Abstract
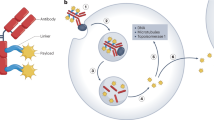
Antibody-mediated tumour targeting and nanoparticle-mediated encapsulation can reduce the toxicity of antitumour drugs and improve their efficacy. Here, we describe the performance of a nanotherapeutic encapsulating a hydrolytically sensitive docetaxel prodrug and conjugated to an antibody specific for EphA2—a receptor overexpressed in many tumours. Administration of the nanotherapeutic in mice led to slow and sustained release of the prodrug, reduced exposure of active docetaxel in the circulation (compared with administration of the free drug) and maintenance of optimal exposure of the drug in tumour tissue. We also show that administration of the nanotherapeutic in rats and dogs resulted in minimal haematological toxicity, as well as the absence of neutropenia and improved overall tolerability in multiple rodent models. Targeting of the nanotherapeutic to EphA2 improved tumour penetration and resulted in markedly enhanced antitumour activity (compared with administration of free docetaxel and non-targeted nanotherapeutic controls) in multiple tumour-xenografted mice. This nanomedicine could become a potent and safe therapeutic alternative for cancer patients undergoing chemotherapy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The datasets generated and/or analysed during the study, and source data for the figures, are available in Figshare at https://doi.org/10.6084/m9.figshare.7786205.
References
Moek, K. L., de Groot, D. J. A., de Vries, E. G. E. & Fehrmann, R. S. N. The antibody–drug conjugate target landscape across a broad range of tumour types. Ann. Oncol. 28, 3083–3091 (2017).
Lee, H. et al. 64Cu-MM-302 positron emission tomography quantifies variability of enhanced permeability and retention of nanoparticles in relation to treatment response in patients with metastatic breast cancer. Clin. Cancer Res. 23, 4190–4202 (2017).
Maeda, H. Toward a full understanding of the EPR effect in primary and metastatic tumors as well as issues related to its heterogeneity. Adv. Drug Deliv. Rev. 91, 3–6 (2015).
Ramanathan, R. K. et al. Correlation between ferumoxytol uptake in tumor lesions by MRI and response to nanoliposomal irinotecan in patients with advanced solid tumors: a pilot study. Clin. Cancer Res. 23, 3638–3648 (2017).
Coulthard, M. G. et al. The role of the Eph–ephrin signalling system in the regulation of developmental patterning. Int. J. Dev. Biol. 46, 375–384 (2002).
Oates, A. C. et al. An early developmental role for Eph–ephrin interaction during vertebrate gastrulation. Mech. Dev. 83, 77–94 (1999).
Yang, N. Y. et al. Crosstalk of the EphA2 receptor with a serine/threonine phosphatase suppresses the Akt-mTORC1 pathway in cancer cells. Cell. Signal. 23, 201–212 (2011).
Tandon, M., Vemula, S. V. & Mittal, S. K. Emerging strategies for EphA2 receptor targeting for cancer therapeutics. Expert Opin. Ther. Targets 15, 31–51 (2011).
Brannan, J. M. et al. Expression of the receptor tyrosine kinase EphA2 is increased in smokers and predicts poor survival in non-small cell lung cancer. Clin. Cancer Res. 15, 4423–4430 (2009).
Brantley-Sieders, D. M. et al. The receptor tyrosine kinase EphA2 promotes mammary adenocarcinoma tumorigenesis and metastatic progression in mice by amplifying ErbB2 signaling. J. Clin. Invest. 118, 64–78 (2008).
Gokmen-Polar, Y. et al. Dual targeting of EphA2 and ER restores tamoxifen sensitivity in ER/EphA2-positive breast cancer. Breast Cancer Res. Treat. 127, 375–384 (2011).
Yuan, W. J. et al. Over-expression of EphA2 and EphrinA-1 in human gastric adenocarcinoma and its prognostic value for postoperative patients. Dig. Dis. Sci. 54, 2410–2417 (2009).
Miyazaki, T., Kato, H., Fukuchi, M., Nakajima, M. & Kuwano, H. EphA2 overexpression correlates with poor prognosis in esophageal squamous cell carcinoma. Int. J. Cancer 103, 657–663 (2003).
Mudali, S. V. et al. Patterns of EphA2 protein expression in primary and metastatic pancreatic carcinoma and correlation with genetic status. Clin. Exp. Metastasis 23, 357–365 (2006).
Dunne, P. D. et al. EphA2 expression is a key driver of migration and invasion and a poor prognostic marker in colorectal cancer. Clin. Cancer Res. 22, 230–242 (2016).
Zeng, G. et al. High-level expression of EphA2 receptor tyrosine kinase in prostatic intraepithelial neoplasia. Am. J. Pathol. 163, 2271–2276 (2003).
Abraham, S. et al. Expression of EphA2 and Ephrin A-1 in carcinoma of the urinary bladder. Clin. Cancer Res. 12, 353–360 (2006).
Liu, Y. et al. Clinical significance of EphA2 expression in squamous-cell carcinoma of the head and neck. J. Cancer Res. Clin. Oncol. 137, 761–769 (2011).
Wang, L. F. et al. Increased expression of EphA2 correlates with adverse outcome in primary and recurrent glioblastoma multiforme patients. Oncol. Rep. 19, 151–156 (2008).
Holm, R., de Putte, G. V., Suo, Z., Lie, A. K. & Kristensen, G. B. Expressions of EphA2 and EphrinA-1 in early squamous cell cervical carcinomas and their relation to prognosis. Int. J. Med. Sci. 5, 121–126 (2008).
Merritt, W. M. et al. Clinical and biological impact of EphA2 overexpression and angiogenesis in endometrial cancer. Cancer Biol. Ther. 10, 1306–1314 (2010).
Thaker, P. H. et al. EphA2 expression is associated with aggressive features in ovarian carcinoma. Clin. Cancer Res. 10, 5145–5150 (2004).
Brantley-Sieders, D. M. et al. Eph/ephrin profiling in human breast cancer reveals significant associations between expression level and clinical outcome. PLoS ONE 6, e24426 (2011).
Cui, X. D. et al. EFNA1 ligand and its receptor EphA2: potential biomarkers for hepatocellular carcinoma. Int. J. Cancer 126, 940–949 (2010).
Ishikawa, M. et al. Higher expression of EphA2 and ephrin-A1 is related to favorable clinicopathological features in pathological stage I non-small cell lung carcinoma. Lung Cancer 76, 431–438 (2012).
Kinch, M. S., Moore, M. B. & Harpole, D. H. Jr. Predictive value of the EphA2 receptor tyrosine kinase in lung cancer recurrence and survival. Clin. Cancer Res. 9, 613–618 (2003).
Annunziata, C. M. et al. Phase 1, open-label study of MEDI-547 in patients with relapsed or refractory solid tumors. Invest New Drugs 31, 77–84 (2013).
Cai, W. et al. Quantitative radioimmunoPET imaging of EphA2 in tumor-bearing mice. Eur. J. Nucl. Med. Mol. Imaging 34, 2024–2036 (2007).
Jackson, D. et al. A human antibody–drug conjugate targeting EphA2 inhibits tumor growth in vivo. Cancer Res. 68, 9367–9374 (2008).
Barile, E. et al. Design, synthesis and bioevaluation of an EphA2 receptor-based targeted delivery system. ChemMedChem 9, 1403–1412 (2014).
Wang, F. et al. The interaction of 4-thiazolidinone derivatives containing indolin-2-one moiety with P-glycoprotein studied using K562 cell lines. Eur. J. Med. Chem. 101, 126–132 (2015).
Wu, B. et al. Design and characterization of novel EphA2 agonists for targeted delivery of chemotherapy to cancer cells. Chem. Biol. 22, 876–887 (2015).
Wykosky, J., Gibo, D. M. & Debinski, W. A novel, potent, and specific ephrinA1-based cytotoxin against EphA2 receptor expressing tumor cells. Mol. Cancer Ther. 6, 3208–3218 (2007).
Patel, A. R., Chougule, M. & Singh, M. EphA2 targeting PEGylated nanocarrier drug delivery system for treatment of lung cancer. Pharm. Res. 31, 2796–2809 (2014).
Patel, K., Doddapaneni, R., Sekar, V., Chowdhury, N. & Singh, M. Combination approach of YSA peptide anchored docetaxel stealth liposomes with oral antifibrotic agent for the treatment of lung cancer. Mol. Pharm. 13, 2049–2058 (2016).
Geddie, M. L. et al. Improving the developability of an anti-EphA2 single-chain variable fragment for nanoparticle targeting. MAbs 9, 58–67 (2017).
Rhoden, J. J., Dyas, G. L. & Wroblewski, V. J. A modeling and experimental investigation of the effects of antigen density, binding affinity, and antigen expression ratio on bispecific antibody binding to cell surface targets. J. Biol. Chem. 291, 11337–11347 (2016).
Zhou, Y. et al. Impact of single-chain Fv antibody fragment affinity on nanoparticle targeting of epidermal growth factor receptor-expressing tumor cells. J. Mol. Biol. 371, 934–947 (2007).
Li, Z. & Gorfe, A. A. Receptor-mediated membrane adhesion of lipid-polymer hybrid (LPH) nanoparticles studied by dissipative particle dynamics simulations. Nanoscale 7, 814–824 (2015).
Kirpotin, D. et al. Sterically stabilized anti-HER2 immunoliposomes: design and targeting to human breast cancer cells in vitro. Biochemistry 36, 66–75 (1997).
Drummond, D. C. et al. Improved pharmacokinetics and efficacy of a highly stable nanoliposomal vinorelbine. J. Pharmacol. Exp. Ther. 328, 321–330 (2009).
Drummond, D. C. et al. Development of a highly active nanoliposomal irinotecan using a novel intraliposomal stabilization strategy. Cancer Res. 66, 3271–3277 (2006).
Drummond, D. C., Noble, C. O., Hayes, M. E., Park, J. W. & Kirpotin, D. B. Pharmacokinetics and in vivo drug release rates in liposomal nanocarrier development. J. Pharm. Sci. 97, 4696–4740 (2008).
Bittman, R. & Verbicky, C. A. Methanolysis of sphingomyelin. Toward an epimerization-free methodology for the preparation of d-erythro-sphingosylphosphocholine. J. Lipid Res. 41, 2089–2093 (2000).
Jain, R. K., Tong, R. T. & Munn, L. L. Effect of vascular normalization by antiangiogenic therapy on interstitial hypertension, peritumor edema, and lymphatic metastasis: insights from a mathematical model. Cancer Res. 67, 2729–2735 (2007).
Maurer-Spurej, E., Wong, K. F., Maurer, N., Fenske, D. B. & Cullis, P. R. Factors influencing uptake and retention of amino-containing drugs in large unilamellar vesicles exhibiting transmembrane pH gradients. Biochim. Biophys. Acta 1416, 1–10 (1999).
Zucker, D., Marcus, D., Barenholz, Y. & Goldblum, A. Liposome drugs’ loading efficiency: a working model based on loading conditions and drug’s physicochemical properties. J. Control. Release 139, 73–80 (2009).
Goldstein, D. et al. nab-Paclitaxel plus gemcitabine for metastatic pancreatic cancer: long-term survival from a phase III trial. J. Natl Cancer Inst. 107, dju413 (2015).
Von Hoff, D. D. et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N. Engl. J. Med. 369, 1691–1703 (2013).
Ho, M. Y. & Mackey, J. R. Presentation and management of docetaxel-related adverse effects in patients with breast cancer. Cancer Manag. Res. 6, 253–259 (2014).
Huang, T. C. & Campbell, T. C. Comparison of weekly versus every 3 weeks paclitaxel in the treatment of advanced solid tumors: a meta-analysis. Cancer Treat. Rev. 38, 613–617 (2012).
Mauri, D. et al. Overall survival benefit for weekly vs. three-weekly taxanes regimens in advanced breast cancer: a meta-analysis. Cancer Treat. Rev. 36, 69–74 (2010).
Jordan, M. A. & Wilson, L. Microtubules as a target for anticancer drugs. Nat. Rev. Cancer 4, 253–265 (2004).
Munzone, E. & Colleoni, M. Clinical overview of metronomic chemotherapy in breast cancer. Nat. Rev. Clin. Oncol. 12, 631–644 (2015).
Kareva, I., Waxman, D. J. & Lakka Klement, G. Metronomic chemotherapy: an attractive alternative to maximum tolerated dose therapy that can activate anti-tumor immunity and minimize therapeutic resistance. Cancer Lett. 358, 100–106 (2015).
Starobova, H. & Vetter, I. Pathophysiology of chemotherapy-induced peripheral neuropathy. Front. Mol. Neurosci. 10, 174 (2017).
Mathew, A. E., Mejillano, M. R., Nath, J. P., Himes, R. H. & Stella, V. J. Synthesis and evaluation of some water-soluble prodrugs and derivatives of taxol with antitumor activity. J. Med. Chem. 35, 145–151 (1992).
Zhigaltsev, I. V. et al. Development of a weak-base docetaxel derivative that can be loaded into lipid nanoparticles. J. Control. Release 144, 332–340 (2010).
Blair, H. A. & Deeks, E. D. Albumin-bound paclitaxel: a review in non-small cell lung cancer. Drugs 75, 2017–2024 (2015).
Hrkach, J. et al. Preclinical development and clinical translation of a PSMA-targeted docetaxel nanoparticle with a differentiated pharmacological profile. Sci. Transl. Med. 4, 128ra139 (2012).
Chipman, S. D., Oldham, F. B., Pezzoni, G. & Singer, J. W. Biological and clinical characterization of paclitaxel poliglumex (PPX, CT-2103), a macromolecular polymer–drug conjugate. Int. J. Nanomed. 1, 375–383 (2006).
Sparreboom, A. et al. Comparative preclinical and clinical pharmacokinetics of a cremophor-free, nanoparticle albumin-bound paclitaxel (ABI-007) and paclitaxel formulated in Cremophor (Taxol). Clin. Cancer Res. 11, 4136–4143 (2005).
Gradishar, W. J. et al. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J. Clin. Oncol. 23, 7794–7803 (2005).
Gradishar, W. J. et al. Significantly longer progression-free survival with nab-paclitaxel compared with docetaxel as first-line therapy for metastatic breast cancer. J. Clin. Oncol. 27, 3611–3619 (2009).
Coffman, K. T. et al. Differential EphA2 epitope display on normal versus malignant cells. Cancer Res. 63, 7907–7912 (2003).
Safavy, A. et al. Synthesis and biological evaluation of paclitaxel-C225 conjugate as a model for targeted drug delivery. Bioconjug. Chem. 14, 302–310 (2003).
Polakis, P. Antibody drug conjugates for cancer therapy. Pharmacol. Rev. 68, 3–19 (2016).
Beck, A., Goetsch, L., Dumontet, C. & Corvaia, N. Strategies and challenges for the next generation of antibody–drug conjugates. Nat. Rev. Drug Discov. 16, 315–337 (2017).
Nielsen, U. B. et al. Therapeutic efficacy of anti-ErbB2 immunoliposomes targeted by a phage antibody selected for cellular endocytosis. Biochim. Biophys. Acta 1591, 109–118 (2002).
Zhou, Y., Zou, H., Zhang, S. & Marks, J. D. Internalizing cancer antibodies from phage libraries selected on tumor cells and yeast-displayed tumor antigens. J. Mol. Biol. 404, 88–99 (2010).
Nellis, D. F. et al. Preclinical manufacture of anti-HER2 liposome-inserting, scFv-PEG-lipid conjugate. 2. Conjugate micelle identity, purity, stability, and potency analysis. Biotechnol. Prog. 21, 221–232 (2005).
Nellis, D. F. et al. Preclinical manufacture of an anti-HER2 scFv-PEG-DSPE, liposome-inserting conjugate. 1. Gram-scale production and purification. Biotechnol. Prog. 21, 205–220 (2005).
Horie, R. & Watanabe, T. CD30: expression and function in health and disease. Semin. Immunol. 10, 457–470 (1998).
Pool, G. L., French, M. E., Edwards, R. A., Huang, L. & Lumb, R. H. Use of radiolabeled hexadecyl cholesteryl ether as a liposome marker. Lipids 17, 448–452 (1982).
Kalra, A. V. et al. Preclinical activity of nanoliposomal irinotecan is governed by tumor deposition and intratumor prodrug conversion. Cancer Res. 74, 7003–7013 (2014).
Acknowledgements
This work was supported in part by a grant from the Department of Defense (grant number OC150083 to C.P.R.). This work was also partially supported by National Cancer Institute grant P30CA016056, involving the use of Roswell Park’s Bioanalytics, Metabolomics and Pharmacokinetics Shared Resource (J.H.W. and S.A.S.). We also acknowledge L. Zalutskaya for support with both preparation and submission of the manuscript.
Author information
Authors and Affiliations
Contributions
W.S.K., D.B.K., Z.R.H., S.K.T., L.L., K.O., G.K., A.A.L., C.P.R., V.A. and D.C.D. designed the studies. D.B.K., Z.R.H., S.K.T., C.O.N., M.E.H., M.L.G., J.D.M. and D.C.D. designed the molecule. W.S.K., D.B.K., Z.R.H., S.K.T., C.O.N., M.E.H., L.L., A.K., T.K., S.O., C.P., N.D., S.A.S., J.H.W., M.L.G., J.S., S.G., N.K., R.B., Y.Z. and A.J.S. collected the data. W.S.K., D.B.K., Z.R.H., S.K.T., C.O.N., M.E.H., L.L., A.K., J.K., K.O., T.K., S.O., G.K., S.A.S., J.H.W., C.P.R., R.B., Y.Z., A.J.S. and D.C.D. analysed the data. W.S.K., V.A. and D.C.D. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
All of the authors affiliated with Merrimack Pharmaceuticals were employees of Merrimack Pharmaceuticals at the time of the study, with D.C.D., A.K., J.D.M., Z.R.H., A.J.S., L.L. and G.K. currently owning stock or stock options in Merrimack Pharmaceuticals.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary methods, results, figures and tables.
Rights and permissions
About this article
Cite this article
Kamoun, W.S., Kirpotin, D.B., Huang, Z.R. et al. Antitumour activity and tolerability of an EphA2-targeted nanotherapeutic in multiple mouse models. Nat Biomed Eng 3, 264–280 (2019). https://doi.org/10.1038/s41551-019-0385-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0385-4
This article is cited by
-
Eph receptors and ephrins in cancer progression
Nature Reviews Cancer (2024)
-
Downregulation of EphA2 stability by RNF5 limits its tumor-suppressive function in HER2-negative breast cancers
Cell Death & Disease (2023)
-
Ratiometric co-delivery of doxorubicin and paclitaxel prodrug by remote-loading liposomes for the treatment of triple-negative breast cancer
Drug Delivery and Translational Research (2022)
-
Targeted Delivery of Combination Therapeutics Using Monoclonal Antibody 2C5-Modified Immunoliposomes for Cancer Therapy
Pharmaceutical Research (2021)
-
The EphA2 and cancer connection: potential for immune-based interventions
Molecular Biology Reports (2020)