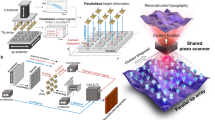
Abstract
Fast, high-resolution mapping of heterogeneous interfaces with a wide elastic modulus range is a major goal of atomic force microscopy (AFM). This goal becomes more challenging when the nanomechanical mapping involves biomolecules in their native environment. Over the years, several AFM-based methods have been developed to address this goal. However, none of these methods combine sub-nanometer spatial resolution, quantitative accuracy, fast data acquisition speed, wide elastic modulus range and operation in physiological solutions. Here, we present detailed procedures for generating high-resolution maps of the elastic properties of biomolecules and polymers using bimodal AFM. This requires the simultaneous excitation of the first two eigenmodes of the cantilever. An amplitude modulation (AM) feedback acting on the first mode controls the tip–sample distance, and a frequency modulation (FM) feedback acts on the second mode. The method is fast because the elastic modulus, deformation and topography images are obtained simultaneously. The method is efficient because only a single data point per pixel is needed to generate the aforementioned images. The main stages of the bimodal imaging are sample preparation, calibration of the instrument, tuning of the microscope and generation of the nanomechanical maps. In addition, with knowledge of the deformation, bimodal AFM enables reconstruction of the true topography of the surface. It takes ~9 h to complete the whole procedure.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Binnig, G., Quate, C. F. & Gerber, C. Atomic force microscope. Phys. Rev. Lett. 56, 930–933 (1986).
Dufrêne, Y. F. et al. Imaging modes of atomic force microscopy for application in molecular and cell biology. Nat. Nanotechnol. 12, 295–307 (2017).
Garcia, R., Magerle, R. & Perez, R. Nanoscale compositional mapping with gentle forces. Nat. Mater. 6, 405–411 (2007).
Proksch, R. et al. Practical loss tangent imaging with amplitude-modulated atomic force microscopy. J. Appl. Phys. 119, 134901 (2016).
Wang, D. & Russell, T. P. Advances in atomic force microscopy for probing polymer structure and properties. Macromolecules 51, 3–24 (2018).
Rodriguez, T. R. & Garcia, R. Compositional mapping of surfaces in atomic force microscopy by excitation of the second normal mode of the microcantilever. Appl. Phys. Lett. 84, 449–451 (2004).
Martinez, N. F., Patil, S., Lozano, J. R. & Garcia, R. Enhanced compositional sensitivity in atomic force microscopy by the excitation of the first two flexural modes. Appl. Phys. Lett. 89, 2–4 (2006).
Proksch, R. Multifrequency, repulsive-mode amplitude-modulated atomic force microscopy. Appl. Phys. Lett. 89, 1–4 (2006).
Garcia, R. & Herruzo, E. T. The emergence of multifrequency force microscopy. Nat. Nanotechnol. 7, 217–226 (2012).
Raman, A. et al. Mapping nanomechanical properties of live cells using multi-harmonic atomic force microscopy. Nat. Nanotechnol. 6, 809–814 (2011).
Sahin, O., Magonov, S., Su, C., Quate, C. F. & Solgaard, O. An atomic force microscope tip designed to measure time-varying nanomechanical forces. Nat. Nanotechnol. 2, 507–514 (2007).
Zhang, S., Aslan, H., Besenbacher, F. & Dong, M. Quantitative biomolecular imaging by dynamic nanomechanical mapping. Chem. Soc. Rev. 43, 7412–7429 (2014).
Santos, S., Lai, C.-Y., Olukan, T. & Chiesa, M. Multifrequency AFM: from origins to convergence. Nanoscale 9, 5038–5043 (2017).
Platz, D., Tholén, E. A., Pesen, D. & Haviland, D. B. Intermodulation atomic force microscopy. Appl. Phys. Lett. 92, 153106 (2008).
Jesse, S., Kalinin, S. V., Proksch, R., Baddorf, A. P. & Rodriguez, B. J. The band excitation method in scanning probe microscopy for rapid mapping of energy dissipation on the nanoscale. Nanotechnology 18, 435503 (2007).
Amo, C. A., Perrino, A. P., Payam, A. F. & Garcia, R. Mapping elastic properties of heterogeneous materials in liquid with Angstrom-scale resolution. ACS Nano 11, 8650–8659 (2017).
Heinz, W. F. & Hoh, J. H. Spatially resolved force spectroscopy of biological surfaces using the atomic force microscope. Trends Biotechnol. 17, 143–150 (1999).
Dufrêne, Y. F., Martínez-Martín, D., Medalsy, I., Alsteens, D. & Müller, D. J. Multiparametric imaging of biological systems by force–distance curve-based AFM. Nat. Methods 10, 847–854 (2013).
Young, T. J. et al. The use of the PeakForceTM quantitative nanomechanical mapping AFM-based method for high-resolution Young’s modulus measurement of polymers. Meas. Sci. Technol. 22, 125703 (2011).
Dokukin, M. E. & Sokolov, I. Quantitative mapping of the elastic modulus of soft materials with HarmoniX and PeakForce QNM AFM modes. Langmuir 28, 16060–16071 (2012).
Pfreundschuh, M., Martinez-Martin, D., Mulvihill, E., Wegmann, S. & Muller, D. J. Multiparametric high-resolution imaging of native proteins by force-distance curve-based AFM. Nat. Protoc. 9, 1113–1130 (2014).
Amo, C. A. & Garcia, R. Fundamental high-speed limits in single-molecule, single-cell, and nanoscale force spectroscopies. ACS Nano 10, 7117–7124 (2016).
Stark, M., Stark, R. W., Heckl, W. M. & Guckenberger, R. Inverting dynamic force microscopy: from signals to time-resolved interaction forces. Proc. Natl. Acad. Sci. USA 99, 8473–8478 (2002).
Shamitko-Klingensmith, N., Molchanoff, K. M., Burke, K. A., Magnone, G. J. & Legleiter, J. Mapping the mechanical properties of cholesterol-containing supported lipid bilayers with nanoscale spatial resolution. Langmuir 28, 13411–13422 (2012).
Platz, D., Forchheimer, D., Tholén, E. A. & Haviland, D. B. Interaction imaging with amplitude-dependence force spectroscopy. Nat. Commun. 4, 1360 (2013).
Payam, A. F., Martin-Jimenez, D. & Garcia, R. Force reconstruction from tapping mode force microscopy experiments. Nanotechnology 26, 1–12 (2015).
Dong, M., Husale, S. & Sahin, O. Determination of protein structural flexibility by microsecond force spectroscopy. Nat. Nanotechnol. 4, 514–517 (2009).
Lü, J., Yang, J., Dong, M. & Sahin, O. Nanomechanical spectroscopy of synthetic and biological membranes. Nanoscale 6, 7604–7608 (2014).
Martinez-Martin, D., Herruzo, E. T., Dietz, C., Gomez-Herrero, J. & Garcia, R. Noninvasive protein structural flexibility mapping by bimodal dynamic force microscopy. Phys. Rev. Lett. 106, 1–4 (2011).
Herruzo, E. T., Perrino, A. P. & Garcia, R. Fast nanomechanical spectroscopy of soft matter. Nat. Commun. 5, 3126 (2014).
Kocun, M., Labuda, A., Meinhold, W., Revenko, I. & Proksch, R. Fast, high resolution, and wide modulus range nanomechanical mapping with bimodal tapping mode. ACS Nano 11, 10097–10105 (2017).
Labuda, A., Kocun, M., Meinhold, W., Walters, D. & Proksch, R. Generalized Hertz model for bimodal nanomechanical mapping. Beilstein J. Nanotechnol. 7, 970–982 (2016).
Rabe, U. et al. Imaging and measurement of local mechanical material properties by atomic force acoustic microscopy. Surf. Interface Anal. 33, 65–70 (2002).
Killgore, J. P. et al. Viscoelastic property mapping with contact resonance force microscopy. Langmuir 27, 13983–13987 (2011).
Tetard, L., Passian, A. & Thundat, T. New modes for subsurface atomic force microscopy through nanomechanical coupling. Nat. Nanotechnol. 5, 105–109 (2010).
Kawai, S. et al. Systematic achievement of improved atomic-scale contrast via bimodal dynamic force microscopy. Phys. Rev. Lett. 103, 1–4 (2009).
Herruzo, E. T. & Garcia, R. Theoretical study of the frequency shift in bimodal FM-AFM by fractional calculus. Beilstein J. Nanotechnol. 3, 198–206 (2012).
Herruzo, E. T., Asakawa, H., Fukuma, T. & Garcia, R. Three-dimensional quantitative force maps in liquid with 10 piconewton, angstrom and sub-minute resolutions. Nanoscale 5, 2678–2685 (2013).
Ebeling, D. & Solares, S. D. Amplitude modulation dynamic force microscopy imaging in liquids with atomic resolution: comparison of phase contrasts in single and dual mode operation. Nanotechnology 24, 135702 (2013).
Ooe, H. et al. Amplitude dependence of image quality in atomically-resolved bimodal atomic force microscopy. Appl. Phys. Lett. 109, 141603 (2016).
Kawai, S., Eren, B., Marot, L. & Meyer, E. Graphene synthesis via thermal polymerization of aromatic quinone molecules. ACS Nano 8, 5932–5938 (2014).
Naitoh, Y. et al. Subatomic-scale force vector mapping above a Ge(001) dimer using bimodal atomic force microscopy. Nat. Phys. 13, 663–667 (2017).
Ebeling, D., Eslami, B. & Solares, S. D. J. Visualizing the subsurface of soft matter: simultaneous topographical imaging, depth modulation, and compositional mapping with triple frequency atomic force microscopy. ACS Nano 7, 10387–10396 (2013).
Thompson, H. T., Barroso-Bujans, F., Herrero, J. G., Reifenberger, R. & Raman, A. Subsurface imaging of carbon nanotube networks in polymers with DC-biased multifrequency dynamic atomic force microscopy. Nanotechnology 24, 135701 (2013).
Perrino, A. P., Ryu, Y. K., Amo, C. A., Morales, M. P. & Garcia, R. Subsurface imaging of silicon nanowire circuits and iron oxide nanoparticles with sub-10 nm spatial resolution. Nanotechnology 27, 275703 (2016).
Li, J. W., Cleveland, J. P. & Proksch, R. Bimodal magnetic force microscopy: separation of short and long range forces. Appl. Phys. Lett. 94, 163118 (2009).
Dietz, C., Herruzo, E. T., Lozano, J. R. & Garcia, R. Nanomechanical coupling enables detection and imaging of 5 nm superparamagnetic particles in liquid. Nanotechnology 22, 125708 (2011).
Schwenk, J. et al. Bimodal magnetic force microscopy with capacitive tip-sample distance control. Appl. Phys. Lett. 107, 132407 (2015).
Nowak, D. et al. Nanoscale chemical imaging by photoinduced force microscopy. Sci. Adv. 2, e1501571 (2016).
Ambrosio, A., Devlin, R. C., Capasso, F. & Wilson, W. L. Observation of nanoscale refractive index contrast via photoinduced force microscopy. ACS Photonics 4, 846–851 (2017).
Nguyen, H. K., Ito, M. & Nakajima, K. Elastic and viscoelastic characterization of inhomogeneous polymers by bimodal atomic force microscopy. Jpn. J. Appl. Phys. 55, 08NB06 (2016).
Dietz, C. Sensing in-plane nanomechanical surface and sub-surface properties of polymers: local shear stress as function of the indentation depth. Nanoscale 10, 460–468 (2018).
Bubendorf, A., Walheim, S., Schimmel, T. & Meyer, E. A robust AFM-based method for locally measuring the elasticity of samples. Beilstein J. Nanotechnol. 9, 1–10 (2018).
Patil, S., Martinez, N. F., Lozano, J. R. & Garcia, R. Force microscopy imaging of individual protein molecules with sub-pico Newton force sensitivity. J. Mol. Recognit. 20, 516–523 (2007).
Lamour, G., Yip, C. K., Li, H. & Gsponer, J. High intrinsic mechanical flexibility of mouse prion nanofibrils revealed by measurements of axial and radial Young’s moduli. ACS Nano 8, 3851–3861 (2014).
Ricci, M., Quinlan, R. A. & Voïtchovsky, K. Sub-nanometre mapping of the aquaporin–water interface using multifrequency atomic force microscopy. Soft Matter 13, 187–195 (2017).
Al-Rekabi, Z. & Contera, S. Multifrequency AFM reveals lipid membrane mechanical properties and the effect of cholesterol in modulating viscoelasticity. Proc. Natl. Acad. Sci. USA 115, 2658–2663 (2018).
Lai, C. Y., Santos, S. & Chiesa, M. Systematic multidimensional quantification of nanoscale systems from bimodal atomic force microscopy data. ACS Nano 10, 6265–6272 (2016).
Bose, K., Lech, C. J., Heddi, B. & Phan, A. T. High-resolution AFM structure of DNA G-wires in aqueous solution. Nat. Commun. 9, 1959 (2018).
González-Domínguez, I., Gutiérrez-Granados, S., Cervera, L., Gòdia, F. & Domingo, N. Identification of HIV-1-based virus-like particles by multifrequency atomic force microscopy. Biophys. J. 111, 1173–1179 (2016).
Cartagena-Rivera, A. X., Wang, W. H., Geahlen, R. L. & Raman, A. Fast, multi-frequency, and quantitative nanomechanical mapping of live cells using the atomic force microscope. Sci. Rep. 5, 1–11 (2015).
Guan, D., Charlaix, E., Qi, R. Z. & Tong, P. Noncontact viscoelastic imaging of living cells using a long-needle atomic force microscope with dual-frequency modulation. Phys. Rev. Appl. 8, 1–10 (2017).
Martinez, N. F. et al. Bimodal atomic force microscopy imaging of isolated antibodies in air and liquids. Nanotechnology 19, 384011 (2008).
Loganathan, M. & Bristow, D. A. Bi-harmonic cantilever design for improved measurement sensitivity in tapping-mode atomic force microscopy. Rev. Sci. Instrum. 85, 043703 (2014).
Dietz, C., Schulze, M., Voss, A., Riesch, C. & Stark, R. W. Bimodal frequency-modulated atomic force microscopy with small cantilevers. Nanoscale 7, 1849–1856 (2015).
Penedo, M. et al. Selective enhancement of individual cantilever high resonance modes. Nanotechnology 26, 485706 (2015).
Ruppert, M. G. & Moheimani, S. O. R. High-bandwidth multimode self-sensing in bimodal atomic force microscopy. Beilstein J. Nanotechnol. 7, 284–295 (2016).
Lozano, J. R. & Garcia, R. Theory of multifrequency atomic force microscopy. Phys. Rev. Lett. 100, 8–11 (2008).
Stark, R. W. Bistability, higher harmonics, and chaos in AFM. Mater. Today 13, 24–32 (2010).
Aksoy, M. D. & Atalar, A. Force spectroscopy using bimodal frequency modulation atomic force microscopy. Phys. Rev. B 83, 075416 (2011).
Lozano, J. R., Kiracofe, D., Melcher, J., Garcia, R. & Raman, A. Calibration of higher eigenmode spring constants of atomic force microscope cantilevers. Nanotechnology 21, 465502 (2010).
Forchheimer, D., Platz, D., Tholén, E. A. & Haviland, D. B. Model-based extraction of material properties in multifrequency atomic force microscopy. Phys. Rev. B 85, 1–7 (2012).
An, S., Solares, S. D., Santos, S. & Ebeling, D. Energy transfer between eigenmodes in multimodal atomic force microscopy. Nanotechnology 25, 475701 (2014).
Santos, S. Phase contrast and operation regimes in multifrequency atomic force microscopy. Appl. Phys. Lett. 104, 143109 (2014).
Solares, S. D. & Chawla, G. Triple-frequency intermittent contact atomic force microscopy characterization: simultaneous topographical, phase, and frequency shift contrast in ambient air. J. Appl. Phys. 108, 054901 (2010).
Chawla, G. & Solares, S. D. Mapping of conservative and dissipative interactions in bimodal atomic force microscopy using open-loop and phase-locked-loop control of the higher eigenmode. Appl. Phys. Lett. 99, 074103 (2011).
Naitoh, Y., Ma, Z., Li, Y. J., Kageshima, M. & Sugawara, Y. Simultaneous observation of surface topography and elasticity at atomic scale by multifrequency frequency modulation atomic force microscopy. J. Vac. Sci. Technol. B 28, 1210–1214 (2010).
Garcia, R. & Proksch, R. Nanomechanical mapping of soft matter by bimodal force microscopy. Eur. Polym. J. 49, 1897–1906 (2013).
Lozano, J. R. & Garcia, R. Theory of phase spectroscopy in bimodal atomic force microscopy. Phys. Rev. B 79, 014110 (2009).
Uchihashi, T., Kodera, N. & Ando, T. Guide to video recording of structure dynamics and dynamic processes of proteins by high-speed atomic force microscopy. Nat. Protoc. 7, 1193–1206 (2012).
Sneddon, I. N. The relation between load and penetration in the axisymmetric Boussinesq problem for a punch of arbitrary profile. Int. J. Eng. Sci. 3, 47–57 (1965).
Knoll, A., Magerle, R. & Krausch, G. Tapping mode atomic force microscopy on polymers: where is the true sample surface? Macromolecules 34, 4159–4165 (2001).
Wang, D., Fujinami, S., Nakajima, K. & Nishi, T. True surface topography and nanomechanical mapping measurements on block copolymers with atomic force microscopy. Macromolecules 43, 3169–3172 (2010).
San Paulo, A. & Garcia, R. High-resolution imaging of antibodies by tapping-mode atomic force microscopy: attractive and repulsive tip-sample interaction regimes. Biophys. J. 78, 1599–1605 (2000).
Perrino, A. P. & Garcia, R. How soft is a single protein? The stress–strain curve of antibody pentamers with 5 pN and 50 pm resolutions. Nanoscale 8, 9151–9158 (2016).
Lai, C.-Y., Santos, S. & Chiesa, M. Reconstruction of height of sub-nanometer steps with bimodal atomic force microscopy. Nanotechnology 27, 075701 (2016).
Lai, C.-Y., Santos, S. & Chiesa, M. General interpretation and theory of apparent height in dynamic atomic force microscopy. RSC Adv. 5, 80069–80075 (2015).
Casuso, I., Kodera, N., Le Grimellec, C., Ando, T. & Scheuring, S. Contact-mode high-resolution high-speed atomic force microscopy movies of the purple membrane. Biophys. J. 97, 1354–1361 (2009).
Gottlieb, S. et al. Thermal scanning probe lithography for the directed self-assembly of block copolymers. Nanotechnology 28, 175301 (2017).
Lorenzoni, M. et al. Assessing the local nanomechanical properties of self-assembled block copolymer thin films by peak force tapping. Langmuir 31, 11630–11638 (2015).
Umeda, N., Ishizaki, S. & Uwai, H. Scanning attractive force microscope using photothermal vibration. J. Vac. Sci. Technol. B 9, 1318 (1991).
Herruzo, E. T. & Garcia, R. Frequency response of an atomic force microscope in liquids and air: magnetic versus acoustic excitation. Appl. Phys. Lett. 91, 89–92 (2007).
Labuda, A. et al. Calibration of higher eigenmodes of cantilevers. Rev. Sci. Instrum. 87, 073705 (2016).
Rabe, M., Verdes, D. & Seeger, S. Understanding protein adsorption phenomena at solid surfaces. Adv. Colloid Interface Sci. 162, 87–106 (2011).
Butt, H.-J. & Jaschke, M. Calculation of thermal noise in atomic force microscopy. Nanotechnology 6, 1–7 (1995).
Acknowledgements
We are grateful for financial support from the European Research Council (ERC–AdG–340177; 3DNanoMech) and grants CSD2010-00024 and MAT2016-76507-R from the Ministerio de Economía y Competitividad. This work received funding from the European Union’s Horizon 2020 Research and Innovation Programme under Marie Sklodowska-Curie grant agreement 721874 (SPM2.0). We also acknowledge fellowships FPU15/04622 (C.A.A.) and BES-2017-081907 (V.G.G.) from the Ministerio de Educación.
Author information
Authors and Affiliations
Contributions
S.B., V.G.G. and A.P.P. performed the experiments. C.A.A. deduced the analytical expressions. S.B. and V.G.G. drafted the procedure. R.G. designed the experiments, supervised development of the theory, wrote the introduction and edited the manuscript. All the authors discussed the results and revised the manuscript.
Corresponding author
Ethics declarations
Competing interests
R.G. holds two US patents on bimodal AFM (7,921,466 B2 and 7,958,563 B2). The bimodal configuration presented in the Protocol is not described in those patents. The other authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Amo, C. A., Perrino, A. P., Payam, A. F. & Garcia, R. ACS Nano 11, 8650–8659 (2017): https://pubs.acs.org/doi/10.1021/acsnano.7b04381
Herruzo, E. T., Perrino, A. P., & Garcia, R. Nat. Commun. 5, 3126 (2014): https://www.nature.com/articles/ncomms4126
Martinez-Martin, D., Herruzo, E. T., Dietz, C., Gomez-Herrero, J. & Garcia, R. Phys. Rev. Lett. 106, 198101 (2011): https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.106.198101
Lozano, J. R. & Garcia, R. Phys. Rev. Lett. 100, 076102 (2008): https://journals.aps.org/prl/abstract/10.1103/PhysRevLett.100.076102
Supplementary information
Rights and permissions
About this article
Cite this article
Benaglia, S., Gisbert, V.G., Perrino, A.P. et al. Fast and high-resolution mapping of elastic properties of biomolecules and polymers with bimodal AFM. Nat Protoc 13, 2890–2907 (2018). https://doi.org/10.1038/s41596-018-0070-1
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-018-0070-1
This article is cited by
-
Multi-scale characterisation of a ferroelectric polymer reveals the emergence of a morphological phase transition driven by temperature
Nature Communications (2021)
-
Mapping mechanical properties of biological materials via an add-on Brillouin module to confocal microscopes
Nature Protocols (2021)
-
Polypeptide templating for designer hierarchical materials
Nature Communications (2020)
-
Capturing transient antibody conformations with DNA origami epitopes
Nature Communications (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.