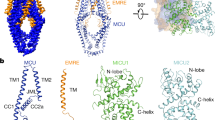
Abstract
Mitochondrial calcium uptake is critical for regulating ATP production, intracellular calcium signalling, and cell death. This uptake is mediated by a highly selective calcium channel called the mitochondrial calcium uniporter (MCU). Here, we determined the structures of the pore-forming MCU proteins from two fungi by X-ray crystallography and single-particle cryo-electron microscopy. The stoichiometry, overall architecture, and individual subunit structure differed markedly from those described in the recent nuclear magnetic resonance structure of Caenorhabditis elegans MCU. We observed a dimer-of-dimer architecture across species and chemical environments, which was corroborated by biochemical experiments. Structural analyses and functional characterization uncovered the roles of key residues in the pore. These results reveal a new ion channel architecture, provide insights into calcium coordination, selectivity and conduction, and establish a structural framework for understanding the mechanism of mitochondrial calcium uniporter function.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gunter, T. E. & Pfeiffer, D. R. Mechanisms by which mitochondria transport calcium. Am. J. Physiol. 258, C755–C786 (1990).
Rizzuto, R., De Stefani, D., Raffaello, A. & Mammucari, C. Mitochondria as sensors and regulators of calcium signalling. Nat. Rev. Mol. Cell Biol. 13, 566–578 (2012).
Szabadkai, G. & Duchen, M. R. Mitochondria: the hub of cellular Ca2+ signaling. Physiology (Bethesda) 23, 84–94 (2008).
Deluca, H. F. & Engstrom, G. W. Calcium uptake by rat kidney mitochondria. Proc. Natl Acad. Sci. USA 47, 1744–1750 (1961).
Vasington, F. D. & Murphy, J. V. Ca ion uptake by rat kidney mitochondria and its dependence on respiration and phosphorylation. J. Biol. Chem. 237, 2670–2677 (1962).
Kirichok, Y., Krapivinsky, G. & Clapham, D. E. The mitochondrial calcium uniporter is a highly selective ion channel. Nature 427, 360–364 (2004).
De Stefani, D., Rizzuto, R. & Pozzan, T. Enjoy the trip: calcium in mitochondria back and forth. Annu. Rev. Biochem. 85, 161–192 (2016).
Rizzuto, R., Brini, M., Murgia, M. & Pozzan, T. Microdomains with high Ca2+ close to IP3-sensitive channels that are sensed by neighboring mitochondria. Science 262, 744–747 (1993).
Giacomello, M. et al. Ca2+ hot spots on the mitochondrial surface are generated by Ca2+ mobilization from stores, but not by activation of store-operated Ca2+ channels. Mol. Cell 38, 280–290 (2010).
Baughman, J. M. et al. Integrative genomics identifies MCU as an essential component of the mitochondrial calcium uniporter. Nature 476, 341–345 (2011).
De Stefani, D., Raffaello, A., Teardo, E., Szabò, I. & Rizzuto, R. A forty-kilodalton protein of the inner membrane is the mitochondrial calcium uniporter. Nature 476, 336–340 (2011).
Chaudhuri, D., Sancak, Y., Mootha, V. K. & Clapham, D. E. MCU encodes the pore conducting mitochondrial calcium currents. eLife 2, e00704 (2013).
Kovács-Bogdán, E. et al. Reconstitution of the mitochondrial calcium uniporter in yeast. Proc. Natl Acad. Sci. USA 111, 8985–8990 (2014).
Lee, S. K. et al. Structural insights into mitochondrial calcium uniporter regulation by divalent cations. Cell Chem. Biol. 23, 1157–1169 (2016).
Csordás, G. et al. MICU1 controls both the threshold and cooperative activation of the mitochondrial Ca2+ uniporter. Cell Metab. 17, 976–987 (2013).
Mallilankaraman, K. et al. MCUR1 is an essential component of mitochondrial Ca2+ uptake that regulates cellular metabolism. Nat. Cell Biol. 14, 1336–1343 (2012).
Kamer, K. J., Grabarek, Z. & Mootha, V. K. High-affinity cooperative Ca2+ binding by MICU1-MICU2 serves as an on-off switch for the uniporter. EMBO Rep. 18, 1397–1411 (2017).
Perocchi, F. et al. MICU1 encodes a mitochondrial EF hand protein required for Ca2+ uptake. Nature 467, 291–296 (2010).
Liu, J. C. et al. MICU1 serves as a molecular gatekeeper to prevent in vivo mitochondrial calcium overload. Cell Reports 16, 1561–1573 (2016).
Patron, M. et al. MICU1 and MICU2 finely tune the mitochondrial Ca2+ uniporter by exerting opposite effects on MCU activity. Mol. Cell 53, 726–737 (2014).
Sancak, Y. et al. EMRE is an essential component of the mitochondrial calcium uniporter complex. Science 342, 1379–1382 (2013).
Tsai, M. F. et al. Dual functions of a small regulatory subunit in the mitochondrial calcium uniporter complex. eLife 5, e15545 (2016).
Raffaello, A. et al. The mitochondrial calcium uniporter is a multimer that can include a dominant-negative pore-forming subunit. EMBO J. 32, 2362–2376 (2013).
Chaudhuri, D., Artiga, D. J., Abiria, S. A. & Clapham, D. E. Mitochondrial calcium uniporter regulator 1 (MCUR1) regulates the calcium threshold for the mitochondrial permeability transition. Proc. Natl Acad. Sci. USA 113, E1872–E1880 (2016).
Tomar, D. et al. MCUR1 Is a scaffold factor for the MCU complex function and promotes mitochondrial bioenergetics. Cell Reports 15, 1673–1685 (2016).
Bick, A. G., Calvo, S. E. & Mootha, V. K. Evolutionary diversity of the mitochondrial calcium uniporter. Science 336, 886 (2012).
Mammucari, C., Gherardi, G. & Rizzuto, R. Structure, activity regulation, and role of the mitochondrial calcium uniporter in health and disease. Front. Oncol. 7, 139 (2017).
Oxenoid, K. et al. Architecture of the mitochondrial calcium uniporter. Nature 533, 269–273 (2016).
Song, J., Liu, X., Zhai, P., Huang, J. & Lu, L. A putative mitochondrial calcium uniporter in A. fumigatus contributes to mitochondrial Ca2+ homeostasis and stress responses. Fungal Genet. Biol. 94, 15–22 (2016).
Shigetomi, E., Kracun, S., Sofroniew, M. V. & Khakh, B. S. A genetically targeted optical sensor to monitor calcium signals in astrocyte processes. Nat. Neurosci. 13, 759–766 (2010).
Hou, X., Pedi, L., Diver, M. M. & Long, S. B. Crystal structure of the calcium release-activated calcium channel Orai. Science 338, 1308–1313 (2012).
Yang, W., Lee, H. W., Hellinga, H. & Yang, J. J. Structural analysis, identification, and design of calcium-binding sites in proteins. Proteins 47, 344–356 (2002).
Saotome, K., Singh, A. K., Yelshanskaya, M. V. & Sobolevsky, A. I. Crystal structure of the epithelial calcium channel TRPV6. Nature 534, 506–511 (2016).
Dong, Z. et al. Mitochondrial Ca2+ uniporter is a mitochondrial luminal redox sensor that augments MCU channel activity. Mol. Cell 65, 1014–1028.e7 (2017).
Lee, Y. et al. Structure and function of the N-terminal domain of the human mitochondrial calcium uniporter. EMBO Rep. 16, 1318–1333 (2015).
Arduino, D. M. et al. Systematic identification of MCU modulators by orthogonal interspecies chemical screening. Mol. Cell 67, 711–723 (2017).
Liao, M., Cao, E., Julius, D. & Cheng, Y. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504, 107–112 (2013).
Doyle, D. A. et al. The structure of the potassium channel: molecular basis of K+ conduction and selectivity. Science 280, 69–77 (1998).
Yan, Z. et al. Structure of the rabbit ryanodine receptor RyR1 at near-atomic resolution. Nature 517, 50–55 (2015).
Sobolevsky, A. I., Rosconi, M. P. & Gouaux, E. X-ray structure, symmetry and mechanism of an AMPA-subtype glutamate receptor. Nature 462, 745–756 (2009).
Fan, G. et al. Gating machinery of InsP3R channels revealed by electron cryomicroscopy. Nature 527, 336–341 (2015).
Zalk, R. et al. Structure of a mammalian ryanodine receptor. Nature 517, 44–49 (2015).
Efremov, R. G., Leitner, A., Aebersold, R. & Raunser, S. Architecture and conformational switch mechanism of the ryanodine receptor. Nature 517, 39–43 (2015).
Wu, J. et al. Structure of the voltage-gated calcium channel Cav1.1 at 3.6 Å resolution. Nature 537, 191–196 (2016).
Tang, L. et al. Structural basis for Ca2+ selectivity of a voltage-gated calcium channel. Nature 505, 56–61 (2014).
Zhu, S. & Gouaux, E. Structure and symmetry inform gating principles of ionotropic glutamate receptors. Neuropharmacology 112, 11–15 (2017).
Doublie, S. Preparation of selenomethionyl proteins for phase determination. Methods Enzymol. 276, 523–530 (1997).
Ho, S. N., Hunt, H. D., Horton, R. M., Pullen, J. K. & Pease, L. R. Site-directed mutagenesis by overlap extension using the polymerase chain reaction. Gene 77, 51–59 (1989).
Walter, T. S. et al. Lysine methylation as a routine rescue strategy for protein crystallization. Structure 14, 1617–1622 (2006).
Pardon, E. et al. A general protocol for the generation of nanobodies for structural biology. Nat. Protocols 9, 674–693 (2014).
McMahon, C. et al. Yeast surface display platform for rapid discovery of conformationally selective nanobodies. Nat. Struct. Mol. Biol. 25, 289–296 (2018).
Chao, G. et al. Isolating and engineering human antibodies using yeast surface display. Nat. Protocols 1, 755–768 (2006).
Kabsch, W. XDS. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010).
Otwinowski, Z. & Minor, W. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997).
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007).
Adams, P. D. et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010).
Emsley, P., Lohkamp, B., Scott, W. G. & Cowtan, K. Features and development of Coot. Acta Crystallogr. D Biol. Crystallogr. 66, 486–501 (2010).
Pape, T. & Schneider, T. R. HKL2MAP: a graphical user interface for macromolecular phasing with SHELX programs. J. Appl. Crystallogr. 37, 843–844 (2004).
Bricogne, G. et al. BUSTER (Global Phasing Ltd, Cambridge, UK, 2017).
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010).
Smart, O. S., Neduvelil, J. G., Wang, X., Wallace, B. A. & Sansom, M. S. HOLE: a program for the analysis of the pore dimensions of ion channel structural models. J. Mol. Graph. 14, 354–360, 376 (1996).
Tallini, Y. N. et al. Imaging cellular signals in the heart in vivo: Cardiac expression of the high-signal Ca2+ indicator GCaMP2. Proc. Natl Acad. Sci. USA 103, 4753–4758 (2006).
Felle, H., Porter, J. S., Slayman, C. L. & Kaback, H. R. Quantitative measurements of membrane potential in Escherichia coli. Biochemistry 19, 3585–3590 (1980).
Veenhoff, L. M. & Poolman, B. Substrate recognition at the cytoplasmic and extracellular binding site of the lactose transport protein of Streptococcus thermophilus. J. Biol. Chem. 274, 33244–33250 (1999).
Zhang, J., Feng, Y. & Forgac, M. Proton conduction and bafilomycin binding by the V0 domain of the coated vesicle V-ATPase. J. Biol. Chem. 269, 23518–23523 (1994).
Feng, L., Campbell, E. B., Hsiung, Y. & MacKinnon, R. Structure of a eukaryotic CLC transporter defines an intermediate state in the transport cycle. Science 330, 635–641 (2010).
Booth, D. S., Avila-Sakar, A. & Cheng, Y. Visualizing proteins and macromolecular complexes by negative stain EM: from grid preparation to image acquisition. J. Vis. Exp. 58, 3227 (2011).
Li, X., Zheng, S., Agard, D. A. & Cheng, Y. Asynchronous data acquisition and on-the-fly analysis of dose fractionated cryoEM images by UCSFImage. J. Struct. Biol. 192, 174–178 (2015).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Mi, W. et al. Structural basis of MsbA-mediated lipopolysaccharide transport. Nature 549, 233–237 (2017).
Zheng, S. Q. et al. MotionCor2: anisotropic correction of beam-induced motion for improved cryo-electron microscopy. Nat. Methods 14, 331–332 (2017).
Rohou, A. & Grigorieff, N. CTFFIND4: Fast and accurate defocus estimation from electron micrographs. J. Struct. Biol. 192, 216–221 (2015).
Ru, H. et al. Molecular mechanism of V(D)J recombination from synaptic RAG1–RAG2 complex structures. Cell 163, 1138–1152 (2015).
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012).
Scheres, S. H. A Bayesian view on cryo-EM structure determination. J. Mol. Biol. 415, 406–418 (2012).
Bai, X. C., Rajendra, E., Yang, G., Shi, Y. & Scheres, S. H. Sampling the conformational space of the catalytic subunit of human γ-secretase. eLife 4, e11182 (2015).
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nat. Methods 11, 63–65 (2014).
Lyumkis, D., Brilot, A. F., Theobald, D. L. & Grigorieff, N. Likelihood-based classification of cryo-EM images using FREALIGN. J. Struct. Biol. 183, 377–388 (2013).
Pettersen, E. F. et al. UCSF Chimera—a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004).
Acknowledgements
We thank the staff at Beamline 24ID-C/E, 23ID-B/D, and 17ID (APS, Argonne National Laboratory), 12-2 (SSRL, SLAC National Laboratory), and 5.0.2 (Advanced Light Source). We thank Z. Li at HMS and C. Xu and K. Song at UMass cryo-EM facility for help with EM data collection. This work was made possible by support from Stanford University, the Klingenstein-Simons Fellowship, and the Harold and Leila Y. Mathers Charitable Foundation to L.F., and from a National Science Foundation Graduate Research Fellowship to N.M.F. NE-CAT is supported by NIH P41 GM103403 and S10 RR029205. We thank A. Manglik for discussions and R. Lewis for suggestions and comments.
Reviewer information
Nature thanks M. Prakriya, D. Stokes and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
C.F. and M.F. performed protein purification, biochemistry, crystallization, data collection, crystallography and functional assays. B.J.O. performed negative-stain EM, cryo-EM, and data processing. N.M.F. performed protein engineering, crystallization, and data collection. Y.X. performed protein purification and biochemistry. J.Z. performed crystallography. M.G.C. helped with initial EM screening. X.X. helped with initial characterization. K.P. assisted with crystallography. M.L. oversaw the EM studies and contributed to EM data collection and processing. L.F. conceived the project, oversaw the X-ray structural, biochemical, and functional studies and contributed to experimental work and crystallography. L.F. and M.L. wrote the manuscript with input from all co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Sequence alignment, sample preparations and functional characterizations.
a, Sequence alignment of MCU. The protein sequences of MCUs from Fusarium graminearum (FgMCU), Metarhizium acridum (MaMCU), Homo sapiens (HsMCU) and Caenorhabditis elegans (CeMCU) were aligned using Clustal Omega with manual adjustment. The domain organization is shown above the sequences. b, FgMCU in DDM on size-exclusion chromatography. c, MaMCU in DDM on size-exclusion chromatography. Data in b, c are representative of five independent experiments with similar results. d, Ca2+ uptake activity of FgMCU in E. coli. Fluorescence changes upon addition of 0.5 mM Ca2+ are shown for wild-type (red), D351A/E354A (green), control (black), and with ruthenium red (blue). e, Cation flux activity of FgMCU in reconstituted liposomes. Fluorescence changes are shown for FgMCU (red) and with Gd3+ (blue). Data in d, e are representative of three independent experiments with similar results.
Extended Data Fig. 2 Crystal packing and molecule interactions in the crystal.
a, The crystal lattice structure of MaMCU-Rub. One MaMCU channel is coloured blue and its associated rubredoxin is coloured cyan. In the crystal, the transmembrane domain lacks clear protein-mediated contacts. Presumably, relatively disordered protein regions or detergent micelles mediate the contact. b, The crystal lattice structure of MaMCU in complex with nanobody. One MCU is shown in blue and the bound nanobodies are shown in cyan. c, The crystal lattice structure of the MaMCU soluble domain, shown in ribbons. Each chain is coloured differently. Two helices that formed a coiled-coil were derived from two different chains, in a domain-swapped manner. d, Close-up view of the interaction between nanobody and McMCU in the crystal. Nanobody binds near the end of TM1 and is distant from the selectivity filter (shown as coloured sticks). Nanobody is coloured grey and MaMCU is colored cyan. e, Mercury and selenium sites identified by MR-SAD. Two protomers of MaMCU in one asymmetric unit are shown as ribbons in green and blue, respectively. The mercury sites identified from ‘marker mutants’ are shown as red spheres; one selenium site from the labelled wild-type protein is shown in the same manner. f, 2Fo−Fc electron density map of MaMCU (cyan mesh, 1σ) with the structure model shown in ribbon. g, Close-up view of 2Fo−Fc electron density map of individual transmembrane helices (cyan mesh, 1.5σ) with side chains shown in sticks.
Extended Data Fig. 3 Comparison of the NMR structure of cMCU with the crystal structure of MaMCU, and the structure of the soluble domain of MaMCU.
a, Left, pentameric NMR structure of cMCU. Right, dimer-of-dimers structure of MaMCU. Box outlines the equivalent region including the transmembrane domain and coiled-coil. b, Comparison of a subunit of cMCU by NMR with that of MaMCU by crystallography. The helical part of the pore-lining TM2 is superimposed. The rest of the protein shows markedly different structure. Right, expanded view of the selectivity filter region. The most conserved WDxxEP residues (shown as sticks) are organized differently in these two structures. c, Ribbon representation of the soluble domain of MaMCU. Each subunit is coloured in blue, green, gold, or cyan, with the portions before and after the transmembrane domain of each subunit in slightly different colours. d, Structural comparison of the NTDs of MaMCU (purple) and human MCU (cyan). The two structures have the same fold and superimpose well onto each other. e, A pair of human MCU NTDs (cyan and blue) superimposes well onto one of dimers formed by MaMCU NTDs (purple and pink).
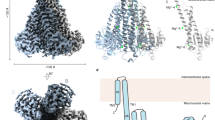
Extended Data Fig. 4 Negative-stain EM and cryo-EM of MaMCU and FgMCU.
a, Representative negative-stain EM image and 2D averages of MaMCU in A8–35 (from 28 micrographs with similar results), MaMCU in nanodiscs (from 28 micrographs with similar results), FgMCU in PMAL-C8 (from 20 micrographs with similar results), and FgMCU in nanodiscs (from 40 micrographs with similar results). The box dimension of the 2D averages is 215 Å. b, Representative cryo-EM image (from 1,862 micrographs with similar results), 2D averages, and image processing workflow of MaMCU in A8–35. The box dimension of the 2D averages is 172 Å. c, Representative cryo-EM image (from 4,055 micrographs with similar results), 2D averages, and image processing workflow of FgMCU in PMAL-C8. The box dimension of the 2D averages is 176 Å. d, Different views of the cryo-EM maps of MaMCU in A8–35 and FgMCU in PMAL-C8, both low-pass filtered to 7 Å. 2D averages and map calculation were not repeated.
Extended Data Fig. 5 Single-particle cryo-EM analysis of FgMCU in nanodiscs.
a, Representative cryo-EM image (from 5,332 micrographs with similar results). b, 2D averages of cryo-EM particle images. The box dimension is 187 Å. c, Image processing workflow. Two cryo-EM data sets were first independently subjected to 3D classification without symmetry, and the particle images in good classes were combined and further processed with two-fold symmetry, generating a 4.9 Å-resolution cryo-EM map. Subsequently, a final round of 3D classification focused on the transmembrane domain sorted out two major classes, which generated two final maps at 5.0 and 4.8 Å resolution for the transmembrane domain. The conformations in these two maps are designated as type 1 and type 2. 2D averages and map calculations were not repeated.
Extended Data Fig. 6 Cryo-EM maps of two conformations of FgMCU in nanodiscs.
a, Gold-standard FSC curves of the type 1 final cryo-EM map, calculated with a soft mask to include only the transmembrane domain (TMD, solid line) or the entire MCU molecule without nanodisc (dashed line). The estimated resolutions based on an FSC = 0.143 criterion are indicated. b, Cross-sectional view of the type 1 final cryo-EM map filtered to 5.0 Å resolution and coloured according to local resolution. c, Angular distribution of the particle images used for calculating the type 1 final cryo-EM map. d–f, Same as a–c, but for the type 2 conformation. g, Two cross-sectional views of the transmembrane domains of the type 1 and type 2 cryo-EM maps, superimposed onto an MaMCU-based homology model of FgMCU. The missing density and extra densities in the type 2 map are indicated. Map calculations in a and d were not repeated.
Extended Data Fig. 7 Tetrameric assembly of MaMCU, overall architecture of a metazoan MCU and symmetry mismatch of MaMCU.
a, The design of cysteine pair mutations for crosslinking transmembrane subunits. L314 on TM1 and M348 on TM2 were mutated to cysteines (side chains shown as sticks). b, Site-directed disulfide cross-linking of MaMCU in detergent solution. L314 on TM1 and M348 on TM2 were mutated to create a cysteine pair at the protomer interface on an otherwise cysteine-free background. The double cysteine mutant was purified and oxidized by copper phenanthroline (CuP) for various time durations. The cross-link product was analysed by SDS–PAGE. c, Site-directed disulfide cross-linking of MaMCU in the membrane. Isolated membrane from the MaMCU L314C/M348C mutant was treated with CuP for an increasing amount of time. The cross-linking product was analysed by western blot using an antibody against the His tag on the protein. d, The size-exclusion profile of MaMCU with or without crosslinking. MaMCU L314C/M348C mutant treated or untreated with CuP in the membrane was purified and analysed by size-exclusion chromatography. The peak fractions were subjected to SDS–PAGE and visualized by Coomassie blue staining. e, Site-directed disulfide cross-linking of mosquito MCU in detergent solution. A pair of residues (T294 on TM1 and V341 on TM2) that were equivalent to those used in MaMCU crosslinking at the protomer interface were mutated to cysteine. The purified double cysteine mutant was oxidized by CuP and analysed by SDS–PAGE. The tetramer position on the SDS–PAGE gel is the same as that of MaMCU in b. f, 2D averages of negatively stained FgMCU in nanodiscs, mosquito MCU (T294C/V341C) in nanodiscs, and mosquito MCU (T294C/V341C) in amphipol A8–35. 2D averages were not repeated. g, Geometry of markers in transmembrane domain and NTD of MaMCU. Site-specific mercury marker on transmembrane domain (red spheres) and Cα position of Ile115 in NTD (blue spheres) are indicated. Data in b–e are representative of three independent experiments with similar results. For gel source data, see Supplementary Fig. 1.
Extended Data Fig. 8 Expression levels of MaMCU mutants and ruthenium red sensitivity of selected mutants.
a, The expression level of wild-type MaMCU and those with a point mutation in the transmembrane domain. Green fluorescent protein-tagged protein was analysed on SDS–PAGE and the protein was visualized by in-gel fluorescence. The experiments were repeated twice independently with similar results. For gel source data, see Supplementary Fig. 1. b–m, The effect of substituting residues near the intermembrane surface of MaMCU on its ruthenium red sensitivity. Ca2+ uptake activity of the wild-type or mutant MaMCU was assayed in an E. coli-based assay (0.5 mM Ca2+, 1 μM RuR). The Ca2+ uptake in the absence (blue) or in the presence (black) of ruthenium red is reflected by fluorescence changes. The experiments were repeated three times independently with similar results.
Supplementary information
Supplementary Figure 1
Source images for gel electrophoresis in Extended Data Figures 7 and 8.
Rights and permissions
About this article
Cite this article
Fan, C., Fan, M., Orlando, B.J. et al. X-ray and cryo-EM structures of the mitochondrial calcium uniporter. Nature 559, 575–579 (2018). https://doi.org/10.1038/s41586-018-0330-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0330-9
This article is cited by
-
The Role of PGC-1α-Mediated Mitochondrial Biogenesis in Neurons
Neurochemical Research (2023)
-
Structure and mechanism of the mitochondrial Ca2+ uniporter holocomplex
Nature (2020)
-
Discovery of EMRE in fungi resolves the true evolutionary history of the mitochondrial calcium uniporter
Nature Communications (2020)
-
Visualizing structural transitions of ligand-dependent gating of the TRPM2 channel
Nature Communications (2019)
-
Super-resolution imaging of non-fluorescent reactions via competition
Nature Chemistry (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.