Abstract
Pesticides can pose environmental risks, and a common neonicotinoid pesticide, thiamethoxam, decreases homing success in honey bees. Neonicotinoids can alter bee navigation, but we present the first evidence that neonicotinoid exposure alone can impair the physical ability of bees to fly. We tested the effects of acute or chronic exposure to thiamethoxam on the flight ability of foragers in flight mills. Within 1 h of consuming a single sublethal dose (1.34 ng/bee), foragers showed excitation and significantly increased flight duration (+78%) and distance (+72%). Chronic exposure significantly decreased flight duration (−54%), distance (−56%), and average velocity (−7%) after either one or two days of continuous exposure that resulted in bees ingesting field-relevant thiamethoxam doses of 1.96–2.90 ng/bee/day. These results provide the first demonstration that acute or chronic exposure to a neonicotinoid alone can significantly alter bee flight. Such exposure may impair foraging and homing, which are vital to normal colony function and ecosystem services.
Similar content being viewed by others
Introduction
Pollinators play an important environmental role by providing essential ecosystem services1. In particular, the honey bee, Apis mellifera L., 1758, is an important global pollinator of crops and native plants1. The decline of managed honey bee colonies has therefore raised concern about ecological impacts, crop production, food security and human welfare2. Although beekeepers can multiply colonies to offset some of these losses, beekeeping is becoming increasingly difficult and expensive2. Multiple factors, including disease and pesticides, contribute to poor honey bee health3. Among pesticides, attention has focused on the neonicotinoids4, neurotoxic insecticides that are globally used on multiple crops5. Neonicotinoids are environmentally persistent and systemic: they can be found in the nectar, pollen, and guttation droplets that bees collect6, 7. Moreover, exposure to even low concentrations of neonicotinoids can harm bee health via synergistic interactions between multiple stressors3, 7, 8.
Neonicotinoids and their degradation products are agonists of insect nicotinic acetylcholine receptors5 and have a wide variety of neural effects8, 9. These compounds can therefore harm bee foraging10,11,12,13, homing14,15,16,17, locomotion18, 19, navigation20, and colony health17. Although neonicotinoids are partially restricted in Europe21, they are still commonly used worldwide5, 22, and thus their sublethal impacts deserve further study. We focused on thiamethoxam (TMX), a second generation neonicotinoid that is widely used5 and persistent6, and is thus frequently found in multiple environmental substrates such as nectar, pollen, guttation, water, and bee hives6, 23,24,25.
Henry et al.16, 17 demonstrated that TMX reduced forager return rates to the nest, raising the interesting possibility that TMX impairs navigation, flight ability, or both. Subsequently, researchers demonstrated that sublethal doses of three different neonicotinoids (clothianidin, imidacloprid and thiacloprid) could impair honey bee navigation20. TMX may similarly impair navigation26, but we tested the hypothesis that it reduces the physical ability of bees to fly. Flight is essential for colony fitness and health because bees fly to collect all of their food and water. Blanken et al.27 recently showed that bees exposed to Varroa destructor and imidacloprid over 13 weeks had a decreased ability to fly. Because there was no effect of imidacloprid alone on bee flight ability27 it was not clear if neonicotinoids alone can reduce bee flight ability. TMX can alter forager flight muscle temperature28, and the results of Henry et al.16, 17 suggested that TMX could impair honey bee flight: we therefore focused on TMX, using tethered bees flying on flight mills to test their physical ability to fly29,30,31,32,33, measuring flight distance, duration, and velocity in exposed and control bees.
Results
Acute exposure elicited excitation: increased flight duration and distance
Each bee flew twice (before and after treatment) in this experiment. Control bees (both phases) flew for 1390 ± 168 seconds (mean ± s.e.m., 23 min) and covered 2145 ± 294 m (2.1 km) at an average velocity of 1.49 ± 0.05 m/s (5.4 km/h) and maximum velocity of 1.78 ± 0.05 m/s (6.4 km/h).
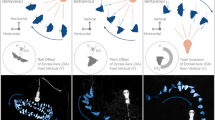
There was a significant effect of the interaction treatment × flight period on duration (p = 0.024, Fig. 1a, main effects in Table 1) because TMX-treated bees flew 78% longer after they consumed TMX (LS Means contrast test: F 1,37 = 10.91, p = 0.002, Fig. 1a). As expected, control bees flew for similar durations in both phases (contrast test: F 1,37 = 0.10, p = 0.75).
The effects of (a) acute or (b) chronic exposure to thiamethoxam (TMX) on forager flight ability. (a) In the acute experiment, we recorded flight duration, distance, mean velocity and maximum velocity before and after treatment; white bars are the control group ( ), grey bars are the TMX group (
), grey bars are the TMX group ( ); the different letters indicate significant differences (LS Means contrast tests comparing before and after periods; N
control group, before
= 16, N
control group, after
= 16, N
TMX group, before
= 23, N
TMX group, after
= 23). (b) In the chronic experiment, we grouped the TMX daily doses (N
TMX daily doses
= 46) in 5 TMX daily dose ranges (0, ≤1.95, ≤2.90, ≤3.71, ≤4.53 ng/bee/day). We pooled data from both days of exposure (1 or 2 days) because there was no significant effect of the number of days of exposure. Different shading reflects different daily dose ranges of TMX and different letters indicate significant differences (Least-Square Means contrast tests; N
control
= 94, N
32.5 ppb
= 44, N
45 ppb
= 75). In the x-axis, we report the upper value of each bin range of TMX daily doses. Error bars show standard errors.
); the different letters indicate significant differences (LS Means contrast tests comparing before and after periods; N
control group, before
= 16, N
control group, after
= 16, N
TMX group, before
= 23, N
TMX group, after
= 23). (b) In the chronic experiment, we grouped the TMX daily doses (N
TMX daily doses
= 46) in 5 TMX daily dose ranges (0, ≤1.95, ≤2.90, ≤3.71, ≤4.53 ng/bee/day). We pooled data from both days of exposure (1 or 2 days) because there was no significant effect of the number of days of exposure. Different shading reflects different daily dose ranges of TMX and different letters indicate significant differences (Least-Square Means contrast tests; N
control
= 94, N
32.5 ppb
= 44, N
45 ppb
= 75). In the x-axis, we report the upper value of each bin range of TMX daily doses. Error bars show standard errors.
Similarly, there was a significant effect of the interaction treatment × flight period on distance (p = 0.025, Fig. 1a, Table 1). Control bees flew similar distances in the before and after phases (contrast test: F 1,37 = 0.10, p = 0.75), but treated bees flew 72% farther after they consumed TMX (contrast test: F 1,37 = 10.59, p = 0.002).
There were no significant effects of treatment, flight period or their interaction on mean velocity or maximum velocity (p > 0.45, Fig. 1a, Table 1).
Chronic exposure to TMX reduced flight ability
In this experiment, each bee flew only once after either one or two days of chronic exposure to TMX. The number of days of exposure had no significant effects on flight (p ≥ 0.07, Table 1). Control bees flew for 2036 ± 218 seconds (34 min) and travelled 3178 ± 357 m (3.2 km) with an average velocity of 1.51 ± 0.03 m/s (5.4 km/h) and a maximum velocity of 1.79 ± 0.03 m/s (6.4 km/h). The daily doses of TMX ingested significantly reduced flight ability for each flight parameter (p < 0.002, Table 1).
The TMX daily dose ingested significantly decreased flight duration (p < 0.0001, Table 1). For each 1 ng of TMX ingested daily by a forager, flight duration decreased by 20% (Mixed ModelREML estimate, based on N TMX daily doses = 46). When we grouped bees by the actual TMX daily dose consumed (Fig. 1b), the foragers that ingested 1.96–2.90 ng/bee/day spent significantly less time flying than control bees (−54%, contrast test: F 1,169 = 4.82, p = 0.029).
The TMX daily dose intake significantly reduced the total flight distance (p < 0.0001, Table 1). For each 1 ng of TMX ingested daily by the forager, their flight distance decreased by 23% (Mixed ModelREML estimate, based on N TMX daily doses = 46). When we grouped bees by the TMX daily dose actually consumed (Fig. 1b), those that ingested 1.96–2.90 ng/bee/day flew significantly shorter distances compared to control (−56%, contrast test: F 1,207 = 1.10, p = 0.019).
The daily dose of TMX ingested significantly reduced the mean velocity of the flights (p = 0.002, Table 1). We estimated that foragers flew 0.15 km/h slower for each 1 ng of TMX ingested daily (Mixed ModelREML estimate, based on N TMX daily doses = 46). When we grouped bees by the TMX daily dose actually consumed (Fig. 1b), doses from 2.90–3.71 ng/bee/day significantly reduced mean velocity as compared to control (−7%, contrast test: F 1,202 = 4.43, p = 0.037).
TMX significantly reduced flight maximum velocity (p = 0.002, Table 1). For each 1 ng of TMX ingested daily by the forager, their flight maximum velocity decreased by 0.15 km/h (Mixed ModelREML estimate, based on N TMX daily doses = 46). When we grouped bees by the TMX daily dose consumed (Fig. 1b), doses from 1.96–2.90 ng/bee/day significantly reduced maximum velocity as compared to control (−6%, contrast test: F 1,197 = 5.00, p = 0.026).
Daily consumption of the higher TMX sucrose solution was significantly higher than consumption of pure sucrose solution (+7%; control = 73 ± 15 mg/bee/day; 32.5 ppb = 75 ± 16 mg/bee/day; 45 ppb = 78 ± 14 mg/bee/day; Kruskal-Wallis Rank Sums, χ2 = 7.40, p = 0.02; Wilcoxon paired-sample test, 0 versus 45 ppb: Z = 2.66, p = 0.008).
There were no significant effects of the interaction TMX daily doses × days of exposure (p > 0.11). There was no significant effect of TMX treatment (Fisher exact test, p = 0.065) on the number of bees that did not fly (23% over all treatments).
Discussion
We present the first results demonstrating that sublethal acute or chronic neonicotinoid exposure is sufficient to significantly alter honey bee flight ability — affecting flight distance, flight duration, and flight velocity. Essentially, TMX had an excitatory short-term effect and a depressive longer-term effect. In the acute experiment, foragers consumed a single sublethal dose of 1.34 ng and subsequently increased their mean flight duration and flight distance by 78% and 72% in comparison with control bees, respectively (Fig. 1a). However, this increase in flight duration and distance is likely not beneficial because, at similar doses, TMX and other neonicotinoids cause flight disorientation20, 26, 34. Bees that fly more erratically for greater distances may thereby decrease their probability of returning home. This decline in the proportion of TMX-treated bees returning to the nest has been demonstrated, at the colony level, in two experiments by Henry et al.16, 17. In a similar study, Thompson et al.35 found no significant effect of TMX on honey bee homing ability. However, the study of Thompson et al.35 was carried out at a smaller spatial and temporal scale (i.e. ca. 1–2 field exposure units, with a single 2-ha treated field as compared to 63 field exposure units, with a total of 288-ha treated fields in Henry et al.17).
Chronic exposure is possible because foragers that consume a single sublethal dose of TMX can survive16 and return to forage at the same contaminated food sources. In fact, recent evidence shows that honey bees prefer sucrose solutions containing TMX or other neonicotinoids over pure sucrose36. Bees may therefore increase their consumption of contaminated food, exacerbating pesticide exposure. In our chronic experiment, bees drank significantly more sucrose solution when it contained TMX (+7%). We found that chronic exposure, which led to daily intakes of 1.96–2.90 ng TMX/bee/day, significantly decreased flight duration, distance, and velocity (Fig. 1b). TMX had the same significant negative effects on bees after one or two days of exposure (Table 1). Thus, chronic exposure to field-relevant daily doses of TMX (<2.94 ng/bee daily) over a single day was sufficient to impair bee flight ability.
Honey bees can forage up to 13.5 km from the colony, depending on forage availability and quality, and colony health37. Steffan-Dewenter and Kuhn38 and Visscher and Seeley39 estimated the mean foraging distance to be 1.5 and 2.3 km from the colony, corresponding to round-trip foraging flights of 3.0–4.6 km. Our control foragers flew an average of 2.1 km (acute experiment) and 3.2 km (chronic experiment). These results are similar to previous flight mill studies that used our same acute feeding procedure: control bees flew 1.8 km30, 2.0 km29 and 2.2 km27.
Chronic TMX exposure to 1.96–2.90 ng/bee per day reduced bee flight distance by 56% (corresponding to a 1.8 km reduction), which could lead to a decline of the overall foraging area by 79% (calculations given in the Supplementary Methods). Each 1 ng of TMX ingested per day reduced flight distance by 23% (model regression coefficient), corresponding to an estimated decline of the overall foraging area by 37% (calculations in the Supplementary Methods). TMX (0.2–2 ng/bee) can alter foragers thorax temperature up to 1 day after exposure28. This effect of TMX on thoracic flight muscles could impair flight because bee flight power is related to muscle temperature40. Tison et al.34 showed decreased honey bee foraging after chronic exposure to the neonicotinoid thiacloprid (4.5 ppm). Based upon our results, we likewise predict that bees foraging on neonicotinoid-treated fields for just one or two days will then fly more slowly and in a reduced area. This behavioural alteration should reduce the pollination service provided to plants, nectar and pollen collection for the colony, and the nutritional biodiversity of collected pollen for the colony.
Blanken et al.27 demonstrated a synergistic effect between imidacloprid (5.98 ng/mL of sucrose solution, over a 13-week period) and Varroa infestation on bee flight ability. They showed that foragers from colonies exposed to high levels of Varroa significantly decreased flight distance (−3% and −1% when respectively fed 1 M and 2 M pure sucrose solution, significant sucrose concentration effect). This effect was larger when the foragers were captured from colonies infected with V. destructor and chronically exposed to the neonicotinoid imidacloprid (−30% and −17% flight distance decreases when respectively fed 1 M and 2 M sucrose solution). However, there was no significant effect of imidacloprid alone on honey bee flight ability, and flight velocity was not affected even when bees were exposed to both Varroa and imidacloprid. We used healthy colonies that were not measurably infested with Varroa. Our results are therefore the first demonstration that a neonicotinoid can impair flight ability in bees that do not come from colonies heavily parasitized with Varroa.
Acute exposure to TMX caused excitation (hyperactivity, increased flight ability), while the chronic exposure produced depression (hypoactivity, reduced flight ability). Why did acute vs. chronic exposures lead to opposite effects? Short-term hyperactivity may lead to longer-term muscular exhaustion or energetic depletion. Neonicotinoids can impair bee energy metabolism41, and neonicotinoid contaminated bees have a reduced nutritional status (glycogen, lipid, and protein content) in the field42. The increase in sucrose consumption observed in our study may reflect bees attempting to compensate for energy deficits. For example, TMX doses of 0.2–2 ng/bee increased or decreased forager thorax temperatures depending on dose and time from exposure28. Cockroaches, Blattella germanica, showed the same response pattern to the neonicotinoid imidacloprid: individuals were hyper-responsive and hyperactive shortly after treatment, but decreased activity later on43. Suchail et al.44 also found differences between the acute and chronic effects of a neonicotinoid and its metabolites on bees. In their study, imidacloprid rapidly caused excitation, hyper-responsiveness and hyperactivity after intoxication. However, these symptoms gradually disappeared and, after several hours, the bees showed a decreased activity, becoming hypo-responsive and hypoactive. After a chronic exposure to the neonicotinoid imidacloprid (10 ppb), bumblebee foraging activity increased in the short-term, but was impaired in the long-term13. Finally, TMX degrades over time and its metabolites (including the main metabolic by-product clothianidin) could interact or differentially affect flight performance as a result of chronic exposure5. Clothianidin is also commonly used as a pesticide and acts on nicotinic acetylcholine receptors (nAChR). However, clothianidin targets different nAChR subtypes than TMX5 and possibly has dissimilar effects. The direct effects of acute and chronic exposure to neonicotinoids and similar compounds on pollinator flight ability therefore deserve further study.
Methods
This study was conducted from September 2012 to September 2014 at University of California San Diego (UCSD), Division of Biological Sciences (La Jolla, CA, USA) with 19 healthy honey bee colonies (A. mellifera ligustica Spinola, 1806, 10 frames per colony) housed at an apiary in the UCSD Biology Field Station. We used standard inspection techniques45 to confirm that our colonies did not have measurable Varroa infestations.
Preparing and flying bees
We tested active foragers captured upon their return to the nest. Their flight ability was tested using a modified flight mill (Fig. 2) that we built based upon the designs and software of Smith and Jones46. Details on the flight mill, the honey bee preparation and the flying procedure are in the Supplementary Methods.
The flight mill used to test the flight ability of tethered forager bees. Foragers were attached to the wire flight mill arm through their tube harness, previously placed on top of their thorax. Once on the flight mill, bees could fly and their flight parameters were recorded by the sensor. The red LED is only triggered to light by the small triggering magnet opposite the bee, and therefore this weak red flash is not visible to the tested individual.
Pesticide doses and concentrations
Field-relevant pesticide doses and concentrations vary widely across space and time8. In our experiments, we used foragers fed with sucrose solution, and thus TMX levels in nectar provide the most realistic residue levels. However, honey bees can be exposed to higher concentrations of TMX in guttation droplets (100 ppm25), that foragers can collect from TMX seed-treated plants such as corn and oilseed rape47, although this may be a minor route of exposure. Even higher concentrations of TMX have been reported in bee tissue (310 ppb23).
We based the acute and chronic experiments and their respective analyses on the actual dose of TMX consumed by each bee. All TMX doses tested were lower than the worst case scenario thresholds, and did not increase mortality as compared to controls. The worst case scenario calculations and dose-thresholds for acute and chronic exposures were respectively defined by the European Food Safety Authority (EFSA)48.
In the acute exposure experiment, we fed the bees a single dose of TMX (1.34 ng). This dose was used by Henry et al.16 who found that it impaired forager homing ability. Using the same dose allowed us to test the hypothesis that neonicotinoids could directly impair forager flight ability. This dose is 3.7 times lower than the LD50 of TMX49 and does not significantly increase mortality as compared to controls16. Although 1.34 ng was subsequently criticized for not being field realistic50, we calculated (based upon EFSA48) that foragers can acutely consume up to 1.80 ng TMX/bee in 1 h of foraging for nectar (10% sugar w/w, oilseed rape51, 52) with a 15 ppb TMX concentration (transplant-drip application24). This worst case scenario considered the field-realistic amount of nectar that foragers consume in 1 h of foraging activity based upon their energy requirements. We consider 15 ppb to be a fairly high TMX concentration24, but even higher concentrations of TMX in nectar were found by Sanchez-Bayo and Goka53 (17 ppb), Dively and Kamel24 (19 ppb, including TMX metabolites), and Stoner and Eitzer54 (20 ppb, see reviews by Bonmatin et al.6 and Godfray et al.7). Transplant-drip applications are typically a short-term contamination route for bees, we therefore used this 15 ppb level to calculate the worst case acute exposure scenario: a 1 h short-term exposure to the contaminated nectar48. In the acute experiment, we thus tested a sublethal dose that is lower than the worst-case scenario (<1.80 ng/bee/1 h) in which bees foraged for 1 h on nectar that was contaminated by TMX after a transplant-drip application.
In the chronic exposure experiment, we tested a broad range of TMX daily doses (N TMX daily doses = 46, RangeDaily doses 1.26–4.53 ng/bee/day, MeanDaily doses = 3.1 ± 0.1 ng/bee/day) that resulted from feeding bees different concentrations of TMX. These daily doses reflected actual TMX consumption per bee cage. To identify the lowest TMX dose that significantly altered bee flight, we grouped the TMX daily doses into five bins that each spanned the same dose range (0, <1.95, <2.90, <3.71, and <4.53 ng/bee/day). EFSA estimated that foragers could consume up to 6.66 ng TMX/bee/day in a worst-case scenario48. This calculation considers the field-realistic amount of nectar consumed by foragers based upon their energy requirements for daily foraging activity, the sucrose content of nectar (i.e. oilseed rape, 10%, w/w51, 52) and the highest TMX concentration found in nectar to which bees could be chronically exposed for at least 2 days (i.e. seed treatment, 5 ppb48). In our experiments, foragers consumed TMX daily doses that were always lower than 6.66 ng TMX/bee/day. Furthermore, the foragers grouped in the first three bins (Fig. 1) consumed TMX daily doses that were lower than 2.94 ng/bee/day, which is the field-relevant amount of TMX that foragers can ingest when foraging on seed-treated oilseed rape producing nectar containing 20% sugar and 5 ppb TMX48. All tested bees remained alive throughout the experiment. Foragers have a lower sucrose requirement when incubated in cages, compared to the field, because of their reduced locomotor activity in restricted environments. This leads to lower daily sucrose consumption in cages. To test field-relevant TMX daily doses approaching a realistic worst-case scenario, foragers were provided with TMX solutions that were more concentrated (32.5 ppb or 45 ppb) than those typically found in field nectar after seed treatments. However, we focused on analyses on the field-realistic TMX daily doses consumed by our bees.
We used analytical grade TMX (CAS#153719-23-43, Sigma Aldrich 37924-100MG-R) prepared as a 25 mg/L stock solution in double-distilled H20, and maintained at 4 °C inside a bottle completely wrapped in aluminium foil to avoid light degradation6. The solutions that we fed to bees were prepared daily by diluting the stock solution with 2.0 M glucose or 1.8 M sucrose solution for the acute and chronic experiments, respectively. These pesticide concentrations were not verified with additional chemical analyses. The rationale for using these different sugars and these concentrations is given below.
Acute experiment
We compared the flights of bees before and after treatment. Immediately after the first flight, bees were given one of two treatments: either 10 µL of pure 2.0 M glucose solution (control treatment) or 2.0 M glucose solution with TMX (acute pesticide treatment, see above). We waited 40 min for pesticide absorption before testing their flight (similar to Henry et al.16). The density of 2.0 M glucose solution at 20 °C and 1 ATM is 1.131 kg/L55, and thus this dose corresponds to a solution of 118 ppb, 134 µg/L and 459 nmol/L. We used glucose because it is rapidly metabolized by bees and provides faster energy recovery than sucrose29.
After feeding, we placed each bee into a separate cage to prevent food exchange with other bees and maintained them in an incubator at 30 ± 1 °C, 60–70% RH, with no food for 40 min before testing their final flight. We tested 37 bees from nine colonies.
Chronic experiment (1-day and 2-day exposures)
Bees can be chronically exposed if they continue to forage over multiple days at a food source with pesticide. We therefore tested the chronic effects of TMX. We determined how continuous exposures over different days of exposure (1 day or 2 days) would affect flight. Unlike the acute experiment, all flights occurred after pesticide treatment because we allowed bees to chronically feed from sucrose solution with pesticide.
After collection, forager bees were incubated with 1.8 M sucrose solution ad libitum containing either 0, 32.5 or 45.0 ppb of TMX, corresponding respectively to 0, 40, 55 µg/L and 0, 137, 190 nmol/L. The density of 1.8 M sucrose solution at 20 °C and 1 ATM is 1.230 kg/L55. Each day, we weighed the sucrose syringe and calculated the average sucrose and TMX consumption per cage per 24 hours and, consequently, per bee. Separately, we used 10 cages maintained in identical conditions but without bees, to measure the average mass loss (<1%) due to evaporation from the syringes. We accounted for this evaporative loss in our calculations. We tested 213 bees from 19 colonies.
Statistical analyses
To analyse the results of the acute experiment, we used Repeated-Measures Analysis of Variance (ANOVA) with a REML algorithm to test the following fixed effects: treatment (control vs. pesticide-treated bees), flight period (before vs. after treatment), and their interaction on the duration (min), distance (m), mean velocity and maximum velocity (km/h) of bee flights. Colony was included as a random grouping variable. We log-transformed distance and duration and used residuals analysis to confirm that our data met parametric assumptions. Significant effects were further analysed with post-hoc Least-Square Means contrast tests.
For the chronic experiment, we used a Mixed Model and tested one continuous effect (TMX daily doses, N TMX daily doses = 46), one fixed effect (days of exposure, 1 vs. 2), the interaction TMX daily doses × days of exposure, and colony (N Colony = 19) as a random grouping variable (REML algorithm). Based on their actual TMX consumption, the bees were grouped into five bins that spanned the same dose range (Fig. 1b; 0, ≤1.95, ≤2.90, ≤3.71, ≤4.53 ng/bee/day). These ranges differ slightly from a span of 0.82 ng because we used actual consumption values to delineate the bin boundaries. We then determined the minimum dose that was significantly different from control using the Least-Square Means contrast tests and visual data inspection. To estimate the dose effect of TMX on flight parameters, we used Mixed Model estimates and assumed a linear relationship between dose and flight parameters. We log-transformed distance and duration and used residuals analysis to confirm that our data met parametric assumptions. We used the Freeman-Halton extension of the Fisher exact probability test (2 × 3, two-tailed) to test the effect of TMX treatment on the number of bees that did not fly56, 57. Sucrose consumption data were not normally distributed, and we therefore used a Kruskal-Wallis Rank Sums test to assess the effect of treatment on sucrose consumption and made limited post-hoc comparisons with Wilcoxon paired-sample tests.
We used JMP v10.0 statistical software and report mean ± 1 standard error (s.e.m.). We used an alpha value of 0.05. We applied stepwise model simplification, building models with all interactions and then removing them if they were not significant. The main results of the acute and chronic experiments are summarized in Table 1.
References
Klein, A.-M. et al. Importance of pollinators in changing landscapes for world crops. Proc. Biol. Sci. 274, 303–313, doi:10.1098/rspb.2006.3721 (2007).
Potts, S. G. et al. Global pollinator declines: trends, impacts and drivers. Trends Ecol. Evol. 25, 345–353, doi:10.1016/j.tree.2010.01.007 (2010).
Sánchez-Bayo, F. et al. Are bee diseases linked to pesticides? — A brief review. Environ. Int. 89–90, 7–11, doi:10.1016/j.envint.2016.01.009 (2016).
Stokstad, E. Field research on bees raises concern about low-dose pesticides. Science (80). 335, 1555–1555, doi:10.1126/science.335.6076.1555 (2012).
Simon-Delso, N. et al. Systemic insecticides (neonicotinoids and fipronil): trends, uses, mode of action and metabolites. Environ. Sci. Pollut. Res. 22, 5–34, doi:10.1007/s11356-014-3470-y (2014).
Bonmatin, J.-M. et al. Environmental fate and exposure; neonicotinoids and fipronil. Environ. Sci. Pollut. Res. Int. 22, 35–67, doi:10.1007/s11356-014-3332-7 (2014).
Godfray, H. C. J. et al. A restatement of recent advances in the natural science evidence base concerning neonicotinoid insecticides and insect pollinators. Proc. R. Soc. B 282, 20151821–2015, doi:10.1098/rspb.2015.1821 (2015).
Pisa, L. W. et al. Effects of neonicotinoids and fipronil on non-target invertebrates. Environ. Sci. Pollut. Res. Int. 22, 68–102, doi:10.1007/s11356-014-3471-x (2014).
Belzunces, L. P., Tchamitchian, S. & Brunet, J.-L. Neural effects of insecticides in the honey bee. Apidologie 43, 348–370, doi:10.1007/s13592-012-0134-0 (2012).
Schneider, C. W., Tautz, J., Grünewald, B. & Fuchs, S. RFID tracking of sublethal effects of two neonicotinoid insecticides on the foraging behavior of Apis mellifera. PLoS One 7, e30023, doi:10.1371/journal.pone.0030023 (2012).
Yang, E. C., Chuang, Y. C., Chen, Y. L. & Chang, L. H. Abnormal foraging behavior induced by sublethal dosage of imidacloprid in the honey bee (Hymenoptera: Apidae). J. Econ. Entomol 101, 1743–8, doi:10.1603/0022-0493-101.6.1743 (2008).
Scholer, J. & Krischik, V. Chronic exposure of imidacloprid and clothianidin reduce queen survival, foraging, and nectar storing in colonies of bombus impatiens. PLoS One 9, e91573, doi:10.1371/journal.pone.0091573 (2014).
Gill, R. J. & Raine, N. E. Chronic impairment of bumblebee natural foraging behaviour induced by sublethal pesticide exposure. Funct. Ecol. 28, 1459–1471, doi:10.1111/1365-2435.12292 (2014).
Vandame, R., Meled, M., Colin, M.-E. & Belzunces, L. P. Alteration of the homing-flight in the honey bee Apis mellifera L. exposed to sublethal dose of deltamethrin. Environ. Toxicol. Chem. 14, 855–860, doi:10.1897/1552-8618(1995)14[855:AOTHIT]2.0.CO;2 (1995).
Bortolotti, L. et al. Effects of sub-lethal imidacloprid doses on the homing rate and foraging activity of honey bees. Bull. Insectology 56, 63–67 (2003).
Henry, M. et al. A common pesticide decreases foraging success and survival in honey bees. Science (80-.) 336, 348–350, doi:10.1126/science.1215039 (2012).
Henry, M. et al. Reconciling laboratory and field assessments of neonicotinoid toxicity to honeybees. Proc. R. Soc. B 282, 20152110, doi:10.1098/rspb.2015.2110 (2015).
Williamson, S. M., Willis, S. J. & Wright, Ga Exposure to neonicotinoids influences the motor function of adult worker honeybees. Ecotoxicology 23, 1409–18, doi:10.1007/s10646-014-1283-x (2014).
Charreton, M. et al. A locomotor deficit induced by sublethal doses of pyrethroid and neonicotinoid insecticides in the honeybee Apis mellifera. PLoS One 10, e0144879, doi:10.1371/journal.pone.0144879 (2015).
Fischer, J. et al. Neonicotinoids interfere with specific components of navigation in honeybees. PLoS One 9, e91364, doi:10.1371/journal.pone.0091364 (2014).
Erickson, B. Regulation: Europe bans three neonicotinoids linked to honeybee population declines. Chem. Eng. News Arch 91, 11 (2013).
Douglas, M. R. & Tooker, J. F. Large-scale deployment of seed treatments has driven rapid increase in use of neonicotinoid insecticides and preemptive pest management in U.S. Field crops. Environ. Sci. Technol. 49, 5088–5097, doi:10.1021/es506141g (2015).
Hladik, M. L., Vandever, M. & Smalling, K. L. Exposure of native bees foraging in an agricultural landscape to current-use pesticides. Sci. Total Environ. 542, 469–477, doi:10.1016/j.scitotenv.2015.10.077 (2016).
Dively, G. P. & Kamel, A. Insecticide residues in pollen and nectar of a cucurbit crop and their potential exposure to pollinators. J. Agric. Food Chem. 60, 4449–4456, doi:10.1021/jf205393x (2012).
Girolami, V. et al. Translocation of neonicotinoid insecticides from coated seeds to seedling guttation drops: a novel way of intoxication for bees. J. Econ. Entomol 102, 1808–15, doi:10.1603/029.102.0511 (2009).
Stanley, D. & Raine, N. Investigating sublethal pesticide effects on bumblebee navigation and foraging. In IUSSI 2014 proceedings 45 at http://ses.library.usyd.edu.au:80/handle/2123/11202 (2014).
Blanken, L. J., Van Langevelde, F. & Van Dooremalen, C. Interaction between Varroa destructor and imidacloprid reduces flight capacity of honeybees. Proc. R. Soc. B 282, 20151738, doi:10.1098/rspb.2015.1738 (2015).
Tosi, S. et al. Effects of a neonicotinoid pesticide on thermoregulation of African honey bees (Apis mellifera scutellata). J. Insect Physiol. 93–94, 56–63, doi:10.1016/j.jinsphys.2016.08.010 (2016).
Gmeinbauer, R. & Crailsheim, K. Glucose utilization during flight of honeybee (Apis mellifera) workers, drones and queens. J. Insect Physiol. 39, 959–967, doi:10.1016/0022-1910(93)90005-C (1993).
Brodschneider, R., Riessberger-Gallé, U. & Crailsheim, K. Flight performance of artificially reared honeybees (Apis mellifera). Apidologie 40, 441–449, doi:10.1051/apido/2009006 (2009).
Hrassnigg, N. & Crailsheim, K. Metabolic rates and metabolic power of honeybees in tethered flight related to temperature and drag (Hymenoptera: Apidae). Entomol. Gen. 24, 23–30, doi:10.1127/entom.gen/24/1999/23 (1999).
Sotavalta, O. On the fuel consumption of the honeybee (Apis mellifica L.) in flight experiments. Ann. ent. fenn (1954).
Jungmann, R., Rothe, U. & Nachtigall, W. Flight of the honey bee I. J. Comp. Physiol. B 158, 711–718, doi:10.1007/BF00693009 (1989).
Tison, L. et al. Honey bees’ behavior is impaired by chronic exposure to the neonicotinoid thiacloprid in the field. Environ. Sci. Technol. 50, 7218–7227, doi:10.1021/acs.est.6b02658 (2016).
Thompson, H., Coulson, M., Ruddle, N., Wilkins, S. & Harkin, S. Thiamethoxam: Assessing flight activity of honeybees foraging on treated oilseed rape using radio frequency identification technology. Environ. Toxicol. Chem. 35, 385–393, doi:10.1002/etc.3183 (2016).
Kessler, S. C. et al. Bees prefer foods containing neonicotinoid pesticides. Nature 521, 74–76, doi:10.1038/nature14414 (2015).
Eckert, J. E. The flight range of the honey bee. J. Agric. Res. 47, 257–285 (1933).
Steffan-Dewenter, I. & Kuhn, A. Honeybee foraging in differentially structured landscapes. Proc. Biol. Sci. 270, 569–575, doi:10.1098/rspb.2002.2292 (2003).
Visscher, P. K. & Seeley, T. D. Foraging strategy of honeybee colonies in a temperate deciduous forest. Ecol. Soc. Am. 63, 1790–1801 (1982).
Woods, Wa, Heinrich, B. & Stevenson, R. D. Honeybee flight metabolic rate: does it depend upon air temperature? J. Exp. Biol. 208, 1161–73, doi:10.1242/jeb.01510 (2005).
Derecka, K. et al. Transient exposure to low levels of insecticide affects metabolic networks of honeybee larvae. PLoS One 8, e68191, doi:10.1371/journal.pone.0068191 (2013).
Mogren, C. L. & Lundgren, J. G. Neonicotinoid-contaminated pollinator strips adjacent to cropland reduce honey bee nutritional status. Sci. Rep. 6, 29608, doi:10.1038/srep29608 (2016).
Wen, Z. & Scott, J. G. Cross-resistance to imidacloprid in strains of German cockroach (Blattella germanica) and house fly (Musca domestica). Pestic. Sci. 49, 367–371, doi:10.1002/(SICI)1096-9063(199704)49:4<>1.0.CO;2-6 (1997).
Suchail, S., Guez, D. & Belzunces, L. P. Discrepancy between acute and chronic toxicity induced by imidacloprid and its metabolites in Apis mellifera. Environ. Toxicol. Chem. 20, 2482–6, doi:10.1002/etc.v20:11 (2001).
Dietemann, V. et al. Standard methods for varroa research. J. Apic. Res 52, 1–54, doi:10.3896/IBRA.1.52.1.09 (2013).
Smith, T. & Jones, V. P. The Flight Mill http://entomology.tfrec.wsu.edu/VPJ_Lab/Flight-Mill. (2012).
Reetz, J. E. et al. Uptake of Neonicotinoid Insecticides by Water-Foraging Honey Bees (Hymenoptera: Apidae) Through Guttation Fluid of Winter Oilseed Rape. J. Econ. Entomol. tov287 109, 31–40, doi:10.1093/jee/tov287 (2015).
EFSA. Statement on the findings in recent studies investigating sub-lethal effects in bees of some neonicotinoids in consideration of the uses currently authorised in Europe. EFSA J 10, 2725 (2012).
EFSA. Conclusion on the peer review of the pesticide risk assessment for bees for the active substance thiamethoxam. EFSA J 11, 3067, doi:10.2903/j.efsa.2013.3067 (2013).
Cresswell, J. E. & Thompson, H. M. Comment on ‘A Common Pesticide Survival in Honey Bees’. Science (80-.) 337, 1453–b, doi:10.1126/science.1224618 (2012).
Crane, E. Honey. A comprehensive survey. (Heinemann, 1975).
Pierre, J., Mesquida, J., Marilleau, R., Pham-Delègue, M. H. & Renard, M. Nectar secretion in winter oilseed rape, Brassica napus - Quantitative and qualitative variability among 71 genotypes. Plant Breed. 118, 471–476, doi:10.1046/j.1439-0523.1999.00421.x (1999).
Sanchez-Bayo, F. & Goka, K. Pesticide residues and bees - A risk assessment. PLoS One 9, e94482, doi:10.1371/journal.pone.0094482 (2014).
Stoner, K. a. & Eitzer, B. D. Movement of soil-applied imidacloprid and thiamethoxam into nectar and pollen of squash (Cucurbita pepo). PLoS One 7, e39114, doi:10.1371/journal.pone.0039114 (2012).
Bubník, Z. & Kadlec, P. Sucrose solubility. doi:10.1007/978-1-4615-2676-6 (Springer US, 1995).
Freeman, G. H. & Halton, J. H. Note on an Exact Treatment of Contingency, Goodness of Fit and Other Problems of Significance. Biometrika 38, 141–149, doi:10.1093/biomet/38.1-2.141 (1951).
Lowry, R. VassarStats: Website for Statistical Computation http://vassarstats.net/fisher2x3.html (2016).
Acknowledgements
We thank Piotr Medrzycki for his comments on this manuscript. We wish to thank Robert Brodschneider, Vincent Jones, and Teah Smith for their flight mill advice. We would also like to thank Ryan Hoffman for his software help and the UCSD undergraduates who contributed to this study. This research was partly supported by the Marco Polo scholarship, awarded to ST by the University of Bologna, and the UCSD Academic Senate. The funding sources had no involvement in the study design, collection, analysis and interpretation of the data, writing and decision to submit the article for publication.
Author information
Authors and Affiliations
Contributions
S.T. and J.N. conceived of and designed the experiments. J.N. built the flight mills. S.T. collected the data. S.T. and J.N. analysed the data. The manuscript was written through contributions of all authors. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Tosi, S., Burgio, G. & Nieh, J.C. A common neonicotinoid pesticide, thiamethoxam, impairs honey bee flight ability. Sci Rep 7, 1201 (2017). https://doi.org/10.1038/s41598-017-01361-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-01361-8
This article is cited by
-
The impact of chronic exposure to field-level thiamethoxam on sunflower visitation and yield for Apis cerana
Apidologie (2024)
-
Non-optimal ambient temperatures aggravate insecticide toxicity and affect honey bees Apis mellifera L. gene regulation
Scientific Reports (2023)
-
Residual effect of imidacloprid and beta-cyfluthrin on Africanized Apis mellifera L. (Hymenoptera: Apidae) workers
Apidologie (2023)
-
Lethal and Sublethal Dose of Thiamethoxam and Its Effects on the Behavior of a Non-target Social Wasp
Neotropical Entomology (2023)
-
Sulfoxaflor influences the biochemical and histological changes on honeybees (Apis mellifera L.)
Ecotoxicology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.