Abstract
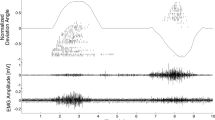
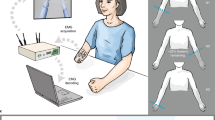
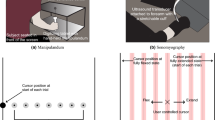
The intuitive control of upper-limb prostheses requires a man/machine interface that directly exploits biological signals. Here, we define and experimentally test an offline man/machine interface that takes advantage of the discharge timings of spinal motor neurons. The motor-neuron behaviour is identified by deconvolution of the electrical activity of muscles reinnervated by nerves of a missing limb in patients with amputation at the shoulder or humeral level. We mapped the series of motor-neuron discharges into control commands across multiple degrees of freedom via the offline application of direct proportional control, pattern recognition and musculoskeletal modelling. A series of experiments performed on six patients reveal that the man/machine interface has superior offline performance compared with conventional direct electromyographic control applied after targeted muscle innervation. The combination of surgical procedures, decoding and mapping into effective commands constitutes an interface with the output layers of the spinal cord circuitry that allows for the intuitive control of multiple degrees of freedom.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Farina, D. & Aszmann, O. Bionic limbs: clinical reality and academic promises. Sci. Transl. Med. 6, 257ps12 (2014).
Kuiken, T. A. et al. Targeted reinnervation for enhanced prosthetic arm function in a woman with a proximal amputation: a case study. Lancet 369, 371–380 (2007).
Tan, D. W. et al. A neural interface provides long-term stable natural touch perception. Sci. Transl. Med. 6, 257ra138 (2014).
Lebedev, M. A. & Nicolelis, M. A. L. Brain-machine interfaces: past, present and future. Trends Neurosci. 29, 536–546 (2006).
Dhillon, G. S., Lawrence, S. M., Hutchinson, D. T. & Horch, K. W. Residual function in peripheral nerve stumps of amputees: implications for neural control of artificial limbs. J. Hand Surg. Am. 29, 605–615 (2004).
Wodlinger, B. & Durand, D. M. Peripheral nerve signal recording and processing for artificial limb control. In 2010 Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. EMBC’10 6206–6209 (2010).
Amsuess, S. et al. Context-dependent upper limb prosthesis control for natural and robust use. IEEE Trans. Neural Syst. Rehabil. Eng. 744–753 (2015).
Gilja, V. et al. A high-performance neural prosthesis enabled by control algorithm design. Nat. Neurosci. 15, 1752–1757 (2012).
Donoghue, J. P. Connecting cortex to machines: recent advances in brain interfaces. Nat. Neurosci. 5 (suppl.), 1085–1088 (2002).
Hochberg, L. R. et al. Reach and grasp by people with tetraplegia using a neurally controlled robotic arm. Nature 485, 372–375 (2012).
Collinger, J. L. et al. High-performance neuroprosthetic control by an individual with tetraplegia. Lancet 381, 557–564 (2013).
Bouton, C. E. et al. Restoring cortical control of functional movement in a human with quadriplegia. Nature 533, 247–250 (2016).
Jiang, N., Dosen, S. & Farina, D. Myoelectric control of artificial limbs: is there the need for a change of focus? IEEE Signal Proc. Mag. 22, 549–558 (2012).
Hoffer, J. & Loeb, G. Implantable electrical and mechanical interfaces with nerve and muscle. Ann. Biomed. Eng. 8, 351–360 (1980).
Kuiken, T. A. et al. Targeted muscle reinnervation for real-time myoelectric control of multifunction artificial arms. JAMA 301, 619–628 (2009).
Gart, M. S., Souza, J. M. & Dumanian, G. A. Targeted muscle reinnervation in the upper extremity amputee: a technical roadmap. J. Hand Surg. Am. 40, 1877–1888 (2015).
Kuiken, T. A. et al. The use of targeted muscle reinnervation for improved myoelectric prosthesis control in a bilateral shoulder disarticulation amputee. Prosthet. Orthot. Int. 28, 245–253 (2004).
Cheesborough, J. E., Dumanian, G. A., Smith, L. H. & Kuiken, T. A. Targeted muscle reinnervation and advanced prosthetic arms. Semin. Plast. Surg. 1, 62–72 (2015).
Zhou, P. et al. Decoding a new neural machine interface for control of artificial limbs. J. Neurophysiol. 98, 2974–2982 (2007).
Tikhonov, A. N. On the stability of inverse problems. Dokl. Akad. Nauk 39, 195–198 (1943).
Birdwell, J. A., Hargrove, L. J., Weir, R. F. & Kuiken, T. A. Extrinsic finger and thumb muscles command a virtual hand to allow individual finger and grasp control. IEEE Trans. Biomed. Eng. 62, 218–226 (2015).
Cipriani, C., Segil, J. L., Birdwell, J. A. & Weir, R. F. Dexterous control of a prosthetic hand using fine-wire intramuscular electrodes in targeted extrinsic muscles. IEEE Trans. Neural Syst. Rehabil. Eng. 22, 828–836 (2014).
Li, Y., Smith, L. H., Hargrove, L. J., Weber, D. J. & Loeb, G. E. Sparse optimal motor estimation (SOME) for extracting commands for prosthetic limbs. IEEE Trans. Neural Syst. Rehabil. Eng. 21, 104–111 (2013).
Farina, D. et al. Non-invasive, accurate assessment of the behavior of representative populations of motor units in targeted reinnervated muscles. IEEE Trans. Neural Syst. Rehabil. Eng. 22, 810–819 (2014).
Kapelner, T. et al. Classification of motor unit activity following targeted muscle reinnervation. In 7th Int. IEEE/EMBS Conf. Neural Eng. (NER) 652–654 (IEEE, 2015).
Holobar, A., Glaser, V., Gallego, J. A., Dideriksen, J. L. & Farina, D. Noninvasive analysis of motor unit behavior in pathological tremor. In Proc. Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. EMBS 7512–7515 (2011).
Djilas, M., Azevedo-Coste, C., Guiraud, D. & Yoshida, K. Spike sorting of muscle spindle afferent nerve activity recorded with thin-film intrafascicular electrodes. Comput. Intell. Neurosci. 2010, 836346 (2010).
Jezernik, S., Grill, W. W. & Sinkjaer, T. Neural network classification of nerve activity recorded in a mixed nerve. Neurol. Res. 23, 429–434 (2001).
Parker, P., Englehart, K. & Hudgins, B. Myoelectric signal processing for control of powered limb prostheses. J. Electromyogr. Kines. 16, 541–548 (2006).
Hargrove, L., Zhou, P., Englehart, K. & Kuiken, T. A. The effect of ECG interference on pattern-recognition-based myoelectric control for targeted muscle reinnervated patients. IEEE Trans. Biomed. Eng. 56, 2197–2201 (2009).
Farina, D., Merletti, R. & Enoka, R. M. The extraction of neural strategies from the surface EMG. J. Appl. Phys. 96, 1486–1495 (2004).
Negro, F., Holobar, A. & Farina, D. Fluctuations in isometric muscle force can be described by one linear projection of low-frequency components of motor unit discharge rates. J. Physiol. 587, 5925–5938 (2009).
Aszmann, O. C. et al. Bionic reconstruction to restore hand function after brachial plexus injury: a case series of three patients. Lancet 6736, 1–7 (2015).
Ursu, D. C., Urbanchek, M. G., Nedic, A., Cederna, P. S. & Gillespie, R. B. In vivo characterization of regenerative peripheral nerve interface function. J. Neural Eng. 13, 26012 (2016).
Muceli, S. et al. Accurate and representative decoding of the neural drive to muscles in humans with multi-channel intramuscular thin-film electrodes. J. Physiol. 593, 3789–3804 (2015).
Ortiz-Catalan, M., Hakansson, B. & Branemark, R. An osseointegrated human-machine gateway for long-term sensory feedback and motor control of artificial limbs. Sci. Transl. Med. 6, 257re6 (2014).
Glaser, V., Holobar, A. & Zazula, D. Real-time motor unit identification from high-density surface EMG. IEEE Trans. Neural Syst. Rehabil. Eng. 21, 949–958 (2013).
Jiang, N., Vujaklija, I., Rehbaum, H., Graimann, B. & Farina, D. Is accurate mapping of EMG signals on kinematics needed for precise online myoelectric control? IEEE Trans. Neural Syst. Rehabil. Eng. 22, 549–558 (2014).
Farina, D., Negro, F. & Dideriksen, J. L. The effective neural drive to muscles is the common synaptic input to motor neurons. J. Physiol. 49, 1–37 (2014).
Negro, F., Muceli, S., Castronovo, A. M., Holobar, A. & Farina, D. Multi-channel intramuscular and surface EMG decomposition by convolutive blind source separation. J. Neural Eng. 13, 26027 (2016).
Farina, D. & Holobar, A. Characterization of human motor units from surface EMG decomposition. Proc. IEEE 104, 353–373 (2016).
Farina, D., Negro, F., Gazzoni, M. & Enoka, R. M. Detecting the unique representation of motor-unit action potentials in the surface electromyogram. J. Neurophysiol. 100, 1223–1233 (2008).
Negro, F. & Farina, D. Factors influencing the estimates of correlation between motor unit activities in humans. PLoS ONE 7, e44894 (2012).
Delp, S. L. et al. OpenSim: Open-source software to create and analyze dynamic simulations of movement. IEEE Trans. Biomed. Eng. 54, 1940–1950 (2007).
Saul, K. R. et al. Benchmarking of dynamic simulation predictions in two software platforms using an upper limb musculoskeletal model. Comput. Methods Biomech. Biomed. Eng. 18, 1445–1458 (2014).
Lu, T. W. & O’Connor, J. J. Bone position estimation from skin marker co-ordinates using global optimisation with joint constraints. J. Biomech. 32, 129–134 (1999).
Thelen, D. G. & Anderson, F. C. Using computed muscle control to generate forward dynamic simulations of human walking from experimental data. J. Biomech. 39, 1107–1115 (2006).
Manal, K. & Buchanan, T. S. A one-parameter neural activation to muscle activation model: estimating isometric joint moments from electromyograms. J. Biomech. 36, 1197–1202 (2003).
Sartori, M., Reggiani, M., Farina, D. & Lloyd, D. G. EMG-driven forward-dynamic estimation of muscle force and joint moment about multiple degrees of freedom in the human lower extremity. PLoS ONE 7, e52618 (2012).
Sartori, M., Farina, D. & Lloyd, D. G. Hybrid neuromusculoskeletal modeling to best track joint moments using a balance between muscle excitations derived from electromyograms and optimization. J. Biomech. 47, 3613–3621 (2014).
Acknowledgements
This work was supported by the European Research Council Advanced Grant DEMOVE (contract #267888) (to D.F.), the Christian Doppler Research Foundation of the Austrian Federal Ministry of Science, Research and Economy (to O.C.A.), the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement number 702491 (NeuralCon) (to F.N.) and Defense Advanced Research Projects Agency (DARPA N66001-15-1-4054) (to J.P.). The authors are grateful to M. Schweisfurth and H. Rehbaum for support in the experimental measurements, M. Castronovo for support in the data analysis, and C. Hofer and S. Salminger for clinical support.
Author information
Authors and Affiliations
Contributions
D.F. and O.C.A. conceived the study. I.V., T.K., M.S. and K.B. performed the acquisition. I.V., T.K., M.S., F.N., N.J., A.A. and J.P. conducted the analysis. D.F., I.V., T.K., M.S., F.N., N.J., K.B., J.P. and O.C.A. interpreted the data. D.F., I.V. and O.C.A. wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Farina, D., Vujaklija, I., Sartori, M. et al. Man/machine interface based on the discharge timings of spinal motor neurons after targeted muscle reinnervation. Nat Biomed Eng 1, 0025 (2017). https://doi.org/10.1038/s41551-016-0025
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41551-016-0025
This article is cited by
-
Neuromorphic hardware for somatosensory neuroprostheses
Nature Communications (2024)
-
ChatGPT and large language models in orthopedics: from education and surgery to research
Journal of Experimental Orthopaedics (2023)
-
A myoelectric digital twin for fast and realistic modelling in deep learning
Nature Communications (2023)
-
Recent advances in flexible noninvasive electrodes for surface electromyography acquisition
npj Flexible Electronics (2023)
-
Transformer-based hand gesture recognition from instantaneous to fused neural decomposition of high-density EMG signals
Scientific Reports (2023)