched by drought treatment even under mild conditions36. We suspected that AtABCG21 and/or AtABCG22 would be related to ABA, but genetic combination of multiple mutations did not show this (Fig. 4). Another experiment showed that the stomatal response to exogenous ABA was intact in atabcg22 mutants, suggesting that AtABCG22 is not involved in ABA uptake into guard cells directly, although AtABCG22 plays a role in initiating stomatal closure due to reduced air humidity37,38. Targeted substrates of AtABCG21 and AtABCG22 are still unknown and remain to be investigated.
Light, especially blue light, causes positive signaling for stomatal opening39. On the other hand, a report that light-dependent stomatal movement of ost1-2, an allele of srk2e, appeared to be unaffected suggested that light-induced stomatal opening is essentially independent of ABA signaling1. In our time-course measurements, we found that atabcg22 mutant plants could actually respond to light, but then showed rapid attenuation of stomatal opening (Fig. 5). When the light was turned on, cellular light-induced signaling in guard cells was triggered instantly, followed by stomatal opening40. Based on the time course-dependent phenotype of the atabcg22 mutant, AtABCG22 may have a unique function in establishing stable stomatal status in the initial hour after light exposure.
Methods
Plant materials and observations
Plants were grown in soil under well-watered conditions at 22 ± 2 °C and 60–70% relative humidity under a 16/8-h light/dark cycle. The atabcg21 mutant was a T-DNA-tagged mutant obtained from the Arabidopsis Biological Resource Center (SAIL_786_G09). The atabcg22 mutant used in this study was reported previously as the atabcg22-2 (salk_113844) allele, and the srk2e and nced3 mutants have been described previously30.
Thermal images were captured using an infrared thermography device (T620; FLIR). Average temperatures were calculated by FLIR Tools. Water-loss experiments were performed using rosette leaves detached from 5-week-old plants, as described previously30.
Visualization of expression sites by GUS staining
For AtABCG21 promoter-driven GUS expression lines, a 2-kb AtABCG21 promoter region was amplified using KOD plus polymerase (Toyobo) with the primers AtABCG21pro_Fw (5′-CACCGACACCTAAACAAATAGACTTCGTGA-3′) and AtABCG21pro_Rv (5′-CTAGAGAAGGAAAGAGAGATAG-3′), cloned into the pENTR/D-TOPO vector (Invitrogen), and then integrated into the GUS-fusion vector, pBGGUS. The plasmid was then electroporated into Agrobacterium tumefaciens to generate transgenic plants by floral dipping. GUS staining and observation of GUS-stained plants were performed as described previously30.
Subcellular localization of AtABCG21
Transient expression assays using mesophyll protoplasts from Arabidopsis were performed as described previously41. To build the 35 S::GFP-AtABCG21 construct, a fragment of the AtABCG21 coding region was amplified by PCR from Arabidopsis cDNA with the primer set AtABCG21_Fw_EcoRV (5′-AACGATATCATGATGCCTCCTAATGAGCA-3′) and AtABCG21_Rv_NotI (5′-ATAGCGGCCGCTCACAAGTTCCTTAGAGCTA-3′). The amplified fragment was ligated between the EcoRV and NotI sites of the pGKX-NsGFP vector42. The same vectors were electroporated into Agrobacterium to generate transgenic plants.
Gas exchange measurements
Stomatal conductance was assayed in rosette leaves of 5- to 7-week-old wild-type and atabcg22 plants using a portable gas exchange system (LI-6400; LI-COR). The air flow was set to 200 μmol s–1, and the humidity of the air was not regulated, but the CO2 concentration of the air was controlled at 400 ppm using a CO2 cylinder during the experiments.
References
Mustilli, A. C., Merlot, S., Vavasseur, A., Fenzi, F. & Giraudat, J. Arabidopsis OST1 protein kinase mediates the regulation of stomatal aperture by abscisic acid and acts upstream of reactive oxygen species production. Plant Cell 14, 3089–3099 (2002).
Yoshida, R. et al. ABA-activated SnRK2 protein kinase is required for dehydration stress signaling in Arabidopsis. Plant Cell Physiol 43, 1473–1483 (2002).
Pandey, S., Zhang, W. & Assmann, S. M. Roles of ion channels and transporters in guard cell signal transduction. FEBS Lett. 581, 2325–2336 (2007).
Kim, T. H., Böhmer, M., Hu, H., Nishimura, N. & Schroeder, J. I. Guard cell signal transduction network: advances in understanding abscisic acid, CO2, and Ca2+ signaling. Annu. Rev. Plant Biol. 61, 561–591 (2010).
Galatis, B. & Mitrakos, K. The ultrastructural cytology of the differentiating guard cells of vigna sinensis. Amer. J. Bot. 67, 1243–1261 (1980).
Wille, A. C. & Lucas, W. J. Ultrastructural and histochemical studies on guard cells. Planta 160, 129–142 (1984).
Palevitz, B. A. & Hepler, P. K. Changes in dye coupling of stomatal cells of Allium and Commelina demonstrated by microinjection of Lucifer yellow. Planta 164, 473–479 (1985).
Rea, P. A. Plant ATP -binding cassette transporters. Annu. Rev. Plant Biol. 58, 347–375 (2007).
Kang, J. et al. Plant ABC Transporters. Arabidopsis Book 9, e0153, https://doi.org/10.1199/tab.0153 (2011).
Yadav, V. et al. ABCG transporters are required for suberin and pollen wall extracellular barriers in Arabidopsis. Plant Cell 26, 3569–3588 (2014).
Yim, S. et al. Postmeiotic development of pollen surface layers requires two Arabidopsis ABCG-type transporters. Plant Cell Rep. 35, 1863–1873 (2016).
Fedi, F. et al. Awake1, an ABC-Type Transporter, Reveals an Essential Role for Suberin in the Control of Seed Dormancy. Plant Physiol. 174, 276–283 (2017).
Choi, H. et al. The role of Arabidopsis ABCG9 and ABCG31 ATP binding cassette transporters in pollen fitness and the deposition of steryl glycosides on the pollen coat. Plant Cell 26, 310–324 (2014).
Pighin, J. A. et al. Plant cuticular lipid export requires an ABC transporter. Science 306, 702–704 (2004).
Bird, D. et al. Characterization of Arabidopsis ABCG11/WBC11, an ATP binding cassette (ABC) transporter that is required for cuticular lipid secretion. Plant J. 52, 485–498 (2007).
Panikashvili, D. et al. The Arabidopsis DESPERADO/AtWBC11 transporter is required for cutin and wax secretion. Plant Physiol. 145, 1345–1360 (2007).
Ukitsu, H. et al. Cytological and biochemical analysis of COF1, an Arabidopsis mutant of an ABC transporter gene. Plant Cell Physiol. 48, 1524–1533 (2007).
Panikashvili, D., Shi, J. X., Schreiber, L. & Aharoni, A. The Arabidopsis ABCG13 transporter is required for flower cuticle secretion and patterning of the petal epidermis. New Phytol. 190, 113–124 (2011).
Hir, R. et al. ABCG9, ABCG11 and ABCG14 ABC transporters are required for vascular development in Arabidopsis. Plant J. 76, 11–824 (2013).
Ko, D. et al. Arabidopsis ABCG14 is essential for the root-to-shoot translocation of cytokinin. Proc. Natl. Acad. Sci. USA 111, 7150–7155 (2014).
Zhang, K. et al. Arabidopsis ABCG14 protein controls the acropetal translocation of root-synthesized cytokinins. Nat. Commun. 5, 3274, https://doi.org/10.1038/ncomms4274 (2014).
Mentewab, A. & Stewart, C. N. Jr. Overexpression of an Arabidopsis thaliana ABC transporter confers kanamycin resistance to transgenic plants. Nat. Biotechnol. 23, 1177–1180 (2005).
Kuromori, T. et al. ABC transporter AtABCG25 is involved in abscisic acid transport and responses. Proc. Natl. Acad. Sci. USA 107, 2361–2366 (2010).
Quilichini, T. D., Friedmann, M. C., Samuels, A. L. & Douglas, C. J. ATP-binding cassette transporter G26 is required for male fertility and pollen exine formation in Arabidopsis. Plant Physiol. 154, 678–690 (2010).
Xu, J. et al. The ABORTED MICROSPORES regulatory network is required for postmeiotic male reproductive development in Arabidopsis thaliana. Plant Cell 22, 91–107 (2010).
Choi, H. et al. An ABCG/WBC-type ABC transporter is essential for transport of sporopollenin precursors for exine formation in developing pollen. Plant J. 65, 181–193 (2011).
Dou, X. Y. et al. WBC27, an adenosine tri-phosphate-binding cassette protein, controls pollen wall formation and patterning in Arabidopsis. J. Integr. Plant Biol. 53, 74–88 (2011).
Kuromori, T., Ito, T., Sugimoto, E. & Shinozaki, K. Arabidopsis mutant of AtABCG26, an ABC transporter gene, is defective in pollen maturation. J. Plant Physiol. 168, 2001–2005 (2011).
Quilichini, T. D., Samuels, A. L. & Douglas, C. J. ABCG26-mediated polyketide trafficking and hydroxycinnamoyl spermidines contribute to pollen wall exine formation in Arabidopsis. Plant Cell 26, 4483–4498 (2014).
Kuromori, T., Sugimoto, E. & Shinozaki, K. Arabidopsis mutants of AtABCG22, an ABC transporter gene, increase water transpiration and drought susceptibility. Plant J. 67, 885–894 (2011).
Iuchi, S. et al. Regulation of drought tolerance by gene manipulation of 9-cis-epoxycarotenoid dioxygenase, a key enzyme in abscisic acid biosynthesis in Arabidopsis. Plant J. 27, 325–333 (2001).
Shitan, N. et al. Involvement of CjMDR1, a plant multidrug-resistance-type ATP-binding cassette protein, in alkaloid transport in Coptis japonica. Proc. Natl. Acad. Sci. USA 100, 751–756 (2003).
Lee, M. et al. The ABC transporter AtABCB14 is a malate importer and modulates stomatal response to CO2. Nat. Cell Biol. 10, 1217–1223 (2008).
Kang, J. et al. PDR-type ABC transporter mediates cellular uptake of the phytohormone abscisic acid. Proc. Natl. Acad. Sci. USA 107, 2355–2360 (2010).
Kang, J. et al. Abscisic acid transporters cooperate to control seed germination. Nat. Commun. 6, 8113, https://doi.org/10.1038/ncomms9113 (2015).
Jammes, F. et al. Acetylated 1,3-diaminopropane antagonizes abscisic acid-mediated stomatal closing in Arabidopsis. Plant J. 79, 322–333 (2014).
Merilo, E., Jalakas, P., Kollist, H. & Brosché, M. The role of ABA recycling and transporter proteins in rapid stomatal responses to reduced air humidity, elevated CO2, and exogenous ABA. Mol. Plant 8, 657–659 (2015).
Merilo, E. et al. Abscisic acid transport and homeostasis in the context of stomatal regulation. Mol. Plant 8, 1321–1333 (2015).
Briggs, W. R. & Huala, E. Blue-light photoreceptors in higher plants. Annu. Rev. Cell Dev. Biol. 15, 33–62 (1999).
Shimazaki, K., Doi, M., Assmann, S. M. & Kinoshita, T. Light regulation of stomatal movement. Annu. Rev. Plant Biol. 58, 219–247 (2007).
Yamada, K. et al. Functional analysis of an Arabidopsis thaliana abiotic stress-inducible facilitated diffusion transporter for monosaccharides. J. Biol. Chem. 285, 1138–1146 (2010).
Qin, F. et al. Arabidopsis DREB2A-interacting proteins function as RING E3 ligases and negatively regulate plant drought stress-responsive gene expression. Plant Cell 20, 1693–1707 (2008).
Acknowledgements
We thank the RIKEN Center for Sustainable Resource Science (CSRS) for transgenic plant and sequencing support. This work was supported by JSPS KAKENHI Grant Number 17K07458 (to TK), and the Kurata Grant awarded by the Kurata Memorial Hitachi Science and Technology Foundation (to TK).
Author information
Authors and Affiliations
Contributions
T.K. designed the experiments, analyzed the data and wrote the article with contributions of all the authors; E.S. and H.O. provided technical assistance and performed the experiments; K.Y.-S. and K.S. supervised and complemented the writing.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kuromori, T., Sugimoto, E., Ohiraki, H. et al. Functional relationship of AtABCG21 and AtABCG22 in stomatal regulation. Sci Rep 7, 12501 (2017). https://doi.org/10.1038/s41598-017-12643-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-12643-6
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.