Abstract
Based on the first-principles evolutionary materials design, we report a stable boron Kagome lattice composed of triangles in triangles on a two-dimensional sheet. The Kagome lattice can be synthesized on a silver substrate, with selecting Mg atoms as guest atoms. While the isolated Kagome lattice is slightly twisted without strain, it turns into an ideal triangular Kagome lattice under tensile strain. In the triangular Kagome lattice, we find the exotic electronic properties, such as topologically non-trivial flat band near the Fermi energy and half-metallic ferromagnetism, and predict the quantum anomalous Hall effect in the presence of spin-orbit coupling.
Similar content being viewed by others
Introduction
Recently, two-dimensional (2D) materials have attracted much attention because of their unusual characteristics, such as Dirac fermions, topological states, and valley polarization1,2,3. The discovery of graphene, a monolayer of carbon atoms in the honeycomb lattice1, has resulted in the synthesis of different 2D materials and various graphene analogues, such as hexagonal BN, transition metal dichalcogenides, silicene, germanene, stanene, and phosphorene3,4,5. Elemental boron exhibits a variety of allotropes with structural complexity6, 7, but a 2D honeycomb lattice is inherently prohibited because boron lacks one valence electron compared to its adjacent carbon in the periodic table. A flat triangular boron lattice has been reported to be unstable to a buckled shape due to excessive electrons occupying the antibonding states8. Since the surplus of electrons can be balanced by introducing hexagonal holes in the triangular lattice, more stable 2D forms composed of triangular and hexagonal motifs have been proposed, including the B α-sheet and its analogues8, 9.
A Kagome lattice also consists of triangular and hexagonal motifs in a network of corner-sharing triangles. Many exotic phenomena have been predicted for the Kagome lattice, such as frustrated magnetic ordering10,11,12, ferromagnetism13, 14, and topologically non-trivial states15,16,17,18. However, the experimental realization of the Kagome lattice is confined to the Kagome layers of pyrochlore oxides19, the Cu ions sitting on a Kagome lattice in Herbertsmithite12, the self-assembled metal-organic molecules on a substrate20, and the cold atoms of an optical Kagome lattice21. A B3 Kagome lattice, in which a triangular motif is made of three B atoms, is known to be dynamically unstable due to the lack of electrons22. Theoretical calculations indicate that, in the case of 2D metal-B systems such as MoB4 23, TiB2 24, FeB6 25, FeB2 26, and MnB6 27, triangular or honeycomb B networks can be stabilized by the electron transfer from metal ions to B networks and the interaction between metal and B layers. Similarly, a bilayer form of MgB6 sandwiching the Mg layer between two B3 Kagome layers was found to be stable22. Despite a number of theoretical attempts to predict 2D boron allotropes, only a few 2D boron sheets have been synthesized on metal substrates, such as a quasi-2D layer of γ-B28 28, a 2D triangular sheet (generally referred to as borophene)29, and borophene with stripe-patterned vacancies30. Given the structural diversity of metal-B systems, a proper choice of substrate and metal elements as guest atoms can open the way to realizing stable 2D boron Kagome sheets that have not been discovered yet.
In this work, we perform an evolutionary crystal structure search for 2D boron phases with the Mg atoms as guest atoms on a silver substrate. We find a new 2D boron sheet consisting of triangular B networks and the Mg atoms embedded in large hexagonal voids. The boron sheet separated from the substrate forms a twisted Kagome lattice and turns into an ideal triangular Kagome lattice under tensile strain, accompanied with a metal-to-half-metal transition. The ferromagnetism of the triangular Kagome lattice is characterized by a nearly flat band at the Fermi level, which is topologically non-trivial and thus induces the quantum anomalous Hall effect in the presence of spin-orbit coupling.
Results
Crystal structure search
First, we explored two-dimensional Mg-B allotropes with low energies on the substrate by using an ab initio evolutionary crystal structure search method, as implemented in the AMADEUS code31. Distinct configurations were generated under the constraint of layer group symmetry, with the number of configurations setting to 20 in the population size of global optimization. For each configuration, the energy minimization was performed by using the generalized gradient approximation of Perdew, Burke, and Ernzerhof (PBE)32 for the exchange-correlation potential and the projector augmented wave pseudopotentials33, as implemented in the VASP code34. The details of calculations are given in Method.
We considered monolayer and bilayer coverages of the B atoms with the Mg atoms as guest atoms for two lateral supercells on the Ag(111) surface, rhombic 2 × 2 and rectangular \(2\times 2\sqrt{3}\) (Fig. 1a). If a 2D triangular sheet, which has been synthesized on a Ag substrate29, is taken as the most stable structure for the monolayer coverage, the B atoms can be deposited up to 12 in the 2 × 2 supercell (Fig. 2a). Since the Mg atoms prefer to occupy the hollow sites on the Ag(111) surface, they can be deposited up to 4 for the monolayer coverage, and three B atoms per Mg atom are depleted. In the bilayer coverage, the Mg atoms pop into the empty space between the two B layers, keeping the same maximum coverage of 4, and each Mg atom depletes six B atoms. Considering such combinations of Mg and B, 2D Mg-B phases can be represented as Mg m B n with 0 ≤ m ≤ 4 and 0 ≤ n ≤ 12–3 m (0 ≤ n ≤ 24–6 m) for the B monolayer (bilayer) coverage in the 2 × 2 supercell.
Phase diagram of Mg-B systems on Ag(111). (a) Rhombic 2 × 2 and rectangular \(2\times 2\sqrt{3}\) supercells on the Ag(111) surface. (b) A phase diagram of 2D Mg-B systems on the substrate. In the 2 × 2 (rhombuses) and \(2\times 2\sqrt{3}\) (rectangles) supercells, filled and open symbols stand for the stable and metastable states, respectively, and red dashed lines represent the B monolayer and bilayer coverages. (c) The convex hull diagram and (d) the lowest energy structure of Mg1B9 for the monolayer coverage are drawn. Yellow, green, and gray balls represent the Mg, B, and Ag atoms, respectively, and red balls denote the B atoms sharing large triangles in the Kagome lattice.
Atomic structures of B-Mg systems on Ag(111). The atomic structures of (a) Mg0B12 (referred to as borophene), (b) Mg4B0, (c) Mg0B24, and (d) Mg1B18 are drawn. The Mg0B24 structure consists of B20 rectified hexagonal bipyramids connected by four B atoms. The atomic structures of Mg3B9 are shown for (e) the metastable and (f) most stable configurations. Yellow, green, and gray balls represent the Mg, B, and Ag atoms, respectively. Red rhombuses represent the 2 × 2 supercell on the Ag(111) surface.
We searched for the lowest-energy configurations for Mg m B n systems and compared their formation energies defined as,
where E tot (A) is the total energy of the system A and x = m/4, y = n/24, and z = 1 − x − y. Here, Mg4B0 and Mg0B24 correspond to the Mg monolayer and the B bilayer on the Ag surface (Fig. 2b and c), respectively, whereas Mg0B0 denotes the bare Ag substrate. The bilayer form of Mg0B24 is energetically more favorable than the Mg0B12 monolayer on the Ag substrate. Thus, the chemical potentials of Mg and B on the substrate are defined as, μ Mg = [E tot (Mg4B0) − E tot (Mg0B0)]/4 and μ B = [E tot (Mg0B24) − E tot (Mg0B0)]/24. The phase diagram of Mg m B n is shown in Fig. 1b, and the configurations on the convex hull are energetically stable against the decomposition into other phases. Among Mg m B n systems, we find that Mg1B9 lies on the convex hull for the monolayer coverage (Fig. 1c). The stability of Mg1B9 was confirmed through the tests for both the 2 × 2 and \(2\times 2\sqrt{3}\) supercells. The Mg1B9 allotrope consists of nine B atoms in slightly buckled triangular networks and one Mg atom occupying a large hole in the unit cell (Fig. 1d). As the boron coverage increases, we obtained a bilayer form of Mg1B18 on the convex hull, in which the Mg atoms are sandwiched between the two B layers, lying in between the empty holes. In the Mg1B18 allotrope, each B layer has the same triangular network as that of Mg1B9, however, its buckling is enhanced due to interlayer interactions (Fig. 2d).
Atomic structure and stability
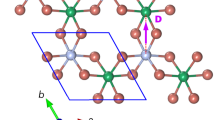
A 2D boron Kagome lattice can be obtained from Mg1B9 by exfoliating the Mg-B sheet from the substrate and removing the guest atoms. The isolation of a 2D boron sheet can be made by using various exfoliation techniques35, 36. In a free-standing Mg1B9 sheet, the Mg ions bind weakly to the boron networks with a smaller binding energy of about 0.7 eV/Mg (Supplementary Table S1), compared with other magnesium borides which were suggested as the potential cathode materials for Mg-ion batteries37. This result indicates that the Mg ions in the Mg1B9 sheet can be dissolved in conventional electrolytes used for Mg batteries38, 39. In the optimized Mg-free B9 sheet, called a twisted Kagome lattice (denoted as B9-tKL), three B atoms are depleted per empty hole, and each void is surrounded by six large triangles (Fig. 3a). As biaxial tensile strain (ε) is applied, the voids are enlarged, and the bonds connecting large triangular units are subsequently broken, resulting in an ideal triangular Kagome lattice (denoted as B9-KL)40, 41, as shown in Fig. 3b. We find that the B9-tKL and B9-KL sheets are perfectly flat, while the remaining Mg ions cause buckling or twisting of the B networks due to the charge transfer. Both B9-tKL and B9-KL belong to the general Kagome system with the subnet 2, in which each triangle of the Kagome arrangement contains a stack of four triangles42. The lattice parameters, plane groups, and Wyckoff positions of B9-tKL and B9-KL are given in Table 1.
Strain effect on atomic structure. Atomic structures for (a) B9-tKL, (b) B9-KL, and (c) B3-KL. Red balls denote the B atoms sharing large triangles in B9-tKL and B9-KL. In (a), gray lines represent the bonds between large triangles, which are broken in B9-KL. (d) The 2D stress (σ 2D) and the angle between two large triangular units (θ) are plotted as a function of biaxial strain.
To estimate the critical strain (ε c ) for the transition from B9-tKL to B9-KL, we calculated the 2D biaxial stress (σ 2D) as a function of strain. In the stress-strain curve, we find two stress drops at ε = 9.5% and 13% (Fig. 3d). The prominent drop at ε = 9.5% is accompanied with bond-breaking relaxations between the large triangular units, whereas the weak drop at ε = 13% is related to a transition to the ferromagnetic state (which will be discussed shortly). When the bonds between the edge B atoms of large triangles are broken, the coordination number of the edge B atoms is reduced from 5 to 4. Then, a charge transfer of 2.5 electrons occurs from six edge atoms to three corner atoms within the unit cell (Table 1). As strain increases above 9.5%, the angle between the large triangular units (θ) increases rapidly and reaches 120° at the critical strain of 16.5%, where B9-KL is formed (Fig. 3d). For strain above 16.5%, B9-KL experiences only elastic deformation and maintains the angle of θ = 120°. For strain up to 24%, overall the calculated stress is below 16N/m, lying in the stress range accessible by using an atomic force microscope, as demonstrated for various 2D materials, such as graphene and transition-metal dichalcogenides43, 44.
The stability of B9-tKL and B9-KL was verified by calculating the full phonon spectra and performing first-principles molecular dynamics simulations at high temperatures (Fig. 4 and Supplementary Figure S1). Among 2D boron sheets composed of triangular and hexagonal motifs, a B3 Kagome lattice (denoted as B3-KL) also has a network of corner-sharing triangles, similar to B9-tKL and B9-KL. In B3-KL, however, each triangle of the Kagome arrangement is made of three B atoms, and a single B atom is depleted in each hexagonal hole (Fig. 3c). It is known that B3-KL is dynamically unstable due to one-electron deficiency to fully occupy the bonding states. On the Ag substrate, we find a Mg3B9 structure consisting of the B3 Kagome lattice and the Mg atoms located underneath the hexagonal holes (Fig. 2e), similar to MgB6 22. However, this allotrope is energetically less stable by 1.64 eV per 2 × 2 cell than the combined structure of Mg1B9 with two additional Mg atoms sandwiched between the Mg1B9 layer and substrate (Fig. 2f).
The valence band of B3-KL consists of five bonding states: two three-center σ-bonding states, two σ-bonding states captured in hexagonal holes, and one delocalized π-bonding state. In B9-KL, six edge atoms in two adjacent large triangular motifs form one six-center σ-bonding state, replacing for two three-center σ-bonding states of B3-KL. While the number of the σ-bonding states is reduced by one, two π-bonding states are fully occupied, in contrast to B3-KL, and the π-non-bonding state is half-filled due to an exchange splitting (Supplementary Figure S2). Meanwhile, one σ-bonding state is further reduced due to the multi-center bonds between the edge atoms of large triangles in B9-tKL. Although one σ-antibonding state is half-filled, two π-bonding and one π-non-bonding states are fully filled. Therefore, it is inferred that the occupation of the delocalized π-orbital states plays a crucial role in stabilizing the flat forms of both B9-tKL and B9-KL, similar to the cases of B α-sheet45 and B clusters46.
Electronic structure
We find that B9-KL exhibits a half-metallic band structure, whereas B9-tKL is metallic but nonmagnetic (Fig. 5a). In B9-tKL, a less dispersive band appears at about −1.0 eV below the Fermi level, and three Dirac-like bands are formed at the K point: one characterized by the p x and p y orbitals in the valence band and two by the p z orbital in both the valence and conduction bands. While strain changes the positions of three Dirac-like bands, a noticeable effect is that the less dispersive band moves upward and becomes flattened. Thus, the density of states at the Fermi level singificantly increases, causing the Stoner instability47. For strain above 13%, the exchange splitting occurs for the flat band, resulting in a half-filled band structure and thereby a ferromagnetic transition, similar to the flat-band ferromagnetism13, 14. With the hybrid functional of Heyd, Scuseria, and Ernzerhof (HSE06) for the exchange-correlation potential48, for B9-KL, we find that the band width of the flat band is 175 meV and the band gap of spin-down electrons is 1.42 eV (Fig. 5b).
Band structure evolution under strain. (a) The PBE band structures of B9-tKL and B9-KL under strain. Circles indicate three Dirac-like bands, and the bands are color-coded according to their orbital characteristics: green for p z , blue for p x and p y , and red for s orbitals. The spin-down bands are shaded in lighter colors. (b) The HSE06 band structure of B9-KL for spin-up and spin-down electrons, with the spin-resolved density of states on the right panel. (c) The contour plot of the charge density for the flat band near the Fermi level at the K point in B9-KL.
To understand the origin of the flat band in B9-KL, we analyzed the wave function associated with the flat band, which is mainly derived from the localized p z orbitals at the corner B atoms of triangles (Fig. 5c). By using maximally localized Wannier functions (MLWFs)49, we derived an effective tight-binding Hamiltonian, with the hopping parameters, t 1 = −2.31 eV, t 2 = −2.10 eV, t 3 = −0.08 eV, t 4 = 0.29 eV, t 5 = 0.52 eV, and t 6 = 0.39 eV (Fig. 3b and Supplementary Method). Nine MLWFs are sufficient enough to represent the bonding characteristics between the p z orbitals and well reproduces the HSE06 band structure near the Fermi level, as illustrated in Fig. 6a. If only the nearest-neighbor hopping terms are considered, a completely flat band is formed at the Fermi level because of the destructive interference of the hopping terms to adjacent hexagonal holes (Supplementary Figure S3).
Tight-binding modeling. (a) The spin-up band structure of B9-KL under 18% strain and (b) its magnified view near the Fermi level along high symmetry lines, with the Hall conductances on the right panels. Gray dots stand for the HSE06 band structure and blue curves represent the band structure derived from the effective tight-binding model with including the SOC. (c) The band structure of the B9-KL ribbon with a width of 50 unit cells. Blue and red curves stand for the chiral bands associated with the different edges.
In the effective tight-binding Hamiltonian, we introduced the spin-orbit coupling (SOC), with the parameters λ SO,3 = 0.004 eV and λ SO,4 = 0.015 eV, which correspond to 5% of the second nearest-neighbor hopping terms, t 3 and t 4, respectively. Although the SOC is small for B systems, it could be enhanced by various methods, such as hydrogenation50, introduction of transition metal adatoms51, and substrate proximity effects52, which have been used for graphene. With including the SOC, we find that the band gap of 57 meV opens at the Γ point and the flat band has the non-trivial Chern number of C = 1 (Fig. 6a). This result implies that the quantum anomalous Hall effect could be realized in B9-KL, provided that the SOC opens the band gap. In fact, the quantization of Hall conductance is found in B9-KL with the SOC (Fig. 6a and b), which is the hallmark of the quantum anomalous Hall effect. In addition, the appearance of chiral edge states inside the bulk band gap is confirmed in a one-dimensional ribbon of B9-KL (Fig. 6c). Similaly, Guterding and coworkers predicted the quantum anomalous Hall and quantum spin Hall effects in doped herbertsmithite18. They achieved the non-trivial band topology by the precise control of doping so that the Fermi level lies at the band touching points, while doping is not required in B9-KL.
Conclusion
In conclusion, we have predicted the ideal planar shape of a triangular B9 Kagome lattice, which contains triangles in triangles. Based on the ab initio evolutionary crystal structure search, we propose that the B9 Kagome lattice can be synthesized on the Ag(111) surface, with selecting the Mg atoms as guest atoms. The triangular B9 Kagome lattice offers the exotic electronic characteristics, such as flat band at the Fermi level and half-metallic ferromagnetism, providing opportunities for spintronics applications. Because of the non-trivial band topology of the flat band, the quantum anomalous Hall effect can be realized in the presence of spin-orbit coupling.
Method
Density functional calculations
We employed the generalized gradient approximation of Perdew, Burke, and Ernzerhof (PBE)32 for the exchange-correlation potential and the projector augmented wave pseudopotentials33, as implemented in the VASP code34. In addition, van der Waals forces were taken into account to describe more accurately interlayer interactions53. In a slab geometry, a vacuum region larger than 15 Å was inserted, with a dipole correction54, ensuring prohibiting interactions between adjacent supercells, and only the topmost layer of the silver substrate was relaxed. The wave functions were expanded in plane waves up to an energy cutoff of 300 eV and the Monkhorst-Pack mesh55 with a grid spacing of 2π × 0.03 Å−1 was used for Brillouin zone integration. The atomic coordinates were optimized until the residual forces were less than 0.02 eV/Å. For the free-standing B9-tKL, the in-plane lattice vectors were relaxed until stress was below 0.5 kbar. At the final stage of optimization, we used a higher energy cutoff of 400 eV.
References
Novoselov, K. S. et al. Two-dimensional gas of massless Dirac fermions in graphene. Nature (London) 438, 197–200 (2005).
Qian, X., Liu, J., Fu, L. & Li, J. Quantum spin Hall effect in two-dimensional transition metal dichalcogenides. Science 346, 1344–1347 (2014).
Geim, A. K. & Grigorieva, I. V. Van der Waals heterostructures. Nature (London) 499, 419–425 (2013).
Novoselov, K. S., Mishchenko, A., Carvalho, A. & Castro Neto, A. H. 2D materials and van der Waals heterostructures. Science 353, aac9439–aac9439 (2016).
Balendhran, S., Walia, S., Nili, H., Sriram, S. & Bhaskaran, M. Elemental Analogues of Graphene: Silicene, Germanene, Stanene, and Phosphorene. Small 11, 640–652 (2015).
Albert, B. & Hillebrecht, H. Boron: Elementary Challenge for Experimenters and Theoreticians. Angew. Chem. Int. Ed. 48, 8640–8668 (2009).
Oganov, A. R. et al. Ionic high-pressure form of elemental boron. Nature (London) 457, 863–7 (2009).
Tang, H. & Ismail-Beigi, S. Novel Precursors for Boron Nanotubes: The Competition of Two-Center and Three-Center Bonding in Boron Sheets. Phys. Rev. Lett. 99, 115501 (2007).
Penev, E. S., Bhowmick, S., Sadrzadeh, A. & Yakobson, B. I. Polymorphism of Two-Dimensional Boron. Nano Lett. 12, 2441–2445 (2012).
Balents, L. Spin liquids in frustrated magnets. Nature (London) 464, 199–208 (2010).
Nisoli, C., Moessner, R. & Schiffer, P. Colloquium: Artificial spin ice: Designing and imaging magnetic frustration. Rev. Mod. Phys. 85, 1473–1490 (2013).
Norman, M. R. Colloquium: Herbertsmithite and the search for the quantum spin liquid. Reviews of Modern Physics 88, 041002 (2016).
Mielke, A. Exact ground states for the Hubbard model on the Kagome lattice. J. Phys. A 25, 4335–4345 (1992).
Tanaka, A. & Ueda, H. Stability of Ferromagnetism in the Hubbard Model on the Kagome Lattice. Phys. Rev. Lett. 90, 067204 (2003).
Ohgushi, K., Murakami, S. & Nagaosa, N. Spin anisotropy and quantum Hall effect in the kagomé lattice: Chiral spin state based on a ferromagnet. Phys. Rev. B 62, R6065–R6068 (2000).
Guo, H.-M. & Franz, M. Topological insulator on the kagome lattice. Phys. Rev. B 80, 113102 (2009).
Tang, E., Mei, J.-W. & Wen, X.-G. High-Temperature Fractional Quantum Hall States. Phys. Rev. Lett. 106, 236802 (2011).
Guterding, D., Jeschke, H. O. & Valentí, R. Prospect of quantum anomalous Hall and quantum spin Hall effect in doped kagome lattice Mott insulators. Scientific Reports 6, 25988 (2016).
Gardner, J. S., Gingras, M. J. P. & Greedan, J. E. Magnetic pyrochlore oxides. Rev. Mod. Phys. 82, 53–107 (2010).
Mao, J. et al. Tunability of supramolecular kagome lattices of magnetic phthalocyanines using graphene-based moir? patterns as templates. J. Am. Chem. Soc. 131, 14136–14137 (2009).
Jo, G.-B. et al. Ultracold Atoms in a Tunable Optical Kagome Lattice. Phys. Rev. Lett. 108, 045305 (2012).
Xie, S.-Y. et al. A novel two-dimensional MgB6 crystal: metal-layer stabilized boron kagome lattice. Phys. Chem. Chem. Phys. 17, 1093–1098 (2015).
Xie, S.-Y. et al. First-principles calculations of a robust two-dimensional boron honeycomb sandwiching a triangular molybdenum layer. Phys. Rev. B 90, 035447 (2014).
Zhang, L. Z., Wang, Z. F., Du, S. X., Gao, H.-J. & Liu, F. Prediction of a Dirac state in monolayer TiB2. Phys. Rev. B 90, 161402 (2014).
Zhang, H., Li, Y., Hou, J., Tu, K. & Chen, Z. FeB6 Monolayers: The Graphene-like Material with Hypercoordinate Transition Metal. J. Am. Chem. Soc. 138, 5644–5651 (2016).
Zhang, H., Li, Y., Hou, J., Du, A. & Chen, Z. Dirac State in the FeB2 Monolayer with Graphene-Like Boron Sheet. Nano Lett. 16, 6124–6129 (2016).
Li, J. et al. Voltage-gated spin-filtering properties and global minimum of planar MnB6, and half-metallicity and room-temperature ferromagnetism of its oxide sheet. J. Mater. Chem. C 4, 10866–10875 (2016).
Tai, G. et al. Synthesis of Atomically Thin Boron Films on Copper Foils. Angew. Chem. Int. Ed. 54, 15473–15477 (2015).
Mannix, A. J. et al. Synthesis of borophenes: Anisotropic, two-dimensional boron polymorphs. Science 350, 1513–1516 (2015).
Feng, B. et al. Experimental realization of two-dimensional boron sheets. Nat. Chem. 8, 563–568 (2016).
Lee, I.-H., Oh, Y. J., Kim, S., Lee, J. & Chang, K. J. Ab initio materials design using conformational space annealing and its application to searching for direct band gap silicon crystals. Comput. Phys. Comm. 203, 110–121 (2016).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized Gradient Approximation Made Simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B 50, 17953–17979 (1994).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B 59, 1758–1775 (1999).
Nicolosi, V., Chhowalla, M., Kanatzidis, M. G., Strano, M. S. & Coleman, J. N. Liquid Exfoliation of Layered. Materials. Science 340, 1226419–1226419 (2013).
Tao, L. et al. Silicene field-effect transistors operating at room temperature. Nat. Nanotech. 10, 1–5 (2015).
Zhao, Y., Ban, C., Xu, Q., Wei, S.-H. & Dillon, A. C. Charge-driven structural transformation and valence versatility of boron sheets in magnesium borides. Phys. Rev. B 83, 035406 (2011).
Saha, P. et al. Rechargeable magnesium battery: Current status and key challenges for the future. Prog. Mater. Sci. 66, 1–86 (2014).
Zhang, R. & Ling, C. Status and challenge of Mg battery cathode. MRS Energy Sustain. 3, E1 (2016).
Norman, R. E. & Stenkamp, R. E. Structure of a copper(II) complex of 2-C-carboxypentonic acid (H3cpa); [Cu9Br2(cpa)6]2−n . xH2O. Acta Cryst. C 46, 6–8 (1990).
Mekata, M. et al. Magnetic ordering in triangulated kagomé lattice compound, Cu9Cl2(cpa)6. nH2O. J. Magn. Magn. Mater. 177–181, 731–732 (1998).
Ziff, R. M. & Gu, H. Universal condition for critical percolation thresholds of kagomé-like lattices. Phys. Rev. E 79, 020102 (2009).
Lee, C., Wei, X., Kysar, J. W. & Hone, J. Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene. Science 321, 385–388 (2008).
Bertolazzi, S., Brivio, J. & Kis, A. Stretching and Breaking of Ultrathin MoS2. ACS Nano 5, 9703–9709 (2011).
Galeev, T. R. et al. Deciphering the mystery of hexagon holes in an all-boron graphene α-sheet. Phys. Chem. Chem. Phys. 13, 11575 (2011).
Zhai, H.-J., Kiran, B., Li, J. & Wang, L.-S. Hydrocarbon analogues of boron clusters — planarity, aromaticity and antiaromaticity. Nat. Mater. 2, 827–833 (2003).
Stoner, E. C. Ferromagnetism. Rep. Prog. Phys. 11, 43–112 (1947).
Heyd, J., Scuseria, G. E. & Ernzerhof, M. Hybrid functionals based on a screened Coulomb potential. J. Chem. Phys 118, 8207–8215 (2003).
Mostofi, A. A. et al. wannier90: A tool for obtaining maximally-localised Wannier functions. Comput. Phys. Comm. 178, 685–699 (2008).
Balakrishnan, J., Kok Wai Koon, G., Jaiswal, M., Castro Neto, A. H. & Özyilmaz, B. Colossal enhancement of spin-orbit coupling in weakly hydrogenated graphene. Nat. Phys. 9, 284–287 (2013).
Weeks, C., Hu, J., Alicea, J., Franz, M. & Wu, R. Engineering a Robust Quantum Spin Hall State in Graphene via Adatom Deposition. Phys. Rev. X 1, 021001 (2011).
Calleja, F. et al. Spatial variation of a giant spin–orbit effect induces electron confinement in graphene on Pb islands. Nat. Phys. 11, 43–47 (2014).
Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 27, 1787–1799 (2006).
Neugebauer, J. & Scheffler, M. Adsorbate-substrate and adsorbate-adsorbate interactions of Na and K adlayers on Al(111). Phys. Rev. B 46, 16067–16080 (1992).
Monkhorst, H. J. & Pack, J. D. Special points for Brillouin-zone integrations. Phys. Rev. B 13, 5188–5192 (1976).
Acknowledgements
This work was supported by Samsung Science and Technology Foundation under Grant No. SSTF-BA1401-08.
Author information
Authors and Affiliations
Contributions
S.K., W.H.H., I.-H.L., and K.J.C. contributed equally to this manuscript. K.J.C. conceived the work and designed the research strategy. S.K. performed theoretical calculations and S.K. and W.H.H. did data analysis. All authors discussed the results and co-wrote the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Kim, S., Han, W.H., Lee, IH. et al. Boron Triangular Kagome Lattice with Half-Metallic Ferromagnetism. Sci Rep 7, 7279 (2017). https://doi.org/10.1038/s41598-017-07518-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-07518-9
This article is cited by
-
Compact Localized States in Engineered Flat-Band \({\mathscr{P}}{\mathscr{T}}\) Metamaterials
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.