Abstract
Cytomegalovirus (CMV) is a latent virus which causes chronic activation of the immune system. Here, we demonstrate that cytotoxic and pro-inflammatory CD4+CD28null T cells are only present in CMV seropositive donors and that CMV-specific Immunoglobulin (Ig) G titers correlate with the percentage of these cells. In vitro stimulation of peripheral blood mononuclear cells with CMVpp65 peptide resulted in the expansion of pre-existing CD4+CD28null T cells. In vivo, we observed de novo formation, as well as expansion of CD4+CD28null T cells in two different chronic inflammation models, namely the murine CMV (MCMV) model and the experimental autoimmune encephalomyelitis (EAE) model for multiple sclerosis (MS). In EAE, the percentage of peripheral CD4+CD28null T cells correlated with disease severity. Pre-exposure to MCMV further aggravated EAE symptoms, which was paralleled by peripheral expansion of CD4+CD28null T cells, increased splenocyte MOG reactivity and higher levels of spinal cord demyelination. Cytotoxic CD4+ T cells were identified in demyelinated spinal cord regions, suggesting that peripherally expanded CD4+CD28null T cells migrate towards the central nervous system to inflict damage. Taken together, we demonstrate that CMV drives the expansion of CD4+CD28null T cells, thereby boosting the activation of disease-specific CD4+ T cells and aggravating autoimmune mediated inflammation and demyelination.
Similar content being viewed by others
Introduction
Multiple sclerosis (MS) is a disabling autoimmune disease of the central nervous system (CNS). Activated autoreactive immune cells infiltrate the brain and spinal cord leading to chronic inflammation, demyelination and ultimately axonal loss1. Although the exact trigger for this activation has not been elucidated yet, a genetic predisposition in combination with environmental factors seems essential to develop MS2. Worldwide, about 2.5 million people are affected, mostly young adults (20–40 y) and females (3:1 ratio), although the disease progression in men can be more severe3.
Naive T cells express CD28 on their cell surface, but due to repeated antigenic stimulation CD28 expression can be lost4,5,6. CD4+CD28null memory T cells arise during chronic activation of the immune system, in a subset of healthy controls (HC) and patients with MS. These cells have a restricted T cell receptor (TCR) diversity (oligoclonal), are costimulation independent, more resistant to apoptosis, and less susceptible to suppression by regulatory T cells (Tregs)7,8,9,10,11,12. Relevant features suggesting their contribution to autoimmune mediated CNS damage in MS include their autoreactive nature; their target tissue infiltration, via e.g. the fractalkine gradient; and their cytotoxic capacities, namely the expression of natural killer (NK) cell receptors and the production of perforin and granzymes11, 13, 14.
So far, the trigger for the selective expansion of CD4+CD28null T cells and their contribution to MS disease pathology is poorly investigated. There is mounting evidence that CD4+CD28null T cell expansion occurs after infection with cytomegalovirus (CMV)9, 15,16,17. CMV is a member of the β-herpesvirus family that establishes lifelong latent infections in ≥70% of the human population18. CMV commits a large portion of its genome to evade recognition and activation of the immune system: e.g. reduction of antigen presentation by interfering with the expression of MHC/HLA molecules, downmodulation of costimulatory molecules, and evasion of NK cell control16, 19,20,21. However, as a result of cross-priming of CMV antigens, CMV-specific T cell responses develop. Moreover, due to the persistent nature of CMV, substantial accumulation of CMV-specific memory T cells (on average 10% of the total memory T cell compartment) can occur18, 22,23,24, albeit with varying degrees, which may be caused by differences in infectious dose25. As a consequence of this large percentage of CMV-specific T cells, immune surveillance could become less effective over time, thereby compromising normal immunity18, 26. Indeed, CMV seropositivity has been correlated with a worse MS disease course, although disease limiting effects have also been stated (Reviewed in ref. 16). The most important finding indicating a disease promoting role is the enrichment of CMV-specific antibodies in MS27. When these antibodies were present in MS patients, this was correlated to a decreased time to relapse, an increase in the number of relapses and enhanced brain atrophy28,29,30. In contrast, another study concluded that the presence of CMV-specific antibodies was associated with a better clinical outcome, an increased age of disease onset and decreased brain atrophy31. A recent meta-analysis on 1341 MS patients and 2042 healthy controls did not yield a conclusive result on the relationship between CMV infection and the occurrence of MS32.
In this study we investigated whether CMV by itself is able to trigger the expansion of CD4+CD28null T cells and aggravate MS disease, using a combination of human data and in vivo animal model systems.
Results
CMV expands CD4+CD28null T cells via repeated antigenic stimulation
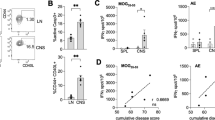
To determine whether CMV infection is linked to expansion of CD4+CD28null T cells (>2% of CD4+ T cells), an association study between CMV serology and the percentage of CD4+CD28null T cells was performed. In our cohort, the percentage of CD4+CD28null T cells is significantly higher in CMV seropositive (CMV+) donors compared to CMV seronegative (CMV−) donors (p < 0.0001, Fig. 1a and b), with no differences between MS and HC, which is in line with other studies9. Furthermore, CMV-specific IgG titers positively correlate with the percentage of CD4+CD28null T cells (ρs = 0.6, p < 0.0001, Fig. 1c). To test whether this correlation is CMV specific, we examined the serology of EBV, another chronic and latent virus which has been implicated in MS33. No significant correlation was found between the percentage of CD4+CD28null T cells and EBNA IgG titers (Fig. 1d). Furthermore, EBV IgG levels did not differ between donors with versus without CD4+CD28null T cell expansion (respectively: 9 ± 4 vs 8 ± 4, p > 0.05). In contrast, donors with CD4+CD28null T cell expansion have significantly higher CMV IgG titers compared to donors without expansion (respectively: 219 ± 92.8 vs 5 ± 0, p < 0.0001).
CMV infection expands CD4+CD28null T cells. Flow cytometry was performed to determine the percentage of CD4+CD28null T cells. CMV and EBV status and immunoglobulin titers were determined via ELFA. CD4+CD28null T cells in (a) CMV seropositive (n = 100) compared to seronegative (n = 127) MS patients and (b) CMV seropositive (n = 24) versus seronegative healthy controls (n = 39). (c) Correlation of CMV IgG levels with the percentage of CD4+CD28null T cells in MS patients and HC (n = 140). (d) Correlation of EBV EBNA IgG titers in 155 MS patients. (e) Repeated stimulation of PBMCs from HC (n = 12) and MS patients (n = 8) with CMV pp65 (e) or IL-2 (f) in vitro, after which the number of CD4+CD28null T cells was determined at different time points. (g) Flow cytometry of splenocytes of MCMV infected mice on day 0, day 8 and day 250 post infection (n = 5/time point). (h) Splenocytes from MCMV-infected WT (n = 5) and CD80/86−/− (n = 4) mice were analysed for the percentage of CD4+CD28null T cells at day 250 post infection. *p < 0.05, **p < 0.01, ****p < 0.0001.
Since CD4+CD28null T cell expansion only occurred in CMV infected individuals and correlated with the level of CMV-specific antibody titers, we investigated whether CMV infection can drive expansion of CD4+CD28null T cells, using in vitro and in vivo models. Since there is no significant difference in the percentage of CD4+CD28null T cells between HC and MS patients, we did not discriminate between both populations in the following experiment. To mimic chronic TCR triggering by CMV, PBMCs from MS patients and HC, who were either CMV+ or CMV− and exhibited CD4+CD28null T cell expansion (exp+) or not (exp−), were repeatedly stimulated with a CMV peptide (CMVpp65) in vitro. The percentage of CD4+CD28null T cells significantly increased over time in CMV+ exp+ donors, as opposed to CMV+ exp− and CMV−exp− donors (Fig. 1e). IL-2 by itself did not induce expansion of CD4+CD28null T cells (Fig. 1f). Repetitive CMV peptide stimulation in vitro did not induce the generation of CD4+CD28null T cells in exp− donors over the duration of the experiment (20 days). To investigate the long term effect of CMV infection on formation and expansion of CD4+CD28null T cells, we used the in vivo MCMV mouse model, the most widely used and relevant model for human CMV infection25. MCMV infected mice showed a significant increase of CD4+CD28null T cells in the spleen over time, with a 2-fold increase at day 8 (p < 0.05) and 20-fold increase at day 250 post-infection compared to non-infected mice (d0, p < 0.0001, Fig. 1g). In non-infected mice, the CD4+CD28null T cell levels were below the threshold for expansion (1 ± 0.2%), indicating that CMV infection induces loss of CD28 in CD4+ T cells in vivo. In summary, repeated in vitro stimulation with CMV peptide expands pre-existing CD4+CD28null T cells, whereas in vivo CMV infection induces CD28 loss in CD4+ T cells and drives expansion of CD4+CD28null T cells.
To determine whether CMV induces the loss of CD28 on CD4+ T cells via repeated antigenic triggering or via interaction with its ligands CD80 and CD86, we infected CD80/86−/− mice with MCMV. MCMV infection induced the expansion of CD4+CD28null T cells to a similar extent in CD80/86−/− mice and WT mice (Fig. 1h), indicating that the loss of CD28 is not caused by binding with their ligands CD80 and CD86. These findings further strengthen our notion that CD28 loss is caused by repeated antigenic triggering via the TCR.
CD4+CD28null T cells are increased in EAE mice and correlate with disease severity
CD4+CD28null T cells are cytotoxic, accumulate in MS lesions and at least a subpopulation is autoreactive in nature14. To test the hypothesis that CD4+CD28null T cells are associated with the severity of neuroinflammation, an EAE experiment was performed. Follow-up time (Fig. 2a) was extended compared to the standard protocol (30 day p.i.), to test whether CD4+CD28null T cells expand during acute and chronic stages of EAE (Fig. 2b). While limited numbers of CD4+CD28null T cells were found in CFA control mice, a significant increase above the 2% threshold for expansion was only found in the EAE mice (EAE: 3 ± 0.7%, p = 0.004 and control: 1.8 ± 0.3%, p > 0.05, Fig. 2b).
CD4+CD28null T cells are increased in EAE mice, as a result of auto-antigenic stimulation. (a) The phenotype of blood-derived mouse CD4+CD28null T cells was measured via flow cytometry. (b) After induction, EAE mice (n = 15) and CFA control mice (n = 10) were scored for maximum 90 days according to their disability. (c) Blood was collected at different time points, to determine the number of CD4+CD28null T cells via flow cytometry. (d) Correlation between CD4+CD28null T cells and EAE score. (e) Historical human MBP specific T cell clones repeatedly stimulated with MBP/PHA (n = 8) were thawed and analysed for the number of CD4+CD28null T cells via flow cytometry. **p < 0.01, ***p < 0.001, ****p < 0.0001.
From previous studies, it is known that human CD4+CD28null T cells produce IFNγ and granzyme B, and that they show low expression of CD62L, CD127 and CD279, 14, 34,35,36. To determine whether mouse CD4+CD28null T cells have a similar phenotype, we analysed these cells, which were present in the peripheral blood of EAE mice. We found that they indeed phenotypically resemble their human counterparts as evidenced by a low expression of CD62L, CD127 and CD27, and production of IFNγ and granzyme B (Fig. 2c), identifying them as proinflammatory and cytotoxic effector memory T cells.
Furthermore, the percentage of CD4+CD28null T cells positively correlated with the EAE disease score (ρs = 0.6, p = 0.0002, Fig. 2d). Long-term follow-up indicated that there was no further expansion of CD4+CD28null T cells in the chronic phase of EAE (after d30).
The increase in CD4+CD28null T cells in EAE mice could result from repeated auto-antigenic stimulation. To test this hypothesis, human MBP-specific T cell clones, generated and sustained in vitro by stimulation rounds with MBP or PHA, were analysed for the presence of CD4+CD28null T cells (Fig. 2e). The number of CD4+CD28null T cells increased after each successive round of stimulation. Thus, repeated MBP stimulation leads to the expansion of CD4+CD28null T cells in vitro, indicating that the expansion of CD4+CD28null T cells in MS patients may result from chronic auto-antigenic stimulation in vivo. Of note, in vitro stimulation with tetanus toxoid also induced expansion of CD4+CD28null T cells (Supplementary Figure S2), indicating that the expansion is not antigen specific, but rather due to the chronicity of the antigen exposure.
CMV infection exacerbates clinical symptoms of EAE
Our results indicate that CD4+CD28null T cells expand after repeated immune activation, either as a result of CMV infection or after the induction of autoimmunity. Here, we investigated whether CMV infection and subsequent expansion of CD4+CD28null T cells correlate with a worse EAE outcome. The interplay between these different factors was investigated by infecting mice with MCMV and subsequently inducing EAE 8 days later. The EAE disease score of mice that were pre-exposed to MCMV was significantly higher compared to the EAE control group (mean cumulative score: 56 ± 4 vs 47 ± 3, p < 0.01; mean maximal score: 3.8 ± 0.26 vs 3.5 ± 0, p < 0.02; mean end score: 3.1 ± 0.35 vs 2.2 ± 0.27 p = 0.002). Furthermore, the MCMV group experienced a relapse between day 26 and day 30 after immunization, whereas EAE control mice did not (Fig. 3a). The percentage of CD4+CD28null T cells in the spleen increased at least eight-fold in each group (CMV: 8 ± 2%, p < 0.001, EAE: 12 ± 3%, p < 0.0001 and CMV+ EAE: 14 ± 2%, p < 0.0001) compared to baseline (1 ± 0.2%) (Fig. 3b). These results provide further evidence that both CMV infection and EAE induction lead to the expansion of CD4+CD28null T cells and that prior CMV infection aggravates EAE symptoms.
MCMV infected mice with EAE have a worse disease course. Mice were infected with MCMV and after 8 days EAE was induced. (a) Daily scoring of the CMV, EAE and MCMV infected EAE groups (n = 8/group). (b) Splenocytes were isolated at day 0 (baseline) and day 30 and CD4+CD28null T cells were measured via flow cytometry. (c) After stimulation of splenocytes with a MOG35–55 peptide, the IFNγ producing CD4+ T cells were measured via flow cytometry and normalized to the non-peptide control. The MOG response significantly correlated with the percentage of CD4+CD28null T cells in the spleen. *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
Since we showed that CMV exacerbates EAE disease, we asked whether this is due to increased autoimmune reactivity. To answer this question, CD4+ T cell reactivity to MOG peptide was measured in the spleen. The MCMV infected EAE group displayed enhanced MOG-specific CD4+ T cell reactivity compared to the control groups (EAE: p < 0.004, CMV: p < 0.002). Furthermore, this MOG response correlated to the percentage of CD4+CD28null T cells in the spleen of these mice (Fig. 3c). Also, we detected splenic CMV-specific CD4+ T cell reactivity in the MCMV infected groups, however they were not increased by EAE induction (data not shown). Viral load measured in the salivary glands at the end of the experiment indicate that the virus was still present in high amounts in both the MCMV and the MCMV infected EAE groups (data not shown). These data indicate that CMV infection increases the percentage of MOG-specific CD4+ T cells, thereby increasing autoimmune mediated neuroinflammation, and that CD4+CD28null T cells take part in this overall MOG response.
CMV infection increases demyelination in EAE
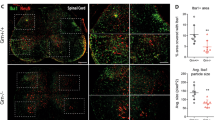
In MS patients, CD4+CD28null T cells accumulate in brain lesions and are in close contact with neural cells14. Since CMV infection leads to a worse EAE disease course, we next questioned whether demyelination of the spinal cord, the predominant location of lesions in this model, is also increased in these animals. No demyelination was found in the spinal cord of MCMV infected mice (Fig. 4a and b). MCMV infected EAE animals exhibited enhanced demyelination compared to the EAE control group (Fig. 4b), indicating that CMV infection accelerates autoimmune-mediated CNS damage. Furthermore, the extent of demyelination is strongly correlated with the percentage of spleen-derived CD4+CD28null T cells (R = 0.71, p < 0.05, Fig. 4c). We further identified the presence of CD4+GranzymeB+ T cells in the spinal cord (Fig. 4d), suggesting that CD4+CD28null T cells, which are granzyme B+, are present in the spinal cord and possibly contribute to CNS damage.
CMV infection increases EAE lesion size. (a) Representative staining of MBP in the CMV control group (left), the EAE control group (middle) and the CMV infected EAE animals (right). (b) The amount of demyelination within the CMV and EAE control groups and the CMV infected EAE group, calculated via dividing the demyelinated area (=loss of MBP) in the white matter of the spinal cord, over the total white matter area for each section. (c) Correlation between the amount of demyelination for each animal and the percentage of peripheral CD4+CD28null T cells. (d) Single and double staining of CD4 and Granzyme B, which points to the presence of CD4+CD28null T cells, in the spinal cord of EAE mice. *p < 0.05, ****p < 0.0001.
Discussion
Here, we demonstrate that CD4+CD28null T cells expand during EAE and positively correlate with disease severity. In addition, we show that CMV by itself is able to enhance activation of disease-specific CD4+ T cells, trigger the expansion of CD4+CD28null T cells and worsen EAE. Overall, our findings support a detrimental role for CMV in autoimmune neuroinflammation.
Our group, together with others have shown that CD4+CD28null T cells are associated with the pathogenesis of chronic inflammatory disorders15, 37, 38. In MS, a direct link with disease severity has not been demonstrated so far. However, indirect evidence, such as their target tissue infiltrating capacity and cytotoxic activity towards oligodendrocytes, certainly alludes to this hypothesis14, 35. In this study, we made use of the widely documented mouse model for MS, EAE. Although this model is certainly not fully equivalent to the human situation, it does recapitulate the inflammatory response that arises in patients with MS, which is the focal point of our study39. Here, we demonstrate that peripheral CD4+CD28null T cells are increased in EAE animals and that the percentage of CD4+CD28null T cells is strongly correlated with the amount of demyelination and disease severity. Mouse-derived CD4+CD28null T cells displayed an effector memory (CD62LlowCD127lowCD27lowIFNγ+) and cytotoxic (granzyme B+) phenotype, indicating that they are similar to their human counterparts9, 14, 34,35,36. Our findings are in line with evidence found in collagen-induced arthritis (CIA), the animal model for RA, where an increase in the number of CD4+CD28−NKG2D+ T cells was observed after immunization40. The increase in peripheral CD4+CD28null T cells in EAE mice could be attributed to repeated auto-antigenic stimulation caused by chronic autoimmune inflammation. Indeed, as evidenced by our in vitro data, repeated MBP stimulation of MBP-specific T cell clones leads to CD4+CD28null T cell expansion. In vivo, we found a direct correlation between the percentage of CD4+CD28null T cells and the anti-MOG response level in the spleen of EAE mice. Together, these findings confirm the autoreactive nature of CD4+CD28null T cells11. After 30 days p.i., there was no further expansion of CD4+CD28null T cells in the blood of EAE mice. Instead, starting from day 60, the memory pool maintained a steady state. This is as expected with regards to the homeostasis of the memory pool: expansion is followed by contraction and ultimately maintenance of the remaining memory T cell pool41.
In contrast to EAE mice, not all MS patients have CD4+CD28null T cell expansion. Therefore, in humans additional components could be important in the generation of CD4+CD28null T cells. Potential triggers include: 1) chronic inflammation42; and 2) viral infections7, of which CMV, as a persistent virus, is a promising candidate. Our data demonstrate that repetitive in vitro CMV peptide stimulation of human PBMCs expands pre-existing CD4+CD28null T cells. IL-2, which enhances T-cell proliferation and differentiation, does not lead to the expansion of CD4+CD28null T cells. EBV, another chronic and latent virus implicated in MS, is not associated with CD4+CD28null T cell expansion. These findings further support the hypothesis that CD4+CD28null T cells arise after CMV infection, which corresponds with previous reports by other groups9, 17. Of note, we did not measure proliferation; therefore the increase in CD4+CD28null T cells after CMV stimulation could be due to survival rather than proliferation. However, van Leeuwen et al. indicated that CD4+CD28null T cells proliferate after addition of CMV antigens, suggesting the latter is true17. In vivo, CMV infection leads to continuous activation, enabling us to study chronic repeated antigenic challenge. Although human CMV and MCMV are different viruses, the MCMV mouse model is widely used and is the most relevant mouse model which mimics human CMV infection25. MCMV virus in the salivary gland is thought to be important for spreading the virus from mouse to mouse. Whereas in all organs the virus is latent in less than a few weeks, in the salivary glands the virus replicates for months43. Thus the amount of virus in the salivary gland is not influencing the titers in other organs, such as spleen and lymph nodes, but is instead set by the initial infection dose, and the local and pre-existing immunity conditions. Using this model, we clearly show formation and expansion of CD4+CD28null T cells in all MCMV infected animals over time. These findings are in line with those of other groups7. Since CMV is unable to infect T cells, CMV cannot directly reduce CD28 expression on T cells, but rather exerts its effects due to its persistent nature. In this study, we show that the loss of CD28 is caused by continued antigenic triggering and not by binding with their ligands CD80 and CD86, since the number of CD4+CD28null T cells did not differ between MCMV-infected CD80/86−/− mice and WT. Furthermore, studies in mice and humans have indicated that the number and phenotypes of CMV-specific T cells correlate with viral load25, 44, 45; higher viral loads drive higher expansions, establishing the antigen-driven aspect of the response. In this study, we used a relatively high dose of MCMV leading to a higher amount of antigen-specific T cells, including CD4+CD28null T cells. In the human population, the dose of CMV is not evenly distributed, leading to variability in the number of antigen specific T cells between individuals. This heterogeneity explains the difference in the percentage of CD4+CD28null T cells among CMV seropositive donors. In this respect, it is of interest to note that the CMV Ig titers correlate with a higher percentage of CD4+CD28null T cells. This implies that individuals with a higher CMV exposure may develop more CD4+CD28null T cells and associated disease. Also, the proportion of CMV-seropositive individuals increases with age46, as does the percentage of CD4+CD28null T cells47.
In MS patients, CMV seropositivity and high IgG titers are correlated with increased percentages of CD4+CD28null T cells. This link between CMV and CD4+CD28null T cell expansion was previously also reported for RA, ankylosing spondylitis and cardiovascular diseases17, 38, 48,49,50,51,52, indicating that CMV infection and CD28null T cell expansion form a common pathogenic background in these diseases9. The logical next step is to confirm the possible link between CMV, CD4+CD28null T cells and autoimmunity. Here, we demonstrate that CD4+CD28null T cells are increased in MCMV, EAE and MCMV+ EAE mice after 30 days p.i. MCMV infected EAE animals had a higher disability score and experienced a relapse, compared to the EAE control mice. Furthermore, MCMV infection increased demyelination in EAE mice, which correlated with higher CD4+CD28null T cell percentages in the periphery. Since we found CD4+GranzymeB+ T cells in the spinal cord of EAE and MCMV infected EAE mice, this suggests that CD4+CD28null T cells accumulate in the CNS to inflict damage in line with our previous observations in post-mortem MS brain material14. Thus, CMV infection exacerbates EAE disease course and does this by boosting the autoimmune response, as indicated by an increased MOG response. Indeed, T cell expansion preferentially occurred in MOG specific T cells, since the overall T cell responsiveness (no peptide control) in the spleen was comparable between all groups (data not shown). This is in accordance with others, where EAE induction combined with viral infection (γ-herpes virus, Semlike Forest virus or Sindbis virus) accelerated or exacerbated disease as a result of enhanced immune cell infiltration and polarization of the adaptive immune response53,54,55. Furthermore, MCMV infection rendered EAE-resistant BALB/c mice susceptible for EAE induction56. In another murine model of MS, namely Theiler’s murine encephalitis virus (TMEV) model, opposite findings were demonstrated; CMV infection attenuated TMEV disease course57. However, the immune response in TMEV is largely CD8 mediated, whereas in EAE and MS CD4+ T cells are the main players58. We believe that the EAE model better represents what is going on in MS, namely a primary autoimmune mediated attack of the CNS, in contrast to the TMEV model, where primary viral induced neurotoxicity induces secondary autoimmunity.
An important question still remains to be answered: is the disease exacerbating effect and enhanced demyelination directly caused by CMV infection itself or attributable to the increased expansion of CD4+CD28null T cells? While technically challenging, an adoptive transfer study is needed to indisputably prove a direct cause-and-effect relationship of CD4+CD28null T cells and disease severity. CMV was previously reported to be present in the CNS, where it could damage local cells and tissues directly33. The ensuing cell death could then enhance autoimmunity as a result of the release and spreading of self-epitopes from degenerating tissue59. However, since demyelination was not present in animals only infected with CMV, it is unlikely that CMV by itself leads to CNS damage as proposed by the epitope spreading hypothesis. On the other hand, reactivation of CMV during ongoing MS could trigger the activation of autoreactive T cells (molecular mimicry) thereby enhancing subsequent demyelination. Of note, CMV-specific T cells were previously identified in MS lesions60. Evidence for cross reactivity between a CMV antigen (UL86981–1003) and the myelin oligodendrocyte glycoprotein epitope (MOG35–55) has been found in rats and non-human primates61, 62. However, our data show that CMV infection alone did not mount a significant MOG response in the spleen, which would have been the case if molecular mimicry was involved. Another possible way by which CMV could directly contribute to autoimmunity is through bystander activation, where the immune response against CMV leads to robust inflammation, triggering the non-specific activation of autoreactive T cells63. We postulate that these bystander activated autoreactive T cells are mainly responsible for exacerbating EAE disease severity.
In summary, CMV infection and EAE induction lead to the expansion of CD4+CD28null T cells. Both CMV infection and CD4+CD28null T cells aggravate autoimmune mediated CNS inflammation, since EAE disease severity, measured by EAE score and the extent of neuroinflammation and demyelination, correlated with increasing amounts of CD4+CD28null T cells and the presence of a CMV infection. Overall, CMV infection drives the expansion of CD4+CD28null T cells, thereby amplifying the activation of disease-specific CD4+ T cells, and exacerbating EAE disease. Future studies will address whether this is also the case in MS patients. However, CMV vaccination to prevent the formation of CD4+CD28null T cells and the adverse effects of the infection itself, could be beneficial for people at risk of developing MS.
Methods
Study subjects
Human
Peripheral blood samples (Li-Heparin coated tubes) were collected from 63 healthy controls (HC) and 227 MS patients in collaboration with the University Biobank Limburg (UBiLim). CMV and Epstein-Barr virus (EBV) status and titers (CMV IgG and EBV EBNA IgG) were determined in serum samples via Vidas ELFA (bioMérieux, Marcy l’Etoile, France) and Architect immunoassay (Abbott, Illinois, USA). Clinical data are presented in Table 1; there were no significant differences between CMV positive or negative donors, neither in MS patients nor in healthy controls.
Mice
Female C57BL/6 mice were purchased from Harlan (Horst, the Netherlands). CD80/86−/− mice64 were bred in LUMC to the C57BL/6 background.
EAE induction
10 week old C57BL/6J mice were immunized subcutaneously with myelin oligodendrocyte glycoprotein 35–55 peptide (MOG35–55) emulsified in complete Freund’s adjuvant (CFA) containing Mycobacterium tuberculosis according to manufacturer’s guidelines (Hooke Laboratories, Lawrence, USA). Directly after immunization and 24 h later, mice were intraperitoneally injected with pertussis toxin. Mice were weighed and evaluated daily for neurological signs of disease using a standard 5-point scale; 0: no symptoms; 1: limp tail; 2: hind limp weakness; 3: complete hind limp paralysis; 4: complete hind limp paralysis and partial front leg paralysis; 5: moribund.
MCMV infection
MCMV-Smith was obtained from the American Type Culture Collection (ATCC, Manassas, VA) and stocks were prepared from the salivary glands of infected BALB/c mice. C57BL/6J WT and CD80/86−/− mice were infected i.p. with 5 × 104 PFU. All mice were maintained under specific pathogen free conditions.
Flow cytometry
Human
All donors included in this study were analysed for the percentage of CD4+CD28null T cells. This was done by isolating peripheral blood mononuclear cells (PBMCs) from whole blood by density gradient centrifugation (Cedarlane lympholyte, Sheffield, UK). Cells were double stained with anti-human CD4 FITC and CD28 PE (both BD Biosciences, Franklin Lakes, NJ). The gating strategy consists of a lymphocyte gate using the forward and side scatter signal, after which CD4+ cells were gated and subsequently CD28 expression was monitored within this gate (Supplementary Figure S1a). Cells were acquired using a FACSAria II cytometer, and data were analysed using BD FACSDiva software. Significant expansion of CD4+CD28null T cells was arbitrary defined as a percentage ≥2% of the total CD4+ T cell population, as this was the minimal percentage of cells that allowed discrimination of a distinctive population14.
Mice
Single cell suspensions were prepared from spleens by mincing the tissue through a 70-μm cell strainer (BD Bioscience). Erythrocytes were lysed in a hypotonic ammonium chloride buffer. The gating strategy consists of a lymphocyte gate using the forward and side scatter signal, after which CD3+CD4+ cells were gated and subsequently CD28 expression was monitored within this gate (Supplementary Figure S1b). Surface and intracellular cytokine staining were used to identify and characterize CD4+CD28null T cells. MOG-specific CD4+ T cell responses were determined after in vitro stimulation with MOG35–55 (10 µg/ml, Hooke laboratories) peptides for 8 hours (6 hours in the presence of Brefeldin A). Fluorochrome-conjugated antibodies specific for CD3, CD4, CD27, CD28, CD62L, CD127, IFN-γ and granzyme B were purchased from BD Biosciences, Biolegend, or eBioscience. Cells were acquired using a BD LSR II flow or FACSAria II cytometer, and data were analysed using FlowJo (TreeStar) or BD FACSDiva software.
Immunohistochemistry
Mice were perfused with Ringer’s solution, spinal cords were dissected and, via a PFA/sucrose gradient, frozen in liquid nitrogen, 30 days after EAE induction. Ten micrometre cryosections were cut on the Leica CM3050S cryostat (Leica Microsystems, Wetzlar, Germany). Sections were fixed, blocked and incubated with antibodies against CD4 (1/100, BD Biosciences, 553043) and granzyme B (1/100, Abcam, Ab4059). Binding of these primary antibodies was visualized with the appropriate Alexa 488 or Alexa 555 (1/500, Life technologies, Merelbeke, Belgium) and nuclear staining was performed with DAPI (Life technologies). Autofluorescence was blocked using 0.1% Sudan Black in 70% ethanol. Demyelination and infiltration were visualized by 3, 30 diaminobenzidine (DAB) staining of myelin basic protein (MBP) with the envision kit according to the manufacturers protocol (dako Glostrup, Denmark) and subsequent hematoxylin counterstaining. In short, peroxidase activity was inhibited with 0.3% H2O2. Slides were blocked in PBS containing 10% protein block (dako Glostrup) and incubated with rat anti-mouse MBP (1/100, Millipore, MAB386) for 1 h at room temperature. Following incubation with a peroxidase labelled polymer, staining was performed with DAB substrate and hematoxylin counterstain. Microscopical analysis was performed using a multiviewer DM 2000 LED microscope and DM 4000 LED microscope with Leica Application Suite software (Leica Microsystems).
Histological quantification
The extent of demyelination was evaluated in spinal cords of three mice per group (MCMV+ EAE, EAE control and MCMV control group). Each mouse displayed a disease score close to the median of the respective group. Every 200 µm, an entire longitudinal spinal cord section was analysed for immune infiltrates and demyelination, with a total of four sections for each animal. Demyelinated area was assessed as loss of MBP staining within the white matter of these four sections covering the entire spinal cord. Microscopical analysis was performed using a multiviewer DM 2000 LED microscope (Leica Microsystems) and Fiji software (NIH ImageJ).
In vitro CMV stimulation assay
PBMCs from 12 HC and 8 MS patients were isolated from whole blood via density gradient centrifugation. These donors differed according to their CMV status and CD4+CD28null T cell expansions (Table 2). PBMCs were cultured in RPMI-1640 medium (Lonza, Basel, Switzerland) supplemented with 10% foetal bovine serum (FBS; Hyclone Europe, Erembodegem, Belgium), 1% nonessential amino acids, 1% sodium pyruvate, 50 U/ml penicillin and 50 mg/ml streptomycin (all Life technologies).
To mimic chronic CMV stimulation, cells were stimulated weekly with CMVpp65 recombinant protein (10 µl/ml, Miltenyi Biotec, Bergisch Gladbach, Germany) or IL-2 (5 U/ml, Roche Diagnostics, Basel, Switzerland) for a maximum of 20 days. At different time points (d0, 1, 6, 9, 12, 15 and 20), the relative number of CD4+CD28null T cells was determined by flow cytometry as described above.
Generation of MBP reactive T cell clones
MBP-specific T cell clones were generated as described previously65. Briefly, MBP-reactive T-cell lines were generated from the blood of MS patients via limiting dilution analysis (LDA), cloned with phytohemagglutinin (PHA) in the presence of allogeneic accessory cells and further expanded by successive rounds of restimulation with MBP or PHA and autologous antigen presenting cells (APCs).
Statistical analysis
Statistical analyses were performed using GraphPad Prism version 6 and SAS 9.3. Parametric analyses include t-tests (2 groups), 1-way ANOVA and 2-way ANOVA (multiple groups). Nonparametric tests encompass Mann-Whitney tests (2 groups) and Kruskal-Wallis tests (multiple groups). Parametric data are shown as mean ± SD, nonparametric data as median ± interquartile range. A p-value < 0.05 was considered significant.
Ethics approval and consent to participate
Experiments involving human samples and data were approved by the Medical Ethics Committee UZ KU Leuven and experiments were performed in accordance with its guidelines and regulations. Informed consents were obtained from all donors.
All animal studies were in accordance with the EU directive 2010/63/EU for animal experiments and were approved by the Ethical Committee Animal Experiments UHasselt.
References
Ransohoff, R. M., Hafler, D. A. & Lucchinetti, C. F. Multiple sclerosis-a quiet revolution. Nature reviews. Neurology 11, 134–142, doi:10.1038/nrneurol.2015.14 (2015).
Legroux, L. & Arbour, N. Multiple Sclerosis and T Lymphocytes: An Entangled Story. J Neuroimmune Pharmacol 10, 528–546, doi:10.1007/s11481-015-9614-0 (2015).
Bearoff, F. et al. Identification of genetic determinants of the sexual dimorphism in CNS autoimmunity. PloS one 10, e0117993, doi:10.1371/journal.pone.0117993 (2015).
Chamberlain, W. D., Falta, M. T. & Kotzin, B. L. Functional subsets within clonally expanded CD8(+) memory T cells in elderly humans. Clin Immunol 94, 160–172, doi:10.1006/clim.1999.4832 (2000).
Effros, R. B. Loss of CD28 expression on T lymphocytes: a marker of replicative senescence. Dev Comp Immunol 21, 471–478 (1997).
Fiorentini, S. et al. Generation of CD28− cells from long-term-stimulated CD8+CD28+ T cells: a possible mechanism accounting for the increased number of CD8+CD28− T cells in HIV-1-infected patients. J Leukoc Biol 65, 641–648 (1999).
Mou, D., Espinosa, J. E., Stempora, L., Iwakoshi, N. N. & Kirk, A. D. Viral-induced CD28 loss evokes costimulation independent alloimmunity. J Surg Res 196, 241–246, doi:10.1016/j.jss.2015.02.033 (2015).
Thewissen, M. et al. CD4+CD28null T cells in autoimmune disease: pathogenic features and decreased susceptibility to immunoregulation. Journal of immunology 179, 6514–6523 (2007).
Maly, K. & Schirmer, M. The story of CD4+CD28− T cells revisited: solved or still ongoing? J Immunol Res 2015, 348746, doi:10.1155/2015/348746 (2015).
Mou, D., Espinosa, J., Lo, D. J. & Kirk, A. D. CD28 negative T cells: is their loss our gain? Am J Transplant 14, 2460–2466, doi:10.1111/ajt.12937 (2014).
Markovic-Plese, S., Cortese, I., Wandinger, K. P., McFarland, H. F. & Martin, R. CD4+CD28− costimulation-independent T cells in multiple sclerosis. The Journal of clinical investigation 108, 1185–1194, doi:10.1172/JCI12516 (2001).
Lovett-Racke, A. E. et al. Decreased dependence of myelin basic protein-reactive T cells on CD28-mediated costimulation in multiple sclerosis patients. A marker of activated/memory T cells. The Journal of clinical investigation 101, 725–730, doi:10.1172/JCI1528 (1998).
Broux, B. et al. IL-15 Amplifies the Pathogenic Properties of CD4+CD28− T Cells in Multiple Sclerosis. Journal of immunology 194, 2099–2109, doi:10.4049/jimmunol.1401547 (2015).
Broux, B. et al. CX(3)CR1 drives cytotoxic CD4(+)CD28(−) T cells into the brain of multiple sclerosis patients. Journal of autoimmunity 38, 10–19, doi:10.1016/j.jaut.2011.11.006 (2012).
Broux, B., Markovic-Plese, S., Stinissen, P. & Hellings, N. Pathogenic features of CD4+CD28− T cells in immune disorders. Trends in molecular medicine 18, 446–453, doi:10.1016/j.molmed.2012.06.003 (2012).
Vanheusden, M., Stinissen, P., t Hart, B. A. & Hellings, N. Cytomegalovirus: a culprit or protector in multiple sclerosis? Trends in molecular medicine 21, 16–23, doi:10.1016/j.molmed.2014.11.002 (2015).
van Leeuwen, E. M. et al. Emergence of a CD4+ CD28− granzyme B+, cytomegalovirus-specific T cell subset after recovery of primary cytomegalovirus infection. Journal of immunology 173, 1834–1841 (2004).
Hanley, P. J. & Bollard, C. M. Controlling cytomegalovirus: helping the immune system take the lead. Viruses 6, 2242–2258, doi:10.3390/v6062242 (2014).
Jackson, S. E., Mason, G. M. & Wills, M. R. Human cytomegalovirus immunity and immune evasion. Virus research 157, 151–160, doi:10.1016/j.virusres.2010.10.031 (2011).
Noriega, V., Redmann, V., Gardner, T. & Tortorella, D. Diverse immune evasion strategies by human cytomegalovirus. Immunologic research 54, 140–151, doi:10.1007/s12026-012-8304-8 (2012).
Arens, R. et al. B7-mediated costimulation of CD4 T cells constrains cytomegalovirus persistence. J Virol 85, 390–396, doi:10.1128/JVI.01839-10 (2011).
O’Hara, G. A., Welten, S. P., Klenerman, P. & Arens, R. Memory T cell inflation: understanding cause and effect. Trends Immunol 33, 84–90, doi:10.1016/j.it.2011.11.005 (2012).
Jackson, S. E., Mason, G. M., Okecha, G., Sissons, J. G. & Wills, M. R. Diverse specificities, phenotypes, and antiviral activities of cytomegalovirus-specific CD8+ T cells. J Virol 88, 10894–10908, doi:10.1128/JVI.01477-14 (2014).
Sylwester, A. W. et al. Broadly targeted human cytomegalovirus-specific CD4+ and CD8+ T cells dominate the memory compartments of exposed subjects. The Journal of experimental medicine 202, 673–685, doi:10.1084/jem.20050882 (2005).
Redeker, A., Welten, S. P. & Arens, R. Viral inoculum dose impacts memory T-cell inflation. European journal of immunology 44, 1046–1057, doi:10.1002/eji.201343946 (2014).
La Rosa, C. & Diamond, D. J. The immune response to human CMV. Future Virol 7, 279–293, doi:10.2217/fvl.12.8 (2012).
Sanadgol, N. et al. Prevalence of cytomegalovirus infection and its role in total immunoglobulin pattern in Iranian patients with different subtypes of multiple sclerosis. The new microbiologica 34, 263–274 (2011).
Horakova, D. et al. Environmental factors associated with disease progression after the first demyelinating event: results from the multi-center SET study. PloS one 8, e53996, doi:10.1371/journal.pone.0053996 (2013).
Weinstock-Guttman, B. et al. Interactions of serum cholesterol with anti-herpesvirus responses affect disease progression in clinically isolated syndromes. Journal of neuroimmunology 263, 121–127, doi:10.1016/j.jneuroim.2013.07.010 (2013).
Zivadinov, R. et al. Humoral responses to herpesviruses are associated with neurodegeneration after a demyelinating event: results from the multi-center set study. Journal of neuroimmunology 273, 58–64, doi:10.1016/j.jneuroim.2014.04.012 (2014).
Zivadinov, R. et al. Positivity of cytomegalovirus antibodies predicts a better clinical and radiological outcome in multiple sclerosis patients. Neurological research 28, 262–269, doi:10.1179/016164106X98134 (2006).
Pakpoor, J., Pakpoor, J., Disanto, G., Giovannoni, G. & Ramagopalan, S. V. Cytomegalovirus and multiple sclerosis risk. Journal of neurology 260, 1658–1660, doi:10.1007/s00415-013-6912-4 (2013).
Olival, G. S. et al. Multiple sclerosis and herpesvirus interaction. Arquivos de neuro-psiquiatria 71, 727–730, doi:10.1590/0004-282X20130160 (2013).
Dumitriu, I. E., Araguas, E. T., Baboonian, C. & Kaski, J. C. CD4+CD28 null T cells in coronary artery disease: when helpers become killers. Cardiovasc Res 81, 11–19, doi:10.1093/cvr/cvn248 (2009).
Zaguia, F. et al. Cytotoxic NKG2C+ CD4 T cells target oligodendrocytes in multiple sclerosis. Journal of immunology 190, 2510–2518, doi:10.4049/jimmunol.1202725 (2013).
Arens, R., Remmerswaal, E. B., Bosch, J. A. & van Lier, R. A. 5(th) International Workshop on CMV and Immunosenescence - A shadow of cytomegalovirus infection on immunological memory. European journal of immunology 45, 954–957, doi:10.1002/eji.201570044 (2015).
Dumitriu, I. E. & Kaski, J. C. The role of T and B cells in atherosclerosis: potential clinical implications. Curr Pharm Des 17, 4159–4171 (2011).
Pierer, M. et al. Association of anticytomegalovirus seropositivity with more severe joint destruction and more frequent joint surgery in rheumatoid arthritis. Arthritis Rheum 64, 1740–1749, doi:10.1002/art.34346 (2012).
Ransohoff, R. M. Animal models of multiple sclerosis: the good, the bad and the bottom line. Nat Neurosci 15, 1074–1077, doi:10.1038/nn.3168 (2012).
Taneja, V. et al. Requirement for CD28 may not be absolute for collagen-induced arthritis: study with HLA-DQ8 transgenic mice. Journal of immunology 174, 1118–1125 (2005).
Marsden, V. S., Kappler, J. W. & Marrack, P. C. Homeostasis of the memory T cell pool. Int Arch Allergy Immunol 139, 63–74, doi:10.1159/000090000 (2006).
Bryl, E., Vallejo, A. N., Weyand, C. M. & Goronzy, J. J. Down-regulation of CD28 expression by TNF-alpha. Journal of immunology 167, 3231–3238 (2001).
Jonjic, S., Mutter, W., Weiland, F., Reddehase, M. J. & Koszinowski, U. H. Site-restricted persistent cytomegalovirus infection after selective long-term depletion of CD4+ T lymphocytes. The Journal of experimental medicine 169, 1199–1212 (1989).
Gamadia, L. E. et al. The size and phenotype of virus-specific T cell populations is determined by repetitive antigenic stimulation and environmental cytokines. Journal of immunology 172, 6107–6114 (2004).
van Leeuwen, E. M. et al. IL-7 receptor alpha chain expression distinguishes functional subsets of virus-specific human CD8+ T cells. Blood 106, 2091–2098, doi:10.1182/blood-2005-02-0449 (2005).
Pawelec, G., McElhaney, J. E., Aiello, A. E. & Derhovanessian, E. The impact of CMV infection on survival in older humans. Curr Opin Immunol 24, 507–511, doi:10.1016/j.coi.2012.04.002 (2012).
Thewissen, M. et al. Premature immunosenescence in rheumatoid arthritis and multiple sclerosis patients. Ann N Y Acad Sci 1051, 255–262, doi:10.1196/annals.1361.066 (2005).
Duftner, C. et al. Prevalence, clinical relevance and characterization of circulating cytotoxic CD4+CD28− T cells in ankylosing spondylitis. Arthritis research & therapy 5, R292–300, doi:10.1186/ar793 (2003).
Hooper, M. et al. Cytomegalovirus seropositivity is associated with the expansion of CD4+CD28− and CD8+CD28− T cells in rheumatoid arthritis. J Rheumatol 26, 1452–1457 (1999).
van Bergen, J. et al. Functional killer Ig-like receptors on human memory CD4+ T cells specific for cytomegalovirus. Journal of immunology 182, 4175–4182, doi:10.4049/jimmunol.0800455 (2009).
Jonasson, L., Tompa, A. & Wikby, A. Expansion of peripheral CD8+ T cells in patients with coronary artery disease: relation to cytomegalovirus infection. J Intern Med 254, 472–478 (2003).
Olsson, J. et al. Age-related change in peripheral blood T-lymphocyte subpopulations and cytomegalovirus infection in the very old: the Swedish longitudinal OCTO immune study. Mech Ageing Dev 121, 187–201 (2000).
Casiraghi, C. et al. Gammaherpesvirus latency accentuates EAE pathogenesis: relevance to Epstein-Barr virus and multiple sclerosis. PLoS pathogens 8, e1002715, doi:10.1371/journal.ppat.1002715 (2012).
Jerusalmi, A., Morris-Downes, M. M., Sheahan, B. J. & Atkins, G. J. Effect of intranasal administration of Semliki Forest virus recombinant particles expressing reporter and cytokine genes on the progression of experimental autoimmune encephalomyelitis. Molecular therapy: the journal of the American Society of Gene Therapy 8, 886–894 (2003).
Rainey-Barger, E. K., Blakely, P. K., Huber, A. K., Segal, B. M. & Irani, D. N. Virus-induced CD8+ T cells accelerate the onset of experimental autoimmune encephalomyelitis: implications for how viral infections might trigger multiple sclerosis exacerbations. Journal of neuroimmunology 259, 47–54, doi:10.1016/j.jneuroim.2013.03.011 (2013).
Milovanovic, J. A., Stojanovic, A., Milovanovic, B., Jonjic, M., Popovic, S., Arsenijevic, B. & Lukic, N. M. Latent Murine Cytomegalovirus Infection Contributes to EAE Pathogenesis. Serbian Journal of Experimental and Clinical Research 15, 183–190, doi:10.2478/sjecr-2014-0023 (2014).
Pirko, I. et al. CMV infection attenuates the disease course in a murine model of multiple sclerosis. PloS one 7, e32767, doi:10.1371/journal.pone.0032767 (2012).
Nelson, A. L., Bieber, A. J. & Rodriguez, M. Contrasting murine models of MS. Int MS J 11, 95–99 (2004).
Halenius, A. & Hengel, H. Human cytomegalovirus and autoimmune disease. BioMed research international 2014, 472978, doi:10.1155/2014/472978 (2014).
Scotet, E. et al. Frequent enrichment for CD8 T cells reactive against common herpes viruses in chronic inflammatory lesions: towards a reassessment of the physiopathological significance of T cell clonal expansions found in autoimmune inflammatory processes. European journal of immunology 29, 973–985, doi:10.1002/(SICI)1521-4141(199903)29:03<973::AID-IMMU973>3.0.CO;2-P (1999).
Brok, H. P. et al. The human CMV-UL86 peptide 981–1003 shares a crossreactive T-cell epitope with the encephalitogenic MOG peptide 34–56, but lacks the capacity to induce EAE in rhesus monkeys. Journal of neuroimmunology 182, 135–152, doi:10.1016/j.jneuroim.2006.10.010 (2007).
Zheng, M. M. & Zhang, X. H. Cross-reactivity between human cytomegalovirus peptide 981–1003 and myelin oligodendroglia glycoprotein peptide 35–55 in experimental autoimmune encephalomyelitis in Lewis rats. Biochemical and biophysical research communications 443, 1118–1123, doi:10.1016/j.bbrc.2013.12.122 (2014).
Delogu, L. G., Deidda, S., Delitala, G. & Manetti, R. Infectious diseases and autoimmunity. Journal of infection in developing countries 5, 679–687 (2011).
Borriello, F. et al. B7-1 and B7-2 have overlapping, critical roles in immunoglobulin class switching and germinal center formation. Immunity 6, 303–313 (1997).
Hellings, N. et al. Longitudinal study of antimyelin T-cell reactivity in relapsing-remitting multiple sclerosis: association with clinical and MRI activity. Journal of neuroimmunology 126, 143–160 (2002).
Acknowledgements
We thank patients and healthy controls for blood donations and Anne Bogaers, Igna Rutten, Kim Ulenaers, Veronique Pousset (Hasselt University and UBiLim), and Anita Knevels (MS rehabilitation centre) for assistance in collecting the blood samples. We also thank Katrien Wauterickx and Christel Bocken for technical assistance. This work was financially supported by grants from the Flemish Fund for Scientific Research (FWO Vlaanderen), special research fund UHasselt (BOF) and Interuniversity Attraction Poles (IUAP-P7-39 (T-time)).
Author information
Authors and Affiliations
Contributions
M.V. designed and performed the experiments, collected and analysed the data, and wrote the manuscript. B.B., S.P.M.W. and E.P. assisted with the experiments and edited the manuscript. B.V.W. and V.S. provided the human samples and acquired the corresponding data. N.H. and R.A. contributed to conception and design of the research, and were involved in drafting and revising the manuscript. L.M.P. and P.S. provided scientific support and revisions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Vanheusden, M., Broux, B., Welten, S.P.M. et al. Cytomegalovirus infection exacerbates autoimmune mediated neuroinflammation. Sci Rep 7, 663 (2017). https://doi.org/10.1038/s41598-017-00645-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-00645-3
This article is cited by
-
Understanding the link between neurotropic viruses, BBB permeability, and MS pathogenesis
Journal of NeuroVirology (2024)
-
Immunopathogenesis of viral infections in neurological autoimmune disease
BMC Neurology (2023)
-
Comprehensive Investigations Relationship Between Viral Infections and Multiple Sclerosis Pathogenesis
Current Microbiology (2023)
-
Integrated single cell analysis of blood and cerebrospinal fluid leukocytes in multiple sclerosis
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.