Abstract
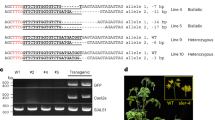
Developing herbicide-tolerant varieties by genome editing holds great promise for addressing the worsening weed problems in wheat cultivation1. Here, we generated transgene-free wheat germplasms harbouring herbicide tolerance mutations that confer tolerance to sulfonylurea-, imidazolinone- and aryloxyphenoxy propionate-type herbicides by base editing the acetolactate synthase (ALS) and acetyl-coenzyme A carboxylase genes. These stackable herbicide tolerance traits provide a potentially powerful tool for weed management. In addition, we found that base editing at the wheat ALS Pro-174 codon (TaALS-P174) endowed wheat with sufficient resistance to nicosulfuron herbicide in MS growth medium to allow selection. When the TaALS-P174 editor was coupled with editors for other targets of interest, co-editing occurred in the nicosulfuron-resistant plants, and selection for resistance in growth medium enriched the frequency of coupled targets by several-fold. This selectable co-editing system has the potential to greatly bolster adoption of base editing for crop improvement applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Data availability
All data supporting the findings of this study are available in the article or its Supplementary Information, or are available from the corresponding author upon reasonable request. Sequence data in this article can be found in the Ensembl Genomes database (http://plants.ensembl.org/Triticum_aestivum/Info/Index) under the following accession codes: TaALS-A (TraesCS6A02G288000), TaALS-B (TraesCS6B02G317400), TaALS-D (TraesCS6D02G268700), TaACCase-A (TraesCS2A02G069400), TaACCase-B (TraesCS2B02G082500) and TaACCase-D (TraesCS2D02G068100)
References
Zhang, C. X., Li, X. J., Huang, H. J. & Wei, S. H. Alert and prevention of the spreading of Aegilops tauschii, a worst weed in wheat field. Acta Phytophylacica Sin. 34, 103–106 (2007).
Global Status of Commercialized Biotech/GM Crops: 2016 Brief 52 (ISAAA, 2016).
Ball, D. A., Young, F. L. & Ogg, A. G. Selective control of jointed goatgrass (Aegilops cylindrica) with imazamox in herbicide-resistant wheat. Weed Technol. 13, 77–82 (1999).
Sudianto, E. et al. Clearfield® rice: Its development, success, and key challenges on a global perspective. Crop Prot. 49, 40–51 (2013).
Alister, C. & Kogan, M. Efficacy of imidazolinone herbicides applied to imidazolinone-resistant maize and their carryover effect on rotational crops. Crop Prot. 24, 375–379 (2005).
Sosnoskie, L. M., Culpepper, A. S., York, A. C., Beam, J. B. & MacRae, A. W. Sequential applications for mesosulfuron and nitrogen needed in wheat. Weed Technol. 23, 404–407 (2009).
Yu, Q., Ahmad-Hamdani, M., Han, H., Christoffers, M. J. & Powles, S. B. Herbicide resistance-endowing ACCase gene mutations in hexaploid wild oat (Avena fatua): insights into resistance evolution in a hexaploid species. Heredity 110, 220–231 (2013).
Komor, A. C., Kim, Y. B., Packer, M. S., Zuris, J. A. & Liu, D. R. Programmable editing of a target base in genomic DNA without double-stranded DNA cleavage. Nature 533, 420–424 (2016).
Yu, Q. & Powles, S. B. Resistance to AHAS inhibitor herbicides: current understanding. Pest Manag. Sci. 70, 1340–1350 (2014).
Shimatani, Z. et al. Targeted base editing in rice and tomato using a CRISPR–Cas9 cytidine deaminase fusion. Nat. Biotechnol. 35, 441–443 (2017).
Chen, Y. et al. CRISPR/Cas9-mediated base-editing system efficiently generates gain-of-function mutations in Arabidopsis. Sci. China Life Sci. 60, 520–523 (2017).
Tian, S. W. et al. Engineering herbicide-resistant watermelon variety through CRISPR/Cas9-mediated base-editing. Plant Cell Rep. 37, 1353–1356 (2018).
Zong, Y. et al. Efficient C-to-T base editing in plants using a fusion of nCas9 and human APOBEC3A. Nat. Biotechnol. 36, 950–954 (2018).
Poppell, C. A., Hayes, R. M. & Mueller, T. C. Dissipation of nicosulfuron and rimsulfuron in surface soil. J. Agric. Food Chem. 50, 4581–4585 (2002).
Zong, Y. et al. Precise base editing in rice, wheat and maize with a Cas9-cytidine deaminase fusion. Nat. Biotechnol. 35, 438–440 (2017).
Zhang, Y. et al. Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat. Commun. 7, 12617 (2016).
Leach, L. J. et al. Patterns of homoeologous gene expression shown by RNA sequencing in hexaploid bread wheat. BMC Genom. 15, 276 (2014).
Kong, L. G. et al. Differential responses of two types of winter wheat (Triticum aestivum L.) to autumn- and spring-applied mesosulfuron-methyl. Crop Prot. 28, 387–392 (2009).
Kawai, K. et al. A novel mutant acetolactate synthase gene from rice cells, which confers resistance to ALS-inhibiting herbicides. J Pesticide Sci. 32, 89–98 (2007).
Lee, K. Y. et al. The molecular basis of sulfonylurea herbicide resistance in tobacco. EMBO J. 7, 1241–1248 (1988).
Ostlie, M. et al. Development and characterization of mutant winter wheat (Triticum aestivum L.) accessions resistant to the herbicide quizalofop. Theor. Appl. Genet. 128, 343–351 (2015).
Kim, D. et al. Genome-wide target specificities of CRISPR RNA-guided programmable deaminases. Nat. Biotechnol. 35, 475–480 (2017).
Xiao, A. et al. CasOT: a genome-wide Cas9/gRNA off-target searching tool. Bioinformatics 30, 1180–1182 (2014).
Shan, Q. W. et al. Targeted genome modification of crop plants using a CRISPR–Cas system. Nat. Biotechnol. 31, 686–688 (2013).
Xing, H. L. et al. A CRISPR/Cas9 toolkit for multiplex genome editing in plants. BMC Plant Biol. 14, 327 (2014).
Acknowledgements
This work was supported by grants from the National Key Research and Development Program of China (2016YFD0101804), the National Natural Science Foundation of China (31788103, 31471785, 31872933 and 31570369), the Chinese Academy of Sciences (QYZDY-SSW-SMC030 and KFJ-STS-ZDTP-024) and Beijing Municipal Science and Technology (D171100007717001 and Z171100001517001).
Author information
Authors and Affiliations
Contributions
C.G., L.J. and J. Li designed the experiments; R.Z., Z.C., S.C. and Y.B. performed most of the experiments; J. Liu generated mutant plants. Y.Z. and K.C. analysed the results; C.G., L.J. and J. Li supervised the project; C.G., L.J., J. Li and R.Z. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
C.G. and L.J. are inventors on patent applications covering generation of herbicide torlerance traits in wheat described in this work. C.G., R.Z. and J. Liu are inventors on patent applications describing generation of a selectable marker in wheat using base editing.
Additional information
Journal peer review information: Nature Plants thanks Donald Weeks, Bing Yang and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figures 1–8 and Supplementary Tables 1–13.
Rights and permissions
About this article
Cite this article
Zhang, R., Liu, J., Chai, Z. et al. Generation of herbicide tolerance traits and a new selectable marker in wheat using base editing. Nat. Plants 5, 480–485 (2019). https://doi.org/10.1038/s41477-019-0405-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41477-019-0405-0
This article is cited by
-
Precise fine-turning of GhTFL1 by base editing tools defines ideal cotton plant architecture
Genome Biology (2024)
-
Application of genome editing techniques to regulate gene expression in crops
BMC Plant Biology (2024)
-
Crop bioengineering via gene editing: reshaping the future of agriculture
Plant Cell Reports (2024)
-
Gene editing tool kit in millets: present status and future directions
The Nucleus (2024)