Abstract
Large cutaneous ulcers are, in severe cases, life threatening1,2. As the global population ages, non-healing ulcers are becoming increasingly common1,2. Treatment currently requires the transplantation of pre-existing epithelial components, such as skin grafts, or therapy using cultured cells2. Here we develop alternative supplies of epidermal coverage for the treatment of these kinds of wounds. We generated expandable epithelial tissues using in vivo reprogramming of wound-resident mesenchymal cells. Transduction of four transcription factors that specify the skin-cell lineage enabled efficient and rapid de novo epithelialization from the surface of cutaneous ulcers in mice. Our findings may provide a new therapeutic avenue for treating skin wounds and could be extended to other disease situations in which tissue homeostasis and repair are impaired.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Data from the microarray, RNA-seq and miRNA microarray analyses have been deposited in the Gene Expression Omnibus (GEO) and ArrayExpress under accession numbers GSE85803, GSE106419 and E-MTAB-5055, respectively. All other relevant data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Gonzales, K. A. U. & Fuchs, E. Skin and its regenerative powers: an alliance between stem cells and their niche. Dev. Cell 43, 387–401 (2017).
Sun, B. K., Siprashvili, Z. & Khavari, P. A. Advances in skin grafting and treatment of cutaneous wounds. Science 346, 941–945 (2014).
Gurtner, G. C., Werner, S., Barrandon, Y. & Longaker, M. T. Wound repair and regeneration. Nature 453, 314–321 (2008).
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Srivastava, D. & DeWitt, N. In vivo cellular reprogramming: the next generation. Cell 166, 1386–1396 (2016).
Driskell, R. R. et al. Distinct fibroblast lineages determine dermal architecture in skin development and repair. Nature 504, 277–281 (2013).
Moll, R., Divo, M. & Langbein, L. The human keratins: biology and pathology. Histochem. Cell Biol. 129, 705–733 (2008).
Boukamp, P., Breitkreutz, D., Stark, H. J. & Fusenig, N. E. Mesenchyme-mediated and endogenous regulation of growth and differentiation of human skin keratinocytes derived from different body sites. Differentiation 44, 150–161 (1990).
Van Muijen, G. N., Warnaar, S. O. & Ponec, M. Differentiation-related changes of cytokeratin expression in cultured keratinocytes and in fetal, newborn, and adult epidermis. Exp. Cell Res. 171, 331–345 (1987).
Okazaki, M., Yoshimura, K., Fujiwara, H., Suzuki, Y. & Harii, K. Induction of hard keratin expression in non-nail-matrical keratinocytes by nail-matrical fibroblasts through epithelial–mesenchymal interactions. Plast. Reconstr. Surg. 111, 286–290 (2003).
Grimm, D. J. et al. In vitro and in vivo gene therapy vector evolution via multispecies interbreeding and retargeting of adeno-associated viruses. J. Virol. 82, 5887–5911 (2008).
Snippert, H. J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143, 134–144 (2010).
Hirsch, T. et al. Regeneration of the entire human epidermis using transgenic stem cells. Nature 551, 327–332 (2017).
De Luca, M., Pellegrini, G. & Green, H. Regeneration of squamous epithelia from stem cells of cultured grafts. Regen. Med. 1, 45–57 (2006).
Schmitz, A. et al. Assessing the in vivo epidermal barrier in mice: dye penetration assays. J. Invest. Dermatol. 135, 1–4 (2015).
Robson, M. C. et al. The safety and effect of topically applied recombinant basic fibroblast growth factor on the healing of chronic pressure sores. Ann. Surg. 216, 401–408 (1992).
Akita, S., Akino, K., Imaizumi, T. & Hirano, A. Basic fibroblast growth factor accelerates and improves second-degree burn wound healing. Wound Repair Regen. 16, 635–641 (2008).
Chapman, S., Liu, X., Meyers, C., Schlegel, R. & McBride, A. A. Human keratinocytes are efficiently immortalized by a Rho kinase inhibitor. J. Clin. Invest. 120, 2619–2626 (2010).
Chandler, R. J., Sands, M. S. & Venditti, C. P. Recombinant adeno-associated viral integration and genotoxicity: insights from animal models. Hum. Gene Ther. 28, 314–322 (2017).
Kotterman, M. A. & Schaffer, D. V. Engineering adeno-associated viruses for clinical gene therapy. Nat. Rev. Genet. 15, 445–451 (2014).
Melino, G., Memmi, E. M., Pelicci, P. G. & Bernassola, F. Maintaining epithelial stemness with p63. Sci. Signal. 8, re9 (2015).
Louis Jeune, V., Joergensen, J. A., Hajjar, R. J. & Weber, T. Pre-existing anti-adeno-associated virus antibodies as a challenge in AAV gene therapy. Hum. Gene Ther. Methods 24, 59–67 (2013).
Rheinwatd, J. G. & Green, H. Serial cultivation of strains of human epidermal keratinocytes: the formation of keratinizing colonies from single cells. Cell 6, 331–343 (1975).
Harajly, M. et al. p53 restoration in induction and maintenance of senescence: differential effects in premalignant and malignant tumor cells. Mol. Cell. Biol. 36, 438–451 (2016).
Provost, J. J. et al. Urokinase plasminogen activator receptor induced non-small cell lung cancer invasion and metastasis requires NHE1 transporter expression and transport activity. Cell Oncol. (Dordr.) 35, 95–110 (2012).
Shimizukawa, R. et al. Establishment of a new embryonic stem cell line derived from C57BL/6 mouse expressing EGFP ubiquitously. Genesis 42, 47–52 (2005).
Roesch, K. et al. The transcriptome of retinal Müller glial cells. J. Comp. Neurol. 509, 225–238 (2008).
Dassule, H. R., Lewis, P., Bei, M., Maas, R. & McMahon, A. P. Sonic hedgehog regulates growth and morphogenesis of the tooth. Development 127, 4775–4785 (2000).
Song, K. et al. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 485, 599–604 (2012).
Muzumdar, M. D., Tasic, B., Miyamichi, K., Li, L. & Luo, L. A global double-fluorescent Cre reporter mouse. Genesis 45, 593–605 (2007).
Kurashige, C. et al. Roles of receptor activity-modifying protein 1 in angiogenesis and lymphangiogenesis during skin wound healing in mice. FASEB J. 28, 1237–1247 (2014).
Hardman, M. J., Sisi, P., Banbury, D. N. & Byrne, C. Patterned acquisition of skin barrier function during development. Development 125, 1541–1552 (1998).
Matsuki, M. et al. Defective stratum corneum and early neonatal death in mice lacking the gene for transglutaminase 1 (keratinocyte transglutaminase). Proc. Natl Acad. Sci. USA 95, 1044–1049 (1998).
Acknowledgements
This work was supported by MEXT KAKENHI Grant numbers JP26293381(Grant-in-Aid for Scientific Research (B) to M.K.), JP23689073 (Grant-in-Aid for Young Scientists (A) to M.K.), JP21689046 (Grant-in-Aid for Young Scientists (A) to M.K.), Kyorin University research promotion award to M.K. (2013), JSPS Overseas Research Fellowships (2015–17) to M.K., and the Uehara Memorial Foundation Research Fellowship for Research Abroad (2017–18) to M.K. M.K. thanks H. Green for support materials. T.H. thanks F. Sugiyama for support materials. M.N.S. is supported by NIH-NCI CCSG: P30 014195 and The Leona M. and Harry B. Helmsley Charitable Trust. Work in the laboratory of J.C.I.B. was supported by the G. Harold and Leila Y. Mathers Charitable Foundation, The Leona M. and Harry B. Helmsley Charitable Trust, The Moxie Foundation, The Evergreen Foundation, Fundacion Dr. Pedro Guillen and Universidad Católica San Antonio de Murcia (UCAM).
Reviewer information
Nature thanks S. Akita, V. Horsley, A. Lombardo and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
M.K., K.H. and J.C.I.B. conceived and designed the experiments. M.K. conceptualized the study and designed and performed most of the experiments and analysis. T.A., T.H., M.S., M.Y. and R.H.-B. prepared the animals and embryos. T.A. and M.Y. helped with histological imaging. K.S., Y.T. and A.S. helped with preparation of plasmid constructs and AAVs. Y.T. and H.E. helped with 3D organotypic cultures. T.H. helped with tumorigenic assay and luciferase detection. M.N.S. analysed the RNA-seq datasets. M.K., D.D.O., J.W., A.O., P.R. and J.C.I.B. prepared the figures and wrote the manuscript. P.M., E.N.D., K.H. and J.C.I.B. coordinated and oversaw the study.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Selection of factors for reprogramming of human mesenchymal cells into iSEPs.
a, Gene-expression analyses from DNA microarrays of human primary keratinocytes and primary hDFs. Pink or red indicates higher expression levels, green indicates lower expression levels. b, MicroRNA microarray analyses from three pairs of primary human keratinocytes and primary hDFs obtained from different tissues of a single subject. c, Reversal participation analysis of a candidate gene set (19 genes) for keratinocyte specification. d, Changes in the expression levels of candidate transcription factors after calcium-induced terminal differentiation of keratinocytes. e, Changes in the expression levels of keratinocyte markers after transduction of each candidate gene as assessed by quantitative PCR (qPCR). f, Schematic of the experimental design for the generation of iSEPs from primary hDFs. g, Morphological analysis of primary hDFs and human primary keratinocytes. Arrows indicate keratinocyte colonies on feeder cells. Scale bars, 200 μm. Similar results were observed five times for both cell types. h, Representative bright field images showing the morphology of colonies obtained during the selection of factors for the generation of iSEPs. Combinations of factors were optimized on the basis of colony morphology. Scale bars, 200 μm. i, Representative bright field images (left and middle) showing the morphology of colonies at passage 0 (P0) and passage 3 (P3) obtained after the transduction of 28 factors. The KRT14-RFP vector was transduced with the 28 factors (right). Red scale bars, 500 μm; yellow scale bars, 200 μm. j, RT–PCR analyses of keratinocyte markers. h, i, j, Images are from one experiment. k, Growth curve of primary keratinocytes and 28TF-iSEPs on feeder cells. Data are mean of technical triplicates. l, Representative images showing H&E staining of human skin and 3D organotypic culture of 28TF-iSEPs-hDF. Scale bars, 100 μm. Similar results were observed in two organotypic cultures. m, Transgene-specific PCR analyses of genomic DNA of 28TF-iSEPs. Each plasmid was used for comparative controls. Images are from one experiment. n, Schematic of the experiment for comparative assessment of transduction efficiency and cytotoxicity between concentrated enhanced GFP (eGFP)-expressing retroviruses and lentiviruses. o, Higher eGFP expression could be obtained with lower cytotoxicity with retroviruses than lentiviruses. Consistent findings were observed in three technical replicates for microscopic findings and flow cytometric analyses. Results of MTT (3-[4,5-dimethylthiazol-2-yl]-2,5 diphenyl tetrazolium bromide) assay represent the mean of three technical replicates. Overlaid dot plots indicate the distribution of the data. p, Schematic of the experiment for generation of iSEPs-ADSCs. q, Representative bright field images showing colony morphologies during factor reduction. Scale bars, 200 μm. r, Transgene-specific PCR analyses of genomic DNA. Plasmids were used as controls. Integration of plasmid-derived sequences is described. q, r, Images are from one experiment. s, Representative images showing the morphological analysis and H&E staining of 3D organotypic culture of iSEPs-ADSCs generated by the transduction of DNP63A and GRHL2. White scale bars, 200 μm; black scale bar, 100 μm. Similar results were confirmed in two independent experiments. j, m, r, For gel source data, see Supplementary Fig. 1. o, For gating strategy example, see Supplementary Fig. 2.
Extended Data Fig. 2 Optimization of factors for reprogramming of hADSCs into iSEPs.
a, Bright field images showing the representative morphology of colonies that emerged during generation of iSEPs after transduction of 67 combinations of factors tested in addition to the minimum factors DNP63A and GRHL2. Scale bars, 1,000 μm. b, Representative H&E staining images of 3D organotypic cultures of iSEPs induced with 35 combinations of factors. Scale bars, 100 μm. c, Rhodanile staining of cells generated with selected combinations of factors 14 days after transduction. d, Schematic representation of the experiment for testing the combinations of DG and non-MYC factors. e, Representative bright field images showing cell morphologies of generated iSEPs. Yellow scale bars, 1,000 μm; red scale bars, 500 μm. f, Quantification of iSEPs generated with different combinations of factors on day 22. Values represent the mean of three technical replicates. Overlaid dot plots indicate the distribution of the data. g, Growth curves of iSEPs and human primary keratinocytes. Cells with GRHL1 could not be isolated. Data are average of triplicates. h, H&E staining and immunohistochemical analysis of 3D organotypic cultures. Scale bars, 100 μm. i, Schematic representation of the experiment for testing the combinations of DGM+1 factors. j, Representative bright field images showing cell morphologies of generated iSEPs. Yellow scale bars, 1,000 μm; red scale bars, 500 μm. k, Quantification of iSEPs generated with different combinations of factors on day 19. Values represent mean of three technical replicates. Overlaid dot plots indicate the distribution of the data. l, Growth curves of iSEPs and human primary keratinocytes (hPK1 and hPK4), and the cumulative population doublings of iSEPs on day 60. Data are the mean of triplicates. m, H&E staining and immunohistochemical analysis of 3D organotypic cultures. Scale bars, 100 μm. a, b, h, m, Findings were confirmed in two technical replicates. c, e, j, Findings were confirmed in two independent experiments.
Extended Data Fig. 3 Defining the procedure of in vivo experiments.
a, Lineage tracing using Krt14cre;LSLtdTomato mice. b, H&E staining and immunohistochemical analysis of ear biopsies from Krt14cre;LSLtdTomato mice. c, Exterior and stereoscopic view of Krt14cre;LSLtdTomato mice. d, Appearance and stereoscopic view of Krt14cre;LSLtdTomato mice after resection of skin. White arrows indicate signals from glands such as the thyroid and mammary glands. e, Appearance and stereoscopic view of resected mammary gland. f, Appearance and H&E staining of resected axillary skin and subcutaneous tissues including the mammary gland. g, Stereoscopic and immunohistochemical analysis of resected skin and subcutaneous tissues including the mammary gland. h, Appearance and stereoscopic analysis after chamber attachment. tdTomato signals were not detected inside the chamber when attached at the interscapular area and lateral thoracic area, whereas signals were detected when attached close to the axilla. The procedure was optimized not to include the mammary gland within the wound chamber. i, Schematic representation of the subcutaneous injection of AAVs into the back skin of mice. Eighteen serotypes of AAVs expressing eGFP under the CAG promoter were tested. j, Fluorescence images showing GFP signals of the injected skin and subcutaneous tissue 1 week after the injection of eGFP-expressing AAVs of different serotypes. AAVDJ resulted in the highest levels of GFP fluorescence. Scale bar, 200 μm. k, Schematic representation of the inoculation of AAVs in a skin chamber on the back of a mouse. Tested AAVs included: (1) AAVDJ with or without the addition of surfactant, (2) AAVs previously identified as optimal for skin and wounds (AAV2 and AAV5), and (3) new AAV serotypes (AAV9 and AAV10). Highly concentrated retroviruses were used as a control. l, H&E and fluorescence analyses of ulcers 1 week after the inoculation of eGFP-expressing viruses. AAVDJ without surfactant yielded by far the highest gene transduction efficiency. Scale bar, 200 μm. m, In vivo luminescent images 1 week after the administration of luciferase-expressing AAVs through tail vein injection, interscapular subcutaneous injection or inoculation into a chamber on the back. Systemic directivity of AAVDJ is mainly focused to the liver. B, brain; Ce, caecum; Co, colon; Es + St, oesophagus and stomach; Ey, eye; F, fat; H, heart; K + Ad, kidney and adrenal gland; L, liver; Lu, lung; Ov + U, ovary and uterus; Pa, pancreas; SI, small intestine; Sk, skin; Sp, spleen. Similar findings were obtained in five (b), three (c–g, l) and two (h, j, m) independent experiments.
Extended Data Fig. 4 Generation of epithelial tissue after DGTM-AAV administration in vivo.
a, H&E analysis of epithelial tissue 18 days after DGTM-AAV administration. Both high magnification (left) and low magnification images (right) are shown. Viral titres are indicated on the left (×1010 GC/animal). Black scale bars, 500 μm; red scale bars, 2 mm. Data from 14 out of 25 animals treated with different titres of DGTM-AAV as shown (summarized data is shown in Fig. 1d). b, Lineage tracing using Pdgfracre;R26Rconfetti mice. c, A representative image showing GFP, YFP, RFP fluorescence and Pdgfra staining of the subcutaneous tissue isolated from the back skin of a Pdgfracre;R26Rconfetti mouse. Mesenchymal cells were differentially labelled with GFP, YFP, RFP or not labelled. Similar findings were confirmed in five animals. c, Representative visual, stereoscopic and histological images showing single-cell-derived epithelial cell clusters 14 days after DGTM-AAVs administration. Similar findings were confirmed in two animals (out of three animals treated with the same procedure). d, e, Representative visual, stereoscopic, and histological images showing participation of YFP-labelled and non-labelled epithelial cell clusters to a single epithelial tissue on day 14 (d) and day 18 (e). Similar findings were confirmed in all three animals treated with the same procedure for each day. f, Representative visual and stereoscopic images of three mice (left) showing that multicolour-labelled cell clusters contribute to epithelial tissues covering the ulcer surface inside the chamber. Histological images (right) of one animal showing the participation of GFP, YFP, RFP and unlabelled cells to generated epithelial tissue. Similar findings were confirmed in all three animals. g, For the estimation of cell number in deep fascia at the time of surgery, deep fascia was dissected out just after the attachment of the chamber. h, Stereoscopic images showing a horizontal image of the deep fascia after DAPI staining (left). Imaging analysis for the counting of nucleated cells (right). g, h, All three samples were processed with similar findings. i, Estimated cell numbers in the deep fascia just after the attachment of chamber for three animals. Data represents mean ± s.d. of from ten stereoscopic images. Overlaid dot plots indicate the distribution of the data. c–h, White scale bar, 5 mm; black scale bars, 1 mm; red scale bars, 200 μm; yellow and magenta scale bars, 100 μm. External diameter of the chamber, 12.5 mm.
Extended Data Fig. 5 Reprogramming of mouse mesenchymal cells with different combinations of DGTM-AAVs.
a, Mesenchymal cells were sorted out from mouse adipose-derived stromal cell fractions by PdgfracreER-driven tdTomato signals with the proportion of 63.2 ± 16.0% (n = 12, established from different animals). For gating strategy example, see Supplementary Fig. 2. Scale bar, 200 μm. b, Fluorescence images of the cells 3 days after addition of GFPNLS-AAV with indicated titre in vitro. GFPNLS expression increases with increase of titre. Scale bars, 200 μm. Similar findings were confirmed in two experiments. c, Results of MTT cell-viability assay of the cells 3 days after the addition of GFPNLS-AAV at the indicated titre in vitro. Overlaid dot plots indicate the distribution of the data (n = 6, technical replicates). d, Schematic of the experimental design. e, Time course stereoscopic and fluoroscopic analysis of the emergence of an iSEPs colony. Yellow scale bar, 5 mm; red scale bar, 1 mm; white scale bar, 200 μm. f, Immunocytochemical analysis of the iSEPs colony in e. e, f, Similar findings were confirmed in two sets of eight wells of samples. g, Time course immunocytochemical analysis of iSEPs colony emergence. iSEPs colonies are strongly positive for Krt14 and Pdgfra on initial emergence. With time, Pdgfra signal intensity decreases. Scale bars, 200 μm. Similar findings were confirmed in a set of eight wells of samples. h, Appearance (left) and stereoscopic analysis (right) of transplanted cell sheet. tdTomato signals indicates the area of cell sheet survival. Yellow scale bars, 1.0 cm. i, Findings of H&E and immunohistological analysis of transplanted cell sheet. Arrows indicate the position of magnified findings in j. Orange scale bars, 2 mm. j, H&E and immunohistological analysis of transplanted cell sheet. White scale bars, 100 μm. h, i, j, Similar findings were confirmed in five animals in two sets of experiments. k, Schematic representation of the experimental design at the lower titre of virus. l, DGTM-AAVs transduced wells. Arrows indicates epithelial shaped colonies. The dotted outline is shown magnified on the right. White scale bar, 5 mm; yellow scale bars, 1 mm. m, Immunocytochemical analysis of epithelial shaped colonies. White scale bars, 1 mm. l, m, Similar findings were confirmed in two sets of three wells (six-well plate). n, Time course numbers of epithelial colonies in eight wells (24-well plate) treated with DGTM-AAVs. Similar findings were confirmed in two series of experiments. o, Immunocytochemical analysis of epithelial cells obtained after transduction of different combinations of factors into Pdgfra+ mADSCs. Scale bar, 50 μm. Images are representative of one experiment. p, Proliferation of epithelial cells obtained after transduction of different combinations of factors into Pdgfra+ mADSCs. Data are mean of triplicates. q, Schematic representation of clonogenicity assessment by expansion of single-cell-derived clones. r, Analysis of ulcers 28 days after administration of nine combinations of AAV and no virus control (ten animals for each group, including samples shown in Fig. 1b, e). Green arrows indicate epithelial tissues confirmed by histological analysis. External diameter of chamber, 12.5 mm. Data are summarized in Fig. 2c–e. Findings were confirmed in a set of experiment. s, Epithelial tissue generated with DTM-, DGM-, DGT-, and DM-AAVs in vivo. Arrows indicate generated epithelial tissue. Black and white scale bars, 2 mm; yellow scale bars, 200 μm. Similar findings were confirmed in three (DTM), six (DGM), two (DGT) or one (DM) animals (out of ten animals for each group).
Extended Data Fig. 6 DGTM-AAVs enable generation of epithelium with the ability to cover ulcers.
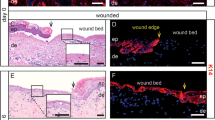
a, Appearance (top left) and immunohistochemical analysis (top middle) of ulcers, 28 days after the administration of DGTM-AAV in PdgfracreER;LSLtdTomato mice, analysed for KRT14 and tdTomato expression. Yellow arrows indicate the periphery of the generated epithelium. The white dotted line indicates the approximate position of the histological section. The white dotted outline indicates position of the magnified panels (bottom). The solid white box indicates the position of the magnified panels of epidermis and generated epithelium (right). White scale bars, 3 mm; black scale bar, 100 μm. Similar findings were confirmed in three animals. b, Appearance of the ulcer before (top left) and after (bottom left) biopsy of generated epithelium 28 days after administration of DGTM-AAV in PdgfracreER;LSLtdTomato mice and immunohistochemical analysis (top middle) of biopsied generated epithelium. Intensity of the tdTomato signal in the biopsied sample is higher than in the chamber (see a) because fixation without the chamber allowed the sample to contract. Yellow arrows indicate the position of the biopsy. The white dotted outline indicates the position of the magnified panels (lower middle). The white solid outline indicates the position of the magnified panels of generated epithelium (far right). White scale bars, 3 mm; black scale bar, 100 μm. Similar findings were confirmed in five biopsies. c, Experimental design of flow cytometric analysis of generated epithelium. d, Representative appearance of an ulcer 18 days after the administration of DGTM-AAV in PdgfracreER;LSLtdTomato mice (left) and primary cultured cells on feeder (middle) and no feeder condition (right). Yellow arrows indicate generated epithelium subjected to flow cytometric analysis. White scale bar, 3 mm; yellow scale bars, 200 μm. Similar findings were confirmed in three animals. e, Flow cytometric analysis of three primary cultured cells from the surface of an ulcer 18 days after administration of DGTM-AAV in PdgfracreER;LSLtdTomato mice (day 18 iSEPs). Primary keratinocytes from the back skin of Krt14cre;LSLtdTomato mice (top) and wild-type mice (bottom) were prepared for positive and negative controls, respectively. For gating strategy example, see Supplementary Fig. 2. f, Schematic of stepwise operations performed with representative images. A wound was created and treated with DGTM-AAVs. After the generation of epithelial tissue, the initial skin chamber was removed and replaced with a larger one (twice the area of the small chamber). After the large chamber was removed, early contraction of the generated epithelium was prevented by a rubber ring. White scale bars, 5 mm. g, Chronological changes in surface area of generated epithelia in the large chamber. Coloured lines represent animals treated with DGTM-AAV (n = 10). The black line represents animals without AAV (n = 5). h, Representative images showing the gross appearance of ulcers in large chambers at different time points. White scale bars, 5 mm. Similar findings were confirmed in ten animals for DGTM-AAV+ group and five animals for the no-virus group. i, Appearance (top left), stereoscopic analysis (bottom left), H&E staining (top right) and immunohistochemical analysis (bottom right) of skin and subcutaneous tissue including generated epithelium on day 93. Yellow arrows indicate the periphery of the generated epithelium. Dotted lines in the left panels indicate the approximate position of the histological sections shown on the right. Stereoscopic analysis revealed areas of generated tissues that are not clear by appearance. Dotted outlines indicate position of magnified panels. Scale bars, 5 mm. Similar findings were confirmed in all animals monitored over days 90–110 (n = 10). j, A representative H&E staining of liver tissue 3 months after the administration of DGTM-AAVs for induction of lineage conversion. Scale bar, 1.0 cm. Similar findings were confirmed in five animals. k, Complete blood count analysis after administration of AAV-DGTM (n = 4 for experimental and control groups). l, Blood chemistry analysis after administration of DGTM-AAVs (n = 3 for experimental and control groups). k, l, Data are mean ± s.d.; statistical differences are analysed with a two-sided Student’s t-test. m, Schematic of the skin island in chamber assay performed with representative images (intact skin group). n, Schematic of wounding, epithelialization and the skin island in chamber assay performed with representative images (epithelialized skin group). o, Schematic of skin biopsy, primary culture, cell-sheet transplantation and the skin island in chamber assay performed with representative images (cell sheet group). p, Schematic of chamber attachment and DGTM-AAV administration, chamber detachment, and the skin island in chamber assay performed with representative images (generated epithelium group). q, Appearances of skin island in chamber assay performed for pure cell sheet transplanted areas. Skin, purely composed of a cell sheet disappeared with time within the ulcer. Similar findings were confirmed in two animals. With these findings, we allowed the transplants to contract for another 1–2 weeks before subjecting the cell sheets to the skin island assay in the cell sheet group. r, Chronological changes in surface area of epithelial tissues after skin island creation and chamber attachment. Mean of four groups (left top), mean (thick line) with respective animals (thin line) for intact skin (top middle, n = 8), epithelialized skin (top right, n = 8), cell sheet (bottom left, n = 6), and generated epithelium (bottom right, n = 8) groups. s, Appearances of skin island in animals with maximum and minimum epithelialized areas for each group on day 14 of the skin island in chamber assay. The abilities of generated epithelium to laterally expand within an ulcer were more variable than seen with controls. m–q, s, External diameter of chamber, 14.3 mm. t, H&E staining and immunohistochemical analyses (K10 and K13) of generated tissues by DGTM-AAV at different time points (day 18, n = 18; days 28–30, n = 20; days 90–11, n = 10). Samples collected from the same generated epithelial tissues by partial biopsy (days 28–30) and thorough histological investigation (days 90–110) are indicated with asterisks (bottom two rows of the middle and left columns). In these two animals, K13 expression was confirmed in a biopsied sample at day 28, but after 3 months, K13 expression had been extinguished. Results are summarized in Fig. 4b. u, H&E staining and immunohistochemical analysis of mouse fetal skin at different gestational ages. Mouse fetal skin was transiently positive for K13. Similar findings were confirmed in three embryos from two different mothers for each gestational age.
Extended Data Fig. 7 In vivo reprogramming in a clinically relevant context.
a, Schematic of investigation of influences of DGTM-AAVs administration on the vascularity of the wound bed. b, Appearance (top left) and immunohistochemical analysis of microvessel densities (right top and bottom) of an ulcer 7 days after the administration of DGTM-AAVs for CD31. Black and white boxes indicate the position of the magnified panels (lower). Yellow and red dotted lines indicate tissues above and below the deep fascia, respectively. Microvessel density was histologically evaluated for tissues above and below deep fascia. Scale bar, 1 mm. c, d, Analysis of microvessel densities for tissues above (c) and below (d) deep fascia in five animals for each group. Between six and eight sections were analysed for each animal. Mean microvessel densities are presented. Overlaid dot plots indicate the distribution of the data. Differences between groups were analysed with one-way ANOVA with a Tukey’s multiple comparison test. DGTM-AAVs showed no influences on vascularity. e, Schematic of the experimental design for the investigation of the efficiency of in vivo reprogramming during the generation of epithelial tissues in old wounds (7 days after the creation of the ulcer). f, Representative appearances of an ulcer at each time point (on day of ulcer creation (day 0), upon administration of DGTM-AAVs (day 7) and 18 days after administration of DGTM-AAVs (day 25)). Scale bar, 3 mm. g, Appearances of ulcers 18 days after administration of DGTM-AAVs in ten animals (including one shown in f) and histological images of small generated epithelial tissues that are unidentifiable by appearance. Yellow arrows indicate visible generated epithelial tissues. Red arrows indicate the position of generated tissues in magnified panels. Red scale bars, 200 μm. h, Schematic of the experimental design for the investigation of efficiency of GFPNLS-AAV administration with or without collagen gel on ulcer. i, Stereoscopic analysis of the centre of an ulcer and the surface of the liver. Different exposure times were used for imaging of ulcer and liver (fluorescence of the ulcer is far stronger than that of the liver surface). Similar findings were confirmed in four animals for each group. j, qPCR analysis of gross AAV genomic copies in ulcer tissues (left) and AAV genomic copies per mouse diploid genome (right) in animals 3 days after administration of GFPNLS-AAV with or without collagen gel (four animals for each group). The displayed values are the minimum (bottom range), mean (holizontal line), and maximum (top range). Overlaid dot plots indicate the distribution of the data. k, qPCR analysis of AAV genome copies per mouse diploid genome in liver in animals 3 days after administration of GFPNLS-AAV with or without collagen gel (four animals for each group). Three different lobes of liver tissues were analysed for each animal. The displayed values are the minimum (bottom range), mean (holizontal line), and maximum (top range) for each animal. Overlaid dot plots indicate the distribution of the data. j, k, Differences between groups were analysed with two-sided Student’s t-test. l, Schematic of the experimental design for epithelialization of ulcer in large chamber. m, Representative appearances of an ulcer treated by DGTM-AAVs and collagen gel administration (protocol I, n = 2, similar findings in both), an ulcer treated by DGTM-AAVs and collagen gel administration with application of Rock inhibitor (protocol II, n = 2, similar findings in both), an ulcer treated by DGTM-AAVs and collagen gel administration with application of FGF2 (protocol III, n = 3, similar findings in all), and an ulcer treated by DGTM-AAVs and collagen gel administration with application of Rock inhibitor and FGF2 (protocol IV, n = 3, similar findings in all). Yellow arrows indicate the initial emergence of visually identifiable epithelial tissues. External diameter of chamber, 14.3 mm.
Extended Data Fig. 8 in vivo iSEPs have high clonogenic ability and tumorigenic potential compatible with non-malignant epithelial cells.
a, Schematic representation of clonogenicity assessment by expansion of single-cell-derived clones performed for neonatal primary keratinocytes (NPKs), primary keratinocytes from adult mice and in vivo iSEPs. b, Representative images of large (left), medium (middle), and small (right) single-cell-derived colonies of primary keratinocytes and iSEPs in 96-well plate. Similar findings were obtained from 122 (from three NPKs and five primary keratinocytes) and 169 (five 3M iSEPs and five 6M iSEPs) clones. c, Representative images of plates stained with Rhodanile blue staining. Similar differences between primary keratinocyte and 3M iSEPs were confirmed in five sets of primary keratinocytes and iSEPs isolated from the same animal. d, Occupied areas of single-cell-derived colonies in 12-well plates. iSEPs (n = 10) constantly show higher clonogenic ability than primary keratinocytes (n = 8). The displayed values are the minimum (bottom whisker), 25th percentile (bottom of box), median (line in box), 75th percentile (top of box), and maximum (top whisker). Overlaid dot plots indicate the distribution of the data (n = 12 for NPK1–3 and 6M iSEPs 1–5, n = 15 for PK1, n = 21 for PK2, n = 18 for PK3–4, n = 14 for PK5, n = 18 for 3M iSEPs 1, n = 16 for 3M iSEPs 2, n = 29 for 3M iSEPs 3, n = 24 for 3M iSEPs 4, and n = 22 for 3M iSEPs 5). e, Representative images of soft-agar assay. In vivo iSEPs showed no colony formation in DMEM (DMEM + 10% FBS), KFM and CKFM. Scale bar, 200 μm. Similar findings were confirmed in one HeLa, five primary keratinocytes, five 3M iSEPs, and five 6M iSEPs. f, Number of colonies (>30 μm) in one well (six-well plate) (n = 3, technical replicates). Averages of triplicates are shown. Overlaid dot plots indicate the distribution of the data. g, Bright field and fluorescence images showing the representative morphology of mES cells from C57BL/6-TfN(act-EGFP) mice, fixed HeLa cells, primary keratinocytes from Krt14cre;LSLtdTomato mice, and in vivo iSEPs from PdgfracreER;LSLtdTomato mice after in vivo reprogramming with DGTM-AAV. hMito, human mitochondria. Scale bar, 200 μm. Similar findings were confirmed once for mES and HeLa, five times for primary keratinocytes, and ten times for in vivo iSEPs. h, Schematic representation of the tumorigenic/teratogenic assay by subcutaneous injection of the cells to immunodeficient mice. i, Volume of the nodules 28 days after injection. Top, volume of mES-cell-derived nodules. Asterisks indicate the day on which nodules were collected (*, day 24; **, day 16) owing to animal-welfare concerns about the size or properties of the nodule. j, Appearances of the largest nodules for HeLa, primary keratinocytes, and iSEPs. Similar findings were confirmed in three animals for each. Scale bars, 5 mm. k–n, H&E staining and fluorescence analysis of the largest nodule of 3M iSEPs 1 (k), primary keratinocytes (l), mES cells (m), and HeLa cells (n). For mES cells and HeLa cells teratogenicity and transfacial invasion were confirmed, respectively. Dotted outlines indicate the position of magnified panels (bottom). Similar findings were observed in other samples for each cells (n = 9 for iSEPs, n = 3 for primary keratinocytes, mES, and Hela cells). Red scale bar, 5 mm; blue scale bar, 2 mm; black scale bar, 500 μm; yellow scale bar, 200 μm. o, Schematic representation of biodistribution assessment of iSEPs. luciferase-expressing iSEPs were prepared using retroviral transduction. p, In vivo luminescent images 56 days after the subcutaneous injection of luciferase-expressing iSEPs. q, Luminescent images of dissected organs and tissues. Luciferase expression was confined to site of injection. M, muscle beneath the transplants; Sk + N, nodule and overlaid skin. p, q, Similar findings were confirmed in three animals for each iSEP group.
Extended Data Fig. 9 Exogenous gene expression and transcriptional profiles of iSEPs.
a, Integration analysis of TP63, GRHL2, TFAP2A and MYC. RNA-seq reads were mapped simultaneously to human and mouse transcriptomes and the sum of the normalized transcript counts for all variants of TP63, GRHL2, TFAP2A, and MYC is shown for Pdgfra+ ADSCs, in vitro iSEPs, 1M in vivo iSEPs, 3M in vivo iSEPs, 6M in vivo iSEPs and primary keratinocytes. Human transcripts are derived from the AAV genome. Data from three Pdgfra+ ADSCs, three in vitro iSEPs, three 1M in vivo iSEPs, five 3M in vivo iSEPs, six 6M in vivo iSEPs and three primary keratinocytes. b, Clustered heat map showing the normalized expression of the top expressed genes across all conditions. c, Clustered keratinocyte marker gene expression represented as a heat map. d, Differentially expressed genes were found between primary keratinocytes and Pdgfra+ ADSCs (red), in vivo iSEPs (blue) and in vitro iSEPs (green). Overlap of genes that are significantly up- (top) or downregulated (bottom) in primary keratinocytes are shown. e, f, Top five Gene Ontology (GO) enrichment terms overrepresented for genes upregulated in primary keratinocytes (e) or genes upregulated in in vivo iSEPs (f). g, h, Top five enriched transcription factors known to bind to promoters of genes upregulated in primary keratinocytes (g) or genes upregulated in in vivo iSEPs (h). e–h, Bar plots show −log(adjusted P value) significance of overrepresentation. Enrichment testing was carried out using HOMER for Gene Ontology enrichment testing (hypergeometric test with Benjamini and Yekutieli general multiple testing correction), and WebGestalt for transcription-factor enrichment testing (hypergeometic test with Benjamini–Hochberg multiple testing correction) on genes upregulated in primary keratinocytes (n = 399), or upregulated in in vivo iSEPs (n = 632). b–h, Data for Pdgfra+ mADSCs, in vitro iSEPs, 6M and 3M in vivo iSEPs were from three experiments. i, Normalized expression values were k-means clustered to reveal the top five patterns of gene expression. Cluster A, Mesench (mesenchymal); cluster B, late; cluster C, sustained; cluster D, early; cluster E, transient. j, Heat map showing relative normalized expression of keratinocyte marker genes. k, Analysis of the enrichment of the top Gene Ontology terms are shown for each cluster of genes. General terms with >100 genes were filtered out to remove overly general terms. l, Top five transcription factors enriched in promoters of clustered genes. k, l, Bar plots show –log(adjusted P value) significance of overrepresentation. Enrichment testing was carried out using HOMER for Gene Ontology enrichment testing (hypergeometric test with Benjamini and Yekutieli general multiple testing correction), and WebGestalt for transcription factor enrichment testing (hypergeometic test with Benjamini–Hochberg multiple testing correction) on genes in clusters A (n = 1,966), B (n = 396), C (n = 2,501), D (n = 400), and E (n = 1,386). i–l, Data are from three Pdgfra+ mADSCs, three 1M in vivo iSEPs, two 3M in vivo iSEPs, three 6M in vivo iSEPs.
Extended Data Fig. 10 Description of wound healing with in vivo reprogramming.
During physiological wound healing, epidermal defects are repaired from the other epidermis. On the other hand, in vivo reprogramming allows de novo epithelialization and greatly enhances the capacity for the regeneration of cutaneous defects.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-2 and Supplementary Methods
Supplementary Tables
This file contains Supplementary Tables 1-13
Source data
Rights and permissions
About this article
Cite this article
Kurita, M., Araoka, T., Hishida, T. et al. In vivo reprogramming of wound-resident cells generates skin epithelial tissue. Nature 561, 243–247 (2018). https://doi.org/10.1038/s41586-018-0477-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0477-4
Keywords
This article is cited by
-
Reproducible strategy for excisional skin-wound-healing studies in mice
Nature Protocols (2024)
-
Mechanical stiffness promotes skin fibrosis via Piezo1-Wnt2/Wnt11-CCL24 positive feedback loop
Cell Death & Disease (2024)
-
Daytime radiative cooling dressings for accelerating wound healing under sunlight
Nature Chemical Engineering (2024)
-
Tracing immune cells around biomaterials with spatial anchors during large-scale wound regeneration
Nature Communications (2023)
-
In situ-formable, dynamic crosslinked poly(ethylene glycol) carrier for localized adeno-associated virus infection and reduced off-target effects
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.