Abstract
Nitrogen oxide (NOx) pollution is emerging as a primary environmental concern across Europe. While some large European metropolitan areas are already in breach of EU safety limits for NO2, this phenomenon does not seem to be only restricted to large industrialized areas anymore. Many smaller scale populated agglomerations including their surrounding rural areas are seeing frequent NO2 concentration violations. The question of a quantitative understanding of different NOx emission sources is therefore of immanent relevance for climate and air chemistry models as well as air pollution management and health. Here we report simultaneous eddy covariance flux measurements of NOx, CO2, CO and non methane volatile organic compound tracers in a city that might be considered representative for Central Europe and the greater Alpine region. Our data show that NOx fluxes are largely at variance with modelled emission projections, suggesting an appreciable underestimation of the traffic related atmospheric NOx input in Europe, comparable to the weekend-weekday effect, which locally changes ozone production rates by 40%.
Similar content being viewed by others
Introduction
The nitrogen cycle1 is essential for maintaining the oxidizing capacity of the atmosphere and regulating ozone in the lower atmosphere2. Perturbations due to rapid industrialization and agricultural activities have led to a significant increase of atmospheric nitrogen oxides (NOx) during the 20th century3. A regionally intense buildup of photochemical smog due to the presence of nitrogen oxides, CO and non-methane volatile organic compounds (NMVOC) was first identified in the US and attributed as the main cause of severe ozone pollution in many areas4. Decades of subsequent research activities ranging from detailed laboratory5, 6 and smog chamber7,8,9 studies to large scale field campaigns10,11,12 have led to a reasonably good mechanistic understanding of the formation of tropospheric ozone, which is characterized by a complex nonlinear relationship between NOx and reactive carbon species13. This interdependency gives regulators two key strategies to mitigate ozone pollution. The effectiveness to control ozone thereby very much depends on the ratio between ambient OH reactivity and NOx concentrations14, which can be described by relatively simple analytical relationships15. The development of mechanistic regional16 and global air chemistry models17 has further given regulators and scientists powerful tools to study tropospheric ozone formation16, where the mitigation of NOx emissions has emerged as one of the key air pollution control strategies for ozone18,19,20 and more recently also for particulate matter with a diameter of 1 μm or less (PM1)21. Due to the toxicity, nitrogen dioxide (NO2) is also regulated as a hazardous air pollutant itself22. For example, in Europe regulatory action under the EU Thematic Strategy on Air Pollution is in place to limit urban street canyon NO2 concentrations to 40 μg/m3 per year (or 200 μg/m3/h on less than 18 days/year)23. Current trends across European air quality networks show that regulatory thresholds of NO2 are violated at many stations, which does not seem to be limited to large population centers anymore (ref. 24, SI). In fact many rural areas and smaller towns see NO2 concentration levels rivaling those of large metropolitan areas. Owing to the spatiotemporal variability and uncertainty of different anthropogenic NOx sources, it is difficult to attribute emission uncertainties to specific sectors in complex bottom-up emission inventories25 or top-down remote sensing assessments26. Recently evidence has accumulated that rapid shifts in transportation fuels can have significant impacts on air quality27, 28. In Europe for example the question about the increasing penetration of Diesel cars raises concerns as to what extent such a technological change has been counterproductive to mitigating atmospheric NO2 pollution under new emission regulation standards19, 29. The United States environmental protection agency’s (US EPA) notice of violation of the Clean Air Act to a German automaker regarding Diesel engines has sparked a number of new real world driving (RDE) emission tests across Europe, which show significant manufacturer and vehicle specific variability30, 31. These new data suggest that the impact on up-scaled average fleet emissions needed for accurate air quality predictions remains unclear32.
A number of urban flux measurement sites for energy and CO2 have been established highlighting their potential for surface-atmosphere exchange studies33, 34. In contrast, similar measurements for reactive gases are often still quite limited, owing to the complexity of the required measurement systems. A set of recently conducted urban ground based and airborne NMVOC flux measurements revealed the usefulness to test bottom-up emission inventories and revealed significant discrepancies for some species35,36,37,38,39. Urban flux measurements for NOx are even more scarce32, 40, 41 indicating that constraints on emission sources in urban areas can be quite uncertain. Here we improve upon existing work, by simultaneously measuring NOx, selected tracer NMVOCs, CO and CO2 leading to a well constrained flux dataset, that allows testing our understanding of prominent NOx emission sources.
Results
Obtaining ensemble average statistics on fleet emissions by eddy covariance flux measurements
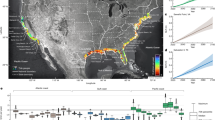
A comprehensive set of eddy covariance measurements for NOx, marker NMVOC, CO and CO2 at an urban location allows a direct comparison of relative flux ratios with bottom-up emission sources. The study site located in Innsbruck (N 47°15′51.50″, E 11°23′6.77″) at the center of the Inn valley, represents one of the most strategically important Alpine crossing points for the transport of goods between Northern and Southern Europe. Each year approximately 6 million vehicles42 pass through the east-west facing valley, which is about 10 km wide surrounded by mountain ridges about 2.5 km high. The valley topography leads to a very predictable and pronounced wind system characterized by a topographic amplification factor (TAF) of about 343. Due to the combination of significant traffic induced NOx emissions and increasingly stringent NO2 limit values, the area is in non-attainment. Local authorities are facing legal proceedings by the European Commission for their failure to control excessive levels of nitrogen oxides (Fig. S2), similar to many areas across Europe23. Tracer flux relationships allow investigating to what extent urban emissions are caused by (a) traffic, (b) urban residential and (c) biomass burning/biofuel activities. Figure 1 shows the diurnal evolution of Weekday (Tuesday-Thursday) and Sunday NOx fluxes and concentrations along with mean traffic count data at the site during the measurement campaign (July – October, 2015). Median measured midday NOx mixing ratios in Innsbruck are comparable to values reported for central London (10–14 ppbv), while corresponding observed fluxes are about a factor of 3–4 lower (i.e. 3000–4000 ng/m2/s vs 700–1440 ng/m2/s)32. These observations are consistent with the idea of an intensification of air pollution proportional to TAF, and a corresponding effective lower air volume that pollutants are being mixed into in steep valleys (Fig. S2, ref. 43). This comparison likely indicates that a much stronger reduction of NOx emissions from the transport sector would be required in the Alps than for example in London in order to achieve current air pollution standards along one of the busiest EU transport corridors across the Alps. Several lines of evidence exclude significant presence of biomass burning during the present study. The average ratio between benzene and toluene fluxes exhibited a typical value (3.3 ± 0.7; R2 = 0.85) characteristic for urban emission sources, dominated by fossil fuel combustion and evaporative/cold-start emissions. The correlation between acetonitrile and benzene, toluene, NOx or CO2 fluxes was low with an R2 of 0.07, 0.08, 0.02 and 0.06 respectively. We also did not observe significant excursions of other species recently suggested as additional biomass burning markers44 such as furfural and furan showing a correlation coefficient of R2 < 0.2 above their background fluxes. We observed an excellent correlation between CO2, benzene and NOx fluxes (CO2/NOx: R2 = 0.86; benzene/NOx: R2 = 0.75). The covariance between between NOx and CO2 (benzene) fluxes yielded values of 0.91 (0.86). We interpret these observations such that benzene, NOx and CO2 emissions are dominated by road traffic with contributions from residential combustion sources.
Statistical plot of measured NOx fluxes, mixing ratios and traffic count data. The center dot shows the ensemble median, where the box around it represents one standard deviation and whiskers the 25 and 75% percentile. Individual extreme values are plotted as open circles. Panels A, B and C represent weekdays (i.e. TUE-THU; composite of 609 individual data points) and panels D, E and F depict Sundays (193 individual data points).
Benchmarking urban source emission ratios and inventories
To gain a more quantitative insight, we investigated flux ratios between NOx and CO2 (FNOx/FCO2; Fig. 2). The advantage of this approach is that it allows determining the actual ensemble average of different emission sources based on measured flux ratios, similar to an end-member un-mixing regression analysis. This allows us to compare our measurements to relative emission strengths reported in emission models and inventories. For large scale emission inventories (e.g. grid cells > 1 km2) this approach also circumvents uncertainties related to assumptions of various downscaling approaches. The observed FNOx/FCO2 ratios follow a diurnal cycle showing a ~40–50% variation throughout a day, which reflects the pronounced fluctuation of traffic activity across the city (e.g. ranging from about 97 vehicles/h at night to 890 vehicles/h during daytime at a traffic count station within the flux footprint). Since we can exclude significant industrial emissions within the flux footprint as well as biomass burning, the variation of FNOx/FCO2 should exhibit the characteristic behavior of city scale sources comprised of (1) a combination of vehicular emissions and (2) residential/domestic combustion sources (e.g. oil and gas heating units). A minimization routine (SI) allowed un-mixing these two end-members of the compositional data, reflecting the actual emission ratios for traffic and urban residential combustion sources (Fig. 2). The fitted model (SI) can reproduce the diurnal cycle and activity factors reasonably well, leading to a NOx/CO2 emission ratio for traffic of 4.2(±0.3) × 10−3 ([mg/m2/h] NOx/[mg/m2/h] CO2), and 0.20(±0.05) × 10−3 ([mg/m2/h] NOx/[mg/m2/h] CO2) for residential combustion sources. These calculated ratios are also depicted in Fig. 2 by horizontal shaded blue and green lines. The activity factors suggest that the ratio is dominated by traffic, comprising about 85% of the activity averaged over the entire day (and >95% during peak traffic). We also investigated NOx/benzene flux source ratios revealing comparable differences as observed for FNOx/FCO2. The CO2 flux weekend-weekday effect and CO/CO2 ratios close to a recent road tunnel study45, all imply that a biogenic influence on CO2 fluxes due to photosynthetic uptake or respiration can be considered negligible at this site. The obtained NOx/CO2 flux ratio for traffic is significantly larger than predicted by a number of state of the art emission inventories and emission standards (Table 1). Generally, we observe 50–70% higher NOx emissions relative to CO2 from road traffic than what is calculated with the most recent traffic emission models. In these detailed bottom – up models, mobile source emissions are treated for different engine sizes and fuels, that, in the past, relied on standardized protocols obtained in test facilities, but were recently updated based on a number of RDE tests30, 31. Exhaust from modern gasoline powered engines, despite higher ignition temperatures than those powered by Diesel, can be effectively treated for NO along with NMVOC (and CO) using a three way catalytic (TWC) converter. TWC treatment can lead to a 10 fold reduction of NO. This has been hard to achieve for Diesel powered cars, which nowadays mostly rely on selective catalytic reduction due to the high air to fuel ratio during combustion. Generally, different combustion and exhaust treatment characteristics result in significantly higher NOx/CO2 emission ratios for Diesel powered cars than for gasoline. The modelled fleet average contribution suggests that at least 90% of urban NOx emissions should originate from Diesel driven vehicles at the present location. 85% is modelled to be emitted by the passenger car fleet based on the COPERT model and TRACCS database (ref. 46, SI). The current Austrian passenger car fleet comprises about 50% Diesel cars and the percentage across Europe grew at a substantially faster rate compared to the US47. Based on our measurements the current average Austrian car fleet emits about 36 times more NOx per CO2 molecule compared to the US TIER II emission standard and a factor of 8–10 more than Euro 6 emission standards31. Factoring in differences in fuel economy between the European and US car fleet, this would equate to about an order of magnitude more NOx emissions per travelled distance compared to newly introduced emission standards. How comparable are these results to other European countries? Diesel engines dominate the European passenger car market: 55% of all newly registered vehicles in the EU were powered by Diesel-fuel in 201242; the penetration of Diesel cars of the two largest European economies bordering the Alps ranges from 30% (Germany) to 70% (France)42, more than an order of magnitude higher than in the US47. When comparing measured NOx/CO2 flux ratios with current inventories used for IPCC atmospheric chemistry/climate48 and air quality models49 we obtain a discrepancy up to about a factor of 3–4 (Table 1). Incidentally, a recent comprehensive model evaluation has suggested significant discrepancies between regionally modelled and observed surface NOx concentrations that seemed worse (e.g. by a factor of 2 during summers) over Europe than over the US25. While a number of uncertainties (agricultural emissions, vertical mixing, biomass burning emissions, deposition) can potentially result in modelled concentration biases in these models, our measurements suggest that road transport related biases likely contribute significantly to these discrepancies as they account for about half of the total NOx emissions across Europe50.
Diurnal cycle of median traffic count data (black line - left axis), measured (blue circles) and model fitted (dashed blue line) mass flux ratio of NOx/CO2 (right axis). The corresponding calculated end members for traffic and residential combustion ratios are indicated by blue and green horizontal lines. The shading reflects one standard deviation. In addition colored dashed lines show predictions using fixed emission ratios from COPERT (magenta) and ACCMIP (red).
What is the impact on atmospheric chemistry?
The weekend – weekday effect (Fig. 1) allows to gain insight into changing NOx and NMVOC fluxes on ozone production in more detail. In Austria, heavy duty vehicle traffic (trucks heavier than 7.5t and all road trains) is banned between Saturday 15:00 and Sunday 22:00 and on public holidays between midnight and 22:00. Traffic count data generally show a pronounced difference in driving habits resulting in a factor of 1.9 ± 0.2 lower vehicle counts on Sunday than on weekdays. We calculated typical ozone production rates for midday-afternoon conditions (11–16 h LT), when photochemistry peaks. The corresponding NOx fluxes are a factor of 2.1 ± 0.2 lower on Sundays, closely matching observations of vehicle activity. Benzene and toluene fluxes, representing the variation of anthropogenic NMVOC emissions, were lower by a factor of 1.8 ± 0.3 and 2.0 ± 0.3 respectively. CO2 fluxes changed by a similar factor of 2.3 ± 0.5. The weekend-weekday comparison provides an independent confirmation that these pollutant emissions are dominated by traffic activity during the day. Average NOx concentrations are a factor of 2.5 ± 0.2 lower during Sundays. Incidentally, the observed weekend – weekday reduction is comparable to the observed measurement-inventory discrepancies or the effect if an entire car fleet was converted from a Euro 5 (0.18 g/km) to a Euro 6 (0.08 g/km) NOx emission standard. Our measurements therefore allows us to benchmark such a hypothetical regulatory action in the real atmosphere. Changes in local ozone production are calculated following procedures outlined before15, 51, where the sensitivity of local ozone production can be approximated by the ratio of radical termination (LN) processes (e.g. NO2 + OH) and photochemical radical production (Q):
Here the δ symbol indicates the relative change between weekday and Sunday, J represents the photolysis rates, and Ox = O3 + NO2. All terms on the right side can be inferred from measurements of the weekend effect, where the anthropogenic change of NMVOCs is assumed to follow benzene. There is evidence of a non-neglegible biogenic NMVOC (BVOC) presence at the site (e.g. 20–50% of the NMVOC reactivity) and these BVOCs do not exhibit any anthropogenically related variation between weekdays and weekend. We apportioned the change of NMVOC reactivity therefore into a biogenic and anthropogenic part using data from the PTR-QiTOF-MS instrument. To achieve this, we estimated the total anthropogenic NMVOC reactivity from known urban concentration ratios52, 53 scaled to benzene and compared this to the measured reactivity of BVOC, in particular the reactive biogenic marker species isoprene and monoterpenes. This allowed to obtain upper (δNMVOC = δbenzene) and lower (δNMVOC = δbenzene × [reactivity anthropogenic NMVOC]/[reactivity BVOC]) bounds for LN/Q. It can further be shown15 that the sensitivity of local ozone production can be related to LN/Q according to:
The calculated range for LN/Q delimits a ratio between 0.82 and 0.92 (theoretical maximum = 1) and places the site in a NOx inhibited chemical regime for ozone production during weekdays15, 51. Incidentally these afternoon values are systematically higher than reported for a rural site in Northern Italy with comparable NOx concentrations, and more closely follow conditions reported for Milan in 199854. Our flux data clearly demonstrate that the weekend effect of ozone production found in Innsbruck is largely a direct result of lower weekend NOx emissions. As a consequence of the dominating nitrogen chemistry for radical loss calculated via eqs 1 and 2, local ozone production will therefore increase by 39% to 70% proportional to a reduction of NOx mixing ratios in Innsbruck. It will decrease between 70% to 85% proportional to a reduction of NMVOC or CO. The impact on gross ozone production (P(O3)) was also investigated independently using the Leeds Master Chemical Mechanism (MCM) as a photochemical box model (refs 13 and 55, SI, Fig. 3). The model simulates an increase of P(O3) by 40% (from 2.5 to 3.5 ppbv/h) on Sundays due to a 2 fold reduction of NOx, giving an explanation for the observed weekend effect, which results in an increase of local afternoon ozone concentrations up to 24% (Fig. 4). The model simulation suggests a maximum increase of P(O3) up to 3 times (2.5 ppbv/h to 7.8 ppbv/h), if solely NOx concentrations were decreased from current levels to about 2 ppbv (Fig. 3). Below 2–4 ppbv of NOx, ozone production would enter the NOx sensitive regime and gradually lead to decreasing P(O3). Next, we setup the MCM as a diurnally constrained 0-dimensional diluting box model (SI) to study the sensitivity with respect to changes in modelled ozone concentrations between weekdays and weekends. The model is thereby fully constrained by methane, CO, measured NMVOC, NO, photolysis rates, and PBL height along with ancilliary meteorological variables (e.g. temperature, humidity). NO2 and ozone are initialized by their measured initial concentrations, but are then allowed to freely adjust during the model run. We chose a spin-up time of 3 days and used model results from the last day of simulation to compare with measured ozone concentrations. Figure 4 shows the diurnal patterns of modelled and observed ozone concentrations. Observed (modelled) peak ozone concentrations are 49 ± 2 (52) ppbv and 43 ± 2 (44) ppbv on weekends and weekdays respectively. Modelled and observed weekend-weekday differences averaged over daytime hours correspond to 7.2 ± 1 and 7.6 ± 1.2. While the model reproduces the general diurnal ozone cycle and midday peak reasonably well, it underestimates absolute nighttime concentrations exhibiting a stronger diurnal cycle. We attribute this shortcoming mainly to poor knowledge and constraints on entrainment and advection processes that dominate observed nighttime distributions of ozone. While this approach has its limitations, we believe it does a sufficient job for investigating relative daytime changes of ozone concentrations caused by the weekend effect. On average, daytime weekday concentrations are about 7–8 ppbv lower than on weekends. These results can be compared to two quite contrasting areas. In Mexico City51, ozone concentrations change very little despite similar relative variations of NOx mixing ratios between weekday and weekend, which can be attributed to a completely NOx saturated environment. On the other hand, a quite pronounced variation is found in the southern California Air basin56, where average ozone concentrations are observed to change almost twice as much compared to the present study. We interpret these observations such that they reflect the change in ozone production efficiency (Fig. 4), when going from environments with NOx saturated conditions to more VOC limited conditions. In this context a number of explanations of the weekend effect on ozone mixing ratios are often discussed51: (1) different timing of NOx emissions on weekends and associated chemical repartitioning, (2) carryover of previous day pollutants at the surface and aloft, (3) higher weekend VOC emissions, (4) higher weekend photolysis frequencies due to lower aerosol loadings, (5) changes in biogenic emissions due to a different radiation regime caused by lower aerosol loadings and (6) lower overall weekend NOx emissions. We show for the first time, that, for Innsbruck, at least 85% of the NOx concentration change, leading to a 7–8 ppbv ozone increase on weekends, can be directly associated with a change of the overall local atmospheric NOx flux.
Discussion
Continued urbanization in conjunction with rapid technological changes in the mobility sector poses a challenge for accurate up to date predictions of pollutant emissions. By constraining the actual fluxes of nitrogen oxides, NMVOC, CO2 and CO into the atmosphere our measurements provide an observationally based explanation why NOx concentrations have hardly declined since the introduction of EURO 3 emission standards in Central Europe. While the technological shift towards Diesel passenger cars might have helped curb CO2 emissions through better fuel economy compared to gasoline powered cars in the past, it created a widespread problem of NO2 pollution across Europe23 that does not seem to be exclusively limited to the largest metropolitan and industrialized areas. The presented flux measurements indicate that traffic related NOx emissions in current operational air quality models can be significantly underestimated by up to a factor of 4 across countries exhibiting a sizeable fraction of Diesel powered cars in their fleet. As Diesel fuels (including bio-diesel) could account for 70% of the growth in transportation fuels by 2040, with significant demand in Asian markets according to industry projections57, a better understanding of the uncertainty in associated changes of NOx fluxes and ozone chemistry will therefore be important for future environmental impact studies. Our measurements show that projected significant decreases in European NOx emissions from the mobile transport sector will lead to conditions improving NO2 exposure limits, but could locally increase ozone levels on the short term. Concomitant reduction measures for NOx and NMVOC (CO) might therefore still prove most effective to avoid parallel increases of local ozone levels due to new NOx emission standards. This might be particularly important in areas where topographic amplification can lead to a stronger accumulation of air pollutants than over flat land and be a relevant consideration for mountainous mega-cities58. Using the observed weekend effect as proxy for underestimated NOx emissions (i.e. a factor 2–4 difference), models would overestimate P(O3) by 30–40% under the observed NOx inhibited – VOC limited regime and underestimate P(O3) downwind, once NOx concentrations fall below 2–4 ppbv. Here we demonstrate that parallel flux measurements of a wide range of chemical species can be used to benchmark urban emission sources, complement traditional approaches and significantly improve uncertainties inherent to bottom-up scaling in atmospheric chemistry models.
Methods
Instrumentation
NMVOC
A PTR-QiTOF instrument (Ionicon, Austria) was operated in hydronium mode at standard conditions in the drift tube of 112 Townsend. The instrument was set up to sample ambient air from a turbulently purged “3/8” Teflon line. Every seven hours, zero calibrations were performed for 30 minutes providing VOC free air from a continuously purged catalytical converter though a setup of software controlled solenoid valves. In addition, known quantities of a suite of VOC from a 1 ppm calibration gas standard (Apel & Riemer, USA) were periodically added to the VOC free air and dynamically diluted into low ppbv mixing ratios. NO x : A dual channel chemiluminescence instrument (CLD 899 Y; Ecophysics) was used for NO and NOx measurements. The instrument was equipped with a metal oxide converter operated at 375C. The instrument was operated in flux mode acquiring data at about 5 Hz, similar to measurements performed over a pasture59 and forest60. A NO standard was periodically introduced for calibration. Zeroing was performed once a day close to midnight. CO 2 , H 2 O: A closed path eddy covariance system (CPEC 200; short inlet, enclosed IRGA design; Campbell Scientific) measured three dimensional winds along with CO2 and H2O. An additional 3D sonic anemometer (CSAT3; Campbell Scientific) was available for turbulence measurements at an alternative height level (Fig. S3). Calibration for CO2 was performed once a day. O 3 : Ozone concentrations were obtained from a closed-path UV photometric analyser (APOA-370, Horiba, Japan); CO: CO measurements were available for a limited amount of time in August 2015. Ambient mole fractions of carbon monoxide (CO) were measured with a quantum cascade laser spectrometer (CWQC-TILDAS-76-D, Aerodyne, USA) with a 76 m path length optical cell at a wavenumber of ca. 2190 cm−1. The QCL was operated at a pressure of ca. 4 kPa using a built-in pressure controller and temperature of the optical bench and housing controlled to 35 °C. Fitting of absorption spectra at 2 Hz, storing of calculated dry mole fractions, switching of zero/calibration valves, control of pressure lock, correction for band broadening and other system controls were realized by the TDLWintel software (Aerodyne, USA) run on a PC synchronized in time with the system collecting the anemometer data using the NTP software (Meinberg, Germany).
Eddy covariance data analysis
The eddy covariance method is derived from the scalar budget equation after Reynolds decomposition, and in its simplest form for horizontally homogeneous flows normal to the surface, where the mean vertical motion of wind (\(\bar{w}\)) can be considered 0, relates the measured surface-atmosphere exchange flux (F) to the covariance between vertical wind and concentration fluctuation according to:
where w′ represents the vertical fluctuation of wind speed, and c′ the concentration fluctuation. Brackets denote the averaging interval. The ensemble average used here is 30 minutes. Fluxes were selected according to standard quality control criteria, such as raw data despiking, correcting for high and low pass filtering biases, applying a stationarity test and test on developed turbulent conditions (e.g. u* filtering)61. In addition we parsed the data to make sure that the flux footprint would reflect a representative urban area (SI).
References
Fowler, D. et al. Effects of global change during the 21st century on the nitrogen cycle. Atmos. Chem. Phys. 15, 13849–13893 (2015).
Monks, P. S. et al. Tropospheric ozone and its precursors from the urban to the global scale from air quality to short-lived climate forcer. Atmos. Chem. Phys. 15, 8889–8973 (2015).
Intergovernmental Panel on Climate Change (IPCC) “Climate Change 2014: Synthesis Report. Contribution of Working Groups I, II and III to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change”, (IPCC, 2014, Geneva, https://www.ipcc.ch/report/ar5/syr/).
Haagen-Smit, A. J., Bradley, C. E. & Fox, M. M. Ozone formation in photochemical oxidation of organic substances. Indust. and Engineering Chemistry 45, 2086–2089 (1953).
Calvert, J. G. Gaseous reactions. Annual Review of Physical Chemistry 11, 41–64 (1960).
Atkinson, R. Kinetics and mechanism of the gasphase reactions of the hydroxylradical with organic compounds under atmospheric conditions. Chem. Rev. 86, 69–201 (1985).
Leone, J. A., Flagan, R. C., Grosjean, D. & Seinfeld, J. H. An outdoor smog chamber and modeling study of toluene-NOx photooxidation. Int. J. of Chem. Kinetics 17, 177–216 (1985).
Martin, M., Pons, M., Wirtz, K., Etzkorn, T. & Senzig, J. An outdoor smog chamber study of the gas-phase chemistry of toluene and xylenes in NOx/air systems, in The oxidizing capacity of the troposphere (Proceedings of the seventh European symposium on physicochemical behavior of atmospheric pollutants, European Commission, Luxembourg, 1996) pp. 350–354.
Karl, M. et al. Kinetic Study of the OH-isoprene and O3-isoprene reaction in the atmosphere simulation chamber SAPHIR. GRL. 31, doi:10.1029/2003GL019189 (2004).
Meagher, J. F., Cowling, E. B., Fehsenfeld, F. C. & Parkhurst, W. J. Ozone formation and transport in the southeastern United States: Overview of the SOS Nashville Middle Tennessee Ozone Study. J. Geophys. Res. 103, 22213–22223 (1998).
Ridley, B. A. & Robinson, E. The Mauna-Loa-Observatory Photochmsitry Experiment. J. Geophys. Res. 97, 10285–10290 (1992).
Jacob, D. J. et al. Transport and Chemical Evolution over the Pacific (TRACE-P) aircraft mission: Design, execution, and first results. J. Geophys. Res. 108, 1–19 (2003).
Jenkin, M. E., Saunders, S. M. & Pilling, M. J. The tropospheric degradation of volatile organic compounds: A protocol for mechanism development. Atmos. Environ. 31, 81–104 (1997).
Brune, W. H. et al. Ozone production chemistry in the presence of urban plumes, Faraday Discussions, doi:10.1039/c5fd00204d (2015).
Kleinman, L. I. et al. Dependence of ozone production on NO and hydrocarbons in the troposphere. GRL 24, 2299–2302 (1997).
Im, U. et al. Evaluation of operational on-line-coupled regional air quality models over Europe and North America in the context of AQMEII phase 2. Part I: Ozone. Atmos. Environm. 115, 404–420 (2015).
Ehhalt, D. & Prather, M. “Atmospheric Chemistry and Greenhouse Gases” (Intergovernmetnal Panel on Climate Change, 3rd assessment report, Chapter 4, 2001; http://www.grida.no/publications/other/ipcc_tar/).
de Gouw, J. A., Parrish, D. D., Frost, G. J. & Trainer, M. Reduced emissions of CO2, NOx, and SO2 from US power plants owing to switch from coal to natural gas with combined cycle technology. Earths Future 2, 75–82 (2014).
Ehlers, C. et al. Twenty years of ambient observations of nitrogen oxides and specified hydrocarbons in air masses dominated by traffic emissions in Germany, Faraday Discussions, doi:10.1039/c5fd00180c (2015).
Carslaw, D. C. & Beevers, S. D. New Directions: Should road vehicle emissions legislation consider primary NO2? Atmos. Environ. 38, 1233–1234 (2004).
Kiendler-Scharr, A. et al. Ubiquity of organic nitrates from nighttime chemistry in the European submicron aerosol. GRL 43, 7735–7744 (2016).
U. S. Environmental Protection Agency, (EPA), “NAAQS Scientific and Technical Information”, https://www.epa.gov/naaqs/nitrogen-dioxide-no2-primary-air-quality-standards retrieved on 07-01-2016, (2016).
Kiesewetter, G. et al. Modelling NO2 concentrations at the street level in the GAINS integrated assessment model: projections under current legislation. Atmos. Chem. Phys. 14, 813–829 (2014).
European Environment Agency, “Air quality in Europe – 2015” (Tech. Rep. 5/2015, ISBN 978-92-9213-702-1, doi:10.2800/62459, 2015).
Giordano, L. et al. Assessment of the MACC reanalysis and its influence as chemical boundary conditions for regional air quality modeling in AQMEII-2. Atmos. Environ. 115, 371–388 (2015).
Heckel, A. et al. Influence of low spatial resolution a priori data on tropospheric NO2 satellite retrievals. Atmos. Meas. Technol. 4, 1805–1820 (2011).
Salvo, A. & Geiger, F. M. Reduction in local ozone levels in urban Sao Paulo due to a shift from ethanol to gasoline use. Nature. Geoscience. 7, 450–458 (2014).
Madronich, S. Atmospheric chemistry: Ethanol and ozone. Nature Geoscience 7, 395–397 (2014).
Carslaw, D. C. Evidence of an increasing NO2/NOx emissions ratio from road traffic emissions. Atmos. Environm 39, 4793–4802 (2005).
Bundesministerium für Verkehr und digitale Infrastruktur (BMVI), “Bericht der Untersuchungskommission “Volkswagen” 2016” (https://www.bmvi.de/SharedDocs/DE/Publikationen/LA/bericht-untersuchungskommission-volkswagen.html, retrieved 2016) [the easiest access to this source is via the URL].
Franco, V., Posada Sanches, F., German, J. & Mock, P. “Real-world exhaust emissions from modern diesel cars, Part 2” (Tech. Report: The International Council on Clean Transportation, www.theicct.org, Washington, DC, 2014).
Lee, J. et al. Measurement of NOx fluxes from a tall tower in central London, UK and comparison with emission inventories. Environ. Sci. & Tech 49, 1025–1034 (2015).
Christen, A. Atmospheric measurement techniques to quantify greenhouse gas emissions from cities. Urban Climate 10, 241–260 (2014).
Nemitz, E., Hargreaves, K. J., McDonald, A. G., Dorsey, J. R. & Fowler, D. Meteorological measurements of the urban heat budget and CO2 emissions on a city scale. Environ. Sci. Tech. 36, 3139–3146 (2002).
Velasco, H. E. et al. Eddy covariance flux measurements of pollutant gases in urban Mexico City. Atmos. Chem. Phys. 9, 7325–7342 (2009).
Valach, A. C., Langford, B., Nemitz, E., MacKenzie, A. R. & Hewitt, C. N. Seasonal and diurnal trends in concentrations and fluxes of volatile organic compounds in central London. Atmos. Chem. Phys. 15, 7777–7796 (2015).
Karl, T. et al. Emissions of volatile organic compounds inferred from airborne flux measurements over a megacity. Atmos. Chem. Phys. 9, 271–285 (2009).
Langford, B., Davison, B., Nemitz, E. & Hewitt, C. N. Mixing ratios and eddy covariance flux measurements of volatile organic compounds from an urban canopy (Manchester, UK). Atm. Chem. Phys 9, 1971–1987 (2009).
Rantala, P. et al. Anthropogenic and biogenic influence on VOC fluex at an urban bachground site in Helsinki, Finland. Atmos. Chem. Phys. 16, 7981–8007 (2016).
Marr, L. C., Moore, T. O., Klapmeyer, M. E. & Killar, M. B. Comparison on NOx fluxes measured by eddy covariance to emission inventories and land use. Environ. Sci. & Techn 47, 1800–1808 (2013).
Vaughan, A. R. et al. Spatially resolved flux measurements of NOx from London suggest significantly hagher emissions than predicted by inventories. Faraday Discussions 189, 455–472 (2016).
EUROSTAT, “Energy, transport and environment indicators” (Tech. Report 201, European Commission, ec.europa.eu, ISBN 978-92-79-49471-0, doi:10.2785/547816, 2015).
Vergreiner, I. & Dreiseitl, E. Valley winds and slope winds – observations and elementary thoughts. Meteorol. Atmos. Phys. 36, 24–286 (1987).
Coggon, M. M. et al. Emissions of nitrogen-containing organic compounds from the burning of herbaceous and arboraceous biomass: Fuel composition dependence and the variability of commonly used nitrile tracers. GRL, doi:10.1002/2016GL070562 (2016).
Popa, M. E. et al. Vehicle emissions of greenhouse gases and related tracers from a tunnel study: CO: CO2, N2O: CO2, CH4: CO2, O2:CO2 ratios, and the stable isotopes 13C, 18O in CO2 and CO. Atmos. Chem. Phys. 14, 210–2123 (2014).
Papadimitriou, G. et al. “TRACCS - Transport data collection supporting the quantitative analysis of measures relating to transport and climate change” (Tech. Rep. EMISIA No. 13.RE.025.V1, 2013).
Harley, R. A., Marr, L. C., Lehner, J. K. & Giddings, S. N. Changes in motor vehicle emissions on diurnal to decadal time scales and effects on atmospheric composition. Environ. Sci. Tech. 39, 5356–5362 (2005).
Lamarque, J. F. et al. Historical (1850–2000) gridded anthropogenic and biomass burning emissions of reactive gases and aerosols: methodology and application. Atmos. Chem. Phys. 10, 7017–7039 (2010).
Vestreng, V. et al. Evolution of NOx emissions in Europe with focus on road transport control measures. Atmos. Chem. Phys 9, 1503–1520 (2009).
Reis, S. et al. A. Road traffic emissions –predictions of future contributions to regional ozone levels in Europe. Atmos. Environ. 34, 4701–4710 (2000).
Stephens, S. et al. Weekly patterns of Mexico City’s surface concentrations of CO, NOx, PM10 and O3 during 1986–2007. Atmos. Chem. Phys. 8, 5313–5325 (2008).
Hellen, H., Hakola, H. & Laurila, T. Determination of source contributions of NMHCs in Helsinki (60 degrees N, 25 degrees E) using chemical mass balance and the Unmix multivariate receptor models. Atmos. Environm 37, 1413–1424 (2003).
Borbon, A. et al. Emission ratios of anthropogenic volatile organic compounds in northern mid-latitude megacities: Observations versus emission inventories in Los Angeles and Paris. J. Geophys. Res. 118, 2041–2057 (2013).
Spirig, C., Neftel, A., Kleinman, L. I. & Hjorth, J. NOx versus VOC limitation of O3 production in the Po valley: Local and integrated vew based on observations, J. Geophys. Res. 107, doi:10.1029/2001JD000561 (2002)
Wolfe, G. & Thornton, J. The Chemistry of atmosphere-forest exchange (CAFÉ) model – Part 1: Model description and characterization. Atmos. Chem. Phys. 11, 77–101 (2011).
Kim, S. W. et al. Modeling the weekly cycle of NOx and CO emissions and their impacts on O3 in the Los Angeles – South Coast air Basin during the CalNex 2010 field campaign. J. Geophys. Res. Atmos. 121, 1340–1360 (2016).
ExxonMobil, “The Outlook for Energy: A view to 2040” (Techn. Report, http://corporate.exxonmobil.com/en/energy/energy-outlook/introduction/a-view-to-2040 retrieved on 1st of August 2016).
Molina, L. T. et al. An overview of the MILAGRO 2006 Campaign: Mexico City emissions and their transport and transformation. Atmos. Chem. Phys. 10, 8697–8760 (2010).
Marx, O., Brümmer, C., Ammann, C., Wolff, V. & Freibauer, A. TRANC – a novel fast-response converter to measure total reactive atmospheric nitrogen. Atmos. Meas. Tech. 5, 1045–1057 (2012).
Rummel, U., Ammann, C., Gut, A., Meixner, F. X. & Andreae, M. O. Eddy covariance measruements of nitric oxide flux within an Amazonian rainforest, J. Geophys. Res. 107, doi:10.1029/2001JD000520 (2002).
Foken, T., Leuning, R., Oncley, S. P., Mauder, M. & Aubinet, M. Corrections and data quality, Eddy Covariance: A Practical Guide to Measurement and Data Analysis (Kluwer Academic Publishers, Dortrecht, ed. 1, 2010), pp. 85–131.
Acknowledgements
T.K. received support from the EC Seventh Framework Program (Marie Curie Reintegration Program, “ALP-AIR”, grant no. 334084).
Author information
Authors and Affiliations
Contributions
Karl, T. and Graus, M. conceived the experiment. Karl, T., Graus, M., Striednig, M., Lamprecht C., Hammerle, A., Wohlfahrt, G., Held, A., von der Heyden, L., and Deventer J. designed and performed the INNAQS field experiment, interpreted the data and wrote the manuscript with inputs from all co-authors; Lee, J. provided input on the interpretation of NOx flux data; Krismer A. provided air quality data, Feichter R. performed and analyzed traffic flow data; Haun C. provided the bottom up stationary emission source data; Karl T. performed the model simulations.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Karl, T., Graus, M., Striednig, M. et al. Urban eddy covariance measurements reveal significant missing NOx emissions in Central Europe. Sci Rep 7, 2536 (2017). https://doi.org/10.1038/s41598-017-02699-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-02699-9
This article is cited by
-
Real-world vehicle emissions as measured by in situ analysis of exhaust plumes
Environmental Science and Pollution Research (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.