Abstract
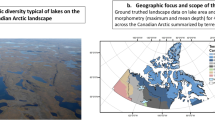
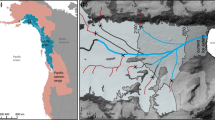
Climate warming at high latitudes has long been expected to exceed that predicted for tropical and temperate climes, but recent warming in the Arctic has exceeded even those expectations1. The geophysical consequences of this warming are reasonably well established2, but the impacts on freshwater fauna are poorly understood. Here we use a large-scale geospatial analysis of the population dynamics of one of the most abundant north temperate freshwater fish species to forecast increased demographic rates, productivity and colonization range in response to IPCC climate warming scenarios. Geospatial lake morphometry data were used to characterize 481,784 lakes in the Canadian Arctic capable of supporting lake trout (Salvelinus namaycush) populations. Lake trout productivity in existing habitat is projected to increase by 20% by 2050 due to climate change, but an expanded habitable zone may result in a 29% increase in harvestable biomass. Although many ecosystems are likely to be negatively impacted by climate warming, the phenotypic plasticity of fish will allow a rapid relaxation of the current environmental constraints on growth in the far north, as well as enhanced colonization of bodies of water in which there are few potential competitors.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
The geospatial data (Canadian Digital Surface Model and Canadian Digital Elevation Model) are available from http://maps.canada.ca/czs/index-en.html. Air temperature data from 881 weather stations across Canada are available from http://climate.weather.gc.ca/climate_normals/results_1981_2010_e.html.
Other data that support the findings of this study have been archived at Knowledge Network for Biocomplexity https://doi.org/10.5063/F1ZP44F1 and https://doi.org/10.5063/F1TX3CPV.
References
Huang, J. et al. Recently amplified arctic warming has contributed to a continual global warming trend. Nat. Clim. Change 7, 875–879 (2017).
Meredith, M. et al. in IPCC Special Report on the Ocean and Cryosphere in a Changing Climate (eds H.-O. Pörtner et al) Ch. 3 (IPCC, Cambridge Univ. Press, 2019).
Thuiller, W. et al. Consequences of climate change on the tree of life in Europe. Nature 470, 531–534 (2011).
Beaugrand, G. A. et al. Prediction of unprecedented biological shifts in the global ocean. Nat. Clim. Change 9, 237–243 (2019).
Forcada, J., Trathan, P. N. & Murphy, E. J. Life history buffering in Antarctic mammals and birds against changing patterns of climate and environmental variation. Glob. Change Biol. 14, 2473–2488 (2008).
Pacifici, M. et al. Assessing species vulnerability to climate change. Nat. Clim. Change 5, 215–225 (2015).
Frainer, A. et al. Climate-driven changes in functional biogeography of Arctic marine fish communities. Proc. Natl Acad. Sci. USA 114, 12202–12207 (2017).
Wessely, J. et al. Habitat-based conservation strategies cannot compensate for climate-change-induced range loss. Nat. Clim. Change 7, 823–827 (2017).
Ryder, R. A. The Morphoedaphic Index—use, abuse and fundamental concepts. Trans. Am. Fish. Soc. 111, 154–164 (1982).
Messager, M. L., Lehner, B., Grill, G., Nedeva, I. & Schmitt, O. Estimating the volume and age of water stored in global lakes using a geo-statistical approach. Nat. Comm. 7, 13603 (2016).
Campana, S. E., Casselman, J. M. & Jones, C. M. Bomb radiocarbon chronologies in the Arctic, with implications for the age validation of lake trout (Salvelinus namaycush) and other Arctic species. Can. J. Fish. Aquat. Sci. 65, 733–743 (2008).
Shuter, B. J., Jones, M. L., Korver, R. M. & Lester, N. P. A general, life history based model for regional management of fish stocks: the inland lake trout (Salvelinus namaycush) fisheries of Ontario. Can. J. Fish. Aquat. Sci. 55, 2161–2177 (1998).
Campana, S. E. Accuracy, precision and quality control in age determination, including a review of the use and abuse of age validation methods. J. Fish. Biol. 59, 197–242 (2001).
Casselman, J. M., Jones, C. M. & Campana, S. E. Bomb radiocarbon age validation for the long-lived, unexploited Arctic fish species Coregonus clupeaformis. Mar. Freshwat. Res. 70, 1–8 (2019).
Lester, N. P., Shuter, B. J. & Abrams, P. A. Interpreting the von Bertalanffy model of somatic growth in fish: the cost of reproduction. Proc. R. Soc. Ser. B 271, 1625–1631 (2004).
Minte-Vera, C. V., Maunder, M. N., Casselman, J. M. & Campana, S. E. Growth functions that incorporate the cost of reproduction. Fish. Res. 180, 31–44 (2016).
Collins, M. et al. in Climate Change 2013: The Physical Science Basis (eds Stocker, T. F. et al.) 1029–1136 (IPCC, Cambridge Univ. Press, 2013)
Climate Change 2014: Synthesis Report (eds Pachauri, R. K. and Meyer, L. A.) (IPCC, Cambridge Univ. Press, 2014).
Islam, D. & Berkes, F. Indigenous peoples' fisheries and food security: a case from northern Canada. Food Secur. 8, 815–826 (2016).
Musick, J. A. Ecology and conservation of long-lived marine animals. Am. Fish. Soc. Symp. 23, 1–10 (1999).
Schloss, C. A., Nunez, T. A. & Lawler, J. J. Dispersal will limit ability of mammals to track climate change in the Western Hemisphere. Proc. Natl Acad. Sci. USA 109, 8606–8611 (2012).
Hirsch, P. E., N’Guyen, A., Muller, R., Adrian‐Kalchhauser, I. & Burkhardt‐Holm, P. Colonizing Islands of water on dry land—on the passive dispersal of fish eggs by birds. Fish. Fish. 19, 502–510 (2018).
Spens, J., Englund, G. & Lundqvist, H. Network connectivity and dispersal barriers: using geographical information system (GIS) tools to predict landscape scale distribution of a key predator (Esox lucius) among lakes. J. Appl. Ecol. 44, 1127–1137 (2007).
Swanson, H. K. et al. Anadromy in Arctic populations of lake trout (Salvelinus namaycush): otolith microchemistry, stable isotopes, and comparisons with Arctic char (Salvelinus alpinus). Can. J. Fish. Aquat. Sci. 67, 842–853 (2010).
Ockendon, N. et al. Mechanisms underpinning climatic impacts on natural populations: altered species interactions are more important than direct effects. Glob. Change Biol. 20, 2221–2229 (2014).
Wilson, K. L., De Gisi, J., Cahill, C. L., Barker, O. E. & Post, J. R. Life‐history variation along environmental and harvest clines of a northern freshwater fish: plasticity and adaptation. J. Anim. Ecol. 88, 717–733 (2019).
Gauthier, G. et al. Long-term monitoring at multiple trophic levels suggests heterogeneity in responses to climate change in the Canadian Arctic tundra. Philos. Trans. R. Soc. B 368, 20120482 (2013).
Thomas, C. D. Climate, climate change and range boundaries. Diversity Distrib. 16, 488–495 (2010).
Healey, M. C. The dynamics of exploited lake trout populations and implications for management. J. Wildl. Manag. 42, 307–328 (1978).
Burr, J. M. Growth, density and biomass of lake trout in Arctic and Subarctic Alaska. Am. Fish. Soc. Symp. 19, 109–118 (1997).
Mills, K. H., Dyck, M. & Harwood, L. A. Proceedings of the second lake trout symposium 2005, Yellowknife, Northwest territories. Can. Tech. Rep. Fish. Aquat. Sci. 2778, 247 (2008).
Hollister, J. W., Milstead, W. B. & Urrutia, M. A. Predicting maximum lake depth from surrounding topography. PLoS ONE 6, e25764 (2011).
Livingstone, D. M., Lotter, A. F. & Walker, I. R. The decrease in summer surface water temperature with altitude in Swiss alpine lakes: a comparison with air temperature lapse rates. Arct. Antarct. Alp. Res. 31, 341–352 (1999).
Shuter, B. J., Schlesinger, D. A. & Zimmerman, A. P. Empirical predictors of annual surface water temperature cycles in North American lakes. Can. J. Fish. Aquat. Sci. 40, 1838–1845 (1983).
Da Fang, X. & Stefan, H. G. Long-term lake water temperature and ice cover simulations/measurements. Cold Reg. Sci. Technol. 24, 289–304 (1996).
Campana, S. E. Physical Characteristics of 55 Canadian Arctic Lake Trout Lakes (Knowledge Network for Biocomplexity archive, 2020); https://doi.org/10.5063/F1ZP44F1
Campana, S. E. Lake Trout Population Characteristics in 55 Canadian Arctic Reference Lakes (Knowledge Network for Biocomplexity, 2020); https://doi.org/10.5063/F1TX3CPV.
Samarasin, P., Minns, C. K., Shuter, B. J., Tonn, W. M. & Rennie, M. D. Fish diversity and biomass in northern Canadian lakes: northern lakes are more diverse and have greater biomass than expected based on species–energy theory. Can. J. Fish. Aquat. Sci. 72, 226–237 (2015).
Campana, S. E., Valentin, A. E., MacLellan, S. E. & Groot, J. B. Image-enhanced burnt otoliths, bomb radiocarbon and the growth dynamics of redfish (Sebastes mentella and S. fasciatus) off the eastern coast of Canada. Mar. Freshw. Res. 67, 925–936 (2016).
Francis, R. I. C. C. Growth in age-structured stock assessment models. Fish. Res. 180, 113–118 (2015).
Smith, M. W. et al. Recommendations for catch-curve analysis. North Am. J. Fish. Managem. 32, 956–967 (2012).
Ricker, W. E. Computation and Interpretation of Biological Statistics of Fish Populations (Bulletin of the Fisheries Research Board of Canada, 1975).
Deriso, R. B. Optimal F 0.1 criteria and their relationship to maximum sustainable yield. Can. J. Fish. Aquat. Sci. 44, 339–348 (1987).
Acknowledgements
This work was supported by the Nunavut Wildlife Management Board, Fisheries and Oceans Canada, US National Science Foundation grant OCE-9985884 and the University of Iceland. We thank S. Armsworthy, P. Bentzen, J. Brazner, C. Campana, P. Campana, S. Campana, S. Casselman, M. Fowler, D. Houlihan, W. Joyce, P. Leblanc, A. MacDonnell and M. Showell for their exceptional assistance in the field and laboratory. B. Shuter and J. Morrongiello provided valuable comments on the MS.
Author information
Authors and Affiliations
Contributions
The study was conceived and designed by S.C., J.C. and C.J., and coordinated by S.C. All authors contributed to fieldwork, data collection and/or data interpretation. G.B. (now retired) prepared and analysed the geospatial data. S.C. drafted the paper with contributions from all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Climate Change thanks Arild Folkvord and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–3, Tables 1–4 and Notes 1–4.
Rights and permissions
About this article
Cite this article
Campana, S.E., Casselman, J.M., Jones, C.M. et al. Arctic freshwater fish productivity and colonization increase with climate warming. Nat. Clim. Chang. 10, 428–433 (2020). https://doi.org/10.1038/s41558-020-0744-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41558-020-0744-x
This article is cited by
-
Major changes in fish thermal habitat diversity in Canada’s Arctic lakes due to climate change
Communications Earth & Environment (2024)
-
Seasonal variation in activity and nearshore habitat use of Lake Trout in a subarctic lake
Movement Ecology (2023)
-
Climate warming accelerates somatic growth of an Arctic fish species in high-latitude lakes
Scientific Reports (2023)
-
Adaptability of lake charr (Salvelinus namaycush) in a changing world: newly recolonized landscapes reveal the significance of traits shaped during the Pleistocene
Environmental Biology of Fishes (2023)
-
An ecothermal paradox: bull trout populations diverge in response to thermal landscapes across a broad latitudinal gradient
Environmental Biology of Fishes (2023)