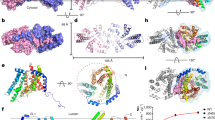
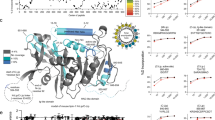
Abstract
Triacylglycerols store metabolic energy in organisms and have industrial uses as foods and fuels. Excessive accumulation of triacylglycerols in humans causes obesity and is associated with metabolic diseases1. Triacylglycerol synthesis is catalysed by acyl-CoA diacylglycerol acyltransferase (DGAT) enzymes2,3,4, the structures and catalytic mechanisms of which remain unknown. Here we determined the structure of dimeric human DGAT1, a member of the membrane-bound O-acyltransferase (MBOAT) family, by cryo-electron microscopy at approximately 3.0 Å resolution. DGAT1 forms a homodimer through N-terminal segments and a hydrophobic interface, with putative active sites within the membrane region. A structure obtained with oleoyl-CoA substrate resolved at approximately 3.2 Å shows that the CoA moiety binds DGAT1 on the cytosolic side and the acyl group lies deep within a hydrophobic channel, positioning the acyl-CoA thioester bond near an invariant catalytic histidine residue. The reaction centre is located inside a large cavity, which opens laterally to the membrane bilayer, providing lipid access to the active site. A lipid-like density—possibly representing an acyl-acceptor molecule—is located within the reaction centre, orthogonal to acyl-CoA. Insights provided by the DGAT1 structures, together with mutagenesis and functional studies, provide the basis for a model of the catalysis of triacylglycerol synthesis by DGAT.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Data availability
The 3D cryo-EM density maps have been deposited into the Electron Microscopy Data Bank under accession numbers EMD-21461 (oleoyl-CoA free), EMD-21481 (with intact oleoyl-CoA) and EMD-21488 (with cleaved oleoyl-CoA). The coordinates have been deposited into the Protein Data Bank (PDB) with accession numbers 6VYI (oleoyl-CoA free) and 6VZ1 (with intact oleoyl-CoA). All other undeposited models and data are available upon request.
References
Cohen, J. C., Horton, J. D. & Hobbs, H. H. Human fatty liver disease: old questions and new insights. Science 332, 1519–1523 (2011).
Cases, S. et al. Identification of a gene encoding an acyl CoA:diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc. Natl Acad. Sci. USA 95, 13018–13023 (1998).
Cases, S. et al. Cloning of DGAT2, a second mammalian diacylglycerol acyltransferase, and related family members. J. Biol. Chem. 276, 38870–38876 (2001).
Lardizabal, K. D. et al. DGAT2 is a new diacylglycerol acyltransferase gene family: purification, cloning, and expression in insect cells of two polypeptides from Mortierella ramanniana with diacylglycerol acyltransferase activity. J. Biol. Chem. 276, 38862–38869 (2001).
Weiss, S. B., Kennedy, E. P. & Kiyasu, J. Y. The enzymatic synthesis of triglycerides. J. Biol. Chem. 235, 40–44 (1960).
Yen, C. L., Stone, S. J., Koliwad, S., Harris, C. & Farese, R. V. Jr. Thematic review series: glycerolipids. DGAT enzymes and triacylglycerol biosynthesis. J. Lipid Res. 49, 2283–2301 (2008).
Yen, C. L., Monetti, M., Burri, B. J. & Farese, R. V. Jr. The triacylglycerol synthesis enzyme DGAT1 also catalyzes the synthesis of diacylglycerols, waxes, and retinyl esters. J. Lipid Res. 46, 1502–1511 (2005).
Wilfling, F. et al. Triacylglycerol synthesis enzymes mediate lipid droplet growth by relocalizing from the ER to lipid droplets. Dev. Cell 24, 384–399 (2013).
Chitraju, C. et al. Triglyceride synthesis by DGAT1 protects adipocytes from lipid-induced ER stress during lipolysis. Cell Metab. 26, 407–418 (2017).
Chitraju, C., Walther, T. C. & Farese, R. V. Jr. The triglyceride synthesis enzymes DGAT1 and DGAT2 have distinct and overlapping functions in adipocytes. J. Lipid Res. 60, 1112–1120 (2019).
Stone, S. J. et al. Lipopenia and skin barrier abnormalities in DGAT2-deficient mice. J. Biol. Chem. 279, 11767–11776 (2004).
Hofmann, K. A superfamily of membrane-bound O-acyltransferases with implications for Wnt signaling. Trends Biochem. Sci. 25, 111–112 (2000).
McFie, P. J., Stone, S. L., Banman, S. L. & Stone, S. J. Topological orientation of acyl-CoA:diacylglycerol acyltransferase-1 (DGAT1) and identification of a putative active site histidine and the role of the N terminus in dimer/tetramer formation. J. Biol. Chem. 285, 37377–37387 (2010).
Cheng, D. et al. Human acyl-CoA:diacylglycerol acyltransferase is a tetrameric protein. Biochem. J. 359, 707–714 (2001).
Caldo, K. M. P. et al. Diacylglycerol acyltransferase 1 is regulated by its N-terminal domain in response to allosteric effectors. Plant Physiol. 175, 667–680 (2017).
Panigrahi, R. et al. Intrinsic disorder in the regulatory N-terminal domain of diacylglycerol acyltransferase 1 from Brassica napus. Sci. Rep. 8, 16665 (2018).
Ma, D. et al. Crystal structure of a membrane-bound O-acyltransferase. Nature 562, 286–290 (2018).
Campaña, M. B. et al. The ghrelin O-acyltransferase structure reveals a catalytic channel for transmembrane hormone acylation. J. Biol. Chem. 294, 14166–14174 (2019).
Grevengoed, T. J., Klett, E. L. & Coleman, R. A. Acyl-CoA metabolism and partitioning. Annu. Rev. Nutr. 34, 1–30 (2014).
Caldo, K. M., Greer, M. S., Chen, G., Lemieux, M. J. & Weselake, R. J. Purification and properties of recombinant Brassica napus diacylglycerol acyltransferase 1. FEBS Lett. 589, 773–778 (2015).
Chang, T. Y., Li, B. L., Chang, C. C. & Urano, Y. Acyl-coenzyme A:cholesterol acyltransferases. Am. J. Physiol. Endocrinol. Metab. 297, E1–E9 (2009).
Röttig, A. & Steinbüchel, A. Acyltransferases in bacteria. Microbiol. Mol. Biol. Rev. 77, 277–321 (2013).
Walther, T. C., Chung, J. & Farese, R. V. Jr. Lipid droplet biogenesis. Annu. Rev. Cell Dev. Biol. 33, 491–510 (2017).
Haas, J. T. et al. DGAT1 mutation is linked to a congenital diarrheal disorder. J. Clin. Invest. 122, 4680–4684 (2012).
Cao, J. et al. Targeting Acyl-CoA:diacylglycerol acyltransferase 1 (DGAT1) with small molecule inhibitors for the treatment of metabolic diseases. J. Biol. Chem. 286, 41838–41851 (2011).
He, S. et al. Discovery of a potent and selective DGAT1 inhibitor with a piperidinyl-oxy-cyclohexanecarboxylic acid moiety. ACS Med. Chem. Lett. 5, 1082–1087 (2014).
Booth, D. S., Avila-Sakar, A. & Cheng, Y. Visualizing proteins and macromolecular complexes by negative stain EM: from grid preparation to image acquisition. J. Vis. Exp. 58, e3227 (2011).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Liao, M., Cao, E., Julius, D. & Cheng, Y. Single particle electron cryo-microscopy of a mammalian ion channel. Curr. Opin. Struct. Biol. 27, 1–7 (2014).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: Fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Ru, H. et al. Molecular mechanism of V(D)J recombination from synaptic RAG1–RAG2 complex structures. Cell 163, 1138–1152 (2015).
Scheres, S. H. A Bayesian view on cryo-EM structure determination. J. Mol. Biol. 415, 406–418 (2012).
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63–65 (2014).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D 66, 213–221 (2010).
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D 60, 2126–2132 (2004).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D 66, 12–21 (2010).
Wallace, A. C., Laskowski, R. A. & Thornton, J. M. LIGPLOT: a program to generate schematic diagrams of protein–ligand interactions. Protein Eng. 8, 127–134 (1995).
Ran, F. A. et al. Genome engineering using the CRISPR–Cas9 system. Nat. Protoc. 8, 2281–2308 (2013).
Gluchowski, N. L. et al. Identification and characterization of a novel DGAT1 missense mutation associated with congenital diarrhea. J. Lipid Res. 58, 1230–1237 (2017).
Pei, J., Kim, B. H. & Grishin, N. V. PROMALS3D: a tool for multiple protein sequence and structure alignments. Nucleic Acids Res. 36, 2295–2300 (2008).
Robert, X. & Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 42, W320–W324 (2014).
Notredame, C., Higgins, D. G. & Heringa, J. T-Coffee: A novel method for fast and accurate multiple sequence alignment. J. Mol. Biol. 302, 205–217 (2000).
Acknowledgements
We thank Z. Li, S. Sterling, R. Walsh and S. Rawson at the Harvard cryo-EM centre and C. Xu, K. Song and K. Lee at the University of Massachusetts cryo-EM facility for electron microscopy data collection; members of the Liao and Farese and Walther laboratories for helpful discussions; B. Schulman and T. Rapoport for comments on the manuscript; and G. Howard for editorial assistance. This work was supported by National Institutes of Health grants R01GM124348 (to R.V.F.), and R01GM097194 (to T.C.W.). X.S. was supported by the American Heart Association postdoctoral fellowship 18POST34030308. T.C.W. is an investigator of the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
X.S., M.L., T.C.W. and R.V.F. conceived the project. X.S. performed protein expression, purification and reconstitution into amphipol, prepared cryo-EM grids, processed cryo-EM data and built the atomic models. M.L. advised on cryo-EM data processing. K.W., N.L.G. and X.S. performed the mutagenesis and activity studies. S.D.E. made genetically modified cell lines. X.S., M.L., T.C.W. and R.V.F. wrote the manuscript. All authors analysed and discussed the results and contributed to the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature thanks David Drew, Savvas N. Savvides and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Purification and characterization of human DGAT1.
a The acyl-transfer reaction catalysed by DGAT1. The enzyme utilizes acyl-CoA as the sole acyl donor and recognizes different lipid molecules (that is, DAG, monoacylcerol (MAG) and fatty alcohol) as the acyl acceptor. The panel shows the reaction with sn-1,2- DAG as the acyl-acceptor and triacylglycerol as the product. The acyl-group in acyl-CoA is coloured in red, and the glycerol backbone in DAG is coloured in blue. b, c, Gel-filtration profile (b) and SDS–PAGE analysis (c) of purified DGAT1 digitonin. Peaks 1 and 2 containing purified DGAT1 by SDS–PAGE analysis were separately collected and pooled. d, Gel-filtration of DGAT1 reconstituted in the amphipol PMAL-C8 that was purified from b. A red (tetramer) and blue (dimer) arrow denote the different oligomerization states of DGAT1. The SDS–PAGE analysis of each peak is shown in the insert. e, Activity analysis of DGAT1-overexpressed microsome or purified enzyme with or without DGAT1 inhibitor (D1i). The protein from the digitonin sample of peak 2 in b and PMAL-C8 sample of the dimer peak in d were used for the assay. The reaction product of triacylglycerol was separated and analysed by TLC. FFA, free fatty acid. f, Quantification of triacylglycerol product shown in e by phosphorimaging. The insert shows the activity of DGAT1 tetramer and dimer from d (mean ± s.d., n = 3 independent experiments). Analysis was performed using two-way ANOVA with Sidak’s post hoc test. g, Representative negative-stain electron micrograph and 2D averages of purified DGAT1 in digitonin (peak 2 in b), and the tetramer and dimer species of DGAT1 in PMAL-C8 (red and blue peaks in d, respectively). The bar in 2D average is 100 Å. Experiments shown in b–e and g were repeated at least three times with similar results. TG, triglyceride.
Extended Data Fig. 2 Cryo-EM image processing of human DGAT1 in PMAL-C8.
a, Representative cryo-EM image of DGAT1 in PMAL-C8. Some DGAT1 particles are outlined by circles. b, The 2D class averages of cryo-EM particle images. The box size of 2D averages is 210 Å. c, Three-dimensional classification and refinement of cryo-EM particles. The initial particle stack was split into two stacks due to the large number of particles for 3D classification. After the first round of classification without imposing symmetry, all of the particles classified into one best class (class 1 contains the most abundant 25% particles) in the final five iterations (indicated as ‘5 cycles’) were kept for further processing. This subset of particles was further classified into three classes by non-alignment classification with C2 symmetry. Afterwards, another round of refinement was performed on each individual class. Among them, class 3 exhibited the highest estimated resolution by Relion and the best side-chain signals by visual inspection and was kept for per-particle contrast transfer function (CTF) and beam-tilt corrections. The resulting cryo-EM map was used for the final cryo-EM maps, contoured at 5σ. d, Unsharpened map (grey) superimposed with the final DGAT1 cryo-EM map (blue) showing the detergent micelle-like signals around DGAT1.
Extended Data Fig. 3 Single-particle cryo-EM analysis of DGAT1 reconstituted in PMAL-C8.
a, Local resolution of the final cryo-EM map of DGAT1. A sliced view of local resolution is shown in the lower panel. b, FSC curves: gold-standard FSC curve between the two half maps with indicated resolution at FSC = 0.143 (red); FSC curve between the atomic model and the final map with indicated resolution at FSC = 0.5 (blue); FSC curve between half map 1 (orange) or half map 2 (green) and the atomic model refined against half map 1. c, Cutaway views of angular distribution of particle images included in the final 3D reconstruction. d, Cryo-EM densities superimposed with the atomic model for individual transmembrane helices (TM1–TM9), resolved N-terminal region (N-ter), and helices in the cytosolic loop region (CL2–CL4). The conserved His415 residue is also labelled. Maps are contoured at 4σ.
Extended Data Fig. 4 Dimer assembly of DGAT1 and phospholipid molecules residing at the dimer interface.
a, Structural details of the DGAT1 dimer interaction. The resolved N-terminal domain (N-ter, amino acids 68–86) of DGAT1 forms a hydrogen-bond network with the opposing subunit depicted in a green box. Dashed lines denote hydrogen-bonding pairs of residues. Hydrophobic interactions mediating the DGAT1 dimer are shown in a red box. b, Densities attributed to phospholipid lipids at the DGAT1 dimer interface are shown as blue surface. The lower panel shows four 1-palmitoyl-2-oleoyl-sn-glycero-3-phosphoethanolamine (POPE) molecules modelled into the density; each pair of POPE densities was symmetry-related and labelled as POPE-A/B and POPE-A′/B′. Maps are contoured at 2.5σ. c, TLC analysis of lipids that co-purified with DGAT1 by iodine staining. The asterisks indicate the presence of phosphatidyl ethanolamine (PE) and DAG in purified DGAT1. PA, phosphatidic acid; PC, phosphatidylcholine. d, e, The SUM159 DGAT1-knockout cell line system analysed by western blot (d) and DGAT1 activity analysis by using lysates from SUM159 DGAT1-knockout cells or cells transiently overexpressing DGAT1 (e). f, Truncation of N termini reduces DGAT1 expression. Fluorescence size-exclusion chromatography (FSEC) and western blot analyses of two N-terminal truncations lacking the first 85 and 90 residues. The asterisks in the gel-filtration profiles denote the DGAT1-containing peak. The dashed line marks the peaking containing free GFP. g, h, Truncation of N-terminal region reduces DGAT1 activity. TLC analysis of triacylglycerol products is shown in g. Each assay was performed in triplicate. The final triacylglycerol products were normalized to the protein expression level with the quantification results shown in h. Mean ± s.d., n = 3 independent experiments. Analysis was performed using one-way ANOVA with Dunnett’s post hoc test. Analysis shown in c was performed once; experiments in d–g were repeated three times with similar results.
Extended Data Fig. 5 Structural comparison of DGAT1 with DltB.
a, Structural superposition of DGAT1 (blue) and DltB (orange, PDB ID: 6BUI)17. The two structures were superimposed with an r.m.s.d. of 1.39 Å over 360 matched Cα positions. b, As a, but the protein structures are shown as cylinders with the conserved histidine residue shown as sticks. The area denoted by the red dashed line exhibits a similar overall architecture in DGAT1 and DltB (MBOAT core) that is not found in other protein structures. Note that beyond this common area, the resolved N-terminal and TM1 regions in DGAT1 appear as extra structural elements beyond the MBOAT-core region as compared to DltB. c, Structure-based sequence alignment of DGAT1 and DltB. The squiggles on the top represent α-helices in DGAT1 (blue) and DltB (orange). Sequences in the black rectangle indicate the most structurally variable region in the two enzymes. In DGAT1, these regions are involved in dimer formation, whereas in DltB, the equivalent two helices (TM1 and TM2) seal off the lateral cavity within the membrane (see main text). Sequence in green rectangle denotes the alanyl-donor binding pocket in DltB. The red triangle denotes the conserved histidine residue. The alignment was performed with PROMALS3D43, and the final alignment figure was generated with ESPript 3.044.
Extended Data Fig. 6 DGAT1 sequence alignment and conservation analysis of MBOAT enzymes.
a, Sequence alignment of DGAT1 enzymes from Homo sapiens, Mus musculus, Bos taurus, Xenopus laevis, Danio rerio, Drosophila melanogaster, Caenorhabditis elegans, Brassica napus, Arachis hypogaea and Chaetomium thermophilum. The colour scheme of amino acids is based on their physicochemical properties. A red triangle denotes the highly conserved histidine residue; black triangles denote highly conserved polar residues in the DGAT1 active centre; black squares denote residues interacting with oleoyl-CoA. The residue numbers for human DGAT1 are indicated at the top. The sequence alignment was performed with T-Coffee45, and the final alignment figure was generated with ESPript 3.044. b, Sequence alignment of MBOAT enzymes that acylate lipids (DGAT1 and ACAT1) or proteins (PORCN and GOAT). Structural information of DGAT1 was incorporated into the alignment, where the regions containing a cluster of conserved residues among MBOAT enzymes were labelled as blue boxes. Note the alignment starts at Arg250 of DGAT1. c, Mapping the conserved blue region shown in b into the DGAT1 structure. The DGAT1 structure is shown as grey cylinders. Blue areas denote the conserved region among MBOATs. Note the acyl-CoA binding tunnel in DGAT1 containing the most conserved region among MBOATs (blue area in b) is highlighted by the dashed circle.
Extended Data Fig. 7 Cryo-EM image processing of human DGAT1 supplemented with acyl-CoA substrate.
a, Representative cryo-EM image of DGAT1 with oleoyl-CoA. Some DGAT1 particles are marked by circles. b, 2D class averages of cryo-EM particle images with the box size of 210 Å. c, Three-dimensional classification and refinement of cryo-EM particle images. The processing flow is similar to the dataset without acyl-CoA treatment (Extended Data Fig. 2). After the second round of 3D classification, all particles classified into class 6 during the final five iterations (indicated as ‘5 cycles’) were kept for next round of non-alignment classification into six classes. Particles in each class (estimated resolution shown in parentheses) were individually refined. After per-particle CTF and beam-tilt corrections, two resulting maps were chosen to represent DGAT1 complexed with the intact and broken oleoyl-CoA molecule.
Extended Data Fig. 8 Single-particle cryo-EM analysis of DGAT1 with acyl-CoA substrate.
a, Local resolution of the final cryo-EM map of DGAT1 (shown as sliced view) complex with an intact oleoyl-CoA molecule. b, FSC curves: gold-standard FSC curve between the two half maps with indicated resolution at FSC = 0.143 (red); FSC curve between the atomic model and the final map with indicated resolution at FSC = 0.5 (blue); FSC curve between half map 1 (orange) or half map 2 (green) and the atomic model refined against half map. c, Cutaway views of angular distribution of particle images included in the final 3D reconstruction. d–f, Similar to a–c, but showing the cryo-EM analysis of the map with a broken oleoyl-CoA density. Note that in e, only the FSC curve between half maps was calculated. g, Cryo-EM densities with the intact oleoyl-CoA density (shown in a–c) superimposed with the atomic model for individual transmembrane helices similar to that shown in Extended Data Fig. 3d. Maps are contoured at 4σ.
Extended Data Fig. 9 Cryo-EM density analysis of the bound acyl-CoA and lipid-like density and mutagenesis study of residues interacting with oleoyl-CoA substrate.
a, A side-by-side density comparison of the bound oleoyl-CoA molecule, oleoyl-CoA with a broken density, and density in the acyl-CoA binding pocket without oleoyl-CoA treatment. b, Cryo-EM density map of the oleoyl-CoA molecule and the surrounding region. Maps are shown at different contour levels to illustrate mobility of the adenosine moiety (red arrowhead) of oleoyl-CoA. By contrast, the phosphopantetheine region (green arrowhead) exhibits a stronger density signal. c, Activities of acyl-CoA binding tunnel alanine mutants of DGAT1 expressed in DGAT1-knockout cells. d, Activities of the bulky side-chain substitutions of residues in the acyl-CoA binding tunnel. Activities in c and d were normalized to the amount of DGAT1 protein expressed (mean ± s.d., n = 3 independent experiments). Analysis was performed using one-way ANOVA with Dunnett’s post hoc test. e, Effect of the acyl chain of the acyl-donor substrate on DGAT1 activity. The analyses used purified DGAT1 in amphipol (mean ± s.d., n = 3 independent experiments). Analysis was performed using one-way ANOVA with Dunnett’s post hoc test comparing C18:1-CoA to other acyl-CoA substrates (coloured in dark grey). f, The zoomed-in views of the lipid-like density in the oleoyl-CoA–free DGAT1 monomer structure shown as surface representation. The protein surface at His415 in the zoomed-in view is coloured in orange. The map is contoured at 3.5σ. g, Cryo-EM density in orange showing both the bound acyl-CoA with a broken signal and the lipid-like density. A black arrowhead denotes the disconnected acyl chain in oleoyl-CoA molecule. A cyan arrowhead indicates the connected density between the acyl chain of oleoyl-CoA and the uncharacterized lipid-like density at a lower contour level.
Extended Data Fig. 10 FSEC, western blot and activity analyses of DGAT1 mutants.
a, FSEC profiles of wild type and all tested mutants in this study. Wild-type DGAT1 and mutants were transiently expressed as GFP-fusion proteins in HEK293 cells. The folding of each mutant was analysed by size-exclusion chromatography monitoring GFP fluorescence. All tested mutants exhibit peaks similar to the wild-type DGAT1 protein, suggesting their correct overall protein folding. b, Western blot analyses of wild-type and tested DGAT1 mutant proteins expressed in SUM159 DGAT1-knockout cells. Untrans., untransfected control. c, Activity of wild type and selected DGAT1 mutants purified by His-tag affinity chromatography purification. The top panel of the raw TLC plates shows the formation of triacylglycerol. The assays were performed with or without DGAT1 inhibitor (D1i) (mean ± s.d., n = 3 independent experiments). One-way ANOVA with Dunnett’s post hoc test was applied. Experiments shown in a–c were repeated twice with similar results.
Supplementary information
Rights and permissions
About this article
Cite this article
Sui, X., Wang, K., Gluchowski, N.L. et al. Structure and catalytic mechanism of a human triacylglycerol-synthesis enzyme. Nature 581, 323–328 (2020). https://doi.org/10.1038/s41586-020-2289-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-020-2289-6
This article is cited by
-
Lipid droplets and cellular lipid flux
Nature Cell Biology (2024)
-
Identification of an alternative triglyceride biosynthesis pathway
Nature (2023)
-
Lipid droplets are a metabolic vulnerability in melanoma
Nature Communications (2023)
-
Mechanism of d-alanine transfer to teichoic acids shows how bacteria acylate cell envelope polymers
Nature Microbiology (2023)
-
Lipid droplet biogenesis and functions in health and disease
Nature Reviews Endocrinology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.