Abstract
We report a fully automated procedure for the optimization and interpretation of reconstructions from cryo-electron microscopy (cryo-EM) data, available in Phenix as phenix.map_to_model. We applied our approach to 476 datasets with resolution of 4.5 Å or better, including reconstructions of 47 ribosomes and 32 other protein–RNA complexes. The median fraction of residues in the deposited structures reproduced automatically was 71% for reconstructions determined at resolutions of 3 Å or better and 47% for those at resolutions worse than 3 Å.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available in the Phenix data repository at https://phenix-online.org/phenix_data/terwilliger/map_to_model_2018. All the parameters, including resolution and specifications of reconstruction symmetry used in this work, are available on this site, along with all of the models produced.
References
Kühlbrandt, W. Science 343, 1443–1444 (2014).
Henderson, R. Arch. Biochem. Biophys. 581, 19–24 (2015).
Wang, R. Y. et al. Nat. Methods 12, 335–338 (2015).
Frenz, B., Walls, A. C., Egelman, E. H., Veesler, D. & DiMaio, F. Nat. Methods 14, 797–800 (2017).
Chen, M., Baldwin, P. R., Ludtke, S. J. & Baker, M. L. J. Struct. Biol. 196, 289–298 (2016).
DiMaio, F. & Chiu, W. Methods Enzymol. 579, 255–276 (2016).
Zhou, N., Wang, H. & Wang, J. Sci. Rep. 7, 2664 (2017).
Terashi, G. & Kihara, D. Nat. Commun. 9, 1618 (2018).
Terwilliger, T. C., Sobolev, O. V., Afonine, P. V. & Adams, P. D. Acta Crystallogr. D Struct. Biol. 74, 545–559 (2018).
Pintilie, G. D., Zhang, J., Goddard, T. D., Chiu, W. & Gossard, D. C. J. Struct. Biol. 170, 427–438 (2010).
Terwilliger, T. C., Adams, P. D., Afonine, P. V. & Sobolev, O. V. J. Struct. Biol. 204, 338–343 (2018).
Afonine, P. V. et al. Acta Crystallogr. D Struct. Biol. 74, 531–544 (2018).
Lawson, C. L. et al. Nucleic Acids Res. 44, D396–D403 (2016).
Merk, A. et al. Cell 165, 1698–1707 (2016).
Berman, H. M. et al. Nucleic Acids Res. 28, 235–242 (2000).
Bartesaghi, A. et al. Science 348, 1147–1151 (2015).
Borgnia, M. J. et al. Mol. Pharmacol. 89, 645–651 (2016).
Terwilliger, T. C. Acta Crystallogr. D Biol. Crystallogr. 59, 45–49 (2003).
Li, Z. et al. Protein Cell 9, 384–388 (2018).
Schoebel, S. et al. Nature 548, 352–355 (2017).
Wang, R. Y. R. et al. Nat. Methods 12, 335–338 (2015).
Pettersen, E. F. et al. J. Comput. Chem. 25, 1605–1612 (2004).
DeLaBarre, B. & Brunger, A. T. Acta Crystallogr. D Biol. Crystallogr. 62, 923–932 (2006).
Terwilliger, T. C. Acta Crystallogr. D Biol. Crystallogr. 66, 285–294 (2010).
Terwilliger, T. C. Acta Crystallogr. D Biol. Crystallogr. 66, 268–275 (2010).
Terwilliger, T. C. Acta Crystallogr. D Biol. Crystallogr. 59, 38–44 (2003).
Terwilliger, T. C. et al. Acta Crystallogr. D Biol. Crystallogr. 69, 2244–2250 (2013).
Shalev-Benami, M. et al. Nat. Commun. 8, 1589 (2017).
Zhang, R., Alushin, G. M., Brown, A. & Nogales, E. Cell 162, 849–859 (2015).
Adams, P. D. et al. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Acknowledgements
This work was supported by the NIH (grant GM063210 to P.D.A. and T.C.T.) and the Phenix Industrial Consortium. This work was supported in part by the US Department of Energy under Contract No. DE-AC02-05CH11231 at Lawrence Berkeley National Laboratory. This research used resources provided by the Los Alamos National Laboratory Institutional Computing Program, which is supported by the US Department of Energy National Nuclear Security Administration under Contract No. DE-AC52-06NA25396. We thank G. Terashi and D. Kihara for providing the MAINMAST and Rosetta models built into segmented maps.
Author information
Authors and Affiliations
Contributions
P.V.A. and O.V.S. developed core tools for map segmentation and secondary-structure restraints; P.D.A. and P.V.A. developed the real space refinement tools; T.C.T. developed the phenix.map_to_model tool and carried out the analyses; and P.D.A. and T.C.T. supervised the work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
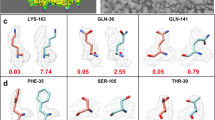
Supplementary Figure 1 Automatic map segmentation.
A. Automatic segmentation of map for β-galactosidase1 at a resolution of 2.2 Å (EMD entry 2984). Symmetry was identified using the Phenix tool phenix.map_symmetry and specifying the symmetry as D2. The map, the sequence of β-galactosidase and the symmetry file were supplied as inputs to the Phenix tool phenix.map_box with the keyword extract_unique=True. The automatically extracted unique part of the map is shown in red and the entire map is shown in yellow. B. Automatic segmentation of map for group II chaperonin2 at a resolution of 4.3 Å (EMD entry 5137). The map, the sequence and symmetry (D8) were supplied as inputs to the Phenix tool phenix.map_box along with the keyword extract_unique=True. The automatically extracted unique part of the map is shown in red and chains A (green) and H (blue) of the deposited model (3Los) are shown. Graphics created with Chimera3. References: 1. Bartesaghi, A. et al. Science 348, 1147–1151 (2015). 2. Zhang, J. et al. Nature 463, 379–383 (2010). 3. Pettersen, E.F. et al. J. Comput. Chem. 25, 1605–1612 (2004).
Supplementary Figure 2 Evaluation of model quality, and comparison with deposited models.
Validation measures for deposited models from PDB (blue columns) and automatically generated models (orange columns), and automatically generated models after removal of an average of 0.4% of side chains and 0.4% of full residues that clashed using the Phenix tool phenix.remove_clashes (gray). A. Clashscore (bad contacts per 1,000 atoms). B. Map correlation. C. Ramachandran outliers (%), D, rotamer outliers (%), E, Bond rmsd (Å), F. Angle rmsd (degrees), G. C-β deviations.
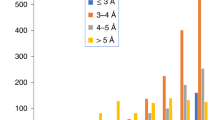
Supplementary Figure 3 Evaluation of model quality as a function of map resolution.
Validation measures for automatically generated models at resolutions of 3.5–4.5 Å (blue) and at 2.2–3.49 Å (orange). A. Clashscore (bad contacts per 1,000 atoms). B. Map correlation. C. Ramachandran outliers (%), D, rotamer outliers (%), E, Bond rmsd (A), F. Angle rmsd (degrees), G. C-β deviations.
Supplementary Figure 4 RNA model building.
RNA model-building into a cut-out region of RNA density from Leishmania ribosome1 at a resolution of 2.5 Å (EMDB entry 7025). A. Model building and real-space refinement using base-paired RNA helices. B. Model building and real-space refinement with the same map using standard nucleic acid model building. Potential hydrogen bonds indicated by dotted red lines. Graphics created with Chimera2. References: 1. Shalev-Benami, M. et al. Nat. Commun. 8, 1589 (2017). 2. Pettersen, E.F. et al. J. Comput. Chem. 25, 1605–1612 (2004).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Table 1 and Supplementary Results
Supplementary Data 1
Summary data for all map_to_model results
Supplementary Data 2
Summary data for Figure 2 map_to_model results
Rights and permissions
About this article
Cite this article
Terwilliger, T.C., Adams, P.D., Afonine, P.V. et al. A fully automatic method yielding initial models from high-resolution cryo-electron microscopy maps. Nat Methods 15, 905–908 (2018). https://doi.org/10.1038/s41592-018-0173-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-018-0173-1
This article is cited by
-
All-atom RNA structure determination from cryo-EM maps
Nature Biotechnology (2024)
-
DeepMainmast: integrated protocol of protein structure modeling for cryo-EM with deep learning and structure prediction
Nature Methods (2024)
-
A deep learning-based method for modeling of RNA structures from cryo-EM maps
Nature Biotechnology (2024)
-
CryoREAD: de novo structure modeling for nucleic acids in cryo-EM maps using deep learning
Nature Methods (2023)
-
StarMap: a user-friendly workflow for Rosetta-driven molecular structure refinement
Nature Protocols (2023)