Abstract
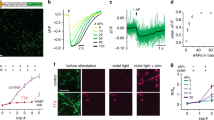
The strength of an excitatory synapse depends on its ability to release glutamate and on the density of postsynaptic receptors. Genetically encoded glutamate indicators (GEGIs) allow eavesdropping on synaptic transmission at the level of cleft glutamate to investigate properties of the release machinery in detail. Based on the sensor iGluSnFR, we recently developed accelerated versions of GEGIs that allow investigation of synaptic release during 100-Hz trains. Here, we describe the detailed procedures for design and characterization of fast iGluSnFR variants in vitro, transfection of pyramidal cells in organotypic hippocampal cultures, and imaging of evoked glutamate transients with two-photon laser-scanning microscopy. As the released glutamate spreads from a point source—the fusing vesicle—it is possible to localize the vesicle fusion site with a precision exceeding the optical resolution of the microscope. By using a spiral scan path, the temporal resolution can be increased to 1 kHz to capture the peak amplitude of fast iGluSnFR transients. The typical time frame for these experiments is 30 min per synapse.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author on request.
References
Pulido, C. & Marty, A. Quantal fluctuations in central mammalian synapses: functional role of vesicular docking sites. Physiol. Rev. 97, 1403–1430 (2017).
Choquet, D. & Triller, A. The dynamic synapse. Neuron 80, 691–703 (2013).
Namiki, S., Sakamoto, H., Iinuma, S., Iino, M. & Hirose, K. Optical glutamate sensor for spatiotemporal analysis of synaptic transmission. Eur. J. Neurosci. 25, 2249–2259 (2007).
Okubo, Y. et al. Imaging extrasynaptic glutamate dynamics in the brain. Proc. Natl. Acad. Sci. USA 107, 6526–6531 (2010).
Brun, M. A. et al. A semisynthetic fluorescent sensor protein for glutamate. J. Am. Chem. Soc. 134, 7676–7678 (2012).
Okumoto, S. et al. Detection of glutamate release from neurons by genetically encoded surface-displayed FRET nanosensors. Proc. Natl. Acad. Sci. USA 102, 8740–8745 (2005).
Tsien, R. Y. Building and breeding molecules to spy on cells and tumors. FEBS Lett. 579, 927–932 (2005).
Hires, S. A., Zhu, Y. & Tsien, R. Y. Optical measurement of synaptic glutamate spillover and reuptake by linker optimized glutamate-sensitive fluorescent reporters. Proc. Natl. Acad. Sci. USA 105, 4411–4416 (2008).
Marvin, J. S. et al. An optimized fluorescent probe for visualizing glutamate neurotransmission. Nat. Methods 10, 162–170 (2013).
Borghuis, B. G., Looger, L. L., Tomita, S. & Demb, J. B. Kainate receptors mediate signaling in both transient and sustained OFF bipolar cell pathways in mouse retina. J. Neurosci. 34, 6128–6139 (2014).
O’Herron, P. et al. Neural correlates of single-vessel haemodynamic responses in vivo. Nature 534, 378–382 (2016).
Brunert, D., Tsuno, Y., Rothermel, M., Shipley, M. T. & Wachowiak, M. Cell-type-specific modulation of sensory responses in olfactory bulb circuits by serotonergic projections from the raphe nuclei. J. Neurosci. 36, 6820–6835 (2016).
Helassa, N. et al. Ultrafast glutamate sensors resolve high-frequency release at Schaffer collateral synapses. Proc. Natl. Acad. Sci. USA 115, 5594–5599 (2018).
Wu, J. et al. Genetically encoded glutamate indicators with altered color and topology. ACS Chem. Biol. 13, 1832–1837 (2018).
Marvin, J. S. et al. Stability, affinity, and chromatic variants of the glutamate sensor iGluSnFR. Nat. Methods 15, 936–939 (2018).
Miesenbock, G., De Angelis, D. A. & Rothman, J. E. Visualizing secretion and synaptic transmission with pH-sensitive green fluorescent proteins. Nature 394, 192–195 (1998).
Granseth, B., Odermatt, B., Royle, S. J. & Lagnado, L. Clathrin-mediated endocytosis is the dominant mechanism of vesicle retrieval at hippocampal synapses. Neuron 51, 773–786 (2006).
Fernández-Alfonso, T., Kwan, R. & Ryan, T. A. Synaptic vesicles interchange their membrane proteins with a large surface reservoir during recycling. Neuron 51, 179–186 (2006).
Voglmaier, S. M. et al. Distinct endocytic pathways control the rate and extent of synaptic vesicle protein recycling. Neuron 51, 71–84 (2006).
Li, H. Concurrent imaging of synaptic vesicle recycling and calcium dynamics. Front. Mol. Neurosci. 4, 1–10 (2011).
Li, Y. & Tsien, R. W. pHTomato, a red, genetically encoded indicator that enables multiplex interrogation of synaptic activity. Nat. Neurosci. 15, 1047–1053 (2012).
Rose, T., Schoenenberger, P., Jezek, K. & Oertner, T. G. Developmental refinement of vesicle cycling at Schaffer collateral synapses. Neuron 77, 1109–1121 (2013).
Balaji, J. & Ryan, T. A. Single-vesicle imaging reveals that synaptic vesicle exocytosis and endocytosis are coupled by a single stochastic mode. Proc. Natl. Acad. Sci. USA 104, 20576–20581 (2007).
Gandhi, S. P. & Stevens, C. F. Three modes of synaptic vesicular recycling revealed by single-vesicle imaging. Nature 423, 607–613 (2003).
Zhu, Y., Xu, J. & Heinemann, S. F. Synaptic vesicle exocytosis-endocytosis at central synapses. Commun. Integr. Biol. 2, 418–419 (2009).
Maschi, D. & Klyachko, V. A. Spatiotemporal regulation of synaptic vesicle fusion sites in central synapses. Neuron 94, 65–73.e3 (2017).
Betz, W. J. & Bewick, G. S. Optical analysis of synaptic vesicle recycling at the frog neuromuscular junction. Science 255, 200–203 (1992).
Laviv, T. et al. Simultaneous dual-color fluorescence lifetime imaging with novel red-shifted fluorescent proteins. Nat. Methods 13, 989–992 (2016).
Campbell, R. E. et al. A monomeric red fluorescent protein. Proc Natl. Acad. Sci. USA 99, 7877–7882 (2002).
Gee, C. E., Ohmert, I., Wiegert, J. S. & Oertner, T. G. Preparation of slice cultures from rodent hippocampus. Cold Spring Harb. Protoc. 2017, 094888 (2017).
Pologruto, T. A., Sabatini, B. L. & Svoboda, K. ScanImage: flexible software for operating laser scanning microscopes. Biomed. Eng. Online 2, 13 (2003).
Suter, B. A. et al. Ephus: multipurpose data acquisition software for neuroscience experiments. Front. Neural Circuits 4, 1–12 (2010).
Negrean, A. & Mansvelder, H. D. Optimal lens design and use in laser-scanning microscopy. Biomed. Opt. Express 5, 1588–1609 (2014).
Jensen, T. P., Zheng, K., Tyurikova, O., Reynolds, J. P. & Rusakov, D. A. Monitoring single-synapse glutamate release and presynaptic calcium concentration in organised brain tissue. Cell Calcium 64, 102–108 (2017).
Hires, S. A., Zhu, Y. & Tsien, R. Y. Optical measurement of synaptic glutamate spillover and reuptake by linker optimized glutamate-sensitive fluorescent reporters. Proc. Natl. Acad. Sci. USA 105, 4411–4416 (2008).
de Lorimier, R. M. et al. Construction of a fluorescent biosensor family. Protein Sci. 11, 2655–2675 (2002).
Wiegert, J. S., Gee, C. E. & Oertner, T. G. Single-cell electroporation of neurons. Cold Spring Harb. Protoc. 2017, 135–138 (2017).
Luisier, F., Vonesch, C., Blu, T. & Unser, M. Fast interscale wavelet denoising of Poisson-corrupted images. Signal Process. 90, 415–427 (2010).
Taschenberger, H., Woehler, A. & Neher, E. Superpriming of synaptic vesicles as a common basis for intersynapse variability and modulation of synaptic strength. Proc. Natl. Acad. Sci. USA 113, E4548–E4557 (2016).
Oertner, T. G., Sabatini, B. L., Nimchinsky, E. A. & Svoboda, K. Facilitation at single synapses probed with optical quantal analysis. Nat. Neurosci. 5, 657–664 (2002).
Grabner, C. P. & Moser, T. Individual synaptic vesicles mediate stimulated exocytosis from cochlear inner hair cells. Proc. Natl. Acad. Sci. USA 115, 12811–12816 (2018).
Helassa, N., Podor, B., Fine, A. & Török, K. Design and mechanistic insight into ultrafast calcium indicators for monitoring intracellular calcium dynamics. Sci. Rep. 6, 38276 (2016).
Zucker, R. S. & Regehr, W. G. Short-term synaptic plasticity. Annu. Rev. Physiol. 64, 355–405 (2002).
Acknowledgements
We thank I. Ohmert and S. Graf for the preparation of organotypic cultures and excellent technical assistance. This study was supported by the German Research Foundation through Research Unit FOR 2419 P4 (T.G.O.) and P7 (J.S.W.), Priority Programs SPP 1665 (T.G.O.) and SPP 1926 (J.S.W.), Collaborative Research Center grant SFB 936 B7 (T.G.O.), and BBSRC grants BB/M02556X/1 (K.T.) and BB/S003894 (K.T.). We thank R. Y. Tsien (University of California, San Diego) for providing pCI syn tdimer2.
Author information
Authors and Affiliations
Contributions
C.D.D., J.S.W., K.T., and T.G.O. designed the experiments and prepared the manuscript. C.D.D. performed synaptic imaging experiments. N.H., S.K., C.C., and M.G. created and characterized novel iGluSnFR variants, C.S. wrote software to acquire and analyze GEGI data.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Journal peer review information: Nature Protocols thanks Edwin R. Chapman, Yulong Li, Jason Vevea and other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key reference using this protocol
Helassa, N. et al. Proc. Natl. Acad. Sci. USA 115, 5594–5599 (2018): https://www.pnas.org/content/115/21/5594
Integrated supplementary information
Supplementary Figure 1 Bleaching of iGluSnFR fluorescence during a 100-trial single-bouton experiment does not affect glutamate-induced responses.
(a) Raw traces (~100 trials) of iGluSnFR signals measured in a single presynaptic terminal in response to single APs elicited every 10 s. Note downward slope of baseline in every trial due to bleaching of iGluSnFR, and slow decrease of F0 over the time course of the experiment (17 min). Partial recovery between trials is likely due to lateral diffusion of unbleached iGluSnFR molecules in the axonal membrane. (b) Decrease in iGluSnFR resting fluorescence (F0) over the time course of the experiment (17 min). (c) Peak amplitude of individual trials expressed as relative change in fluorescence (ΔF/F0) is stable over the time course of the experiment. (d) Peak amplitude (ΔF/F0) is independent of F0.
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1
Rights and permissions
About this article
Cite this article
Dürst, C.D., Wiegert, J.S., Helassa, N. et al. High-speed imaging of glutamate release with genetically encoded sensors. Nat Protoc 14, 1401–1424 (2019). https://doi.org/10.1038/s41596-019-0143-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0143-9
This article is cited by
-
Extrasynaptic NMDA receptors in acute and chronic excitotoxicity: implications for preventive treatments of ischemic stroke and late-onset Alzheimer’s disease
Molecular Neurodegeneration (2023)
-
Specialized astrocytes mediate glutamatergic gliotransmission in the CNS
Nature (2023)
-
Asynchronous glutamate release is enhanced in low release efficacy synapses and dispersed across the active zone
Nature Communications (2022)
-
Vesicular release probability sets the strength of individual Schaffer collateral synapses
Nature Communications (2022)
-
Freeze-frame imaging of synaptic activity using SynTagMA
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.