Abstract
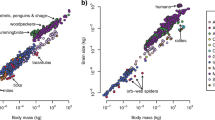
Organisms vary widely in size, from microbes weighing 0.1 pg to trees weighing thousands of megagrams — a 1021-fold range similar to the difference in mass between an elephant and the Earth. Mass has a pervasive influence on biological processes, but the effect is usually non-proportional; for example, a tenfold increase in mass is typically accompanied by just a four- to sevenfold increase in metabolic rate. Understanding the cause of allometric scaling has been a long-standing problem in biology. Here, we examine the evolution of metabolic allometry in animals by linking microevolutionary processes to macroevolutionary patterns. We show that the genetic correlation between mass and metabolic rate is strong and positive in insects, birds and mammals. We then use these data to simulate the macroevolution of mass and metabolic rate, and show that the interspecific relationship between these traits in animals is consistent with evolution under persistent multivariate selection on mass and metabolic rate over long periods of time.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data generated or analysed during this study are included within the article and its supplementary Information files.
References
Vernon, H. M. The physiological evolution of the warm-blooded animal. Sci. Prog. 7, 378–394 (1898).
Krogh, A. Respiratory Exchange of Animals and Man (Longmans, Green and Co., London, 1916).
Schmidt-Nielsen, K. Scaling: Why is Animal Size so Important? (Cambridge Univ. Press, Cambridge, 1984).
West, G. B., Brown, J. H. & Enquist, B. J. A general model for the origin of allometric scaling laws in biology. Science 276, 122–126 (1997).
West, G. B., Brown, J. H. & Enquist, B. J. The fourth dimension of life: fractal geometry and allometric scaling of organisms. Science 284, 1677–1679 (1999).
Glazier, D. S. Beyond the ‘3/4-power law’: variation in the intra- and interspecific scaling of metabolic rate in animals. Biol. Rev. 80, 611–662 (2005).
Glazier, D. S. A unifying explanation for diverse metabolic scaling in animals and plants. Biol. Rev. 85, 111–138 (2010).
White, C. R. & Kearney, M. R. Determinants of inter-specific variation in basal metabolic rate. J. Compr. Physiol. B 183, 1–26 (2013).
White, C. R. & Kearney, M. R. Metabolic scaling in animals: methods, empirical results, and theoretical explanations. Compr. Physiol. 4, 231–256 (2014).
Pettersen, A. K., Marshall, D. J. & White, C. R. Understanding variation in metabolic rate. J. Exp. Biol. 221, jeb166876 (2018).
Uyeda, J. C., Pennell, M. W., Miller, E. T., Maia, R. & McClain, C. R. The evolution of energetic scaling across the vertebrate tree of life. Am. Nat. 190, 185–199 (2017).
Brown, J. H., Gillooly, J. F., Allen, A. P., Savage, V. M. & West, G. B. Toward a metabolic theory of ecology. Ecology 85, 1771–1789 (2004).
Burton, T., Killen, S. S., Armstrong, J. D. & Metcalfe, N. B. What causes intraspecific variation in resting metabolic rate and what are its ecological consequences? Proc. R. Soc. Lond. B 278, 3465–3473 (2011).
Biro, P. A. & Stamps, J. A. Do consistent individual differences in metabolic rate promote consistent individual differences in behavior? Trends Ecol. Evol. 25, 653–659 (2010).
Pettersen, A. K., White, C. R. & Marshall, D. J. Metabolic rate covaries with fitness and the pace of the life history in the field. Proc. R. Soc. Lond. B 283, 20160323 (2016).
McNab, B. K. Ecological factors affect the level and scaling of avian BMR. Comp. Biochem. Physiol. A 152, 22–45 (2009).
McNab, B. K. An analysis of the factors that influence the level and scaling of mammalian BMR. Comp. Biochem. Physiol. A 151, 5–28 (2008).
Kolokotrones, T., Savage, V. M., Deeds, E. J. & Fontana, W. Curvature in metabolic scaling. Nature 464, 753–756 (2010).
White, C. R., Phillips, N. F. & Seymour, R. S. The scaling and temperature dependence of vertebrate metabolism. Biol. Lett. 2, 125–127 (2006).
Speakman, J. R. & Król, E. Maximal heat dissipation capacity and hyperthermia risk: neglected key factors in the ecology of endotherms. J. Anim. Ecol. 79, 726–746 (2010).
Speakman, J. R., McDevitt, R. M. & Cole, K. R. Measurement of basal metabolic rates: don’t lose sight of reality in the quest for comparability. Physiol. Zool. 66, 1045–1049 (1993).
Hirst, A. G., Glazier, D. S. & Atkinson, D. Body shape shifting during growth permits tests that distinguish between competing geometric theories of metabolic scaling. Ecol. Lett. 17, 1274–1281 (2014).
Kooijman, S. A. L. M. Dynamic Energy Budget Theory for Metabolic Organisation 3rd edn (Cambridge Univ. Press, Cambridge, 2010).
Killen, S. S., Atkinson, D. & Glazier, D. S. The intraspecific scaling of metabolic rate with body mass in fishes depends on lifestyle and temperature. Ecol. Lett. 13, 184–193 (2010).
Witting, L. The body mass allometries as evolutionarily determined by the foraging of mobile organisms. J. Theor. Biol. 177, 129–137 (1995).
Glazier, D. S. et al. Ecological effects on metabolic scaling: amphipod responses to fish predators in freshwater springs. Ecol. Monogr. 81, 599–618 (2011).
Glazier, D. S. Is metabolic rate a universal ‘pacemaker’ for biological processes?. Biol. Rev. 90, 377–407 (2015).
Kozłowski, J. & Weiner, J. Interspecific allometries are by-products of body size optimization. Am. Nat. 149, 352–380 (1997).
Lande, R. Quantitative genetic analysis of multivariate evolution, applied to brain:body size allometry. Evolution 33, 402–416 (1979).
Walsh, B. & Blows, M. W. Abundant genetic variation + strong selection = multivariate genetic constraints: a geometric view of adaptation. Annu. Rev. Ecol. Evol. Syst. 40, 41–59 (2009).
Zeng, Z.-B. Long-term correlated response, interpopulation covariation, and interspecific allometry. Evolution 42, 363–374 (1988).
Tieleman, B. I., Versteegh, M. A., Helm, B. & Dingemanse, N. J. Quantitative genetics parameters show partial independent evolutionary potential for body mass and metabolism in stonechats from different populations. J. Zool. 278, 129–136 (2009).
Nilsson, J.-Å., Åkesson, M. & Nilsson, J. F. Heritability of resting metabolic rate in a wild population of blue tits. J. Evol. Biol. 22, 1867–1874 (2009).
Ronning, B., Jensen, H., Moe, B. & Bech, C. Basal metabolic rate: heritability and genetic correlations with morphological traits in the zebra finch. J. Evol. Biol. 20, 1815–1822 (2007).
Careau, V. et al. Genetic correlation between resting metabolic rate and exploratory behaviour in deer mice (Peromyscus maniculatus). J. Evol. Biol. 24, 2153–2163 (2011).
Schimpf, N. G., Matthews, P. G. D. & White, C. R. Discontinuous gas exchange exhibition is a heritable trait in speckled cockroaches Nauphoeta cinerea. J. Evol. Biol. 26, 1588–1597 (2013).
Alton, L. A., Condon, C., White, C. R. & Angilletta, M. J. Jr. Colder environments did not select for a faster metabolism during experimental evolution of Drosophila melanogaster. Evolution 71, 145–152 (2017).
Nespolo, R. F. & Roff, D. A. Testing the aerobic model for the evolution of endothermy: implications of using present correlations to infer past evolution. Am. Nat. 183, 74–83 (2014).
Delph, L. F., Steven, J. C., Anderson, I. A., Herlihy, C. R. & Brodie, E. D. III Elimination of a genetic correlation between the sexes via artificial correlational selection. Evolution 65, 2872–2880 (2011).
Roff, D. A. & Fairbairn, D. J. A test of the hypothesis that correlational selection generates genetic correlations. Evolution 66, 2953–2960 (2012).
Roff, D. A. & Fairbairn, D. J. The evolution of trade-offs under directional and correlational selection. Evolution 66, 2461–2474 (2012).
Sgrò, C. M. & Hoffmann, A. A. Genetic correlations, tradeoffs and environmental variation. Heredity 93, 241–248 (2004).
Glazier, D. S. The 3/4-power law is not universal: evolution of isometric, ontogenetic metabolic scaling in pelagic animals. Bioscience 56, 325–332 (2006).
Kooijman, S. A. L. M. Energy budgets can explain body size relations. J. Theor. Biol. 121, 269–282 (1986).
Maino, J. L., Kearney, M. R., Nisbet, R. M. & Kooijman, S. A. L. M. Reconciling theories for metabolic scaling. J. Anim. Ecol. 83, 20–29 (2014).
Lighton, J. R. B. Measuring Metabolic Rates: A Manual for Scientists (Oxford Univ. Press, Oxford, 2008).
Wilson, A. J. Why h 2 does not always equal V A/V P? J. Evol. Biol. 21, 647–650 (2008).
Gilmour, A. R. et al. ASReml User Guide Release 3.0 (NSW Department of Industry and Investment, 2009).
White, I. Pin function for ASReml-R. http://www.homepages.ed.ac.uk/iwhite//asreml/ (2013).
Revell, L. J. phytools: an R package for phylogenetic comparative biology (and other things). Methods Ecol. Evol. 3, 217–223 (2012).
R Development Core Team R : A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, Austria, 2016).
Bininda-Emonds, O. R. P. et al. The delayed rise of present-day mammals. Nature 446, 507–512 (2007).
Jones, K. E. et al. PanTHERIA: a species-level database of life history, ecology, and geography of extant and recently extinct mammals. Ecology 90, 2648 (2009).
Frappell, P. B. & Butler, P. J. Minimal metabolic rate, what it is, its usefulness, and its relationship to the evolution of endothermy: a brief synopsis. Physiol. Biochem. Zool. 77, 865–868 (2004).
Butler, P. J., Green, J. A., Boyd, I. L. & Speakman, J. R. Measuring metabolic rate in the field: the pros and cons of the doubly labelled water and heart rate methods. Funct. Ecol. 18, 168–183 (2004).
Dlugosz, E. M. et al. Phylogenetic analysis of mammalian maximal oxygen consumption during exercise. J. Exp. Biol. 216, 4712–4721 (2013).
Norin, T. & Clark, T. D. Measurement and relevance of maximum metabolic rate in fishes. J. Fish Biol. 88, 122–151 (2016).
Lynch, M. Methods for the analysis of comparative data in evolutionary biology. Evolution 45, 1065–1080 (1991).
Housworth, E. A., Martins, E. P. & Lynch, M. The phylogenetic mixed model. Am. Nat. 163, 84–96 (2004).
Hadfield, J. D. & Nakagawa, S. General quantitative genetic methods for comparative biology: phylogenies, taxonomies and multi-trait models for continuous and categorical characters. J. Evol. Biol. 23, 494–508 (2010).
Hinchliff, C. E. et al. Synthesis of phylogeny and taxonomy into a comprehensive tree of life. Proc. Natl Acad. Sci. USA 112, 12764–12769 (2015).
Hadfield, J. D. MCMC methods for multi-response generalized linear models: the MCMCglmm R package. J. Stat. Softw. 33, 1–22 (2010).
Schluter, D. Adaptive radiation along genetic lines of least resistance. Evolution 50, 1766–1774 (1996).
Acknowledgements
This research was supported by the Australian Research Council (projects DP110101776, FT130101493, DP170101114 and DP180103925).
Author information
Authors and Affiliations
Contributions
C.R.W., D.O.-B. and D.J.M. designed the study. C.R.W., L.A.A., P.A.A., J.E.B., C.L.B., C.C., T.S.C., A.J., E.P., H.S.W.-S., M.J.A., S.F.C., C.E.F., L.G.H., M.R.K. and S.J.P. collected the data. C.R.W. analysed the data. C.R.W. and D.O.-B. wrote the first version of the manuscript. All authors contributed to and approved the final version.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Supplementary Information
Supplementary Methods, Supplementary References, Supplementary Tables 1–7 and Supplementary Figures 1–4
Rights and permissions
About this article
Cite this article
White, C.R., Marshall, D.J., Alton, L.A. et al. The origin and maintenance of metabolic allometry in animals. Nat Ecol Evol 3, 598–603 (2019). https://doi.org/10.1038/s41559-019-0839-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41559-019-0839-9
This article is cited by
-
Detecting natural selection in trait-trait coevolution
BMC Ecology and Evolution (2023)
-
Putting the energetic-savings hypothesis underground: fossoriality does not affect metabolic rates in amphibians
Evolutionary Ecology (2023)
-
Energy constraints on brain network formation
Scientific Reports (2021)
-
Correlational selection in the age of genomics
Nature Ecology & Evolution (2021)
-
Meta-analysis reveals that resting metabolic rate is not consistently related to fitness and performance in animals
Journal of Comparative Physiology B (2021)