Abstract
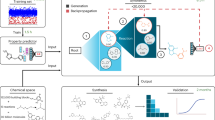
The oral administration of peptide drugs is hampered by their metabolic instability and limited intestinal uptake. Here, we describe a method for the generation of small target-specific peptides (less than 1,600 Da in size) that resist gastrointestinal proteases. By using phage display to screen large libraries of genetically encoded double-bridged peptides on protease-resistant fd bacteriophages, we generated a peptide inhibitor of the coagulation Factor XIa with nanomolar affinity that resisted gastrointestinal proteases in all regions of the gastrointestinal tract of mice after oral administration, enabling more than 30% of the peptide to remain intact, and small quantities of it to reach the blood circulation. We also developed a gastrointestinal-protease-resistant peptide antagonist for the interleukin-23 receptor, which has a role in the pathogenesis of Crohn’s disease and ulcerative colitis. The de novo generation of targeted peptides that resist proteolytic degradation in the gastrointestinal tract should help the development of effective peptides for oral delivery.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The main data supporting the results of this study are available within the paper and its Supplementary Information. The data used to make the graphs in the figures are provided as Supplementary Information. Next-generation sequencing source data are available from figshare at https://doi.org/10.6084/m9.figshare.11921139.v2. Coordinates of the two X-ray structures have been deposited in the Protein Data Bank with accession numbers 6TWB and 6TWC.
References
Goldberg, M. & Gomez-Orellana, I. Challenges for the oral delivery of macromolecules. Nat. Rev. Drug Discov. 2, 289–295 (2003).
Moroz, E., Matoori, S. & Leroux, J. C. Oral delivery of macromolecular drugs: where we are after almost 100 years of attempts. Adv. Drug Deliv. Rev. 101, 108–121 (2016).
Pratley, R. et al. Oral semaglutide versus subcutaneous liraglutide and placebo in type 2 diabetes (PIONEER 4): a randomised, double-blind, phase 3a trial. Lancet 394, 39–50 (2019).
Nielsen, D. S. et al. Orally absorbed cyclic peptides. Chem. Rev. 117, 8094–8128 (2017).
Aguirre, T. A. S. et al. Current status of selected oral peptide technologies in advanced preclinical development and in clinical trials. Adv. Drug Deliv. Rev. 106, 223–241 (2016).
Räder, A. F. B. et al. Orally active peptides: Is there a magic bullet? Angew. Chem. Int. Ed. 57, 14414–14438 (2018).
Naylor, M. R., Bockus, A. T., Blanco, M. J. & Lokey, R. S. Cyclic peptide natural products chart the frontier of oral bioavailability in the pursuit of undruggable targets. Curr. Opin. Chem. Biol. 38, 141–147 (2017).
Gentilucci, L., De Marco, R. & Cerisoli, L. Chemical modifications designed to improve peptide stability: incorporation of non-natural amino acids, pseudo-peptide bonds, and cyclization. Curr. Pharm. Des. 16, 3185–3203 (2010).
Perry, A. F. et al. Hydrocarbon double-stapling remedies the proteolytic instability of a lengthy peptide therapeutic. Proc. Natl Acad. Sci. USA 107, 14093–14098 (2010).
Wang, C. K. & Craik, D. J. Designing macrocyclic disulfide-rich peptides for biotechnological applications perspective. Nat. Chem. Biol. 14, 417–427 (2018).
Banga, A. K. Therapeutic Peptides and Proteins: Formulation, Processing, and Delivery Systems 3rd edn (CRC Press, 2015).
Kristensen, P. & Winter, G. Proteolytic selection for protein folding using filamentous bacteriophages. Fold. Des. 3, 321–328 (1998).
Sieber, V., Plückthun, A. & Schmid, F. X. Selecting proteins with improved stability by a phage-based method. Nat. Biotechnol. 16, 955–960 (1998).
Howell, S. M. et al. Serum stable natural peptides designed by mRNA display. Sci. Rep. 4, 6008 (2015).
Baeriswyl, V. & Heinis, C. Phage selection of cyclic peptide antagonists with increased stability toward intestinal proteases. Protein Eng. Des. Sel. 26, 81–89 (2013).
Kather, I., Bippes, C. A. & Schmid, F. X. A stable disulfide-free gene-3-protein of phage fd generated by in vitro evolution. J. Mol. Biol. 354, 666–678 (2005).
Kale, S. S. et al. Cyclization of peptides with two chemical bridges affords large scaffold diversities. Nat. Chem. 10, 715–723 (2018).
Wang, J., Yadav, V., Smart, A. L., Tajiri, S. & Basit, A. W. Toward oral delivery of biopharmaceuticals: an assessment of the gastrointestinal stability of 17 peptide drugs. Mol. Pharm. 12, 966–973 (2015).
Heinis, C., Rutherford, T., Freund, S. & Winter, G. Phage-encoded combinatorial chemical libraries based on bicyclic peptides. Nat. Chem. Biol. 5, 502–507 (2009).
Bane, C. E. & Gailani, D. Factor XI as a target for antithrombotic therapy. Drug Discov. Today 19, 1454–1458 (2014).
Rentero Rebollo, I., Sabisz, M., Baeriswyl, V. & Heinis, C. Identification of target-binding peptide motifs by high-throughput sequencing of phage-selected peptides. Nucleic Acids Res. 42, e169 (2014).
Gaffen, S. L., Jain, R., Garg, A. V. & Cua, D. J. The IL-23–IL-17 immune axis: from mechanisms to therapeutic testing. Nat. Rev. Immunol. 14, 585–600 (2014).
Bourne, G. T. et al. Oral peptide inhibitors of interleukin-23 receptor and their use to treat inflammatory bowel diseases. US patent US9624268B2 (2015).
Sayago, C. et al. Deciphering binding interactions of IL-23R with HDX–MS: mapping protein and macrocyclic dodecapeptide ligands. ACS Med. Chem. Lett. 9, 912–916 (2018).
Patel, D. V. Protagonist therapeutics expands PTG-200 collaboration agreement with Janssen to include second generation oral IL-23 receptor antagonists. Bloomberg (8 May 2019); https://www.bloomberg.com/press-releases/2019-05-08/protagonist-therapeutics-expands-ptg-200-collaboration-agreement-with-janssen-to-include-second-generation-oral-il-23-receptor
Rentero Rebollo, I. & Heinis, C. Phage selection of bicyclic peptides. Methods 60, 46–54 (2013).
Acknowledgements
We thank B. Mangeat from the Ecole Polytechnique Fédérale de Lausanne (EPFL) Gene Expression Core Facility for help with next-generation sequencing, A. Reynaud, D. Hacker, L. Durrer and S. Quinche from the EPFL Protein Production and Structure Core Facility for help with protein expression and crystallization, and E. Simeoni, I. Desbaillets, G. Ferrand and C. Waldvogel of the EPFL Center of Phenogenomics (CPG) for help with mouse experiments. The financial contributions from the Swiss National Science Foundation grants (project grant 157842, NCCR Chemical Biology and project grant 169526) and the EPFL are gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
X.-D.K. and C.H. conceived the strategy for proteolytic phage display with double-bridged peptides. X.-D.K. established the phage selection procedure, cloned the libraries, performed the phage selections against FXIa, synthesized and characterized the peptides, expressed and purified FXIa and determined the X-ray structures. J.M. expressed and purified IL-23R, performed the phage selections against IL-23R and characterized the peptides. V.C. identified the d-proline mutant. F.P. collected X-ray data and analysed the structures. L.A.A. performed the NMR study. X.-D.K., K.D. and C.H. wrote the manuscript with help from all authors.
Corresponding author
Ethics declarations
Competing interests
J.M., X.-D.K. and C.H. are inventors on a patent protecting the IL-23R antagonists.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary results, methods, figures and tables.
Supplementary Dataset 1
Source data for the figures.
Rights and permissions
About this article
Cite this article
Kong, XD., Moriya, J., Carle, V. et al. De novo development of proteolytically resistant therapeutic peptides for oral administration. Nat Biomed Eng 4, 560–571 (2020). https://doi.org/10.1038/s41551-020-0556-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-020-0556-3
This article is cited by
-
Characterisation of IL-23 receptor antagonists and disease relevant mutants using fluorescent probes
Nature Communications (2023)
-
High-affinity peptides developed against calprotectin and their application as synthetic ligands in diagnostic assays
Nature Communications (2023)
-
Targeting MYC with modular synthetic transcriptional repressors derived from bHLH DNA-binding domains
Nature Biotechnology (2023)
-
Efficient in planta production of amidated antimicrobial peptides that are active against drug-resistant ESKAPE pathogens
Nature Communications (2023)
-
De novo development of small cyclic peptides that are orally bioavailable
Nature Chemical Biology (2023)